Abstract
Objective
Annual fecal immunochemical tests can reduce colorectal cancer incidence and mortality. However, screening is a multi-step process and most patients do not perfectly adhere to guideline-recommended screening schedules. Our objective was to compare the reduction in colorectal cancer incidence and life-years gained based on US guideline-concordant fecal immunochemical test screening to scenarios with a range of delays.
Method
The Colorectal Cancer Simulated Population model for Incidence and Natural history (CRC-SPIN) microsimulation model was used to estimate the effect of systematic departures from fecal immunochemical test screening guidelines on lifetime screening benefit.
Results
The combined effect of consistent modest delays in screening initiation (1 year), repeated fecal immunochemical test screening (3 months), and receipt of follow-up or surveillance colonoscopy (3 months) resulted in up to 1.3 additional colorectal cancer cases per 10,000, 0.4 additional late-stage colorectal cancer cases per 10,000 and 154.7 fewer life-years gained per 10,000. A 5-year delay in screening initiation had a larger impact on screening effectiveness than consistent small delays in repeated fecal immunochemical test screening or receipt of follow-up colonoscopy after an abnormal fecal immunochemical test. The combined effect of consistent large delays in screening initiation (5 years), repeated fecal immunochemical test screening (6 months), and receipt of follow-up or surveillance colonoscopy (6 months) resulted in up to 3.7 additional colorectal cancer cases per 10,000, 1.5 additional late-stage colorectal cancer cases per 10,000 and 612.3 fewer life-years gained per 10,000.
Conclusions
Systematic delays across the screening process can result in meaningful reductions in colorectal cancer screening effectiveness, especially for longer delays. Screening delays could drive differences in colorectal cancer incidence across patient groups with differential access to screening.
Introduction
A program of annual fecal immunochemical tests (FITs) is an effective and cost-effective regimen for colorectal cancer (CRC) screening. 1 Screening may benefit patients by detecting CRC at an earlier stage when prognosis is more favorable and treatment is more tolerable, and by detecting and removing adenomas to prevent their progression to CRC.
US CRC screening guidelines specify an age at initiation of screening, re-screening every year until there is an abnormal FIT, timely colonoscopy follow-up of abnormal FIT tests, and ongoing adenoma surveillance when adenomas are detected by follow-up colonoscopy. 2 Simulation studies used to inform these guidelines compare lifetime screening outcomes under a wide range of screening regimens, with a focus on screening efficacy, and estimate the expected benefits and harms when patients perfectly adhere to screening guidelines. 3
In real life, few patients adhere perfectly to guideline-recommended screening schedules. There is evidence of departures from recommended guidelines at all points in the process, beginning with screening initiation. In an insured population, only 51% of people initiated screening by their 51st birthday, 73% initiated screening by their 52nd birthday, and 89% had initiated screening by their 55th birthday. 4 There are also delays in repeat screening. For example, among screening-eligible people who were enrolled in an integrated health plan who had completed at least one FIT, only 75% repeated screening within the recommended 1-year interval and 83% repeated screening within 2 years. 5 In a 14-year study of seven rounds of biennial stool-based screening, only 14% were consistently screened every 2 years. 6 Delays also occur after receipt of an abnormal FIT. Only 68% of patients enrolled in federally qualified health centers received a follow-up colonoscopy within 3 months of an abnormal stool test (either FIT or guaiac fecal occult blood test), 7 and the time to follow-up colonoscopy after an abnormal FIT varies across health care systems. 8 Finally, delays occur among patients in adenoma surveillance regimens. There is wide variability in the percentage of patients who receive a repeat colonoscopy within 3.5 years of an exam finding three or more adenomas or at least one adenoma with advanced histology; with estimates ranging from 18% of patients in a safety net system to 60% of patients in the highest performing system. 9 International studies also find that most patients do not receive guideline-concordant adenoma surveillance.10–12
In this paper, we use a simulation study to estimate the change in lifetime CRC screening benefit under a range of systematic departures from US guideline-recommended screening, focusing on delays at different points in a program of annual FIT screening.
Methods
We simulated the detection and death from CRC using the CRC-SPIN model, which is supported by the National Cancer Institute's Cancer Intervention and Surveillance Modeling Network. 13 Microsimulation models synthesize existing evidence about the natural history of disease and accuracy of screening tests to examine the impact of screening on multiple outcomes. CRC-SPIN describes the natural history of CRC in the absence of screening and changes in outcomes due to screening via detection and removal of adenomas (primary prevention) and detection of preclinical cancer at a potentially earlier and more treatable stage than symptom-detected cancer. Simulated individuals begin in a disease-free state and potentially develop adenomas that transition to preclinical (asymptomatic) CRC. From the preclinical cancer state, individuals may transition to a clinical (symptomatic) CRC state. In the absence of screening, CRC is detected when an individual transition into the (symptomatic) clinical CRC state. Disease progression depends systematically on age and sex and varies randomly across individuals.
We simulated an average-risk cohort with 10 million individuals born in 1965, who began screening at age 50 in 2015. The model simulated risk that is consistent with recent evidence of increasing CRC risk, 3 which is observable as increased CRC incidence before age 50. 14
Simulated screening
We simulated screening of this cohort assuming a 1-year FIT regimen consistent with 2016 United States Preventive Services Task Force (USPSTF) guidelines and under 12 delay scenarios. Screening was simulated to begin at age 50, in 2015, with the annual FIT. In 2021, after our simulated cohort had begun screening, USPSTF guidelines were updated to recommend screening beginning at age 45. 2 We assumed FIT operating characteristics that were consistent with those used to inform the 2021 USPSTF guidelines which were based on an updated systematic review. 15 Individual-level specificity of FIT was set to 0.97, so that at each FIT an individual that is free from both CRC and adenomas has a 3.0% chance of an abnormal FIT. Individual-level sensitivity was based on the most advanced lesion and was set to 0.05 if the largest adenoma was <6 mm in diameter, 0.15 if the largest adenoma was 6 mm to <10 mm in diameter, 0.22 if the largest adenoma was 10 mm or larger in diameter, and 0.74 if there was at least one preclinical cancer. For guideline-concordant screening, individuals were simulated to receive an FIT test every year until their first abnormal test After an abnormal FIT, follow-up colonoscopy was simulated at 2 weeks. If there were no findings at follow-up colonoscopy, the individual was returned to FIT screening, with the next test in 10 years. If adenomas, but not cancer, were detected at colonoscopy then individuals were simulated to receive adenoma surveillance colonoscopy, based on recent Multi-Society Task Force recommendations, as shown in Table 1. 16 Guideline-recommended screening ends at age 75; individuals in adenoma surveillance remain in the surveillance program, and receive colonoscopies, until age 85.
Adenoma surveillance recommendations based on findings at colonoscopy. 16

Simulated screening delays
As shown in Table 2, we examined the impact of delays at five points in the FIT screening process, assuming consistent delays throughout each individual's lifetime: (1) screening initiation, (2) re-screening after a normal FIT, (3) colonoscopy follow-up of an abnormal FIT, (4) receipt of surveillance colonoscopy, and (5) return to the FIT screening after colonoscopy, either after a normal follow-up colonoscopy or completion of adenoma surveillance. In addition to examining the separate effects of consistent delays at a single step in the screening process, we also examined the combined effect of consistent modest or large delays at every step in the process. When there are consistent large delays, screening begins at age 55, with a FIT test every 18 months (annually scheduled with a 6-month delay) until the first abnormal FIT, and with follow-up colonoscopy 6 months after an abnormal FIT. If there are no findings at follow-up colonoscopy, the next FIT occurs in 15 years (10 years plus a 5-year delay), but if there are findings that result in adenoma surveillance, the next surveillance exam occurs 6 months later than under guideline recommendations (e.g. if any adenoma 10 mm or larger were found, then the next exam would occur in 3 years plus 6 months).
Simulated time between screening events for different steps in the screening process. Bolded screening times reflect delays relative to guideline-concordant screening.
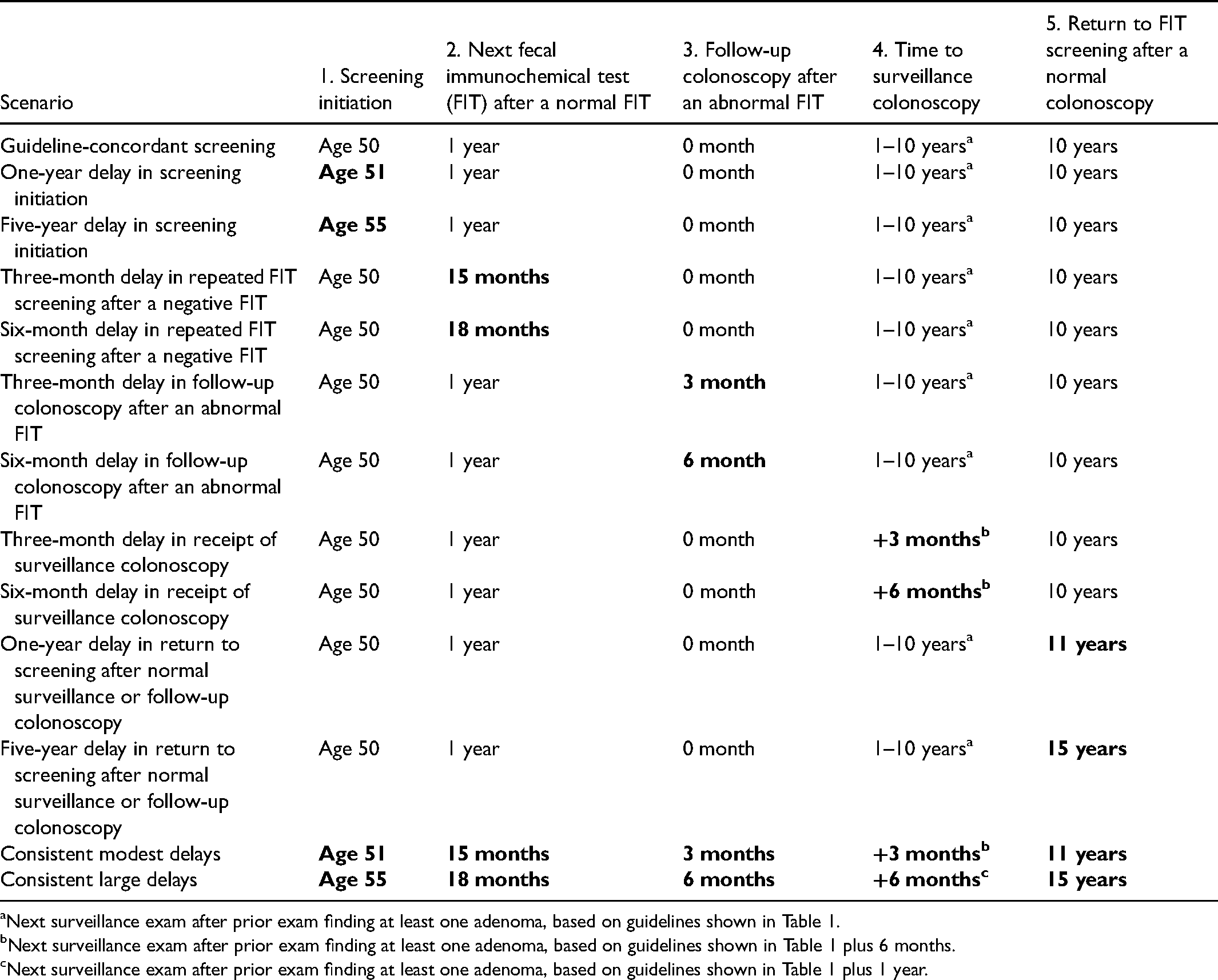
Next surveillance exam after prior exam finding at least one adenoma, based on guidelines shown in Table 1.
Next surveillance exam after prior exam finding at least one adenoma, based on guidelines shown in Table 1 plus 6 months.
Next surveillance exam after prior exam finding at least one adenoma, based on guidelines shown in Table 1 plus 1 year.
A cancer-free individual who is adherent to FIT screening guidelines and never has an abnormal result will be screened 25 times: once a year from the age of 50 to 74 years. An individual simulated to screen with consistent large delays who never has an abnormal FIT will be screened 14 times, with the first test at age 55 and the last at age 74.5.
Outcomes
We estimated three measures of lifetime screening benefit for guideline-concordant screening relative to no screening: (1) CRC prevented; (2) late-stage CRC prevented; and (3) undiscounted life-years gained (LYG). LYG was estimated as the difference in the simulated number of person-years under screening and unscreened conditions. We also estimated the number of colonoscopies per 10,000 as a measure of harm. For each delay scenario, we estimated both the absolute and percentage change in outcome for the delay scenario relative to the guideline-concordant screening scenario.
Results
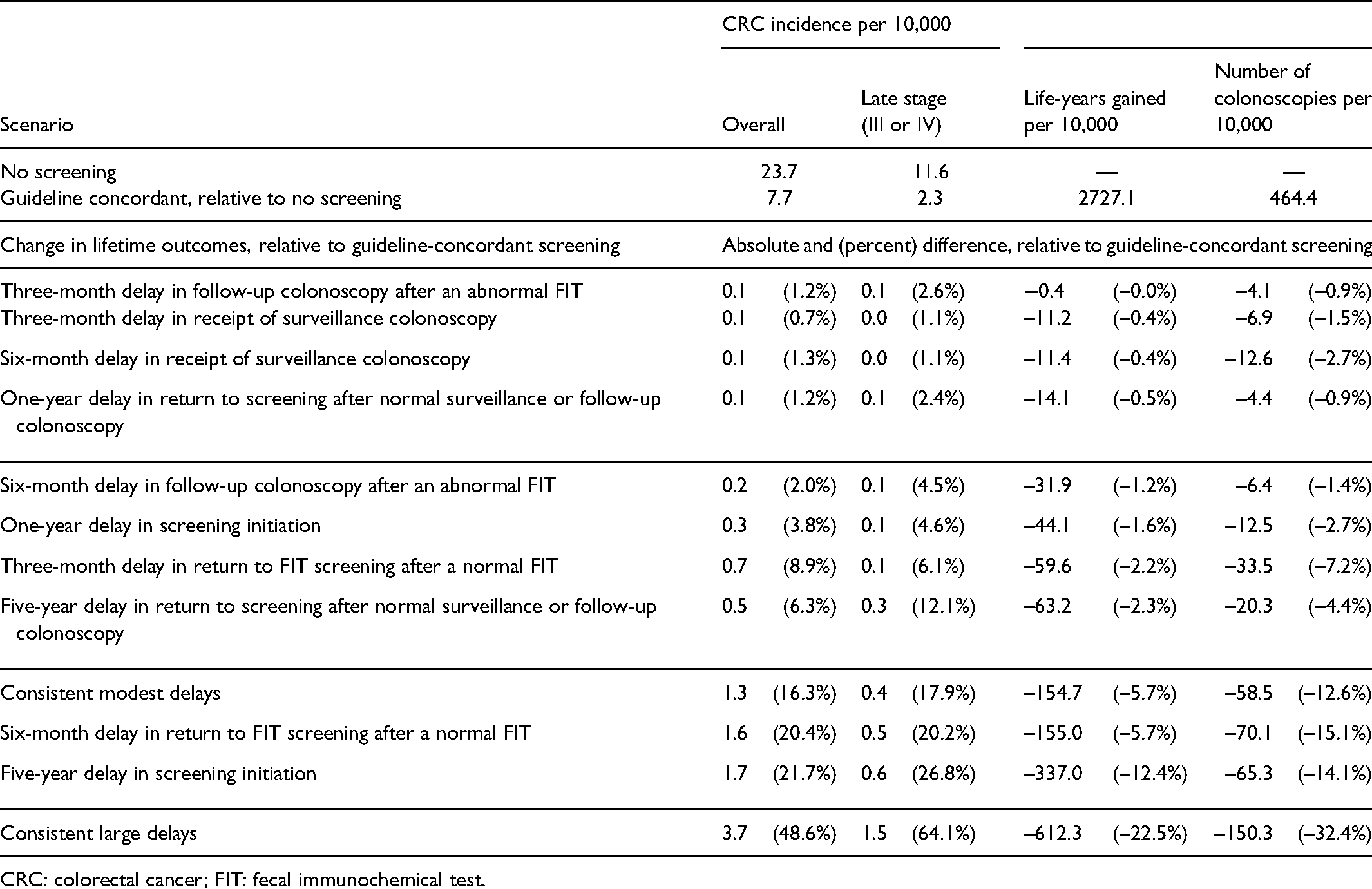
Table 3 shows the predicted impact of guideline-concordant screening on CRC incidence, late-stage incidence, LYG, and the number of colonoscopies performed, and the impact of departures from guideline-concordant screening on these outcomes, ordered from delays that had the least to most impact on LYG. Guideline-concordant screening would dramatically reduce lifetime incidence of both overall CRC and late-stage CRC incidence, resulting in 2727 LYG per 10,000 50-year-old individuals. Departures from screening guidelines would reduce both screening benefits and harms.
Lifetime predicted loss of benefit and harms due to screening delays.
CRC: colorectal cancer; FIT: fecal immunochemical test.
Four delay scenarios had a small impact on screening benefit: consistent 3-month delays in receipt of follow-up colonoscopy after an abnormal FIT, consistent 3- or 6-month delays in receipt of surveillance colonoscopy, and consistent 1-year delays in return to FIT screening after receipt of normal colonoscopy (due to follow-up of an abnormal test or completion of surveillance). Relative to guideline-concordant screening, these four scenarios resulted in at most a 0.5% reduction in LYG (a 14-year reduction in LYG per 10,000), a 1.3% increase in CRC incidence (0.1 additional cases per 10,000) and a 2.4% increase in late-stage CRC incidence (0.1 additional late-stage cases per 10,000). These four scenarios resulted in up to a 2.7% reduction in the number of colonoscopies (12.6 fewer colonoscopies per 10,000), with the largest reduction in colonoscopies performed when there were delays in receipt of surveillance exams.
Four delay scenarios resulted in a 1.2% to 2.3% reduction in LYG (up to 63 fewer LYG per 10,000): consistent 6-month delays in receipt of follow-up colonoscopy after an abnormal FIT, a 1-year delay in initiation of screening (at age 51 rather than 50), consistent 3-month delays in repeated FIT screening after a negative FIT, and a 5-year delay in return to FIT screening after receipt of normal colonoscopy. Relative to guideline-concordant screening, these four delay scenarios resulted in a 2.0% to 8.9% increase in CRC incidence (0.2–0.7 additional cases per 10,000) and a 4.5% to 12.1% increase in late-stage CRC incidence (0.1–0.3 additional late-stage cases per 10,000). These scenarios resulted in a 1.4% to 7.2% decrease in colonoscopies (6.4–33.5 fewer tests per 10,000).
A scenario with consistent modest delays at all steps in the screening processes had a similar effect on outcomes as a consistent large (6-month) delay in return to FIT screening after a normal test; both resulted in a 5.7% reduction in LYG (155 fewer LYG per 10,000) due to an increase in both overall and late-stage CRC incidence. They also resulted in similar reductions in the number of colonoscopies, 12.6% fewer with consistent modest delays and 15.1% fewer with consistent large delays in return to FIT screening (absolute reductions of 58.5 and 70.1 fewer colonoscopies per 10,000, respectively).
A large (5-year) delay in FIT screening initiation resulted in a 12.4% reduction in LYG (337 fewer years per 10,000), a 21.7% increase in CRC incidence, a 26.8% increase in late-stage CRC, and a 14.1% decrease in colonoscopies per 10,000. Consistent large delays in initiation, repeated FIT screening, and receipt of either follow-up or surveillance colonoscopy had a large impact on the effectiveness of screening compared to guideline-concordant screening, with a 48.6% increase in CRC incidence (3.7 additional cases per 10,000), a 64.1% increase in late-stage CRC incidence (1.5 additional cases per 10,000), a 22.5% decrease in LYG (612 fewer LYG per 10,000), and a 32.4% decrease in colonoscopies (150.3 fewer colonoscopies per 10,000).
Discussion
Simulation studies that focus on screening efficacy, with perfect adherence, are important for guiding policy because they estimate the benefits that are possible from screening under ideal circumstances. Estimates of lifetime screening outcomes when there are consistent but modest departures from screening guidelines can inform the extent to which such departures may erode screening efficacy. Non-adherence does not only refer to the complete absence of screening, but also longer than recommended intervals between the various steps in the screening process.
Systematic delays can result in meaningful reductions in CRC screening effectiveness, especially with longer delays. Consistent modest delays in a single step of the screening process, such as a consistent 3-month delay in the follow-up of an abnormal FIT, had a very small effect on predicted screening benefit. However, consistent modest delays across multiple steps of the screening process add up, resulting in substantial decreases in screening benefits. The consistent modest delays we modeled are not difficult to imagine in practice, and most studies examining screening adherence would label these delays “guideline adherent.” Consistent larger delays are also plausible, especially in patient populations that face barriers to screening. Large delays resulted in large reductions in benefits compared to guideline-concordant screening, with CRC cases increasing from 7.7 per 10,000 under guideline-concordant screening to 11.4 per 10,000 when there were consistently large delays.
Our analyses focused on annual FIT testing, consistent with US guidelines. Many countries recommend biennial FIT as a cost-effective approach to CRC screening. 17 Relative to no screening, FIT with an 18-month screening interval (annual screening with consistent 6-month delays in return after a normal FIT) reduced CRC incidence by 6.0 per 10,000 and late-stage CRC by 1.8 per 10,000, and increased LYG by 2390.0 per 10,000. Consistent large delays combined with an 18-month screening interval would increase CRC incidence by 2.1 per 10,000 (35%) and late-stage CRC by 1.2 per 10,000 (66.6%), while decreasing LYG by 549.1 per 10,000 (23.0%). This suggests that the impact of delays would be similar for a biennial screening program, with larger delays producing greater reductions in benefits (and harms), smaller loss of benefits due to delays in receipt of colonoscopy for adenoma surveillance, and larger losses in benefits due to delays in initiation.
Although our analyses examined ongoing delays in screening, the findings are relevant to delays in screening due to the COVID-19 pandemic. Modeling studies estimated that a one-time 12-month pause in CRC screening and follow-up due to the pandemic would cause an 0.8% to 2.0% reduction in LYG. 18 This is similar to our estimated 1.6% decline in LYG due to a 12-month delay in screening initiation and suggests that delays in initiation may account for much of the impact of screening disruption on the loss of LYG. Our analysis suggests that, in the context of limited colonoscopy resources, it may make sense to prioritize screening initiation and follow-up of positive FIT tests above resumption of adenoma surveillance.
Our findings are based on predictions from a simulation model, which is both a strength and a limitation. Simulation allows population-level predictions of outcomes that cannot be directly observed, but requires assumptions about disease processes, patient behaviors, and test characteristics. We simulated relatively simple screening delay scenarios, assuming consistent delays over each individual's entire screening history. We did not address the potential for “catch up” screening after age 75 among people who were not fully adherent to screening or did we examine the impact of recent advances in treatment that could reduce the impact of screening delays on reduction in LYG, although treatment, especially chemotherapy for more advanced CRC, would reduce the quality of life. Finally, we did not examine the impact of delays in treatment after CRC detection.
Compared to previously published simulations examining delays in follow-up colonoscopy after an abnormal FIT, we estimated a smaller effect of short (3-month) delays in follow-up of abnormal FIT tests on LYG than previous simulations, but similar effects on longer (6-month) delays. 19 These differences reflect the use of an updated model, 20 updated assumptions about FIT accuracy, 15 and new adenoma surveillance guidelines. 16 In particular, previous analyses assumed an individual-level FIT sensitivity of 0.076 when the most advanced adenoma was <10 mm in diameter; these newer analyses assumed a lower sensitivity of 0.05 when the most advanced adenoma was <6 mm and a higher sensitivity of 0.15 when the most advanced adenoma was at least 6 mm but <10 mm. Although assumptions about FIT sensitivity affect the estimated benefit loss, we expect the general conclusions to hold: cumulative delays add up and result in a loss of benefit. These simulations inform how delays might reduce screening effectiveness among 50-year-olds who began screening in 2015. We did not examine the impact of departures from current USPSTF guidelines, which recommend screening initiation at age 45, 2 but anticipate that results would be similar.
These simulation-based results are generally consistent with observational screening studies. A meta-analysis found that cancer risk was significantly higher among patients who waited 6 months before receiving a follow-up colonoscopy after an abnormal FIT 21 ; patients who waited a year or more had a higher risk of late-stage disease 22 and cancer mortality. 23 A recent review of interval-detected CRC found that 24% of interval cancers occurred when follow-up was longer than guideline recommendations based on findings at colonoscopy. 24 Another study found that detection of advanced neoplasia was about twice as likely in patients with delayed adenoma surveillance compared to patients with on-time or early surveillance colonoscopy. 10
Departures from screening recommendations may be more likely in some patient groups than others and may drive disparities in CRC incidence and mortality. Estimated CRC screening rates, which reflect screening initiation and repeated testing, are generally lower in racial and ethnic minority populations,25–31 uninsured populations,26,32 and foreign-born US populations.33–35
Conclusions
Relatively small but consistent delays in screening, follow-up of abnormal FIT results, or surveillance erode screening benefits. Large delays, especially delays in initiation of screening, result in greater loss of screening benefit. These findings point to the importance of interventions to increase patient adherence to CRC screening guidelines, 36 including the use of patient decision aids, 37 offering screening regimens that are consistent with patient preferences, 38 and directly notifying gastroenterology providers about patient referrals. 39
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Research reported in this publication was supported by Grant Numbers U01CA199335 and U01CA253913 from the National Cancer Institute as part of the Cancer Intervention and Surveillance Modeling Network (CISNET). Its contents are solely the responsibility of the authors and do not necessarily represent the official views of the National Cancer Institute.
