Abstract
Objective
To classify interval colorectal cancers as false negatives or newly occurring cases in a biennial Fecal immunochemical test (FIT) screening program and by various interscreening intervals.
Setting
Data from the Taiwanese biennial colorectal cancer screening program involving FIT from 2004 to 2014 were used to estimate the incidence rate of asymptomatic colorectal cancer and the rate of its subsequent progression to clinical mode.
Methods
The sensitivity of detecting asymptomatic colorectal cancers excluding newly developed colorectal cancers was compared to the conventional estimate of sensitivity, the complementary FIT interval cancer rate as a percentage of the expected incidence rate ((1-I/E)%). The relative contribution of newly developed or false-negative cases to FIT interval colorectal cancers was estimated by age and interscreening intervals.
Results
The Taiwanese biennial fecal immunochemical test screening program had a conventional sensitivity estimate of 70.2%. After newly developed colorectal cancers were separated from FIT interval cancers, the ability to detect asymptomatic colorectal cancers increased to 75.5%. FIT interval colorectal cancers from the biennial program mainly resulted from newly developed colorectal cancers (68.8%). The corresponding figures decreased to 61.1% for the annual program but increased to 74.7% for the triennial program. The preponderance of newly developed colorectal cancers among FIT interval cancers was more prominent in screenees aged 50–59 than in those aged 60–69.
Conclusions
Newly developed colorectal cancers showed a predominance among the FIT interval colorectal cancers in particular in the younger population screened. It is desirable to identify high-risk individuals to offer them a short interscreening interval or advanced detection methods to reduce their odds of developing interval cancer.
Keywords
Introduction
Colorectal cancer (CRC), the third most common cancer, accounted for 881,000 deaths in 2018.1,2 The early detection of CRC through screening programs based on established evidence is therefore an important weapon in the control of this disease.3,4 Among a variety of screening modalities, stool-based tests focusing on the detection of bleeding phenotypes have increasingly gained attention. 5 Screening strategies using a guaiac fecal occult blood test (gFOBT) have been proven to be effective in reducing mortality among individuals with CRC.4,6–8 A fecal immunochemical test (FIT) was developed in recent decades and has been shown in several programs to be effective in screening patients.9,10
The major goal of applying stool-based tests in population-based screening programs is to detect colorectal neoplasms at an early stage, namely asymptomatic CRC. This aim is highly associated with the sensitivity of the screening tool, which is closely related to the emergence of symptomatic cases between screens, namely interval cancers.11,12 Compared with screen-detected CRCs, interval cancers are characterized by a more advanced stage and a lower survival rate.13,14 The interval cancer rate is thus one of the cardinal indicators of the quality of a population-based screening program.12,15,16
In addition to FIT interval cancer as a result of the failure to detect asymptomatic CRCs that already exist at the time of screening (false-negative CRC, the lower part of Figure 1), CRCs that develop between screening rounds (newly developed CRC, the upper part of Figure 1) may also account for interval cancers in a screening program. 17 Interval cancer caused by newly developed CRC begins from the CRC-free status and progresses first to the asymptomatic CRC status and thereafter to symptomatic CRC status before the next screen. Notably, the CRC-free status is defined by normal or precancerous lesions such as small or advanced adenomas.
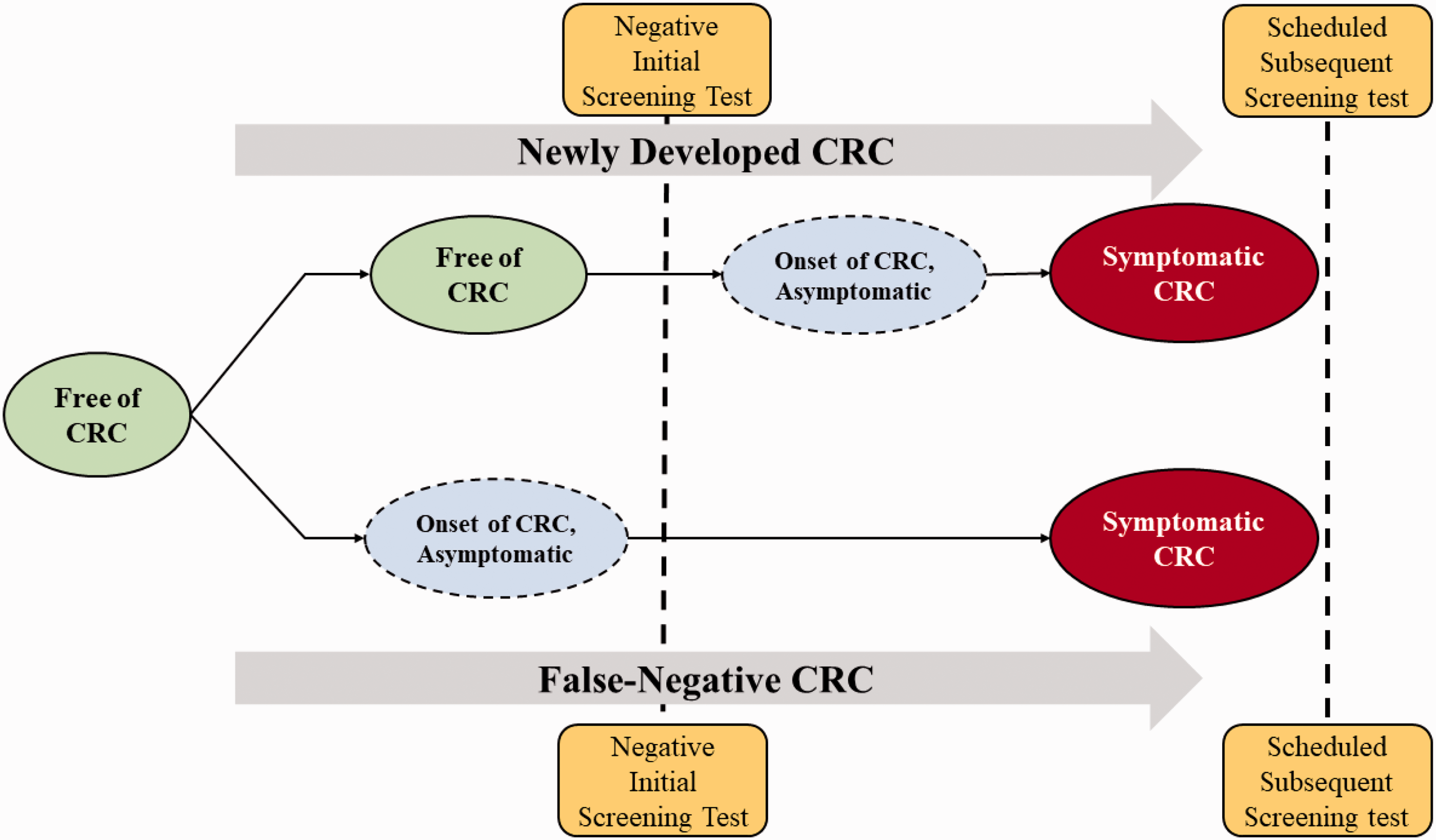
Newly developed and false-negative CRC cases among the FIT-negative interval cancer cases.
While false-negative CRCs are closely related to the ability of a screening tool to detect asymptomatic cancers, newly developed CRCs are strongly associated with the interscreening interval, as the risk of CRC developing from advanced colorectal neoplasms such as serrated lesions is highly associated with the follow-up time. Very few studies have been conducted to differentiate newly developed CRCs from false-negative CRCs among FIT interval cancers as there is no possibility of directly distinguishing the two kinds of case by observation. Mixing up newly developed CRCs with false-negative CRCs may lead to an underestimation of FIT sensitivity. Understanding the relative contribution of newly developed and false negative cases to FIT interval CRCs may help determine the optimal interscreening interval and improve the quality of screening programs.
Thus, in this study, we aimed to classify interval cancers as false negatives or newly occurring, and to determine how these proportions are affected by different interscreening strategies, using the data from the Taiwanese biennial FIT screening program.
Patients/Methods
Natural course of progression of CRC
Figure 1 shows the natural progression of CRC from the CRC-free state to asymptomatic CRC and symptomatic CRC for both false-negative and newly developed cancer cases in relation to the date of screening. The natural course of CRC is determined by the incidence rate and the progression rate. The annual incidence rate of CRC is defined as the number of new asymptomatic cases per year divided by the population size. The annual progression rate is defined as the number of asymptomatic CRCs that progress to the symptomatic phase per year, as evidenced by the presence of clinical symptoms and signs, divided by the number of asymptomatic CRCs. In addition to the sensitivity of FIT, the probability of giving rise to false-negative cancer and newly developed cancer is therefore determined by these two rates.
Supplementary Material A demonstrates different kinetic profiles regarding the probability of asymptomatic and symptomatic CRCs developing after a negative FIT result, stratified by two incidence rates (high and low: 150 and 70 per 100,000) and two rates of progression (high and low: 0.5 and 0.2 per year, corresponding to the average dwelling time of two and five years, respectively). These kinetic curves reveal clinical implications. The magnitude of the incidence rate determines the final number of symptomatic CRC cases during the follow-up period. It is also interesting to note that the higher the progression rate, the sooner the crossover between the two curves occurs: at three years for a progression rate of 0.5 and eight years for a progression rate of 0.2. Using these two cardinal quantities, the incidence rate and progression rate, the proportion of symptomatic CRC cases with different screening intervals can be quantified, given the risk levels of the target population.
Data sources
Data from the Taiwanese nationwide FIT-based CRC screening program collected from 2004 to 2014 were used to estimate the natural progression of CRC. The FIT-based mass screening program in Taiwan was launched in 2004 and involved screening individuals aged 50–69 every two years.10,18,19 The occurrence of CRC, along with the date of diagnosis, in the target population was retrieved from the National Cancer Registry and Death Registry up to 2016.
Information on interval CRCs was derived from the National Cancer Registry for individuals with normal screening results. For the derivation of CRC incidence and rate of progression under the regular screening scenario with a two-year interval, the individuals who attended a prevalent screen, those who attended a subsequent screen with an average interval of two years, and those who developed clinical CRC within three years of the last negative screening result were included in the analysis.
Study design
We evaluated the effect of interscreening intervals ranging from one to three years on interval cancer cases detected within a FIT-based screening program. To ensure the study arms that were exposed to FIT-based screens could be compared with different interscreening intervals and the control arm, we first estimated the incidence rate, progression rate, and sensitivity of FIT for CRC, with consideration of age and sex, using a Markov regression model in conjunction with the empirical data from the Taiwanese nationwide CRC screening program.20–22 The frequency of interval cancers along with the two components, false-negative and newly developed CRC, were then projected on the basis of the estimated results.
The proportions of interval cancers that were newly developed or false-negative CRC cases were derived according to the method described in Supplementary Material B. By using the estimated incidence and progression rates of CRC derived from the empirical data, a three-arm study with one control group and three intervention arms with interscreening intervals of one, two, and three years over a six-year follow-up period was designed by allocating 10,000 subjects to each arm. The total number of interval CRCs and numbers of false-negative and newly developed CRCs were derived for each arm. The effects of a screening program with short and long interscreening intervals were then evaluated on the basis of the number and sources of interval cancers considering age- and sex-specific risk levels of CRC.
We also compared the gFOBT and FIT screening tools in terms of performance in detecting newly developed and false-negative CRC cases using three randomized controlled trials.4,9,23,24 The details of these three trials and the study design are provided in Supplementary Material C.
Statistical analysis
The CRC-free state, asymptomatic CRC state, and interval CRC state constituted the three-state model for CRC progression. For the derivation of the two cardinal quantities, the CRC incidence and progression rates, a three-state Markov model that considered sensitivity was applied. 21 Details of the specification of this model are provided in Supplementary Material D. A proportional hazards regression model with an exponential link was applied to take into account the effect of age and sex on the risk of developing CRC. To assess the impact of the interscreening interval and screening tools on interval CRC, the estimated results for age- and sex-specific rates and sensitivities were further applied to the proposed multi-arm trials depicted in Supplementary Materials B and C. All statistical analyses were performed with SAS 9.4 (SAS Institute, Cary, NC, USA).
Results
Demographic characteristics of the screening attendees
The demographic characteristics are listed in Table 1. Between 1 January 2004 and 31 December 2014, a total of 3,070,511 eligible Taiwanese residents attended the nationwide screening program for CRC. Of these, 10,989 (0.36%) asymptomatic cases were identified at the prevalent screen. A biennial screening program was offered for 1,604,443 (52.3%) patients with negative FIT at the prevalent screen, of whom 3128 (0.19%) were identified as having an asymptomatic case at subsequent screens. A total of 9641 (0.6%) interval CRC cases diagnosed between the two screens were recorded. Male and older (aged between 60 and 69) subjects had a higher risk of CRC.
Demographic characteristics of screening population.
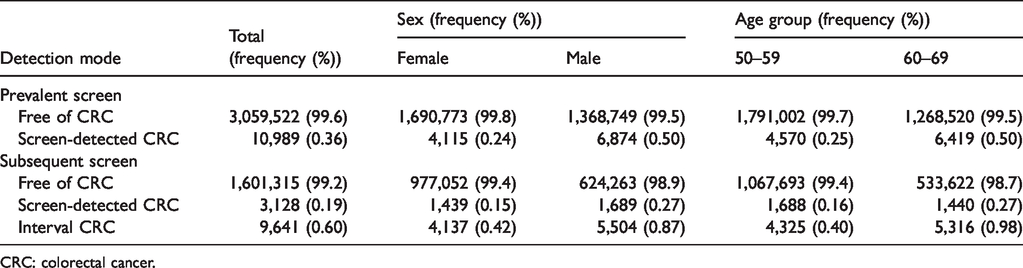
CRC: colorectal cancer.
Age- and sex-specific risk of CRC and dwelling time
Table 2 shows the estimated results for incidence rate, progression rate, and sensitivity based on the empirical data of the Taiwanese nationwide screening program for CRC. The incidence rate was estimated as 0.00151 per year (95% CI, 0.00147–0.00155). The annual progression rate from asymptomatic to symptomatic CRC was 0.36 per year (95% CI, 0.34–0.38), with an average dwelling time in the asymptomatic state of 2.78 (95% CI, 2.63–2.94) years. It is very interesting to see that the sensitivity for detecting CRC with FIT after the newly developed cases were separated from the false-negative cases (among the interval cancers) was estimated to be 75.5% (95% CI, 73.0%–77.8%).
Estimated age- and sex-specific incidence rate, progression rate (plus dwelling time), and sensitivity.
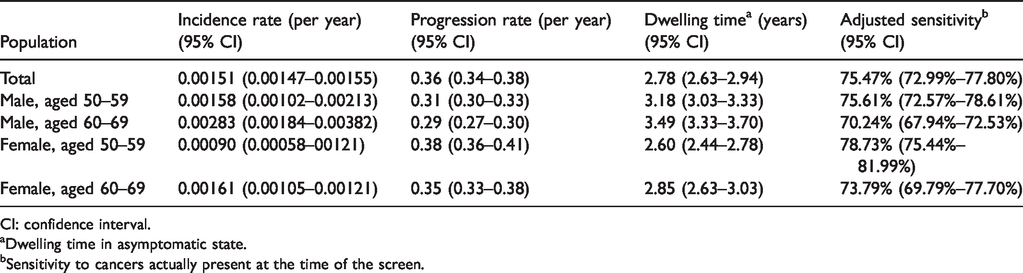
CI: confidence interval.
Dwelling time in asymptomatic state.
Sensitivity to cancers actually present at the time of the screen.
The estimated results in Table 2 also show the age- and sex-specific risk of CRC. The average dwelling times in males were 3.18 years and 3.49 years for the 50–59 and 60–69 age group, respectively. The average times were 2.60 years and 2.85 years for the same age groups, respectively, in the females. The sensitivity of FIT was highest for women aged 50–59 (78.73%), followed by men aged 50–59 (75.61%), women aged 60–69 (73.79%), and men aged 60–69 (70.24%).
Figure 2 demonstrates the effect of age- and sex-specific incidence and progression rates on the probability of having asymptomatic and symptomatic CRC over the follow-up period. Within the same age range, men had a higher probability of having symptomatic CRC than women ((b) vs. (d) and (c) vs. (e), Figure 2). Within the same sex, the elderly subjects had a higher probability of having interval CRC than did the younger subjects ((b) vs. (c) and (d) vs. (e), Figure 2).
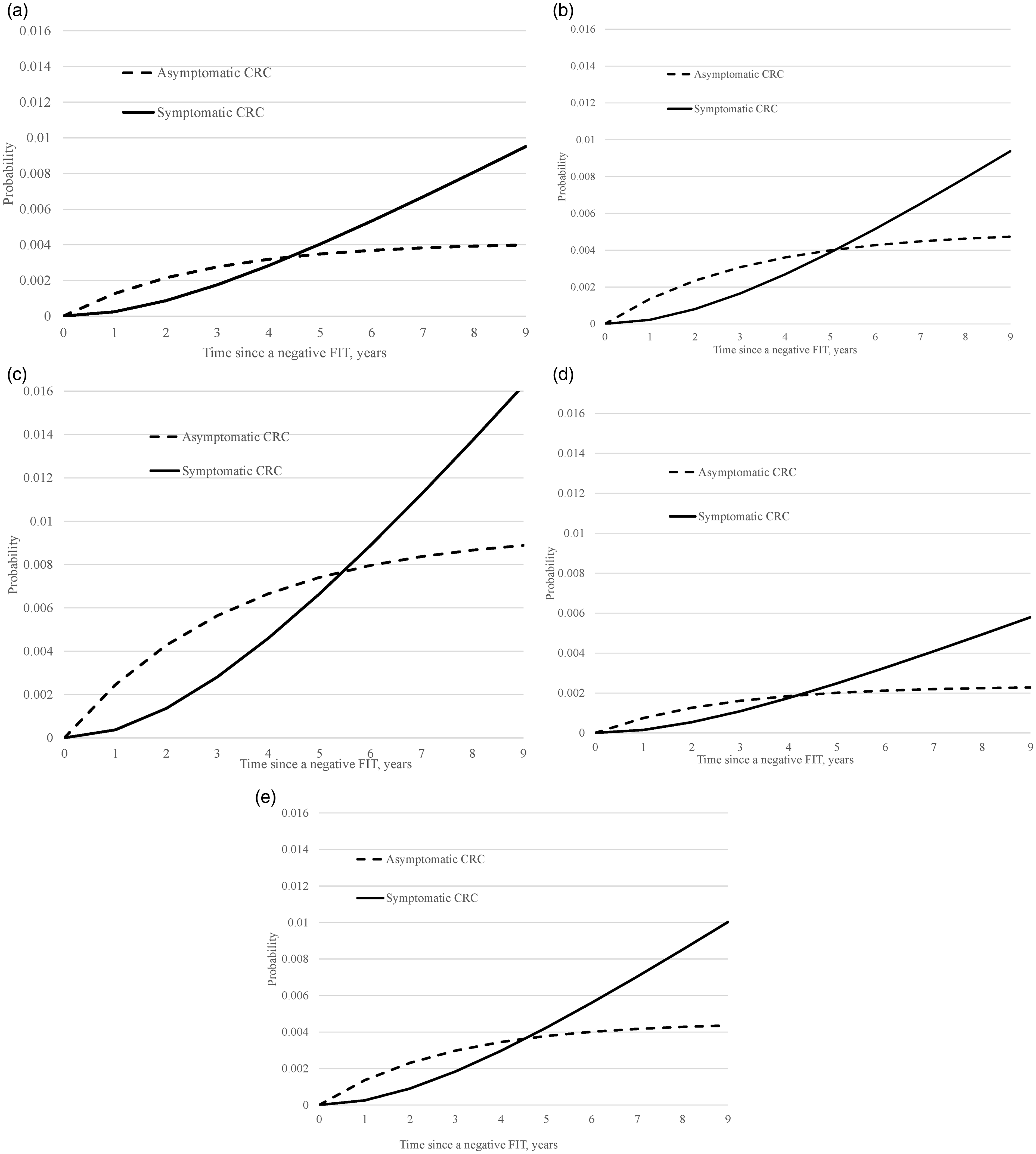
Proportions of asymptomatic and symptomatic CRC cases based on empirical data from the Taiwanese CRC screening program. (a) Overall (incidence 151 per 100,000, rate of progression 0.36 per year). (b) Males, 50–59 years. (c) Males, 60–69 years. (d) Females, 50–59 years. (e) Females, 60–69 years.
Effect of screening interval and dwelling time on interval CRC
Figure 3 shows the estimates based on the incidence rate, transition rate, and FIT sensitivity of the Taiwanese CRC screening program. The risk of developing interval cancer increased with the incremental interscreening interval. False-negative CRC cases accounted for 38.9% of FIT interval cancer cases for the annual program, 31.2% for the biennial program, and 25.3% for the triennial program, and thus newly developed CRCs accounted for 61.1%, 68.8%, and 74.7%, respectively (General Population, Figure 3). It should be noted also that the proportion of newly developed CRCs was higher in the subjects aged 50–59 than in those aged 60–69. The findings did not substantially vary by sex.
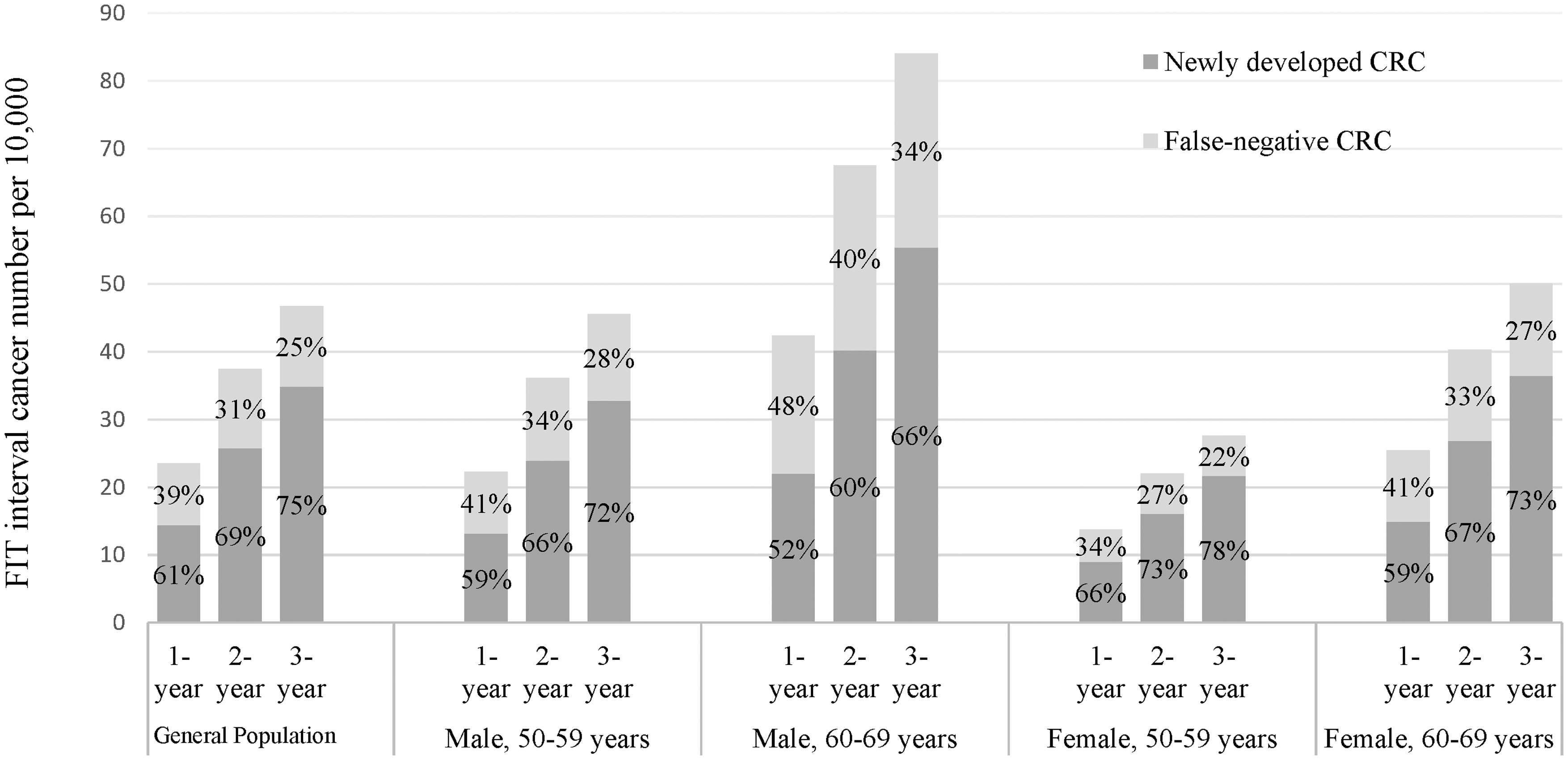
Frequencies and proportions of newly developed and false-negative CRC cases stratified by the interscreening interval and characteristics of the subjects.
Effect of the screening test on interval CRC: gFOBT versus FIT
Figure 4 shows the interval cancer rate as a percentage of the expected incidence rate (I/E ratio) and the percentages of newly developed and false-negative CRC cases. The I/E ratios were 29.8%, 38.5%, 35.7%, and 43.5% for the Taiwan program (current study), the Funen study, 9 the Nottingham study, 4 and the Finland study,23,24 respectively. Compared with the three gFOBT-based randomized controlled trials, the FIT screening program is associated with fewer interval cancer cases.
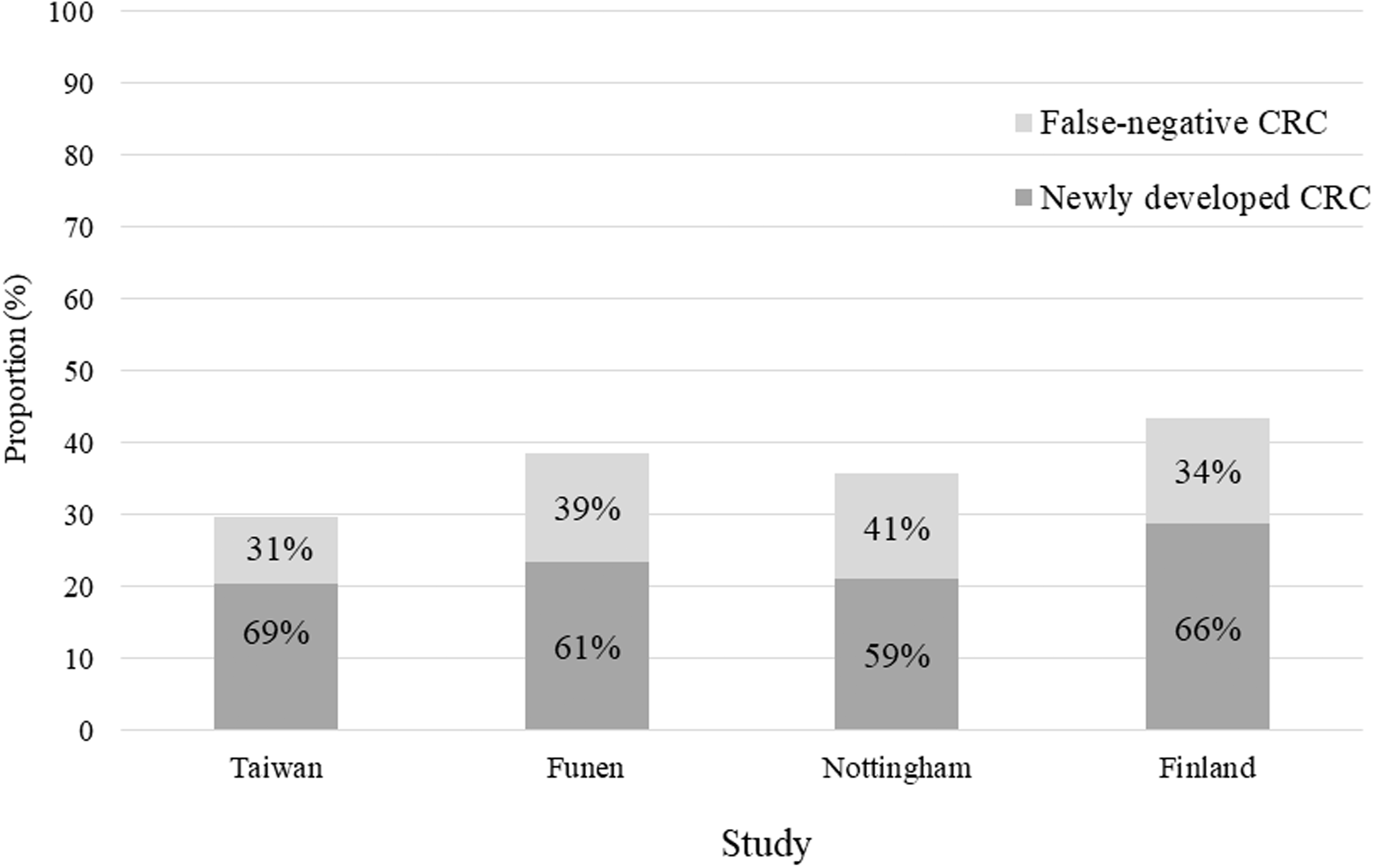
Interval CRC as a percentage of the expected incidence from g-FOBT (Funen, Nottingham, and Finland) and FIT (Taiwan) studies.
Discussion
Our study is the first to divide FIT interval cancer cases into two components, namely, newly developed and false-negative CRC cases. Based on a population-wide FIT-based screening program, we demonstrate that newly developed CRC cases account for 68.8% of FIT interval cancer cases. The sensitivity in detecting asymptomatic CRC after separating out the newly developed CRC cases was 75.5%, whereas the traditional estimate of sensitivity was 70.2%.
Interval cancer, one of the core measures of the quality of cancer screening programs, is substantially affected by sensitivity. Compared with gFOBT, FIT has a higher sensitivity in detecting CRC (depending on the FIT threshold used) and is more likely to reduce the number of interval cancer cases in a screening program.25–27 Regarding the proportion of interval cancers compared to screen-detected cancers, Wieten et al. reported ratios of 1:1.2 and 1:2.6 estimated from screening programs using gFOBT and FIT as the tool, respectively. 28 The results of our simulation, based on the general population, can be used to calculate this ratio. Of 10,000 subjects in the general population, 41 CRCs were detected in the screening program, and 17 FIT interval CRCs were diagnosed between screens. This gives a ratio of 1:2.4 between the FIT interval cancers and screen-detected CRCs. These findings consistently indicate a higher odds of detecting CRC by using FIT compared with gFOBT.
The sensitivity of the Taiwan FIT-based screening program was 70.2% based on the conventional proportional incidence method, which as noted above was lower than the estimated result of 75.5% derived from direct estimation based on the Markov model. This is mainly due to the difference in how the components of interval cancer, namely, newly developed CRC and false-negative CRC cases, were considered for the estimation of sensitivity. Among the observed FIT interval cancers, 68.8% were newly developed CRCs and were classed as missed at screenings when using the conventional proportional incidence method. On the other hand, the Markov model took account of these newly developed CRCs, to derive an accurate evaluation of FIT sensitivity to those cancers which were actually present at the time of screening.
In our study, the sensitivity of FIT was higher for subjects aged 50–59 than for those aged 60–69 and for women than for men. The biological background accounting for this finding may be due to the association between the location of colorectal neoplasm and age and gender. Evidence from previous studies shows a higher incidence rate of proximal CRC among subjects older than 60 years.29,30 Since the sensitivity of FIT was lower for proximal CRC, 18 this difference in CRC risk by location may partly account for our finding.
Since newly developed CRC is estimated to represent the majority of FIT interval cancers, it is interesting to scrutinize the effect of the interscreening interval by sex and age group. The longer the interscreening interval is, the higher the proportion of newly developed CRC. Among the target population, the younger age group had a higher proportion of newly developed CRCs (66.2% and 73.0% for males and females aged 50–59, respectively) than did the older age group (59.6% and 66.7% for males and females aged 60–69, respectively), which can be decreased by the use of a shorter interscreening interval. Our results, therefore, provide insight into age- and sex-based risk stratification by breaking down the evolution of CRC into incidence rate and progression rate.
Sex and age are important determinants for the incidence rate of CRC and the rate of progression from the asymptomatic to symptomatic state. Our results show a higher incidence rate of CRC among the older population, aged 60–69. Although the risk level for CRC was low, the younger age group suffered from a higher proportion of newly developed CRC. The older age group had more interval cancers that were either newly developed or false-negative CRCs due to the increased incidence of asymptomatic CRC. Given these effects of age and sex on the initiation and progression of CRC and the components of interval cancers, a risk-based and age-tailored strategy is required, although uniform recommendations for CRC screening have been used in all of the current population-based programs.31–33
Our study has several strengths. First, a population-based screening program for average-risk individuals was used, and thus the results may be representative of the target population of most FIT-based screening programs. Second, the large sample size of the study cohort provides a reliable estimation of the parameters of CRC progression, the sensitivity of FIT, and the effects of age group and sex. Third, using the empirically estimated results, we evaluated the occurrence of interval cancers and classified these cases as newly developed or false-negative CRCs, as these results could not be obtained by direct observation of FIT interval CRC cases. Finally, the discovery that newly developed CRCs accounted for the majority of FIT interval cancers may also serve as the rationale for developing an individually tailored screening policy for high-risk individuals.
To detect newly developed CRCs at an early stage and reduce the occurrence of false-negative CRCs, several solutions, in addition to shortening the interscreening interval as demonstrated in our study, can be considered. First, within the context of the FIT screening program, lowering the positive cutoff value may facilitate early detection. Increasing the number of stool samples (from one to two) is an alternative approach. 34 The impact of adopting these approaches on manpower demand, acceptability or endoscopy capacity, however, should be carefully evaluated. Second is to use the age- and sex-tailored FIT cutoff. Our recent study has demonstrated that such an approach can effectively improve the sensitivity of 5% for detecting CRC while increasing the endoscopy demand very little. 35 Finally, novel screening tests, such as multitarget stool DNA tests, which have been reported to be more sensitive than FIT in detecting not only advanced adenoma but also sessile serrated adenoma, may help reduce the risk of FIT interval cancers. 36 The high screening-related cost and lower specificity of these DNA tests, however, are barriers to adopting these tests in an organized screening program.
In the current study, we estimated the contribution of false-negative cases and newly developed cases to FIT interval cancer. However, it would be interesting to model whether adenomas, particularly large adenomas, detected at a subsequent screen are attributed as false negatives in the previous screen or whether they are newly developed cases. By making use of the transition parameters derived from an eight-state Markov model of the natural history of adenomas (including diminutive, small, and large adenoma) and asymptomatic and symptomatic CRCs in a previous study conducted in Taiwan, 37 the [(1-I/E ratio) x 100%] for FIT interval cancer was 69.7%, which is very close to our estimate of 70.2%. With the application of these parameters and this model, we performed a simulation study to explore [(1-I/E ratio) x 100%] to quantify FIT interval adenomas (including those missed in the previous but detected at subsequent screen and newly developed cases), as shown in Supplementary Material E.1. It can be seen that [(1-I/E ratio) x 100%] increased from 33.2% at the second screen to 44.5% at the third screen, 57.2% at the fourth screen, and 64.5% at the fifth screen. This finding implies that the FIT test sensitivity for detecting adenomas is approximately 33% given a two-year interval. However, missed adenomas may be identified after two rounds of screens, as shown in Supplementary Material E.1. The sensitivity would increase to 45% when they are detected by one more subsequent screen. As it is uncertain whether some adenomas missed at the prevalent screen are able to be detected after a certain number of screening rounds, a sophisticated natural history model that incorporates the test sensitivity for detecting adenomas would be required for an accurate evaluation. This objective is beyond the scope of the current study and will be the subject of future research.
The lack of a model of the natural history of adenoma does not affect our current finding on the respective contributions of false-negative cases and newly developed CRC cases to FIT interval cancers, as the time required for the development of a large adenoma, not to mention adenoma, even if missed in the previous screen would be still longer and would not make a substantial contribution to FIT interval cancers. The time required for new adenomas to progress to invasive CRC would be even longer. Moreover, Supplementary Material E.2 shows that at the fifth screen the curve plateaus, which implies that adenomas missed after the first screen are expected to be fully detected after three subsequent screens, approximately six years later. This finding suggests that our result on the individual contributions of the two types of cases to FIT interval cancers would not be substantially affected if the occurrence of CRC were modeled to follow the adenoma-carcinoma pathway. The majority of the newly developed FIT interval cancers may occur through the de novo pathway pertaining to the serrated lesion. This explains why the CRC-free status was defined by normal or precancerous lesions, such as small or advanced adenomas.
There is one limitation. Our study was not a randomized controlled trial. To ensure comparability among groups, we established the intervention group and control group according to the incidence and progression rates of CRC and the FIT sensitivity derived from empirical data, and these parameters were comparable with those reported in other studies addressing mass screening for CRC.38,39
In conclusion, newly developed CRCs account for 68.8% of FIT interval CRCs. Particularly in a younger population, it is imperative to identify high-risk individuals to offer a short interscreening interval or more accurate and advanced detection methods to reduce the odds of developing interval cancer.
Supplemental Material
sj-pdf-1-msc-10.1177_0969141320986830 - Supplemental material for Classifying interval cancers as false negatives or newly occurring in fecal immunochemical testing
Supplemental material, sj-pdf-1-msc-10.1177_0969141320986830 for Classifying interval cancers as false negatives or newly occurring in fecal immunochemical testing by Wen-Feng Hsu, Chen-Yang Hsu, Amy Ming-Fang Yen, Sam Li-Sheng Chen, Sherry Yueh-Hsia Chiu, Jean Ching-Yuan Fann, Yi-Chia Lee, Han-Mo Chiu and Hsiu-Hsi Chen in Journal of Medical Screening
Footnotes
Data and materials availability
Aggregate data and codes are available from the author upon request.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: HHC, AMFY, SLSC, YCL, and CYH are supported by Ministry of Science and Technology grants (grant numbers MOST 108–2118-M-002–002-MY3; MOST 108–2118-M-038–001-MY3; MOST 108–2118-M-038–002-MY3). YCL is supported by The Featured Areas Research Center Program within the framework of the Higher Education Sprout Project by the Ministry of Education (MOE) in Taiwan (NTU-107L9003). The funders did not play any roles in the study design, data collection or analysis, decision to publish, or preparation of the manuscript.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
