Abstract
Objective
Testing for occult blood in faeces is widely used in bowel cancer screening around the world. In many programmes, the faecal immunochemical test (FIT) is replacing the traditional guiaic faecal occult blood test (gFOBT). There have been a number of reports on the clinical impact of making this change; yet, no-one has considered the pre-analytical and analytical impact of moving from a gFOBT to a FIT bowel cancer screening programme.
Methods
We interrogated data obtained in a FIT pilot carried out in England in 2014 to assess the timeliness of specimen collection device return time and analysis for gFOBT and FIT, the impact of time to analysis on faecal haemoglobin (f-Hb) concentration, and any differences observed between analyses carried out at two different testing laboratories.
Results
FIT kits were returned on average 5.6 days sooner than gFOBT. The time to analysis for FIT leads to an overall rise in f-Hb concentration within the manufacturer’s stated 14-day stability period.
Conclusion
Both these factors are important considerations for laboratories when considering setting up a bowel cancer screening programme, especially if transitioning from gFOBT to FIT. Our data also support previous evidence of males having a higher f-Hb than females and demonstrate that after adjusting for sex, age and screening hub, neither index of multiple deprivation nor screening episode significantly affected f-Hb.
Keywords
Introduction
Testing for occult blood in faeces is widely used in bowel cancer screening programmes across the world.1,2 Historically, this has been carried out using the guaiac faecal occult blood test (gFOBT), which provides qualitative results. The National Health Service (NHS) bowel cancer screening programme (BCSP) in England introduced the gFOBT to a screening population aged 60–70 in 2006. In 2015, the BCSP was fully rolled out to include people aged 60–74 inclusive. The service was provided by five regional hubs across England.
The faecal immunochemical test for haemoglobin (FIT) uses antibodies derived against the globin component of the haemoglobin (Hb) molecule to quantify the human Hb concentration in a faecal specimen (f-Hb). The benefits of FIT over gFOBT have been well described.3–6 These include the ability to automate and quantitate analysis and the specificity of the antibodies to human Hb.
In 2014, the BCSP carried out a large comparative FIT pilot study to establish the acceptability and diagnostic performance of FIT compared to gFOBT. 3 Two of the five hubs in England were included in the study: the Midlands and North-West Hub (M&NWH) and the Southern Hub (SH). These two hubs cover over half of the eligible population in England, which at the time of sending out invites was 1.1–1.2 million subjects per year. Each hub performed analysis of FIT samples for its served population. The study demonstrated marked improvements in uptake, specifically in groups where uptake was previously low, and higher detection rates for cancer and advanced adenoma were reported at a range of thresholds used for referral for colonoscopy. 3
In this study, we have interrogated the data obtained in the FIT pilot study to
assess the timeliness of specimen collection device return time and analysis for gFOBT and FIT evaluate the effect of device return time and time to analysis on f-Hb results obtained by FIT evaluate the extent to which results obtained differ by hub examine the impact of age and sex on f-Hb results obtained by FIT assess whether there is an association between f-Hb, index of multiple deprivation (IMD) and episode type: First screen with no previous invitation (first time invitees) First screen with previous non-response to invitation (previous non-responders) Second or subsequent screen (previous responders).
We use the term subject to describe someone invited to participate in the screening programme and the term participant to describe someone who has returned their test kit.
The information obtained from this study can in theory be used to help inform programmes (laboratories and policy makers) about the changes that might occur when transitioning from gFOBT to FIT.
Methods
The FIT pilot study is described in detail elsewhere. 3 Briefly, over a six-month period in 2014, gFOBT kits (Immunostics, Ocean Township, NJ, USA, supplied by Alpha Laboratories, Eastleigh, UK) were sent as per usual BCSP practice to the majority of subjects (total 1,123,306). FIT (OC-SENSOR, Eiken Chemical Co., Ltd, Tokyo, Japan) devices were sent to 1 in 28 subjects (total 40,928) from the M&NWH and the SH. The BCSP call/recall system was modified so that the first in every 28 routine invitations sent out for each screening centre was a FIT. This regime was strictly applied to ensure that any additional colonoscopy workload generated was split across screening centres. The pre-invitation letters for gFOBT and FIT were similar in content, the difference being that those invited for FIT received an additional single information sheet explaining that they had been invited as part of the FIT pilot and the rationale for the study. Participants were asked to record on their gFOBT sample(s) or FIT device the date(s) the specimen(s) were collected and were provided with a pre-paid envelope to return their kit or device through the postal system. gFOBT kits were returned in an envelope and the FIT device in a uniquely designed box. Both types were sent out and returned using the same postal process, and all were logged onto the electronic Bowel Cancer Screening System (BCSS) on the day of receipt in the laboratory.
Of the 1,123,306 subjects sent a gFOBT kit, 661,780 (59%) valid results were obtained. Of the 40,928 subjects sent a FIT device, 27,238 (67%) valid FIT results were collected. All participants with a positive gFOBT result or an f-Hb ≥ 20 µg Hb/g faeces were referred for follow-up and managed according to the usual BCSP protocol and were given a clinic appointment for follow-up in their local screening centre. The presence of f-Hb on gFOBT is observed as a blue colour visible to the eye when hydrogen peroxidase is added to the sample and left for 30 s to develop. Each gFOBT kit has three faecal samples spread on two windows (six in total per kit). Five out of six windows need to turn blue for the sample to be reported as positive. Subjects were sent a repeat gFOBT kit if between one and four windows were blue. If negative, a third kit was sent. If the retest had any blue window on either repeat, the subject was classified as positive. Subjects were only included in the final analysis if they had a definitive result, so with gFOBT, if a repeat kit was not returned following a weak positive, then they were defined as having not been adequately screened.
FIT kits that had not been dated or had incorrect dates were analysed and if positive the result was reported. If the result was negative the sample was spoilt, and the subject asked to complete another kit. Samples that were not dated but returned less than 10 days from the date of issue were reported.
Data on participant age, sex, IMD status, previous screening history (episode type), f-Hb concentration and result of further investigation for those with a positive gFOBT and f-Hb ≥ 20 µg Hb/g faeces were extracted from the BCSS. The dataset used in this paper was extracted from the BCSS with reference ODR_1819_103. It has been substantially updated and cleaned since the previous publication, in particular including f-Hb results which became available after the previous publication was written and so will not have exactly the same numbers as previously reported.3,7,8 Compared to the previous paper, in the FIT group, we report on two fewer subjects (40,928 vs. 40,930), 71 more participants with valid FIT results (27,238 vs. 27,167), six more samples with f-Hb ≥20 µg Hb/g faeces (2133 vs. 2127) and one more colonoscopy result (1825 vs. 1824).
Statistical methods
We analysed three measures of time:
number of days between date the testing kit was sent to a subject and the date the kit was logged at the hub (total return time); number of days between the date of specimen collection (written on kit by participant) and logged at the hub (specimen return time); number of days between date of specimen collection and date of specimen being analysed at the hub (time to analysis).
For the date of specimen collection for gFOBT, which includes three samples, the date of the last sample was used. For FIT, the date the sample was taken was used.
For both time intervals, we compared the difference between gFOBT and FIT using the Wilcoxon rank-sum test. We used means, standard deviations (SD) and medians and interquartile ranges (IQR) to describe the results with respect to these time intervals. For the estimation of associations of both time intervals, hub, age, sex, IMD and episode type with f-Hb, the situation was complicated by the fact that the majority of participants had undetectable f-Hb; that is, f-Hb below the manufacturer’s stated limit of detection (LOD) of 4 µg Hb/g faeces. We therefore used hurdle regression,9,10 simultaneously fitting a probit regression of the binary variable of
All analyses were carried out in StataMP version 16.1 on a Windows 8 platform.
Results
Tables 1 and 2 give a breakdown of participants who returned valid gFOBT and FIT kits in terms of their characteristics, demographics and f-Hb statistics. In both the hubs, higher positivity was observed by both methods in males compared to females, in older participants and in more deprived populations. Previous non-responders had the highest positivity using gFOBT and FIT. Higher positivity for gFOBT was observed in the M&NWH. For those with detectable f-Hb, higher values were observed in the SH.
Frequencies and proportions of participants with gFOBT devices, who were adequately screened, and had definite positive results, stratified by demographic characteristics and episode type.
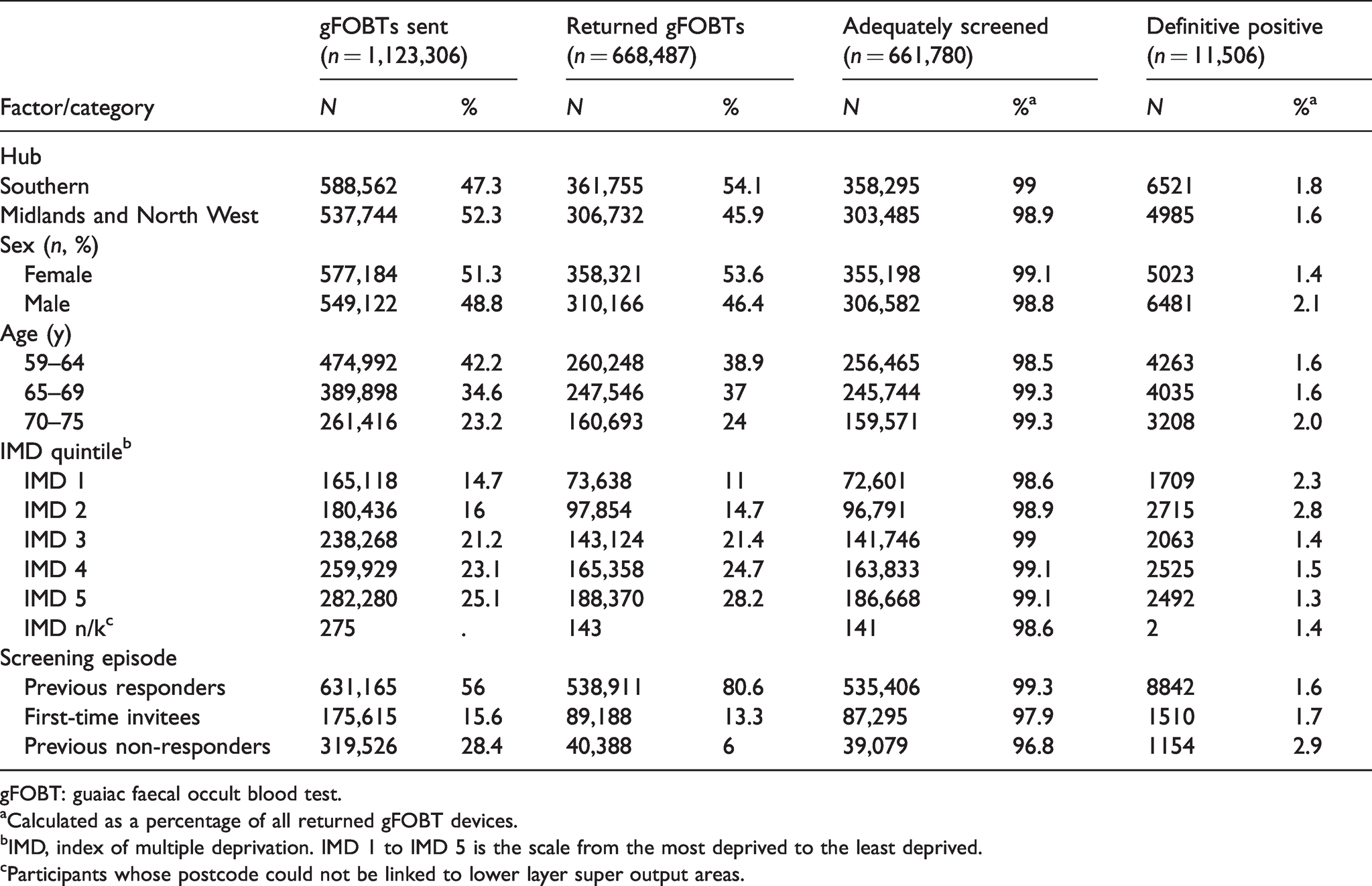
gFOBT: guaiac faecal occult blood test.
aCalculated as a percentage of all returned gFOBT devices.
bIMD, index of multiple deprivation. IMD 1 to IMD 5 is the scale from the most deprived to the least deprived.
cParticipants whose postcode could not be linked to lower layer super output areas.
Frequencies, proportions, geometric means and 80% empirical percentile ranges of participants with analysable FIT devices and detectable faecal haemoglobin concentrations (f-Hb in µg Hb/g faeces), stratified by demographic characteristics and episode type.
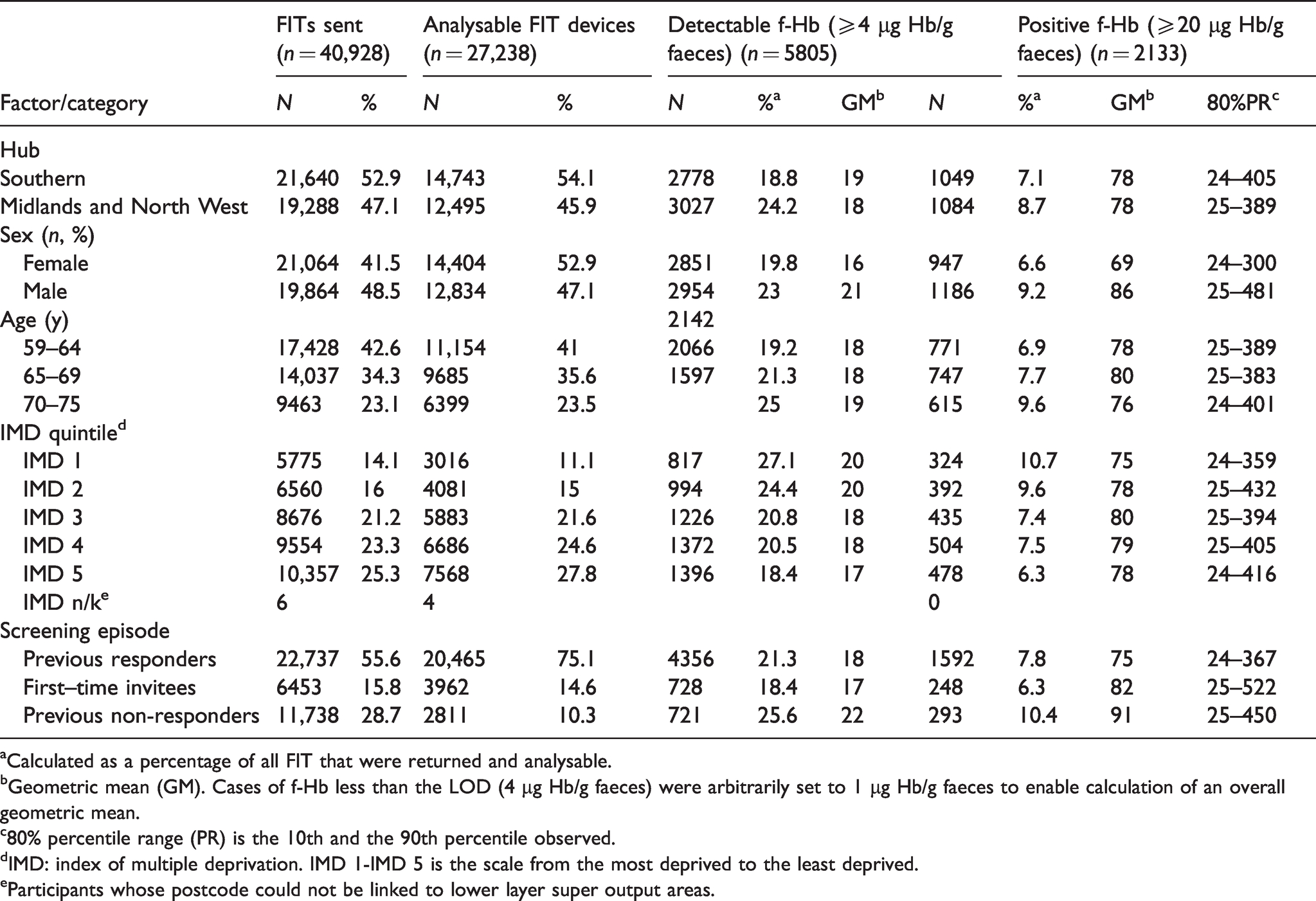
aCalculated as a percentage of all FIT that were returned and analysable.
bGeometric mean (GM). Cases of f-Hb less than the LOD (4 µg Hb/g faeces) were arbitrarily set to 1 µg Hb/g faeces to enable calculation of an overall geometric mean.
c80% percentile range (PR) is the 10th and the 90th percentile observed.
dIMD: index of multiple deprivation. IMD 1-IMD 5 is the scale from the most deprived to the least deprived.
eParticipants whose postcode could not be linked to lower layer super output areas.
There were 627,006 subjects returning a gFOBT kit and 26,121 returning a FIT kit with a recorded date kit sent, date sample taken, date kit logged and date analysed. Table 3 shows means (SD) and medians (IQR) for total return time, specimen return time and time to analysis. Only subjects whose device return time, sample return time and time to analysis are of at least one day and no longer than 60 days are included. All times were significantly longer for gFOBT than for FIT (
Time to return of specimens for gFOBT and FIT.
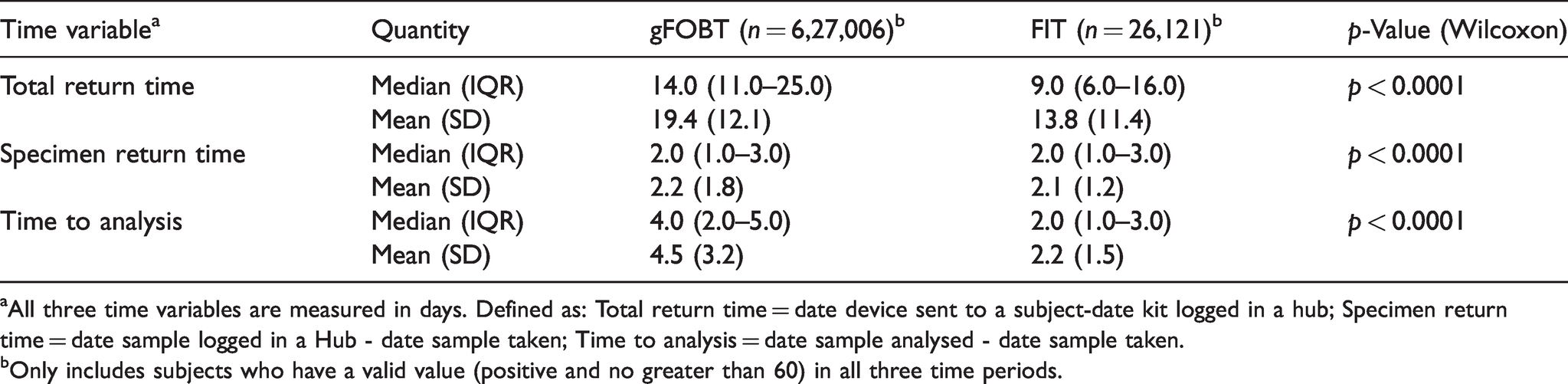
aAll three time variables are measured in days. Defined as: Total return time = date device sent to a subject-date kit logged in a hub; Specimen return time = date sample logged in a Hub - date sample taken; Time to analysis = date sample analysed - date sample taken.
bOnly includes subjects who have a valid value (positive and no greater than 60) in all three time periods.
Detailed day-by-day data on numbers with undetectable f-Hb and average f-Hb for those with detectable f-Hb are given in Table 4. Because of small numbers received after eight days, data are broken down by day until day 7 and then grouped together for greater than or equal to eight days. For the first seven days, the median f-Hb increases for participants with a detectable f-Hb (≥4 µg Hb/g faeces). However, the median f-Hb did not display a particular upwards or downwards trend for participants with a positive f-Hb (≥20 µg Hb/g faeces). A total of 330 kits were received with no sample date, 4 had an illegible date and 65 had an incorrect date.
Detailed f-Hb by individual day between specimen taken and analysisa.
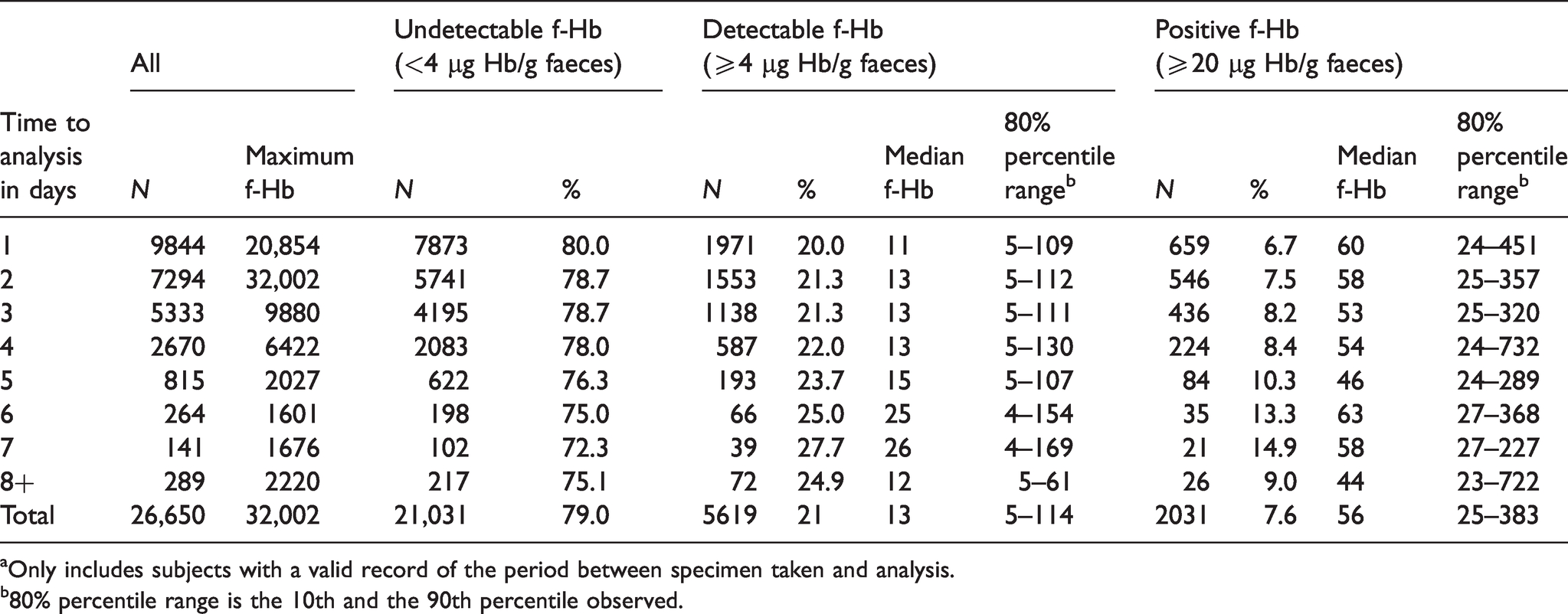
aOnly includes subjects with a valid record of the period between specimen taken and analysis.
b80% percentile range is the 10th and the 90th percentile observed.
In the hurdle regression model, there was a significantly increased probability of having a detectable f-Hb with an increasing time to analysis in days, with a probit regression coefficient 0.02 (95%CI: 0.013–0.026) corresponding to a relative increase of approximately 2% per day to analysis in the probability of a detectable f-Hb. The multivariable hurdle regression results for those with detectable f-Hb are shown in Table 5. The hurdle regression showed a small but statistically significant increase in f-Hb with time to analysis, corresponding to an approximate 2.0% increase per day. There was a small but significant increase in f-Hb with increasing age, corresponding to a 0.6% increase with every 10-year age difference. Males had significantly (around 5.0%) higher f-Hb than females. And f-Hb results were approximately 2% lower in the M&NWH compared to the SH. There was no significant effect of IMD or episode type.
Results from hurdle regression, reported as changes per unit or from baseline category, of natural logarithm of f-Hb on time to analysis and demographic variables.a
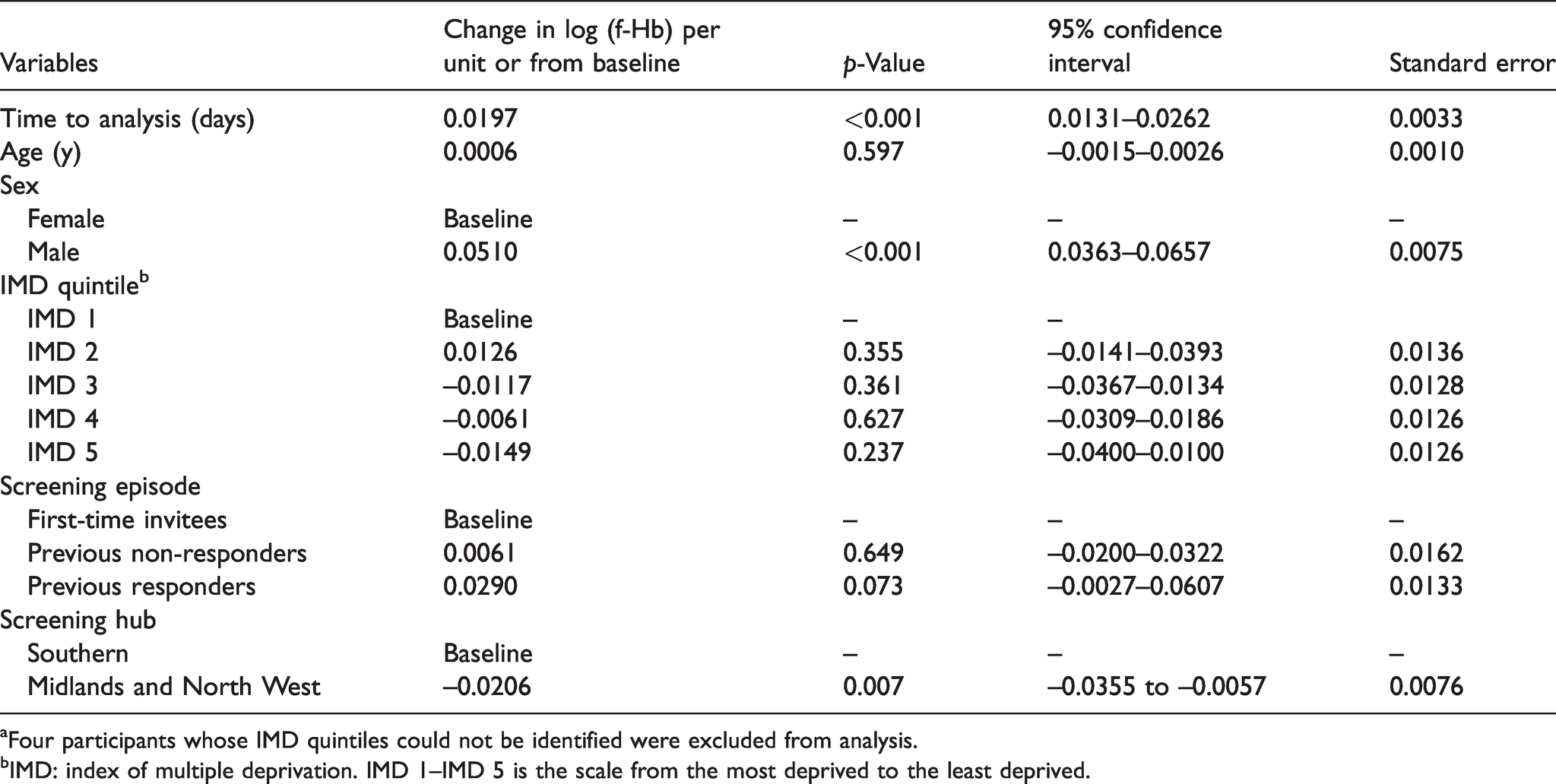
aFour participants whose IMD quintiles could not be identified were excluded from analysis.
bIMD: index of multiple deprivation. IMD 1–IMD 5 is the scale from the most deprived to the least deprived.
Figure 1 shows the f-Hb concentrations against day of sample analysed, post collection. The majority of samples were received and analysed within the first seven days.
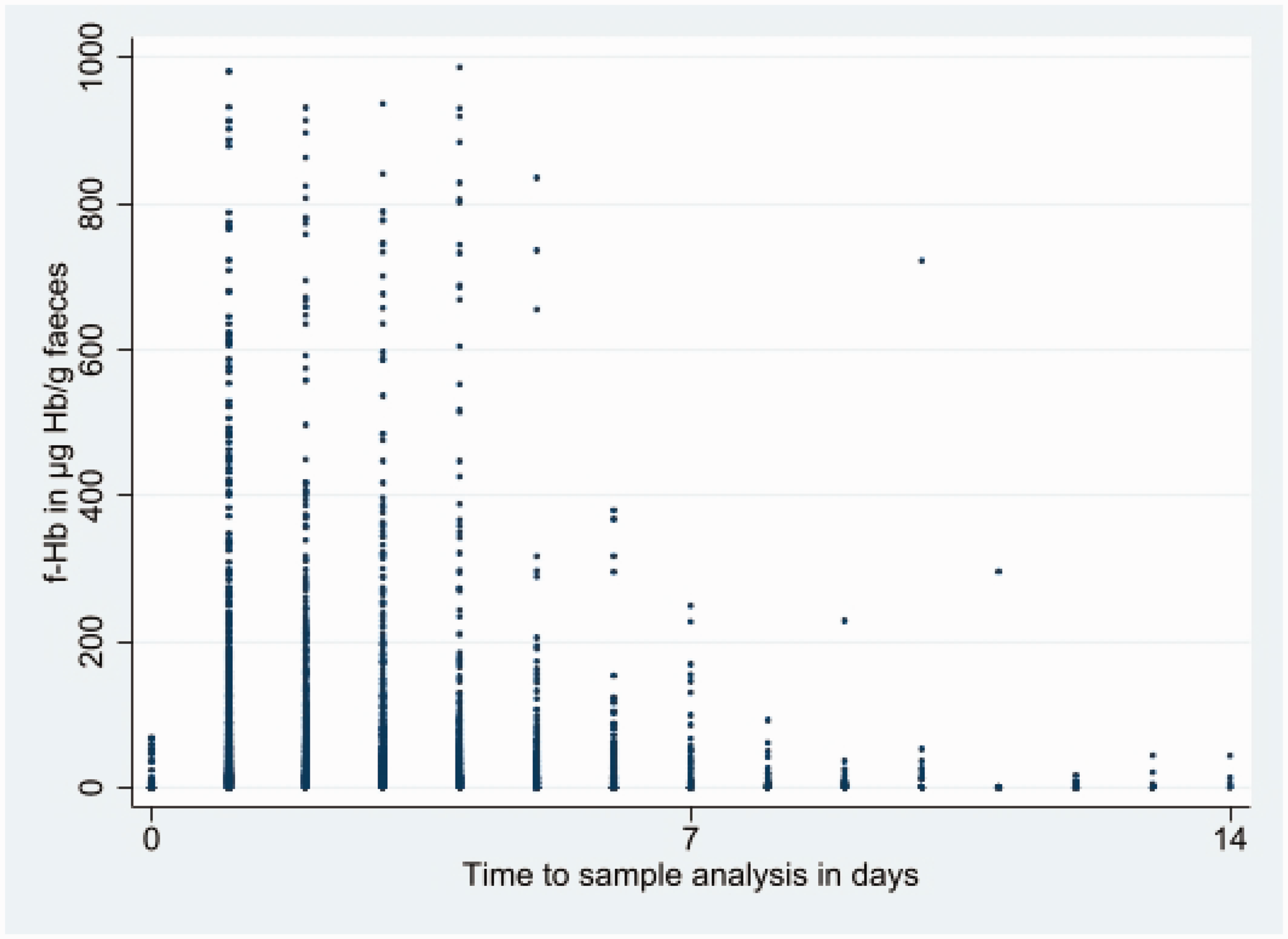
Faecal haemoglobin concentration (f-Hb in µg Hb/g faeces), capped at 1000 µg Hb/g faeces, by time to sample analysis (days), capped at 14 days.
Discussion
To our knowledge, there are only two published pilots of FIT in existing rolled out gFOBT-based screening programmes.3,12 These focus on uptake, positivity and clinical outcomes. Likewise, the published data describing the transition from a gFOBT to a FIT screening programme are also all concerned with positivity, uptake and clinical outcomes.13–16 These factors are all crucially important in terms of a screening programme. What this paper adds to the current knowledge base is the pre-analytical and analytical variations that occur during the transition from gFOBT to FIT and the impact these might have on a screening programme.
The higher f-Hb values in males were to be expected.17,18 Whilst gFOBT positivity was higher in the SH compared to M&NWH, interestingly, the positivity for FIT at the two thresholds analysed was lower in the SH compared to the M&NWH. The reason for this is unclear although it might be related to the threshold at which a gFOBT generates a positive result. Throughout the study, thorough quality assurance procedures were in place which included participation in external quality assurance schemes and a sample swap scheme. These ensured that the results obtained in both hubs for gFOBT and FIT overall were equal, so there is no evidence that local processes impacted the results.
In a multivariate hurdle regression model, there was no significant effect of IMD or episode on f-Hb, although there was a suggestive but not significant increase in f-Hb for participants who had previously participated in bowel cancer screening.
It took significantly less time for a participant to return a FIT kit compared to a gFOBT kit. This is to be expected to a degree because, for FIT, only a single sample is required as compared to three separate samples. However, the mean difference of 5.6 days indicates that it might not just be due to the difference in number of samples required, and we would speculate, based on anecdotal data from participants in the study, that FIT is a more acceptable and straightforward test for people to complete. There is extremely limited data available to assess patient perception of faecal testing although what is available does indicate that patients find FIT easy to complete. In addition, the participants were made aware in the pre-invitation letter that they were part of a study which might have introduced a bias, particularly because they were told to return the kit within 10 days. To fully assess the more rapid return time of FIT, the above data analysis should be repeated in a fully rolled out FIT screening programme.
The more rapid analysis time for FIT can be explained by the fact that the analytical method used a semi-automated analyser, and in addition, the samples being part of a study meant that they were analysed by a dedicated group of staff who were rotated on to the analysers. This did not impact the routine analysis of the gFOBT kits.
The time to analysis for FIT has a positive impact on f-Hb concentration within the manufacturer’s stated 14-day stability period. f-Hb is well known to be unstable, and it is for this reason samples are collected straight into a buffer in the FIT tube because this contains agents that stabilise the Hb, preventing degradation. Evidence shows that over long time periods, there is a decrease in f-Hb, and therefore, laboratories analyse samples as soon as they can, and as such, one might have expected a deterioration rather than an increase in f-Hb with time to analysis. 19 However, it should be noted that times to analysis within this study were almost entirely within the manufacturer’s recommended limit of 14 days. Only eight (0.03%) were analysed after this limit, and exclusion of these did not alter the results. This finding is supported by other studies that have also demonstrated a small increase in f-Hb within the first 14 days when using the same manufacturer’s FIT kits and reagents, including the same buffer formulation that was used in the OC-Sensor device in 2014 for the current study.20,21 Hypothetically, it might be that during the first few days, as time progresses, the Hb molecule opens up exposing more epitopes for binding to the anti-Hb antibody in the reagent. This is speculation, and further work is required to confirm the findings from this study. What is reassuring about this finding is that so long as samples are analysed within the manufacturer’s stated stability period (14 days for OC-Sensor), then a delay of a few days has no detrimental effect on the outcome for the participant. In fact, it could be postulated that a delay of a day or two might increase chances of a positive result being obtained and hence a pre-cancer/cancer being detected.
In summary, we have demonstrated that FIT specimens are collected and returned more rapidly than gFOBT kits in a screening programme. The time to analysis for FIT leads to an overall rise in f-Hb concentration within the manufacturer’s stated 14-day stability period. Both these factors are important considerations for laboratories when considering setting up a bowel cancer screening programme, especially if transitioning from gFOBT to FIT. Our data also support previous evidence of males having a higher f-Hb than females and demonstrate that, after adjusting for the sex effect, neither IMD nor screening episode significantly affects f-Hb.
The clinical benefits of changing from gFOBT to FIT have been well reported. What this study highlights is the impact on testing laboratories, and hopefully, this information can support laboratories and policy makers planning to make the transition to plan resources. The key to demonstrating the reality of the differences observed here will be repeating the analysis in a fully rolled out FIT screening programme.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
