Abstract
Objectives
The purpose of this study was to evaluate the grade distribution of screen-detected ductal carcinoma in situ (DCIS) diagnosed in Ireland, in the context of the clinical trials currently underway to determine if active surveillance is a feasible management option for low-risk DCIS.
Setting
BreastCheck is the national breast screening programme in Ireland, offering screening to women aged 50 to 69 every two years.
Methods
This study was a secondary analysis of data collected by BreastCheck on all screen-detected DCIS diagnosed in the 12 years of nationwide screening. Incidence and detection rates were calculated. Descriptive analysis of the cases was performed and, for comparative analysis, grade of DCIS was analysed as a binary variable (high vs. low/intermediate) in keeping with the inclusion criteria for active surveillance trials. Analysis was performed in IBM Statistical Package for Social Sciences, version 26.
Results
Between 2008 and 2020, 2240 women were diagnosed with DCIS through BreastCheck; 876 (39.1%) were low/intermediate-grade. The overall incidence rate has remained relatively stable during this period. Women with low/intermediate-grade DCIS were younger than women with high-grade DCIS (56 (interquartile range: 56-61) years v 57 (interquartile range: 53–61) years; p < 0.001). They were also more likely to have been diagnosed at an initial screening episode compared with those who had high-grade lesions (42.5% v 29.0%; p < 0.001).
Conclusion
If current clinical trials recommend active surveillance as a feasible option for DCIS, up to 40% of women with screen-detected DCIS may be eligible. These women are younger and often diagnosed on initial screening episode, so may require longer active follow-up.
Introduction
Ductal carcinoma in situ (DCIS) is a pre-invasive lesion of the breast, in which the malignant epithelial cells are confined within the basement membrane and myoepithelial layer of the ductal system. 1 It is considered to be a non-obligate precursor as not all DCIS cases will progress to invasive cancer. 2 Currently, there is no robust method of determining which lesions will progress and which will remain indolent.3,4 Consequently, most women with DCIS are treated by surgical excision, often with adjuvant radiotherapy and endocrine therapy.3,5
Since the introduction of breast cancer screening programmes in recent decades, many countries have reported an increase in the incidence of DCIS,6,7 with the majority of DCIS diagnosed asymptomatically. 6 Ireland's breast cancer screening programme, BreastCheck, was established in 2000 and achieved nationwide coverage by 2008. 8 The programme now offers all women in Ireland between the ages of 50 and 69 years a mammogram every two years. 9 Digital mammography has been used in all screening units since 2008, with technology from Siemens, GE Healthcare, Sectra, and Hologic. Lesions seen on imaging are biopsied. Most DCIS lesions are stereotactically sampled by large-gauge vacuum-assisted core biopsy systems (Suros/EnCor Breast Biopsy Systems). A small number of calcifications are identified sonographically and are usually sampled with a 16-gauge hand-held biopsy needle.
Mammographic screening for breast cancer has resulted in an increased number of DCIS diagnoses, as most lesions are diagnosed in a pre-clinical phase; it is estimated in BreastCheck that non-calcified DCIS represent only 1-2%. Yet, despite the current management approach, the incidence of invasive breast cancer is not declining.7,10 This has led to the view that breast cancer screening is leading to the over-diagnosis and over-treatment of DCIS lesions which may have remained indolent. There is a move towards a more nuanced approach to DCIS management, with de-escalation of treatment, including judicious use of radiotherapy5,11 and lower doses of endocrine therapies. 12 In addition, there are a number of clinical trials underway to evaluate if active surveillance of low-risk lesions is a feasible alternative to surgical management. 13 Although these trials vary in their inclusion criteria and end-points, all stipulate that a lesion must be low/intermediate-grade to be included.14,15
In light of these developments, this study aimed to examine the trends in screen-detected DCIS in Ireland diagnosed through BreastCheck, and to identify which cases may be eligible for active surveillance, should the current clinical trials prove successful.
Methods
The study included all DCIS diagnosed through the screening programme between 2008 and 2020 (the years since nationwide coverage was achieved). BreastCheck has compiled a population-based register which is continuously updated, from which women are invited to attend screening. All women undergoing screening consent to their data being collected for use in research that benefits the programme. These routinely collected data were anonymised and access was granted by BreastCheck. The data provided included: age at time of screening, screening unit, episode of screening, year of screening, grade of DCIS, and code and name of electoral district of residence. Electoral district of residence was used as an indirect measure of socio-economic status by cross-referencing the Pobal Haase-Pratschke (HP) Deprivation Index 2016. 16
Additional data on numbers of women eligible for screening, numbers of women invited for screening, and numbers of women attending for screening were obtained from the publicly available annual reports published by BreastCheck. 17
The age distribution and socio-economic demographics of women diagnosed with DCIS, along with the year, screening episode, and screening location of these cases, are described. Incidence rates and detection rates were calculated based on the numbers of women eligible for screening and women attending for screening. Grade was transformed into a binary variable, high-grade and low/intermediate-grade, in line with the trials’ inclusion criteria. The Mann-Whitney U and Chi-square tests were used to compare the two groups. Statistical analysis was conducted in IBM Statistical Package for Social Sciences (SPSS) version 26.
Ethics exemption was received from University College Dublin Research Ethics.
Results
Between 2008 and 2020, 2240 women were diagnosed with DCIS through BreastCheck (Figure 1); 876 (39.1%) had low/intermediate-grade and 1353 (60.4%) had high-grade DCIS. Of the 764 women diagnosed on an initial screening episode, 372 cases (48.7%) had low/intermediate-grade DCIS and 392 cases (51.3%) had high-grade DCIS. On a subsequent screening episode, 1465 women were diagnosed with DCIS. Of these, 504 (34.4%) had low/intermediate-grade DCIS and 961 cases (65.6%) had high-grade DCIS.
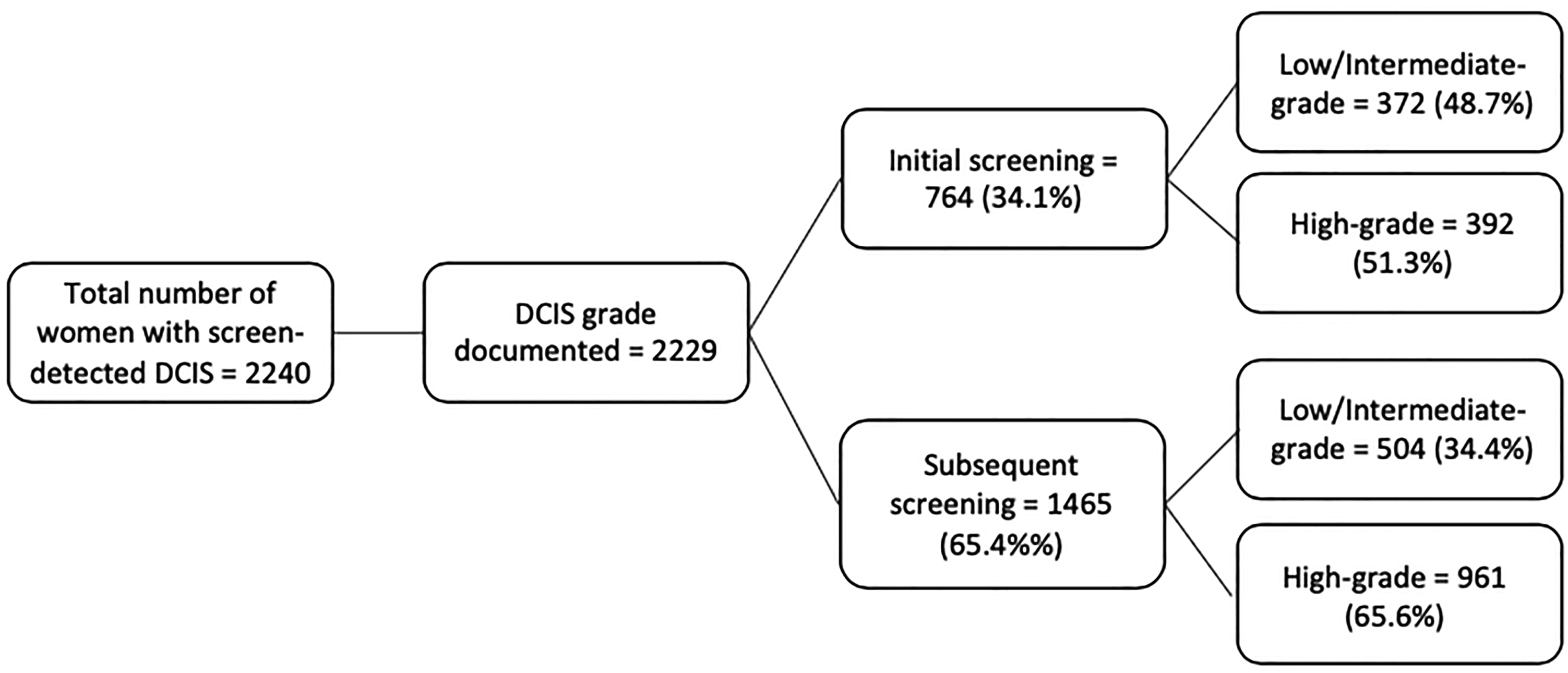
Flowchart of results of screening for ductal carcinoma in situ (DCIS), in Ireland 2008-2019, by screening episode and grade.
The trends in incidence (cases per 10,000 women) are shown in Figure 2. The incidence of screen-detected DCIS has fluctuated around 9-10 per 10,000. In 2008, the incidences of screen-detected low/intermediate-grade and high-grade DCIS were similar (approximately 5 per 10,000). However, the incidence of screen-detected high-grade DCIS has increased since then and has fluctuated around 6 per 10,000, whereas the incidence of screen-detected low/intermediate-grade DCIS has declined over the same period to 3.0 per 10,000 in 2019.
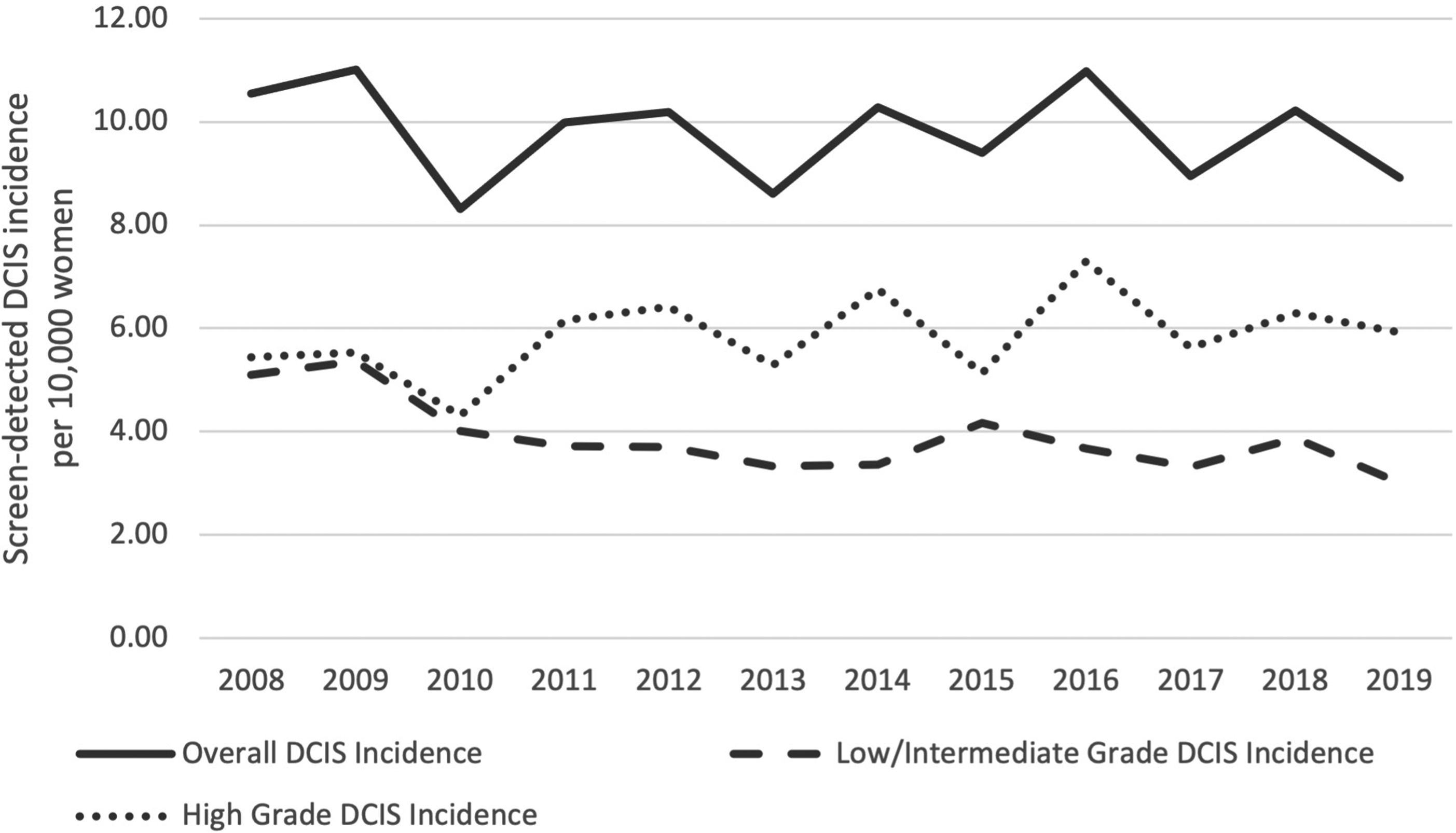
Trends in screen-detected ductal carcinoma in situ (DCIS) incidence in Ireland, 2008-2019.
Table 1 shows the detection rate of DCIS, by screening episode and by histological grade. The overall detection rate of DCIS was 1.4 per 1000 women screened. At initial screening this was 1.8 per 1000 women screened, dropping to 1.2 cases per 1000 women screened at subsequent screening, giving a subsequent to initial screening detection rate ratio of 0.7. The subsequent to initial screening detection rate ratio showed a larger decrease in detection rates between initial and subsequent screening for low/intermediate-grade DCIS compared with high-grade DCIS, with ratios of 0.3, 0.4 and 0.9 for low-, intermediate- and high-grade respectively.
Breastcheck DCIS data: detection rate per 1000 tests by grade and screening episode.
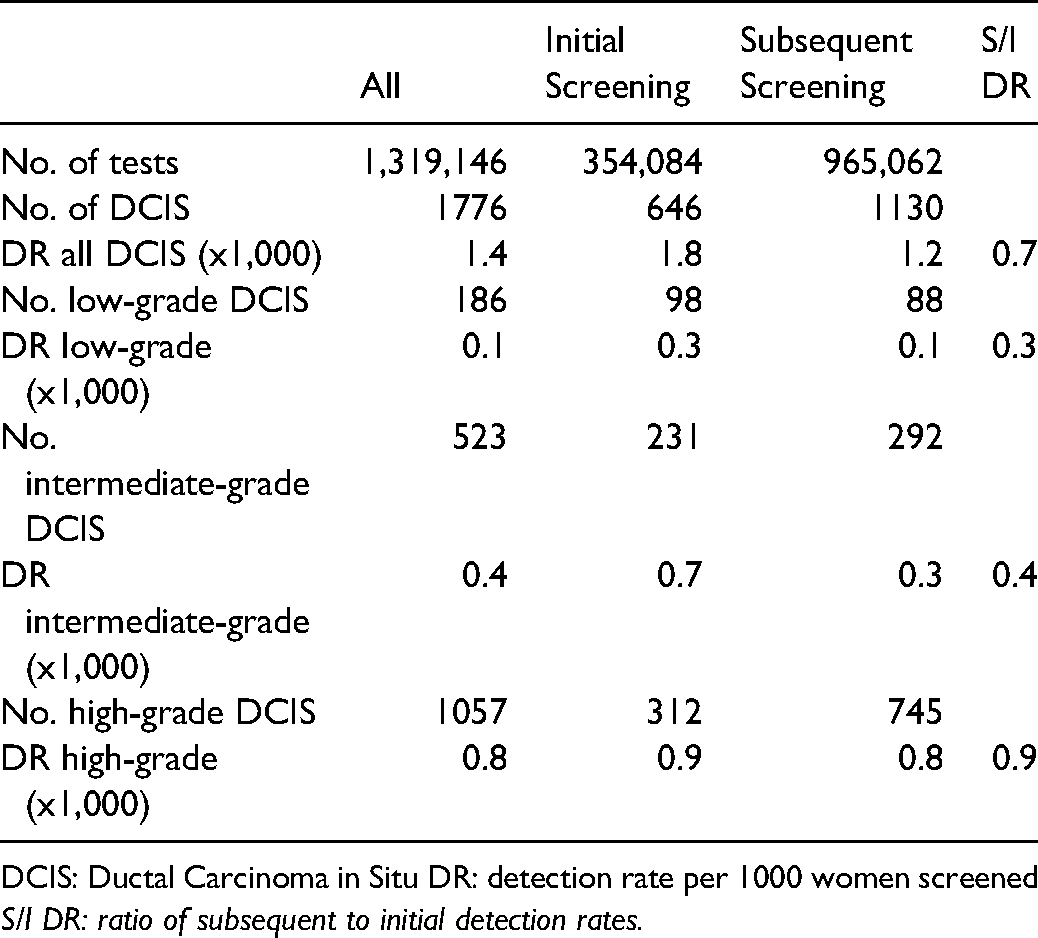
DCIS: Ductal Carcinoma in Situ DR: detection rate per 1000 women screened S/I DR: ratio of subsequent to initial detection rates.
The median age of women at diagnosis was 57 years (interquartile range (IQR) 52 to 61 years). Approximately half (48.7%) of women were of above average or affluent socioeconomic status. DCIS grade was known in 99.5% of women diagnosed. High-grade lesions accounted for 1353 (60.4%), low-grade for 234 (10.4%) and intermediate-grade for 642 (28.7%). Theoretically, 876 (39.1%) women diagnosed with DCIS through BreastCheck during the study period would have been eligible for inclusion in the active surveillance trials. The majority of women (65.6%) were diagnosed on a second or subsequent screening episode, with only one in three (34.4%) diagnosed at initial screen. See Table 2 for clinic-pathological and demographic details.
Breastcheck DCIS data: characteristics of women diagnosed with DCIS through breastCheck.
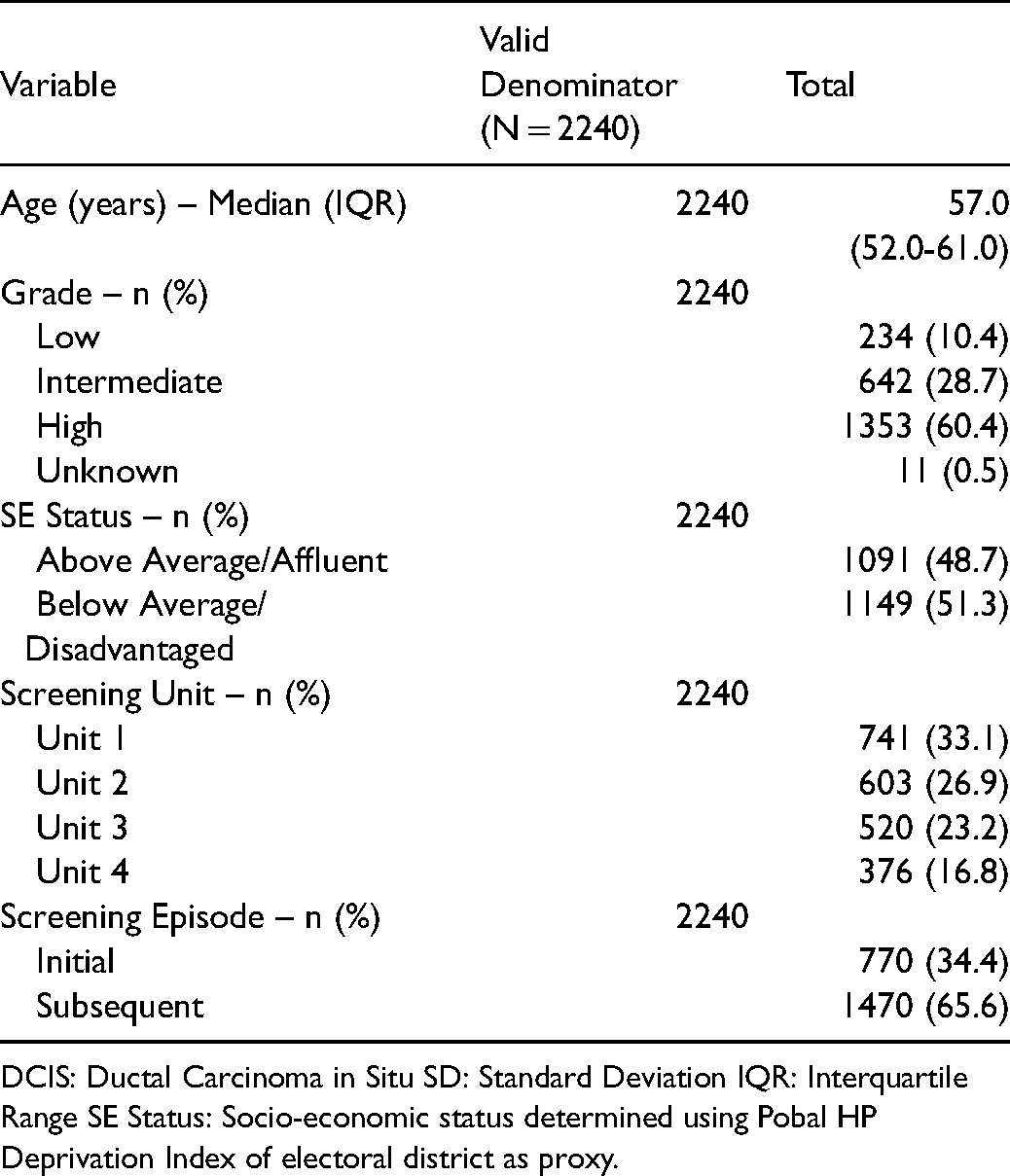
DCIS: Ductal Carcinoma in Situ SD: Standard Deviation IQR: Interquartile Range SE Status: Socio-economic status determined using Pobal HP Deprivation Index of electoral district as proxy.
Comparative analysis was performed to evaluate the two study groups – those who would be eligible for active surveillance (low/intermediate-grade DCIS) and those who would require treatment (high-grade DCIS) (Table 3). Women diagnosed with low/intermediate-grade DCIS were younger than those diagnosed with high-grade DCIS (median 56.0 years (IQR 51.0–61.0) v 57.0 (IQR 52.0–61.0); p < 0.001). The difference in the proportion of women of above average or affluent socio-economic status and of below average or disadvantaged socio-economic status between the two study groups was not statistically significant (p = 0.106). In keeping with the detection rate ratios, low/intermediate-grade was more often diagnosed at an initial screening episode, compared with high-grade DCIS (p<0.001). There was an overall significant difference in DCIS grade by screening unit (p = 0.028).
Breastcheck DCIS data: comparison of characteristics between Low/intermediate-grade and high-grade DCIS.
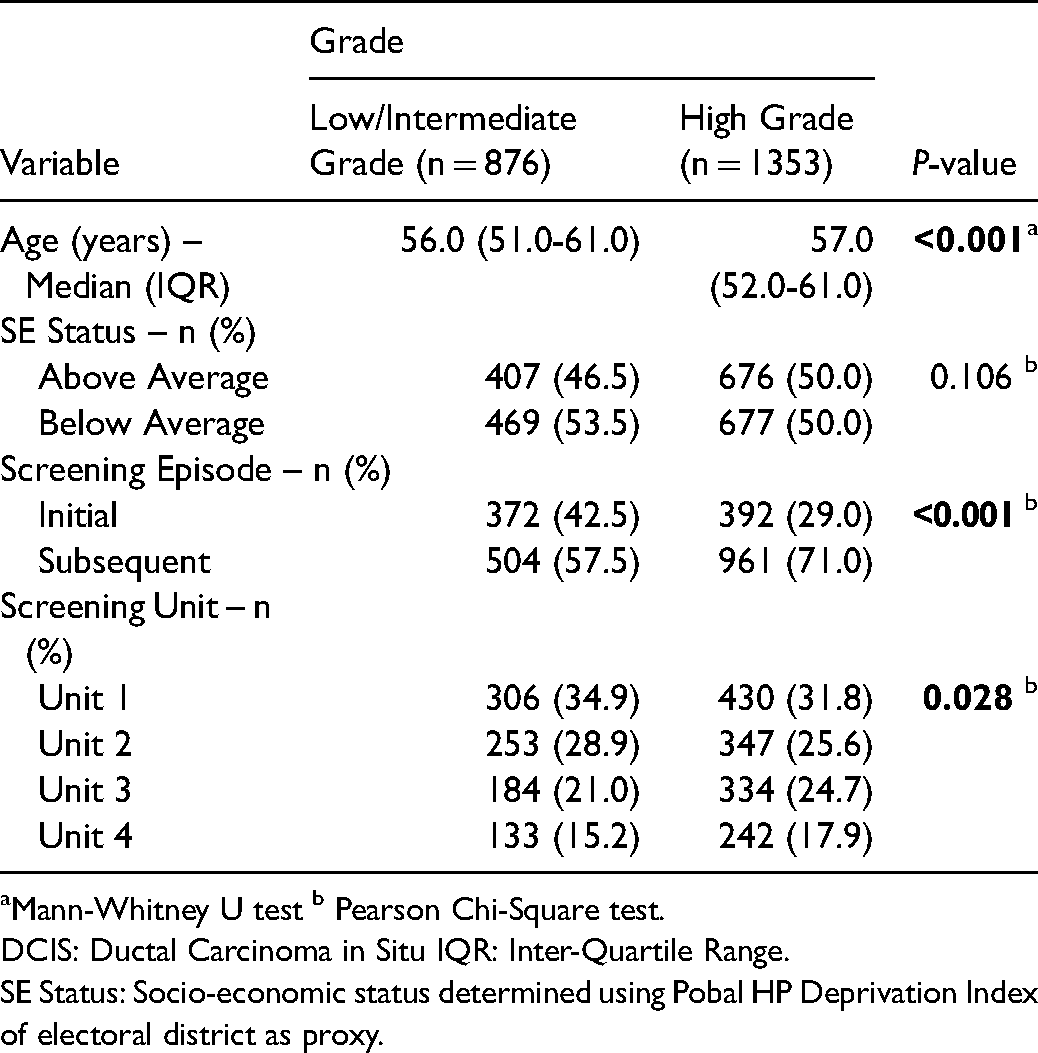
Mann-Whitney U test b Pearson Chi-Square test.
DCIS: Ductal Carcinoma in Situ IQR: Inter-Quartile Range.
SE Status: Socio-economic status determined using Pobal HP Deprivation Index of electoral district as proxy.
Discussion
This study reviewed DCIS detected through a national screening programme over 12 years of nationwide screening, in the context of the active surveillance trials currently underway. 13 The purpose of the study was to (i) determine trends in screen-detected DCIS over the study period and (ii) given the possibility that active surveillance may become an alternative management strategy, to ascertain the proportion of those diagnosed with DCIS who may benefit from such an option and to establish if there are any factors which would need to be considered in planning for such services.
Over 2000 women were diagnosed with DCIS through BreastCheck during the study period. Of those women, 10.4% (234 cases) and 28.7% (642 cases) were diagnosed with low- and intermediate-grade respectively. According to inclusion criteria for the active surveillance trials, grades 1 and 2 lesions are considered low risk and hence eligible for inclusion.14,15 Therefore, based on these criteria, 39.1% of women diagnosed with screen-detected DCIS would be eligible for inclusion on these trials and, depending on the results of these trials, may be eligible for consideration of active surveillance.
The overall incidence of screen-detected DCIS has remained relatively stable since 2008, with the proportion of DCIS in the total rate of diagnosed carcinomas remaining between 18% and 23% since 2006. However, the proportion of high grade DCIS has increased. This is in keeping with the observation that DCIS is more commonly diagnosed in older women with an expected change in the distribution of grade as women proceed from incident rounding screening through subsequent mammographic screens. 18 In our study we also observed differences in grade distribution between the four screening units. This may be partly explained by the fact that in two of the units screening had been operational since 2000 while screening commenced in the other two in 2008, such that the populations are not entirely comparable.
The possibility that inter-observer variation between pathologists may be causing these differences in grade distribution also requires consideration.14,19 Although diagnostic criteria for DCIS grading are well established, further standardisation of the grading systems19,20 and a second review of DCIS biopsies 14 have been highlighted internationally as necessary measures, should active surveillance be implemented. In order to address this in Ireland, a second pathologist review is already performed for all core biopsies as part of routine multidisciplinary team process, and all breast pathologists working in the breast screening programme are specialist trained. In addition, adherence to published guidelines (eg. Royal College of Pathologists (RCPath) guidelines) and mandatory participation in the UK National Health Service (NHS) Breast Pathology External Quality Assessment (EQA) scheme improves consistency of diagnoses. 21 There is a standardised grading system used by all pathologists in the breast screening programme, which is based on nuclear morphology. The features of this are documented in the European Guidelines for Quality Assurance in Breast Cancer Screening and Diagnosis (4th edition). 22
Necrosis is not a criterion for DCIS grading, and whilst it is more common in high-grade DCIS, it does occur in all grades. Although the presence of necrosis is documented in pathology reports, because it is not a diagnostic criterion and because it is not on the list of the UK Breast EQA RCPath core dataset items, 23 it is not entered onto the screening programme datasheet, and hence presence of necrosis was not readily available for inclusion in this study. However, the recently published International Collaboration on Cancer Reporting DCIS guidelines do list necrosis as a core item. 24 Therefore, necrosis may be collected through the screening programme database in future.
Women diagnosed with low/intermediate-grade DCIS were, on average, younger than those diagnosed with high-grade DCIS. This likely reflects the finding that low/intermediate-grade DCIS is more often diagnosed on an initial screening episode, when the women undergoing screening are likely to be younger. Whilst the ratio of subsequent to initial screening episode detection rate (S/I DR ratio) overall is 0.7, the detection rates of low- and intermediate-grade lesions are lower on subsequent screens (S/I DR ratios of 0.3 and 0.4 respectively) whereas the detection rates of high-grade lesions are similar on subsequent and initial screens (S/I DR 0.9). These findings, in keeping with those of other international studies,20,25 suggest that the pre-clinical phase, during which diagnosis through screening is possible, is longer for low/intermediate-grade lesions than for high-grade lesions. This in turn suggests that low/intermediate-grade lesions progress at a slower rate than high-grade lesions, adding to evidence that low/intermediate-grade DCIS should be considered a different entity to high-grade lesions when it comes to treatment decisions and to prevent over-treatment. 20
The active surveillance trials are currently recruiting and in the early stages of follow-up. If these trials show that active surveillance is a safe, feasible, and cost-effective alternative to the current management of DCIS, cancer services internationally will need to be prepared to offer this approach as an option. Services need to be planned to ensure there is capacity to provide the additional mammograms and follow-up appointments that would be required for potentially 4 in 10 women with screen-detected DCIS. The follow-up imaging regimen differs between the trials, with two trials offering bilateral mammograms every 12 months, and the other two trials offering an ipsilateral mammogram every 6 months and contralateral mammograms every 12 months.14,15 The follow-up period in the trials also differs, between 5 and 10 years.14,15 During the period 2008-2019, the incidence of screen-detected low/intermediate grade DCIS was between 3.0 and 5.4 per 10,000 women. As these women tend to be younger they may require 6-12 monthly mammograms for upwards of 5-10 years, depending on the trial findings and the final protocols developed. Whether current population screening programmes would be expanded to incorporate these additional mammograms, or whether new services would need to be established to meet this need, would need to be considered.
There are a number of strengths associated with this study. The data used were collected by a national population breast screening programme. It is a census study of all DCIS detected through screening over a 12-year period, with all 2240 women included, precluding selection bias. This study used routine data which had been collected contemporaneously, requiring no additional data to be collected subsequently, ensuring no recall bias. The grade of DCIS lesion was unknown in only 0.5% cases, and so it was appropriate to use complete case analysis.
The study has a number of limitations. Attendance at screening is voluntary and there may be bias as a result of non-attendance. Women who attend for screening and are diagnosed with DCIS may differ from those who do not attend screening, who will either remain asymptomatic or present with invasive cancer. However uptake in BreastCheck is consistently higher than the European target of 70%. 26 Another potential source of bias in the data may be interobserver variability between pathologists at different screening sites. However, the second review of biopsy specimens should improve consistency of grading.
This study was limited in its design as a cross-sectional study, however, this was the most appropriate study to conduct at this stage and with the available data. It is limited to being a descriptive study, without the possibility of drawing conclusions about causal relationships. Finally, this study is unable to determine how many of the women who would be considered low risk would actually opt for active surveillance if that was a feasible option. Further qualitative research would be beneficial to determine the acceptability of active surveillance, as has been done in other jurisdictions. 27 This research would also be useful to identify potential barriers and facilitators to the acceptance of active surveillance, which could be addressed in the implementation of such an approach.
Conclusions
DCIS rates vary between screening programmes, but this study shows that up to 40% of women diagnosed with DCIS through screening programmes may be eligible for active surveillance, should the current trials show that this is a feasible option. These women will require repeat mammograms and, given the younger age at diagnosis, may need prolonged follow-up. This will require planning to ensure capacity is available for these surveillance scans and further treatment if necessary. The findings of this study may assist with this planning in future.
Until the trials publish their findings, the current approach of surgical excision for management of DCIS is probably the safest option.11,28,29 However, with the possibility of active surveillance, clinicians may decide to use this as an opportunity to re-evaluate how they view DCIS and how they communicate risk to patients, 30 to ensure that screen-detected DCIS is not being over-treated.
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
