Abstract
Objective
Current lung cancer screening criteria based primarily on outcomes from the National Lung Screening Trial may not adequately capture all subgroups of the population at risk. We aimed to evaluate the efficacy of lung cancer screening criteria recommended by the United States Preventive Services Task Force, Centers for Medicare and Medicaid Services, and the National Comprehensive Cancer Network in identifying known cases of lung cancer.
Methods
An investigation of the Stony Brook Cancer Center Lung Cancer Evaluation Center's database identified 1207 eligible, biopsy-proven lung cancer cases diagnosed between January 1996 and March 2016. Age at diagnosis, smoking history, and other known risk factors for lung cancer were used to determine the proportion of cases that would have met current United States Preventive Services Task Force, Centers for Medicare and Medicaid Services, and National Comprehensive Cancer Network eligibility requirements for lung cancer screening.
Results
Of the 1046 ever smokers in the study, 40% did not meet the National Lung Screening Trial age requirements, 20% did not have a ≥30 pack year smoking history, and approximately one-third quit smoking >15 years before diagnosis, thus deeming them ineligible for screening. Applying the United States Preventive Services Task Force, Centers for Medicare and Medicaid Services, and National Comprehensive Cancer Network eligibility criteria to the Stony Brook Cancer Center's Lung Cancer Evaluation Center cases, 49.2, 46.3, and 69.8%, respectively, would have met the current lung cancer screening guidelines.
Conclusions
The United States Preventive Services Task Force and Centers for Medicare and Medicaid Services eligibility criteria for lung cancer screening captured less than 50% of lung cancer cases in this investigation. These findings highlight the need to reevaluate the efficacy of current guidelines and may have major public health implications.
Keywords
Introduction
Lung cancer mortality is higher than all other cancer types, with an overall five-year survival rate of 17%. 1 Early detection has the potential for a dramatic improvement in survival, with rates exceeding 90% when lung cancer is detected at the earliest clinical stage. 2 The landmark National Lung Screening Trial (NLST) was the first large randomized controlled trial to demonstrate a significant reduction in mortality among individuals undergoing low-dose computed tomography compared with chest X-ray. 3 The NLST included more than 53,000 individuals aged 55–74 with at least a 30 pack year smoking history, and the study findings precipitated the United States Preventive Services Task Force (USPSTF) to recommend screening for individuals at increased risk of developing lung cancer. 4 Consequently, in February 2015, the Centers for Medicare and Medicaid Services (CMS) approved reimbursement criteria similar to those applied in the NLST.
Results from several studies that evaluated the performance of the NLST guidelines for identifying cases of lung cancer indicate that the NLST criteria may underestimate the risk of lung cancer in some individuals and may therefore not sufficiently capture the full array of vulnerable subgroups comprising the at-risk population.5–8 As the USPSTF, CMS, and others have established recommendations for lung cancer screening based primarily on the NLST criteria, with some noted variability, there is a need to evaluate the adequacy of their guidelines. We aimed to quantify the percentage of cases that would have met the current USPSTF, CMS, and National Comprehensive Cancer Network (NCCN) eligibility criteria for lung cancer screening, to determine the efficacy of these recommendations for standard clinical practice.
Methods
All biopsy-proven lung cancer cases seen at the Stony Brook Cancer Center's Lung Cancer Evaluation Center (LCEC) between January 1996 and March 2016 were reviewed. Eligible subjects included LCEC cases with available data for age at diagnosis, smoking status (current, former, never), and number of pack years and quit date, if appropriate. Other data abstracted from the LCEC database included history of lung disease, history of other cancer, family history of lung cancer in a parent or other first degree relative, and exposure history to asbestos or silica.
Current lung cancer screening recommendations.
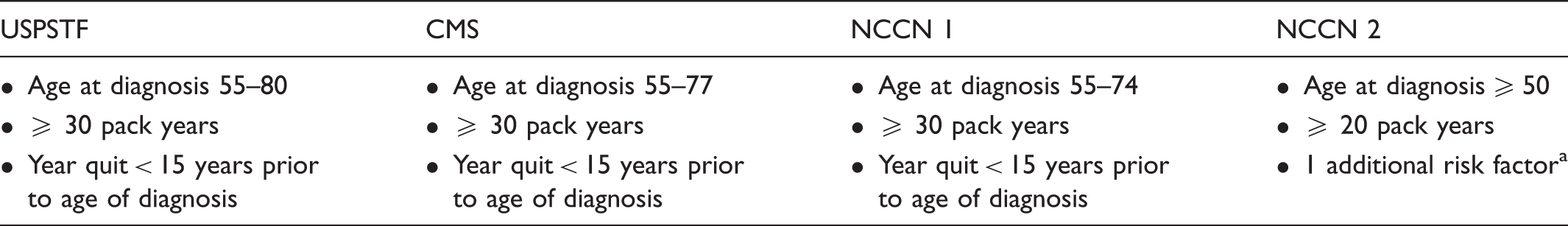
CMS: Centers for Medicare and Medicaid Services; NCCN: National Comprehensive Cancer Network; USPSTF: United States Preventive Services Task Force.
Additional risk factors as defined by NCCN. History of lung disease, personal history of cancer, family history of lung cancer, exposure history (asbestos, silica, radon, cadmium, arsenic, beryllium, chromium, diesel fumes, nickel).
Statistical analyses
The distribution of demographic and other potential risk factors for lung cancer among never smokers and ever smokers (current or former) is presented as frequencies and percentages, with comparisons between the two groups evaluated by chi-square tests. P-values<0.05 are considered to be statistically significant. Among ever smokers, we present the number and percentage of cases meeting each individual screening criterion, as well as the number and proportion of those meeting all of the required criteria for each association. Risk profile scores in this investigation are constructed based on cutoff values for each of the eligibility criteria from the NCCN guidelines, which categorize individuals into three risk groups (high, moderate, and low). 9 This risk stratification schema is consistent with other published risk prediction models for lung cancer,10–13 with quantifiable scores ranging from 1 to 3, representing low, moderate, and high risk, respectively. As any of the other specified risk factors (beyond age and smoking history) are independently considered contributors of “high risk” according to the NCCN criteria, the presence of one or more of these factors is assigned a risk weighting value of 3 for that category. A composite score, cumulated over all criteria, including age, number of pack years, duration since tobacco cessation, and additional risk factors, was used to quantify total risk for each case. For comparative purposes, absolute risks were also computed using the Liverpool Lung Project (LLP) risk prediction models incorporating age, gender, smoking duration/pack years, emphysema/COPD, family history of lung cancer, and personal history of cancer as potential risk factors.
The Stony Brook Institutional Review Board approved this investigation and waived the need for consent.
Results
Demographic and clinical characteristics of N = 1207 patients with biopsy-proven lung cancer.
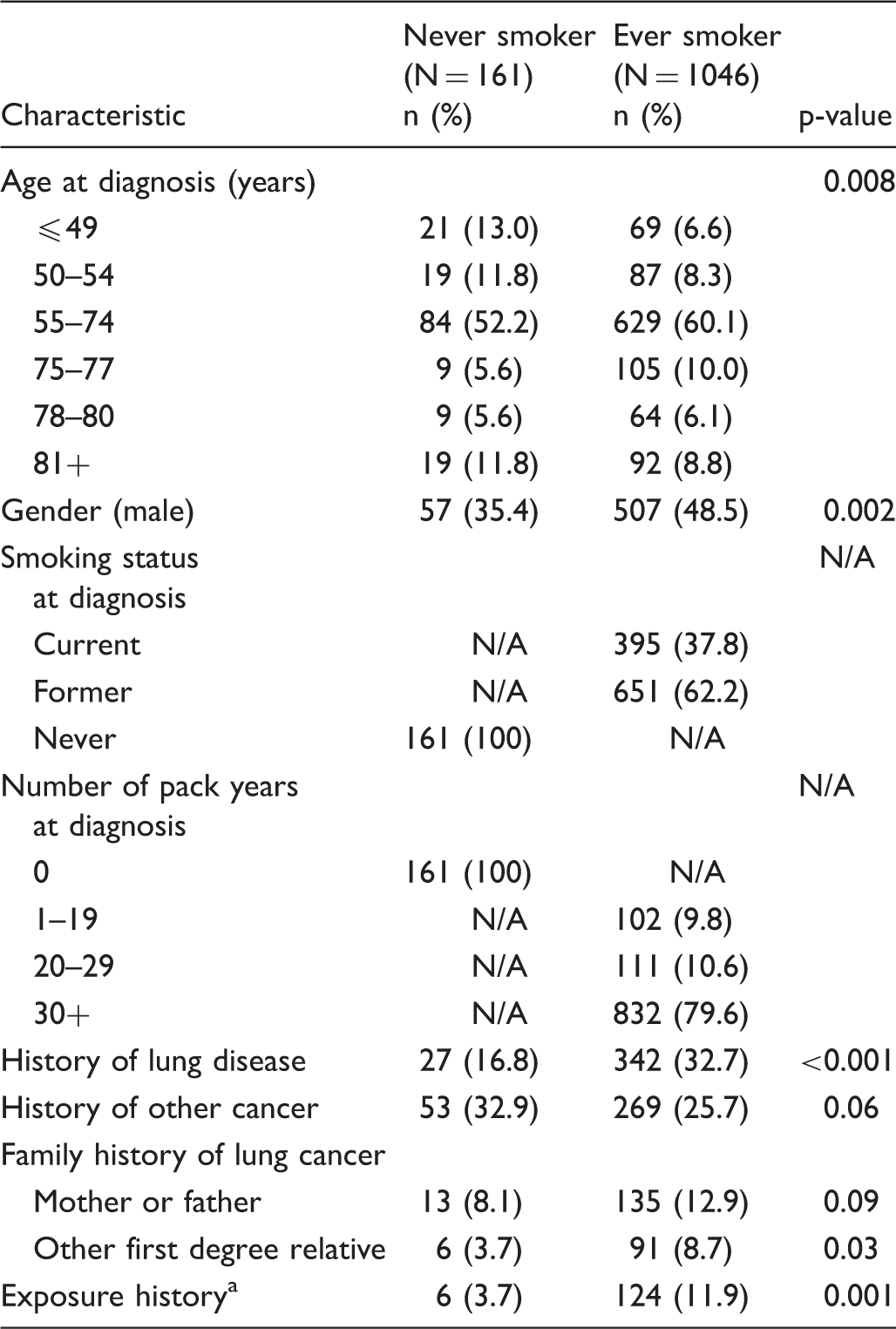
Includes asbestos and silica.
Distribution of N = 1046 ever smokers with lung cancer who meet current screening recommendations.
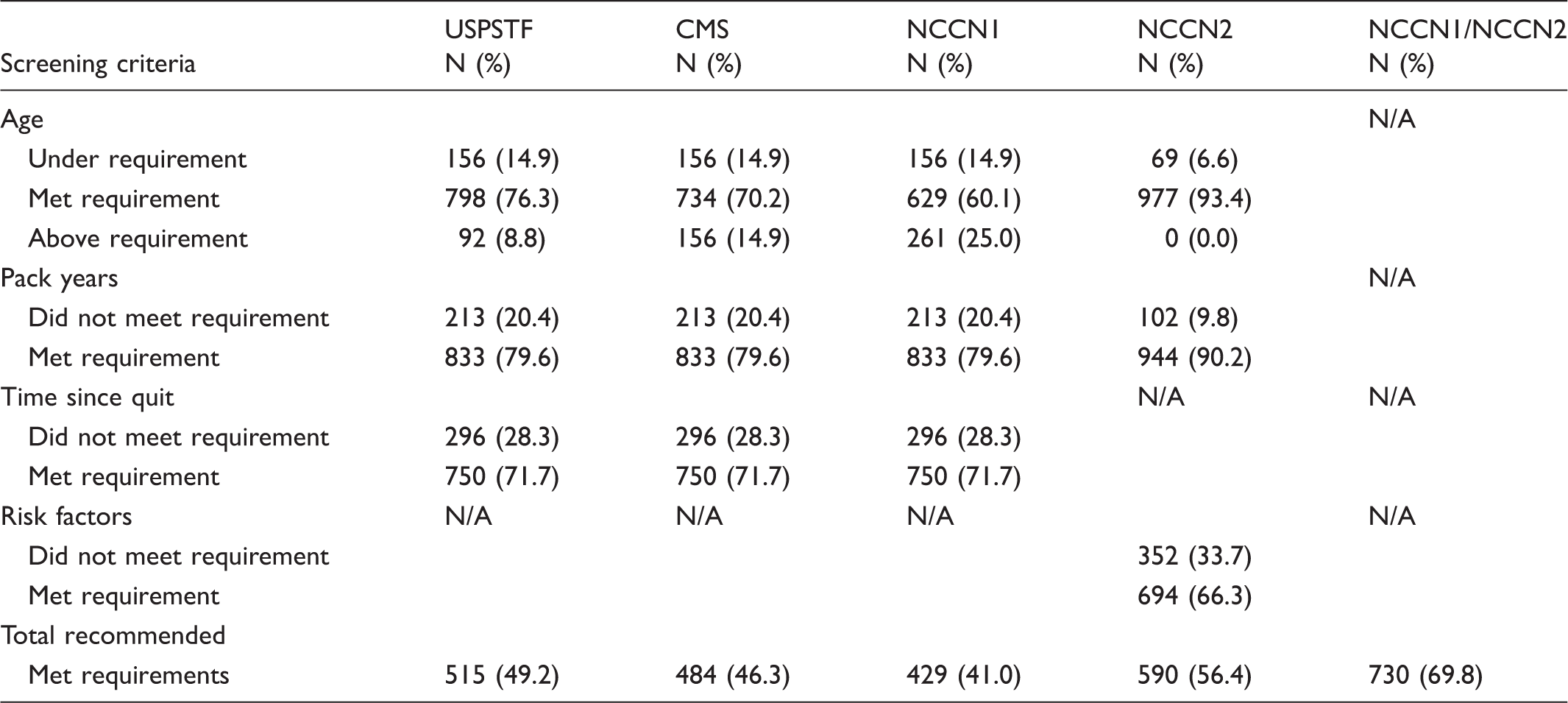
CMS: Centers for Medicare and Medicaid Services; NCCN: National Comprehensive Cancer Network; USPSTF: United States Preventive Services Task Force.
The NCCN defines high-risk individuals eligible for screening using two distinct protocols. The first protocol (NCCN1) uses criteria identical to those used in the NLST and resulted in 41% of LCEC cases meeting such guidelines. The second protocol (NCCN2), which includes smokers with a lesser smoking history (≥20 pack years), wider age range (≥50), and without restrictions on years since quitting, along with one additional risk factor, captured the highest proportion of eligible cases for screening (56%). Approximately two-thirds of LCEC lung cancer cases had at least one additional risk factor (beyond age and smoking history) that would qualify them as a candidate for screening. Taken collectively (NCCN1 or NCCN2), 730/1046 (69.8%) cases would have been eligible for screening.
Stony Brook Lung Cancer Screening (SBLCS) risk profile scoring criteria.
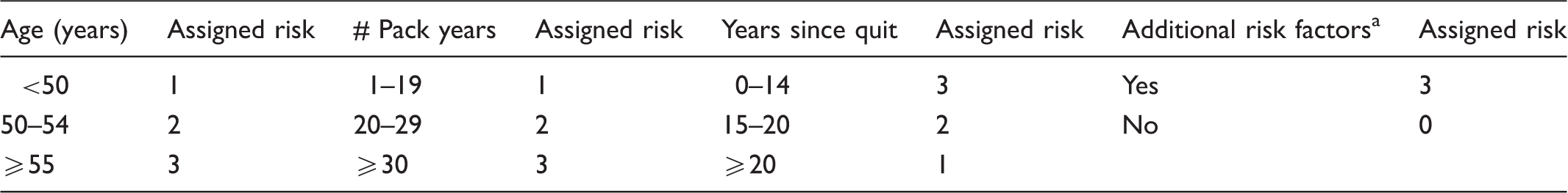
Additional risk factors as defined by NCCN include history of lung disease, personal history of cancer, family history of lung cancer, exposure history (asbestos, silica, radon, cadmium, arsenic, beryllium, chromium, diesel fumes, nickel).
Distribution of Stony Brook Lung Cancer Screening (SBLCS) risk profile scores among N = 1046 lung cancer cases.
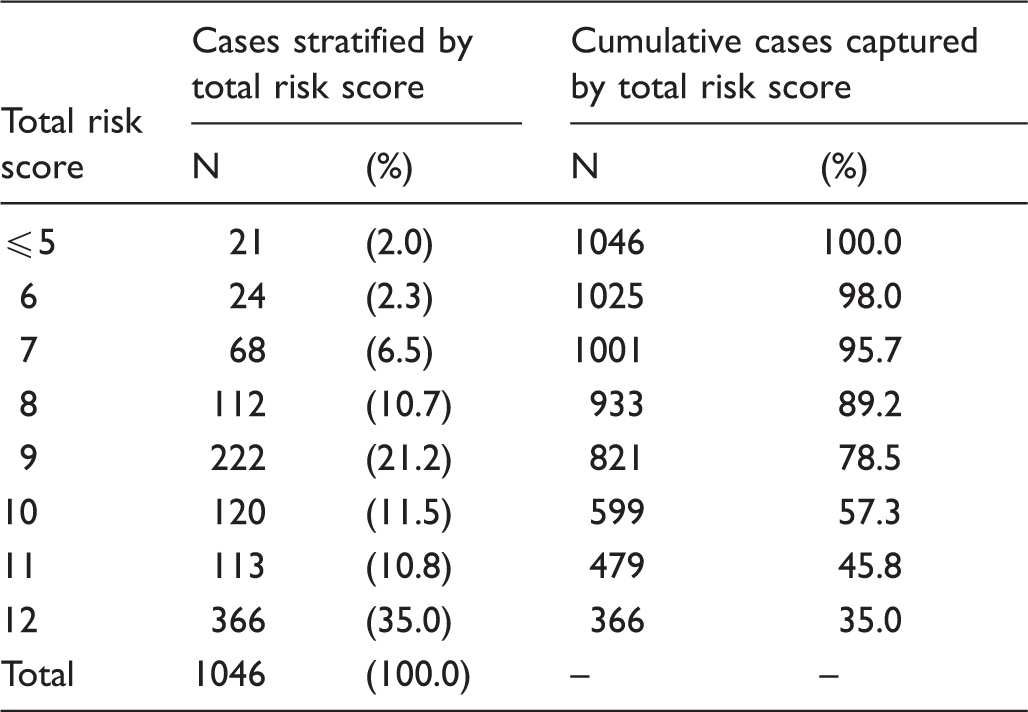
Discussion
In our study, only one-half of ever smokers met the USPSTF criteria for lung cancer screening, and fewer (46%) met the CMS guidelines. The NCCN recommendations captured 70% of cases, as a result of a multifaceted set of guidelines which include considerations of additional risk factors for screening eligibility, coupled with a wider age range, and less stringent criterion for history of tobacco use. These findings highlight the need to reevaluate the current clinical practice guidelines for lung cancer screening, which are based primarily on the eligibility criteria provided by CMS.
A total of 13.3% of lung cancer cases in the present investigation never smoked. This proportion of never smokers in LCEC is consistent with a recent report based on a similar cohort of 1351 pathologically confirmed incident lung cancer cases identified between 1984 and 2011 in Olmsted County, Minnesota, of which 9.3% never smoked. 14 Although a recent study suggested that never smokers with a high enough risk for lung cancer would benefit from screening, a very small percentage would likely ever reach this degree of risk. 15
Among ever smokers, approximately one-fifth of the lung cancer cases in the LCEC smoked <30 pack years, and almost one-third quit smoking ≥15 years ago, thereby making them ineligible for screening based on the current USPSTF and CMS guidelines. These findings are consistent with the Olmsted County data, in which 19.5% of lung cancer cases smoked <30 pack years, and 38.3% reported cessation of tobacco use more than 15 years prior. 14 Among LCEC lung cancer cases, 15% were diagnosed before age 55, and almost 9% after age 80, implying that almost one-quarter of the cases would not meet the current USPSTF screening criteria. These findings are again comparable with those from Olmsted County, where 13% of cases were younger than 55, and 12% older than 80. 14 Due to increased life expectancy in the United States, screening older individuals (aged 74–84) might be beneficial, particularly among females, as their survival benefits after aggressive therapy have been shown to be similar to those of younger women (aged 55–74). 15
Although data are limited on how well the current “clinical practice” inclusion criteria capture the population at risk for lung cancer, several studies have retrospectively evaluated the performance of the NLST inclusion protocols on the detection of lung cancer. Pinsky and Berg reviewed data from the 2010 US census; the National Health Interview Survey; and the Surveillance, Epidemiology and End Results investigation, and found that only 27% of lung cancer patients from these registries met the criteria for screening outlined by the NLST. 6 Using protocols based on the NLST criteria, the Pamplona International Early Lung Action Detection Program and the Pittsburgh Lung Screening Study both yielded suboptimal sensitivity. 8 The Pamplona study screened 3061 participants aged 40 or older, who were current or former smokers with at least a 10 pack year smoking history, of whom only 1112 (36%) met the NLST screening criteria. Of the 54 lung cancer cases detected, 21 (39%) would have been missed using the more restrictive NLST criteria.8,16 The Pittsburgh investigation included 3638 current or former smokers aged between 50 and 79 with at least a 12.5 pack year smoking history, of whom 2161 (59%) met the NLST criteria8,17,18; of the 96 incident lung cancers identified, 19 (20%) would have been missed using the NLST criteria.
The Prostate, Lung, Colorectal and Ovarian (PLCO) Cancer Screening Trial compared chest radiography with standard care among smokers (regardless of tobacco use duration and quantity) aged 55–74. Among a subset of 37,332 participants in the intervention group of the trial, 14,144 (37.9%) met the NLST guidelines. 7 In a further investigation comparing models incorporating additional risk factors to the NLST criteria, the risk prediction models for the more inclusive PLCO trial protocols were significantly more sensitive than the NLST criteria for detecting lung cancer. This study, however, lacked cost analysis, so it remains unclear whether a screening program using this risk modeling would maintain the high cost-effectiveness achieved in the NLST. Nonetheless, of the 678 known lung cancers, the PLCO protocols successfully identified 81 (11.9%) more cases than did the NLST criteria over the six-year follow-up period. 7 Consistent with the PLCO results, 41% of LCEC lung cancer cases would have met the NLST criteria. These findings add to a growing evidence base indicating that the current eligibility criteria for lung cancer screening may lack the sensitivity required to adequately capture the population at risk.
Several studies have reported on using emphysema as a factor to refine the eligibility protocols for lung cancer screening.8,17,19–21 One investigation suggests narrowing the selection criteria, 19 while others support adding radiologic emphysema to screening protocol guidelines to improve the detection of incident lung cancer. 8 In our study, where 19% of ever smokers reported a history of emphysema, adding this factor to the USPSTF guidelines as a complementary option for eligibility would have captured 67 (6.4%) previously ineligible cases for screening, and 77 (7.4%) additional cases for screening eligibility using the CMS criteria. Given the high prevalence of emphysema/COPD among smokers, these findings suggest that including this risk factor in the screening protocols for lung cancer should be considered.
The most recent National Health Interview Survey indicates that cigarette smoking among US adults has declined from 20.9% (45 million persons) in 2005 to 16.8% (40 million persons) in 2014. 22 This trend will directly influence characterization of the lung cancer screening population in the future. Given the limitations of the currently approved screening protocols recommended by CMS and others, which failed to capture approximately one-half of lung cancer cases in this investigation, as well as the noted shift in smoking patterns over the past decade, there is an even more pressing need to refine the risk profiles used to define the at-risk population for lung cancer screening. Additionally, the current guidelines do not take into consideration ethnicity. A very small percentage of NLST participants were considered minority, and recent studies suggest that the CMS screening eligibility criteria do not correspond with lung cancer risk among Black and Hispanic subgroups. 23 An ethnic-specific model for African-Americans has been developed and validated to provide more precise estimates for predicting risk in this population. 24
Although this investigation is strengthened by the inclusion of a large, well-defined patient population with biopsy-proven lung cancer, it has several limitations. First, LCEC cases likely represent a cohort with more advanced disease than individuals who would be targeted for screening, so the percentage of cases meeting the given eligibility criteria in the present study may overestimate the sensitivity of the guidelines. Second, the study was limited by the use of self-reported data on smoking status and other known risk factors. Cases who reported no prior occurrence of disease, or for which the information was not available, were both defined as having a null history, which could result in some unknown level of underreporting. Additionally, the LCEC database only collected exposure histories related to asbestos and silica, thereby potentially missing information on other exposures, such as radon and cadmium. These missing data would not influence the findings related to the USPSTF and CMS guidelines, as neither includes such risk factors among the eligibility criteria; however, the absence of such data may underestimate the sensitivity of the NCCN criteria. It is expected, however, that these exposures would be less common. Another consideration with regard to interpretation of the study findings is the potential effect of a temporal downward trend in tobacco use over the past two decades. 14 Lastly, the LCEC cohort originated from a single institution in a suburban geographic location in the Northeast, and the findings may therefore not be generalizable to the population at large.
The Society of Thoracic Surgeons and American Association of Thoracic Surgery have strongly advocated for the need to expand the current lung cancer screening criteria, which are primarily based on the NLST guidelines.25–27 The present investigation adds to a growing evidence base that highlights the need to reevaluate these restrictive protocols. Our findings suggest that implementing a weighted risk profile approach for defining individuals at high risk may improve sensitivity and capture a higher proportion of at-risk subjects who would otherwise be missed by the current guidelines. The SBLCS risk profile schema provided in this investigation is based primarily on the NCCN cutoffs for low, moderate, and high-risk groups and was not designed to provide absolute risk estimates for the development of lung cancer. The SBLCS scoring structure is presented as a simple tool that could be easily implemented in a clinical setting without necessitating regression-based risk-prediction calculations. To evaluate the performance of the suggested risk profile tool, we applied the LLP risk prediction model 28 to the LCEC data. The results indicated that 96% of cases had >3% risk, a cutoff that has been suggested as sufficiently high for inclusion in a screening program. 28 This finding is consistent with data reported by the LLP, in which 93% of LLP participants had a >3% risk of developing lung cancer. 28 Additional analyses also indicated that the SBLCS scoring system produced significantly well-correlated results with the LLP model (Pearson r = 0.5, p < 0.01). These data suggest the potential usefulness of the SBLCS scoring tool; however, additional research is required to validate the findings in other populations and to optimize the weighted risk assignments for each of the eligibility criteria.
Conclusions
Screening for lung cancer requires well-defined criteria that effectively identify all subgroups of the at-risk population. Lung cancer screening is in its infancy and, although it is beyond the scope of this investigation to recommend policy changes, consideration should be given to redefining the population at risk, particularly as the current screening protocols recommended by the USPSTF and CMS identified fewer than 50% of lung cancer cases in the present investigation.
Footnotes
Acknowledgements
We thank John Reagan and Sean Reagan, student volunteers of the LCEC, for their assistance in the data abstraction.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
