Abstract
Objective
To achieve the lung cancer screening (LCS) mortality benefit in clinical trials, timely, real-world follow-up of abnormal test results is necessary. Presently, annual LCS rates are lower than in trials, and adherence to follow-up after suspicious findings has not been well studied. This study examined timely adherence to follow-up recommendations after positive low-dose computed tomography (LDCT) screenings.
Methods
This retrospective study included individuals from two academic primary care practices in New York City who met United States Preventative Services Task Force LCS eligibility and had a positive LDCT scan between 2013 and 2020. They were recommended for shorter interval follow-up repeat computed tomography (CT), CT biopsy, or positron emission tomography/CT. Adherence was completion of the prescribed imaging by 15 days after the recommended 7-, 30-, and 90-day follow-up and by 30 days after the 180-day recommended follow-up.
Results
Among 106 individuals with a positive LDCT scan, 64 (60%) were adherent to follow-up recommendations. Adherence was 72%, 63%, and 42% for recommended follow-ups of 30, 90, and 180 days, respectively. Being male was a predictor of a lower adherence rate. Among 23 individuals newly diagnosed with lung cancer after a positive LDCT scan, 83% were adherent to follow-up testing and 82% of cancers were Stage 1A or limited stage.
Conclusions
There was variable adherence to the LCS follow-up recommendations despite positive screening CT, suggesting that even in a well-established screening program there may not be an efficient, systematic approach for follow-up. The delays in repeat testing potentially undermine the benefits of early detection.
Introduction
Lung cancer is the leading cause of cancer-related mortality in the United States and accounted for 23% of cancer deaths in 2020. 1 The survival rate varies significantly by stage: early-stage lung cancer is associated with a 61% 5-year survival compared to 7% for the distant stage. 2 The survival benefit of lung cancer screening (LCS) via low-dose computed tomography (LDCT) was first demonstrated in the National Lung Screening Trial (NLST), in which screening led to a 20% relative reduction in mortality compared to chest x-ray over a 6.5-year period. The mortality benefit was confirmed by a subsequent trial, in which computed tomography (CT) scanning decreased mortality by 24% versus no screening over a 10-year period in the Netherlands.3,4
LCS currently carries a grade “B” recommendation by the United States Preventative Services Task Force (USPSTF) for the annual screening of high-risk individuals aged 50 to 80 with ≥20 pack-year smoking history for individuals who currently smoke or those who quit within the last 15 years. An estimated 12 million individuals in the United States are eligible according to the current USPSTF guidelines. This is a 31% increase compared to the prior recommendation 5 in 2013 that had a more limited eligibility criteria of age 55 to 80 with ≥30 pack-year smoking history. National rates of LCS have been slowly increasing but remain low, with only 18% of eligible individuals undergoing LCS in 2018 and 21% in 2019.6,7 While there was a pandemic-associated decrease in breast, colorectal, and prostate cancer screenings, LCS rates remained stable with significant variation between states in 2020.8,9 Continued adherence is crucial as the full benefits of screening are only attained when eligible patients receive both screening and subsequent follow-up in a timely manner.
Among studies evaluating LCS adherence, most have focused on annual adherence to routine screening. A meta-analysis of 21 studies found a pooled annual adherence rate of only 57%, despite most sites using dedicated personnel and interventions. In sub-group analysis, those recommended for short-term follow-up had a significantly higher adherence rate of 74%. However, all adherence rates are much lower than those in clinical trials and modeling studies that were the basis for current guidelines. 5 Thus, the same survival benefit is likely not being achieved. Within these studies, shorter post-screening follow-up intervals have been identified as a potential predictor of adherence.10–15 However, to date there have not been studies that have specifically focused on this high-risk cohort and specific predictors of adherence.
The goal of this study was to evaluate the real-world adherence to recommendations for shorter than annual repeat screening follow-ups after positive findings on LDCT scans in a large academic medical center. Patients were recommended for repeat LDCT, positron emission tomography (PET)/CT, or CT biopsy in follow-up time frames of 7, 30, 90, and 180 days. Secondary outcomes include characterizing the timeliness of follow-up between different recommendations and determining specific factors that may impact adherence to follow-up.
Methods
We conducted a retrospective study of patients seen at two primary care practices in an academic health system located in New York City (NY, USA). Patients were selected from a query of our electronic medical record (EMR) data repository for individuals who received an initial LDCT between January 2013 and September 2020. Eligibility criteria were based on the USPSTF 2021 guidelines (age 50–80 with a 20 pack-year history and <15 years quit date for former smokers) and included those who had a positive finding with recommended follow-up scan (repeat LDCT, PET/CT, or CT biopsy) in 6 months or less. The initial LDCT scan indication was reviewed in the EMR to ensure that these scans were only ordered for asymptomatic screenings and that those who received LDCT scans for other indications, including pneumonia, other lung-related pathology, and metastatic cancer monitoring, were excluded. The data collection for this study was approved by the Icahn School of Medicine at Mount Sinai Institutional Review Board.
The LDCT scan results were abstracted from the EMR with the reading radiologist's assessment and recommended follow-up. The LDCT scoring system and recommendations used for follow-up at this institution were based on the International Early Lung Cancer Action Program protocols. 16
The primary outcome of interest was adherence to recommendations for follow-up tests post-LDCT screening. Primary care providers and pulmonologists received the radiologist LDCT report electronically and contacted patients via varying methods including telephone, EMR patient portal, or during in-person visits to discuss the results and schedule follow-up testing. These were documented via telephone encounters or office visit notes and reviewed as part of this study. Overall, there was no systematic standardized process of communication with patients. The prescribed follow-up scan was ordered electronically by the provider, and patients were both given information and contacted by the radiology office to schedule the scan. There was adequate capacity within the health system for patients to schedule scans. Adherence was defined as receiving the recommended repeat CT, PET/CT, or CT biopsy by 15 days after the recommended 7-, 30-, and 90-day follow-up and by 30 days after the recommended 180-day follow-up. 16 Non-adherence was defined as not receiving any follow-up scan within the prescribed time frames after the positive LDCT, but the EMR was reviewed at future dates to assess if later scans were completed. The number of newly diagnosed lung cancers that resulted from screening was recorded, by histologic type (adenocarcinoma, large cell, small cell lung cancer (SCLC), and squamous cell) and staging (extensive and limited for SCLC and Stage 1A and Stage 1B for all others).
We collected the following demographic characteristics from the EMR: age, sex, race (White, African-American, and other), marital status, type of insurance (private, Medicare, Medicaid, and uninsured), and primary spoken language (English vs. non-English). Income was estimated using US census data by zip code. Personal health characteristics included smoking history (status and pack-year history), specific comorbidities (chronic obstructive pulmonary disease, cardiovascular disease, diabetes, and stroke), and family history of any cancer among first-degree relatives. Our sample consisted of patients belonging to primary care practices. The hospital-associated practice is staffed by attending and resident physicians; the faculty practice is staffed by attending physicians only with a payer mix that has a higher proportion of private insurance plans. Distance from the radiology site was the mileage between the patient's address and the radiology scanning center. Outpatient visits included outpatient office visits for any provider over the previous 12 months. Follow-up communication regarding LCS results included timing and type of communication, either telephone encounter or office visit with documentation addressing LDCT scan results.
We used chi-square, Wilcoxon rank sum test, and t-test to compare baseline characteristics between the adherent and nonadherent groups. Logistic regression analysis was performed to assess potential predictors of screening adherence. All statistical analysis was performed using SAS, version 9.4 (SAS Institute Inc., Cary, NC, USA).
Results
Among the 709 individuals who met USPSTF LCS criteria, 106 (15%) had an initial LDCT with positive findings and were recommended shorter follow-ups. As detailed in Table 1, the mean age was 64.8 ± 6.3 years, 62% were female, and regarding race, 31% were White, 33% were African-American, and 36% were Other. Nearly all listed English as the primary language (93%). The most common insurance was Medicaid (42%), followed by Medicare (33%) and private (25%), with most patients (64%) seen at the hospital-associated practice; 52% were individuals who currently smoked with a median smoking history of 38 pack-years. In total, there were 23 newly diagnosed lung cancers (22%); 82% were Stage 1A (non-SCLC) or limited stage (SCLC), and information on type and staging was not available for one case (Table 2).
Characteristics of the study population, adherent and nonadherent cohorts.
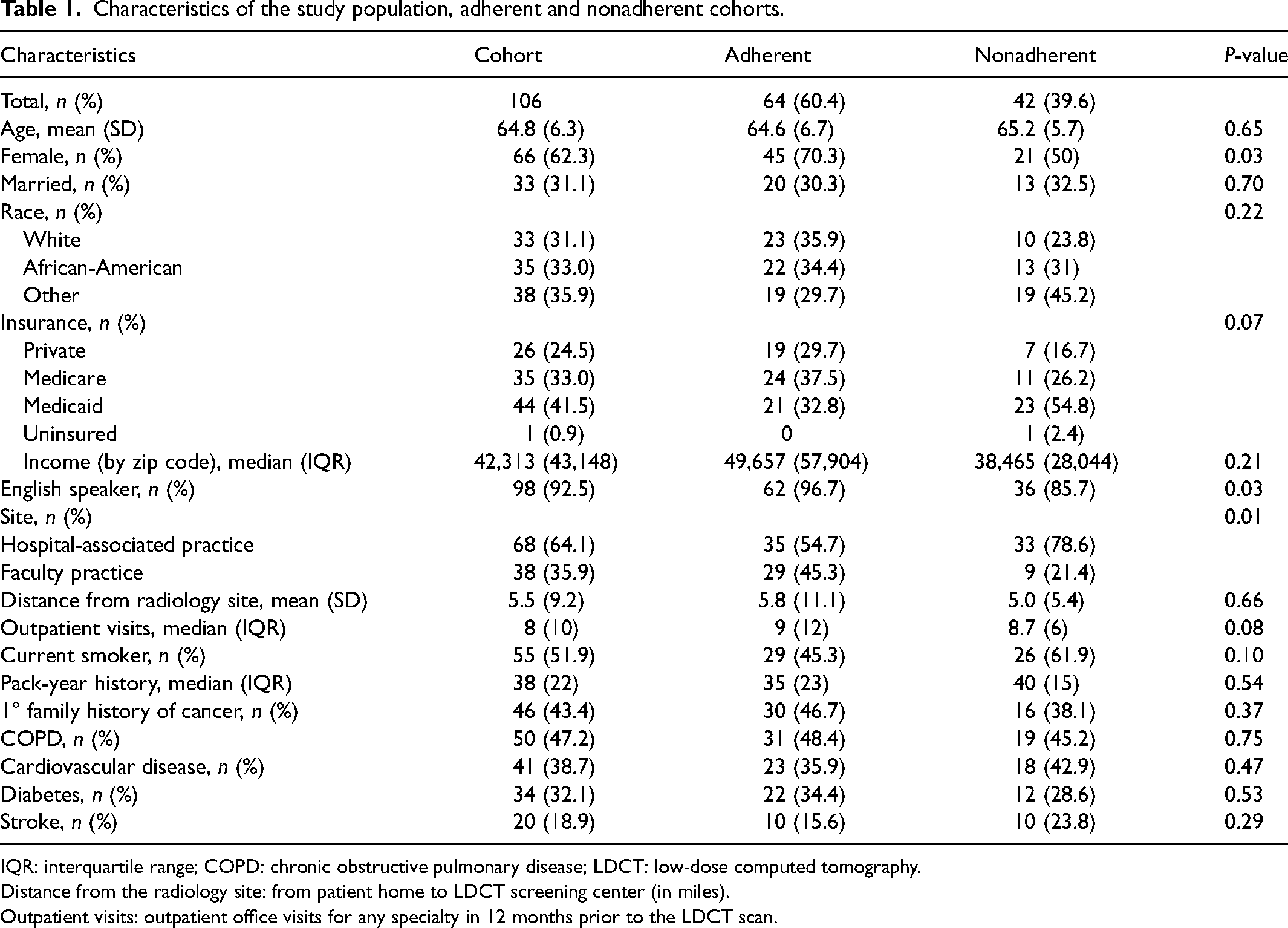
IQR: interquartile range; COPD: chronic obstructive pulmonary disease; LDCT: low-dose computed tomography.
Distance from the radiology site: from patient home to LDCT screening center (in miles).
Outpatient visits: outpatient office visits for any specialty in 12 months prior to the LDCT scan.
Follow-up and new lung cancer diagnoses.
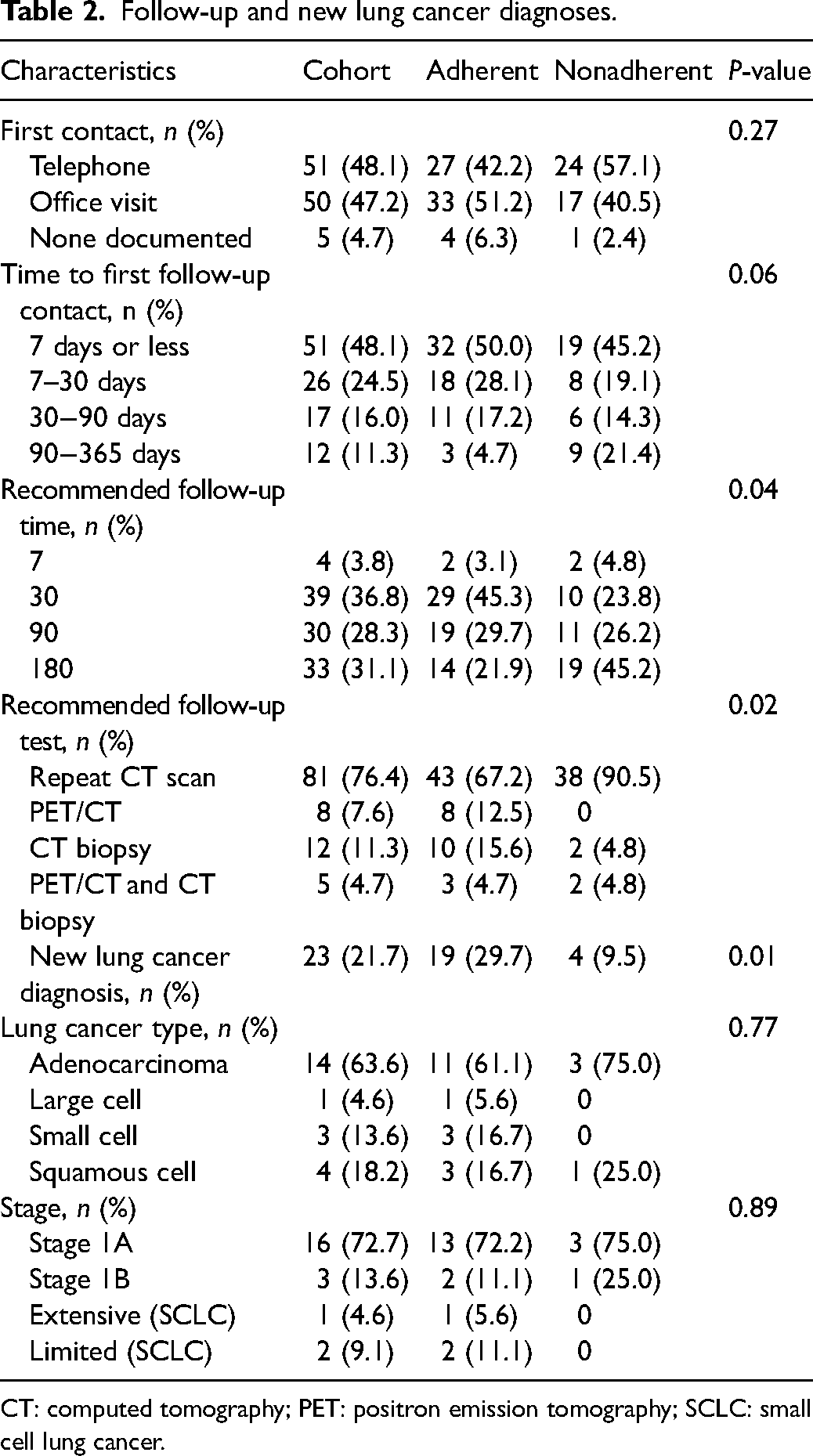
CT: computed tomography; PET: positron emission tomography; SCLC: small cell lung cancer.
The overall rate of adherence to follow-up recommendations was 60%. Compared to those who were nonadherent, a higher proportion of those who were adherent was female sex (70% vs. 50%), primary English speakers (97% vs. 86%), and was seen at the faculty practice (45% vs. 21%), (p < 0.05 for each comparison) (Table 1). Shorter recommended follow-up times, recommended follow-up scan of PET/CT scan or CT biopsy, and new lung cancer diagnoses were associated with higher adherence to recommendations (Table 2).
Median follow-up time was 34 days (interquartile range (IQR): 30), 95 days (IQR: 58), and 277 days (IQR: 185) for recommended follow-ups of 30, 90, and 180 days, respectively. The adherence rate was 71.8% (95% confidence interval (CI): 58.7%−85.9%), 63.3% (95% CI: 46.1%−77.5%), and 42.4% (95% CI: 25.6%−59.3%) for individuals with recommended follow-up times of 30, 90, and 180 days. Of note, 8% of patients in this study did not have a documented follow-up scan and 4% of those who received follow-up scans appropriately did not have any documentation of discussion with their primary care provider after initial LCS.
Among those who were adherent to LCS recommendations, a higher proportion was female (odds ratio (OR) 3.0; 95% CI: 1.1–7.9) (Table 3). English as a primary language, clinic site, race, insurance status, shorter recommended screening follow-up time, and a new lung cancer diagnosis were not found to have a significant association with screening adherence.
Logistic regression analysis of predictors of adherence.
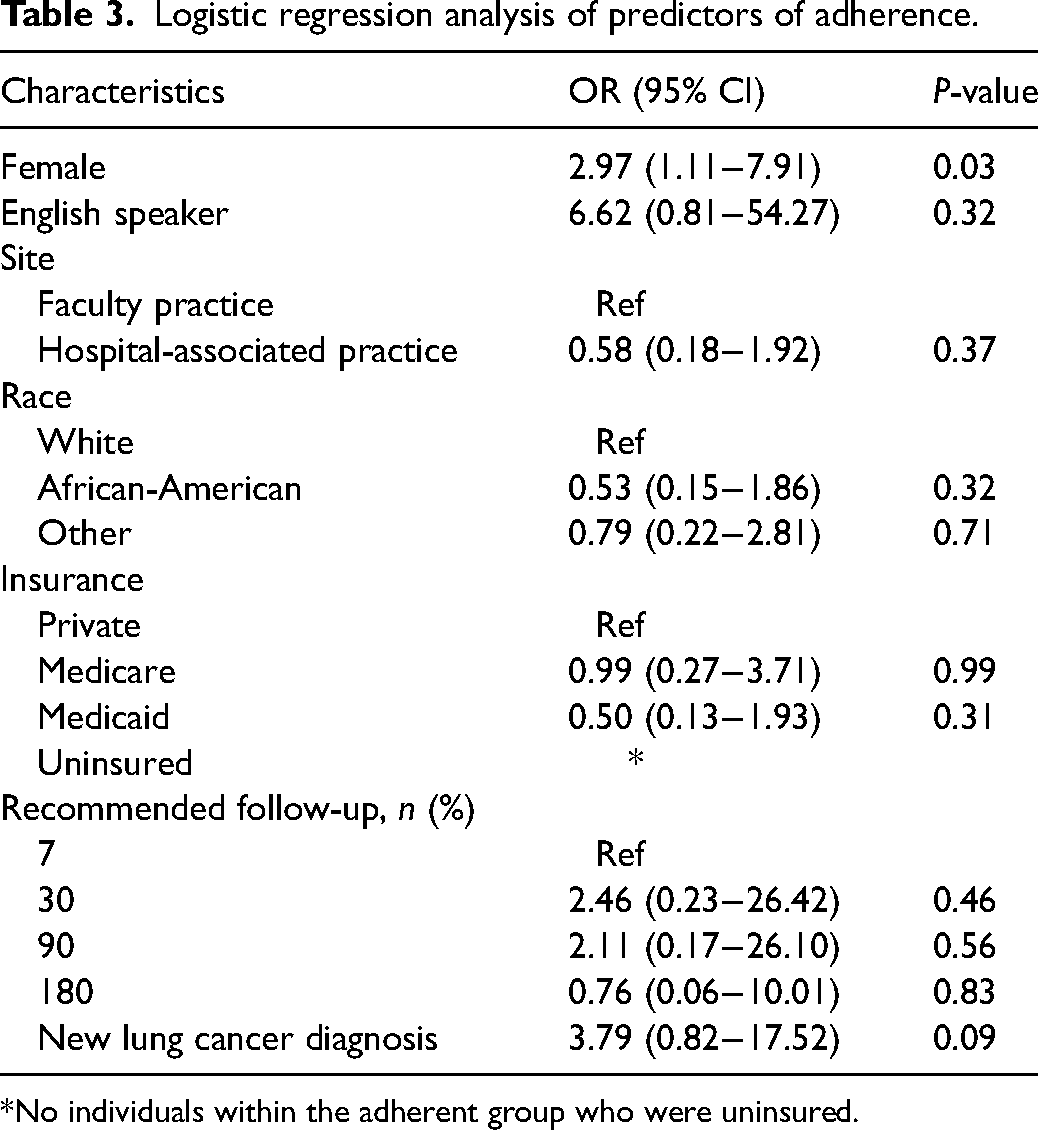
*No individuals within the adherent group who were uninsured.
Discussion
Adherence to follow-up recommendations is critical for achieving the full benefits of LCS. In this real-world study of follow-up after positive LCS findings, there was 60% adherence to recommendations and 22% led to newly diagnosed lung carcinoma. Female sex, English as a primary language, being seen at the faculty practice site, shorter recommended follow-up time, PET/CT or CT biopsy follow-up recommendation, and new lung cancer diagnosis was associated with adherence in univariate analyses. Being female was the only independent predictor of timely follow-up.
The overall adherence rate in our study falls below the 74% average for 3- to 6-month follow-up recommendations seen in other studies. Specifically, within prior sub-group analyses for the 6-month recommendation, the adherence rate has ranged from 54% to 85%, and for 3 months between 83% and 92% across varying settings and definitions of adherence.10,12–15,17 Furthermore, all these rates are significantly below the 95% in the NLST trial, 88% in the NELSON trial and the assumed 100% for the Cancer Intervention and Surveillance Modeling Network modeling studies that provided evidence for the current USPSTF screening recommendations.3–5,18 The failure to obtain prompt follow-up may delay diagnosis and treatment, and potentially allow cancers to progress to more advanced stages. 19 Overall adherence was higher with shorter recommended follow-up, which replicates other studies’ findings and may be related to several causes.10,11,14,15,20 From the healthcare system perspective, clinicians who direct follow-up might prioritize more stringent patient contact and counseling for more significant findings until scans are completed. For patients, previous studies have documented greater anxiety and fear after suspicious or indeterminate LCS results that diminishes over time, potentially in as short as 6 months.21,22 Thus, those with longer follow-up time recommendations may be less motivated to attain appropriate follow-up. Of note, the vast majority of scans and follow-ups took place prior to the COVID-19 pandemic. Additionally, many were in close proximity to the radiology center, and it is not thought that transportation or scheduling challenges would have had a significant impact on receiving the follow-up tests.
Patients with English as their primary language had higher rates of adherence in our study, reflecting potentially fewer communication barriers as seen for other cancers. 23 In a study of 188 individuals who completed initial LCS and were notified of results, nearly half (47%) were unable to correctly identify the timing and next step for LCS follow-up recommendations within 6 months. Higher rates of misidentification were seen among individuals with lower education and income levels, as well as those who had public insurance. 24 Populations including those with limited English proficiency may require more targeted promotion of screenings and a clear explanation of the benefits of translation. 25 Of note, current smoking status was not found to significantly affect adherence, unlike in several other LCS studies.17,26
Sex was found to be a significant predictor of adherence in the multivariate model. The greater adherence among those who are female aligns with the literature showing they are more active users of healthcare, have a greater awareness of cancer screenings, and are more likely to adhere to screening recommendations. 27 Patients of the faculty practice clinic site as compared to hospital-associated were more likely to be White, have English as their primary language, and have private insurance, which may each be a contributory factor to the difference in LCS adherence that was found in univariate analyses but was not present after adjustment in the multivariate model. Other factors that may contribute include that patients at the faculty practice may be a more health-literate population and engaged in their healthcare management. Differences in screening follow-up by race have been noted for lung cancer and are thought to be related to socioeconomic status and patient-provider trust.28,29 Those without employer-based coverage may be underinsured with gaps in coverage or face cost barriers to care, prohibiting receipt of subsequent scans and evaluations. 30 A shorter recommended follow-up time was not found to be an independent predictor as in other studies, which may be because our cohort only included those with positive findings and the shorter differences between recommendations negated the effect.
These findings underscore the importance of improving the proper utilization of downstream testing and interventions after initial LCS. Studies of interventions such as tracking, coordinator support, and patient reminders have improved documentation, tracking, and engagement with patients, but have yielded limited improvement in LCS rates.12,13,17 The use of centralized programs has led to a 10% to 30% improvement in annual screening compared to decentralized screening programs. However, overall rates remain variable and below optimal levels to achieve full mortality benefit.10,27 Thus, there is an urgent need for novel strategies to address this problem. The next steps may include qualitative work to elucidate barriers that may be specific to LCS follow-up, impacting both understanding and completion of timely follow-up, which can be used to guide novel interventions. Additionally, assessing the use of patient-friendly technology such as patient portals and navigation may also yield insight into opportunities for improvement.
This study was unique in that the analyzed cohort included a subset of patients who had positive screening findings and were referred for shorter-term follow-up, as compared to prior studies that predominantly included those with benign findings and recommended annual repeat screening. This study took place in a real-world community setting without additional interventions of a centralized screening follow-up program, which has only been implemented in some centers. However, as a retrospective chart review of patients from a single health system, there are several inherent limitations. The overall sample size was small and thus there are inherent limitations in what conclusions may be drawn from this study. This study population was from a New York City academic medical center, which may limit the generalizability of the study. Our data were limited to what was abstracted from our electronic health record, which does not include follow-up scans conducted at outside facilities, potentially underestimating the true adherence rate. However, as our study was conducted using primary care records, it is unlikely we failed to ascertain many follow-up studies. Finally, we did not assess individuals’ knowledge, motivation, or barriers to LCS follow-up, including whether health insurance and finances had a role in non-adherence.
In conclusion, real-world LCS adherence to follow-up on suspicious findings remains inadequate and there is an opportunity to improve outcomes. Our findings mirror other community settings and demonstrate that while overall LCS uptake has been slowly increasing, the full benefits of screening are not being achieved. Future directions may include exploring tailored interventions to address patient and healthcare barriers to timely follow-up.
Footnotes
Prior presentations
This work was presented at the Society of General Internal Medicine Annual Meeting on 04/09/2022, titled “Timely Follow-Up After Positive Lung Cancer Screening.”
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Mr Kee and Dr Sigel have nothing to disclose. Dr Wisnivesky reports consulting fees from Sanofi, Athea, Banook, PPD, and Prospero, serves on an Advisory Board at the University of Pennsylvania, has stock in Merus Pharmaceutical and grants from Sanofi, Regeneron, Axella, and Arnold Consultants outside the submitted work. Dr Kale participates on a Data Safety Monitoring Board for R01 CA251754.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by a grant from the American Cancer Society, RSG 19-118-01-CPHPS, and National Institute on Minority Health and Health Disparities, R01MD014890.
