Abstract
Objectives
To report complications after colonoscopy and surgery in patients with neoplasia detected through a population based colorectal cancer (CRC) screening programme in the capital region of Sweden.
Methods
Patients who after a positive FOBT screening result underwent colonoscopy from 1 January 2008 to 30 June 2012 were included. Mortality and complications within 30 days after colonoscopy or subsequent surgery were identified through national registers, and complications were assessed through review of medical charts. Complications were graded using the Clavien-Dindo classification.
Results
After 2984 colonoscopies, the complication rate was 1%. The risk of post-polypectomy bleeding was 14/1000. The risk of perforation was 1/1000 after a diagnostic colonoscopy and 2.5/1000 after a colonoscopy with polypectomy. One patient developed a post-polypectomy syndrome. There was one death which was not related to the colonoscopy. After surgery for 37 adenomas and 155 CRCs, the total complication rates were 27% and 50%, respectively. The rate of anastomotic leakage was 13% and 12% after surgery for adenomas and CRC, respectively. There were no deaths after surgery. The overall complication rate after colonoscopy and surgery for adenomas and cancer was 4%.
Conclusions
Overall complication rates were acceptable and mortality low; however, the rate of anastomotic leakage after surgery for both adenomas and CRC was higher than expected.
Introduction
With an incidence of 447,000 new cases in Europe in 2012, colorectal cancer (CRC) is the second most common form of cancer in Europe, and the second most common cause of death from cancer (215,000 cases in 2012). 1 Since 2003, the European Council has recommended implementation of population-based primary screening programmes using faecal occult blood testing (FOBT) for men and women aged 50–74. Faecal immunochemical testing (FIT) is currently the recommended screening test. 2 Screening with endoscopy is a possible alternative to FOBT, and four large randomized controlled trials (RCT) assessing sigmoidoscopy as the primary screening method have demonstrated a reduction in distal CRC incidence and mortality of 41% and 61%, respectively, and RCTs evaluating colonoscopy are currently ongoing.3,4 Regardless of whether FOBT or endoscopy is used for screening, colonoscopy plays a key role in the diagnostic evaluation of positive findings by other methods, and these findings may subsequently require intervention, either endoscopically or surgically. There is a vast literature on complications after colonoscopy,5–9 and some studies on complications after screening colonoscopies, including 30-day morbidity and mortality.9,10 However, the literature on the collected burden of complications from population-based screening programmes for CRC is limited.
Colonoscopy by itself, but also subsequent interventions following a positive colonoscopy, put the screening participants at risk of complications. This risk must be within reasonable limits, in particular following interventions designed to remove pre-malignant lesions, as the true risk of malignant transformation is unknown in each individual case.
In this study, positive FOBT screening results were followed by colonoscopy. We aimed to evaluate 30-day morbidity and mortality caused by colonoscopy, including both diagnostic and therapeutic procedures, and by subsequent surgery in case of cancer, suspected cancer, or in adenomas not suitable for endoscopic removal.
Methods
The programme and registers
In 2008, a population-based screening programme for CRC was launched in the capital region of Sweden, Stockholm, and by 2009 the island of Gotland was included. The region has a total of 2.3 million inhabitants. The programme is based on biennial screening with guaiac-based FOBT (Hemoccult®), and the target population is all residents aged 60–69. Two birth cohorts were successively included in the programme every year, and by 2014 all eligible birth cohorts had been included. The programme is centrally administered and does not involve primary care. Individuals invited to screening are mailed a test-kit, and a reminder after eight weeks if no tests are returned. Cases with positive test results (≥1 of three cards) are referred for a colonoscopy, to be performed within two weeks at designated centres. All data regarding the screening process, including detailed information on the colonoscopy procedure, findings, and histopathological diagnosis, are prospectively collected in the Stockholm-Gotland screening register. The results of the first five years of the programme have been previously reported. 11 From the screening register, data on all patients with a positive FOBT followed by a colonoscopy performed between 1 January 2008 and 30 June 2012 were retrieved.
The National Board of Health and Welfare, a Swedish government agency under the Ministry of Health and Social Affairs, administers a number of registries, including the Swedish National Inpatient Register (IPR) and the Cause of Death Register. Over 99% of all hospital discharges are recorded in the IPR, and previous validation has shown that 85–95 % of all diagnoses in the IPR are correct. 12 We defined complications within 30 days as events requiring over-night hospital care, and extracted data from the IPR regarding diagnoses and proceedings according to the International Classification of Diseases (ICD-10), length of stay, and hospital name. This was done for all in-patient care admissions from 2008 to 2012, on the patients in the screening register who underwent screening colonoscopy between 1 January 2008 and 30 June 2012, thus generating a minimum of six months of follow-up. In addition, for the same cohort of patients, data regarding date of death were extracted from the Cause of Death Register from 2008 to 2013.
The Swedish Colorectal Cancer Registry (SCRCR), a national quality registry established in 1995, prospectively and continuously collects data on all patients with adenocarcinomas of the colon and rectum. The SCRCR holds detailed information on preoperative staging, surgical procedures, and pathological findings, and on oncological treatment, complications, and recurrence within five years after surgery. The validity of registered data is high and SCRCR covers ≥98% of all CRCs in Sweden.13,14
Complications and mortality
Data on all patients in the screening programme who, after their first positive FOBT, underwent a colonoscopy were matched with the IPR and the Cause of Death Register to find all in-care medical events and possible deaths within 30 days after screening colonoscopy. All adverse events deviating from a normal postoperative course were registered after scrutinizing the hospital charts. In patients with multiple complications, the most severe of these was registered according to the Clavien–Dindo classification of surgical complications. 15
In order to detect complications and mortality within 30 days after surgery for adenomas, data on all patients in the screening register with one positive FOBT, and a subsequent colonoscopy, were matched to the IPR to find patients with relevant codes of surgical procedures according to the Swedish version of the Nordic Medico-Statistical Committee Classification of Surgical Procedures 16 within six months after the screening colonoscopy. The following procedure codes were included: JFA00 – JFA98 (Local operations on intestine), JFB00 – JFB 97 (Partial excision of intestine), JFC00 – JFC 51 (Anastomosis of intestine), JFF00 – JFF97 (Exteriorization of intestine and creation of intestinal stomas), JFH 00 – JFH 96 (Total colectomy), JFW96 – JFW 98 (Other operations on intestine), JGA 00 – JGA98 (Proctotomy and local operations on rectum), JGB 00 – JGB97 (Excision of rectum), JGW00 – JGW98 (Other operations on rectum), and JHA00 – JHA30 (Incision, biopsy and excision of lesion of anus and perianal tissue). The limit of six months was selected to capture surgical procedures which may not have been performed promptly due to the lack of malignant diagnosis. Patients meeting these criteria, but who were also found in SCRCR, were not included in the adenoma group. Hospital charts of the remaining patients were reviewed for mortality and complications within 30 days after adenoma surgery. Complications were registered according to the Clavien–Dindo classification of surgical complications. All adverse events deviating from a normal postoperative course were registered, and in patients with multiple complications the most severe one was registered.
Data on all individuals in the screening register with one positive FOBT within the screening programme with a subsequent colonoscopy between 1 January 2008 and 30 June 2012 were matched to the SCRCR to identify patients with a histopathologically verified screening detected CRC. Among these patients, hospital charts of all those undergoing resectional surgery were reviewed for assessment of complications and mortality within 30 days after surgery for CRC.
The study was approved by the Regional Ethics Committee in Stockholm.
Statistical analysis
Fisher’s exact test was used to determine significant differences in complications according to tumour stage in patients undergoing surgery for CRC, and to determine differences in complications between prevalent and incident screening rounds.
Results
Of 3522 screened individuals with a positive FOBT result, 2984 underwent colonoscopy, of whom 37 and 155 patients subsequently underwent surgery for screening detected adenomas and CRC, respectively. After matching data with the IPR, the Cause of Death register, the SCRCR, and with relevant procedure codes, the medical charts were further reviewed for assessment of complications and mortality within 30 days after the respective procedure. An overview of the complications after colonoscopy and surgery for screening detected adenomas and CRC is presented in Figure 1 Complications after colonoscopy and surgery for screening-detected adenomas and CRC. *Rate of post-polypectomy bleeding and perforation, respectively.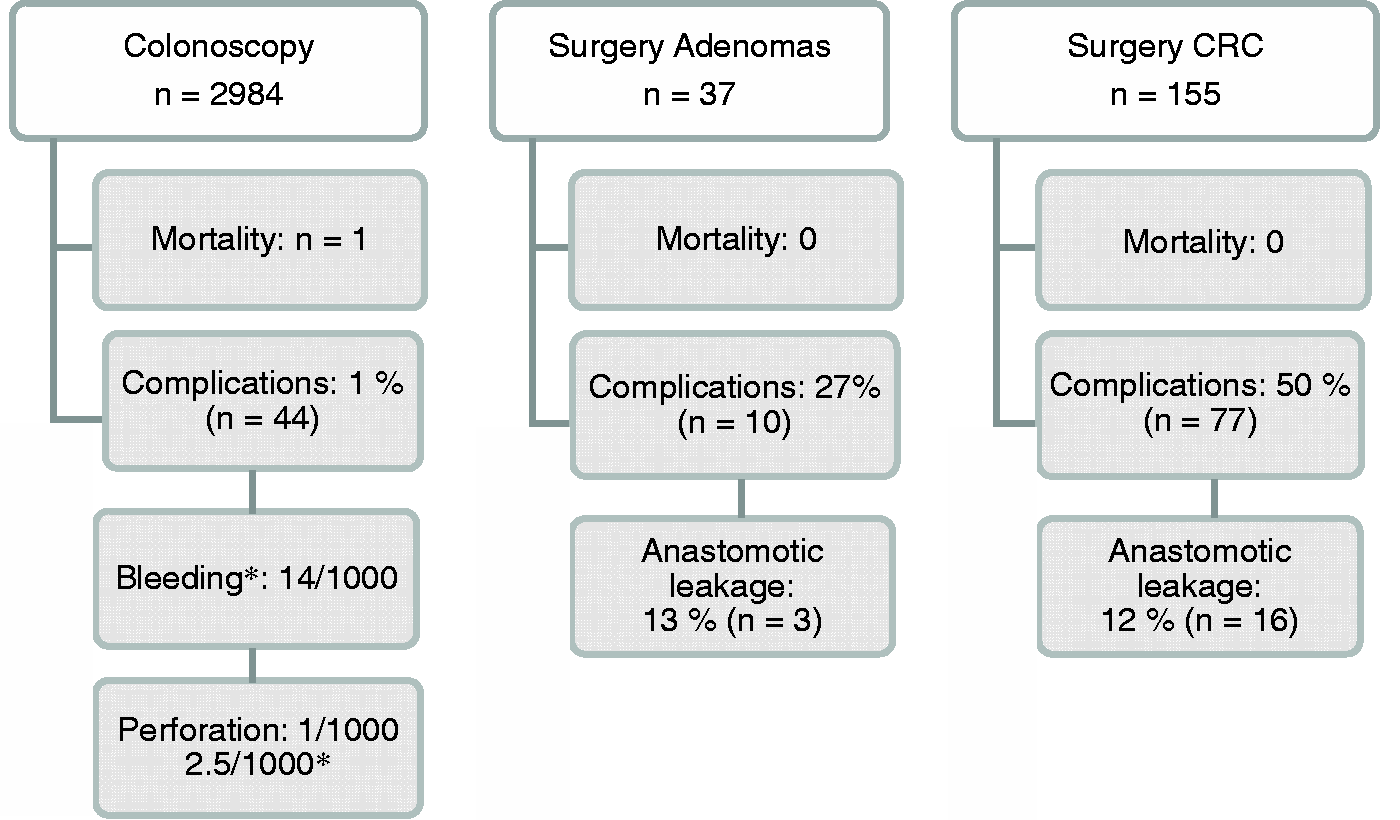
Complications and mortality after screening colonoscopy
The matching procedure identified 105 patients attending in-patient care within 30 days after screening-colonoscopy, 16 of whom were considered to have non-colonoscopy related causes (e.g. visits to a plastic surgeon, hand surgeon, psychiatrist, ophthalmologist, etc.) and were excluded from further analysis. A further 40 patients had no colonoscopy related complications, but were identified as they were subject to surgery for screening detected cancer, surgery for non-screening related causes, and re-colonoscopy because of incomplete bowel laxation at the first colonoscopy, or other causes. Data were missing for four patients.
Complications (
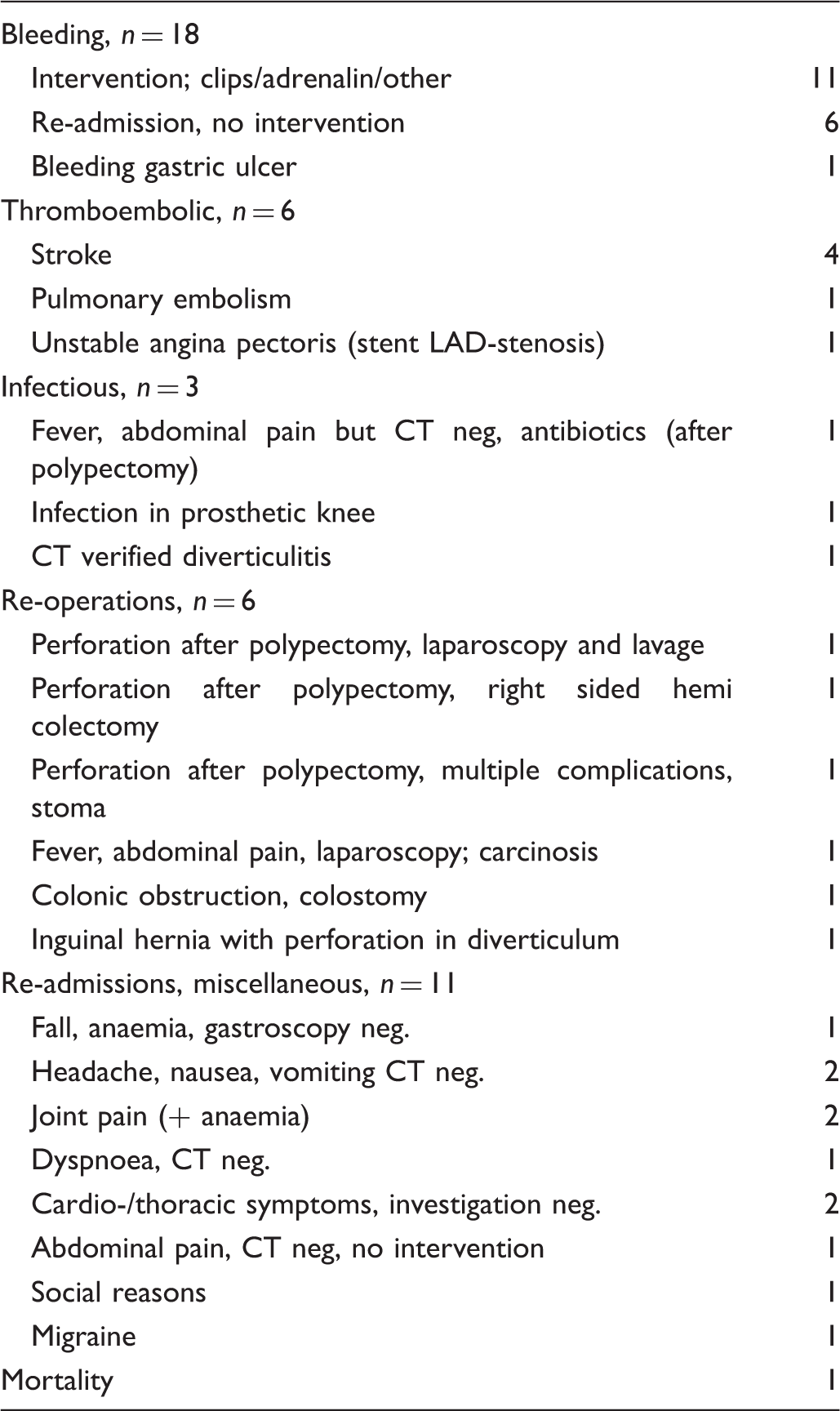
Additional morbidity following screening colonoscopy is presented in Table 1, including four patients with stroke. Two of these patients were on anticoagulants, and one had temporarily omitted anticoagulation therapy prior to colonoscopy. One patient died within 30 days after colonoscopy of non-colonoscopy related causes. This patient was re-admitted because of jaundice and a GI-bleeding caused by an esophagitis and further investigations diagnosed carcinomatosis with multiple hepatic metastases from a neuroendocrine cancer.
Complications and mortality after surgery for adenomas
Fifty-six patients had surgical procedures registered within six months of the screening colonoscopy, but 14 patients only had elective colonoscopies. Five patients underwent surgical procedures unrelated to adenomas. Of these, three were operated on because of tumours in the distal ileum, one for diverticulosis, and one for a perianal abscess. The remaining 37 patients underwent surgery for adenomas motivated by findings at the screening colonoscopy.
Complications (

Note: No deaths occurred.
Complications and mortality after surgery for CRC
Complications (
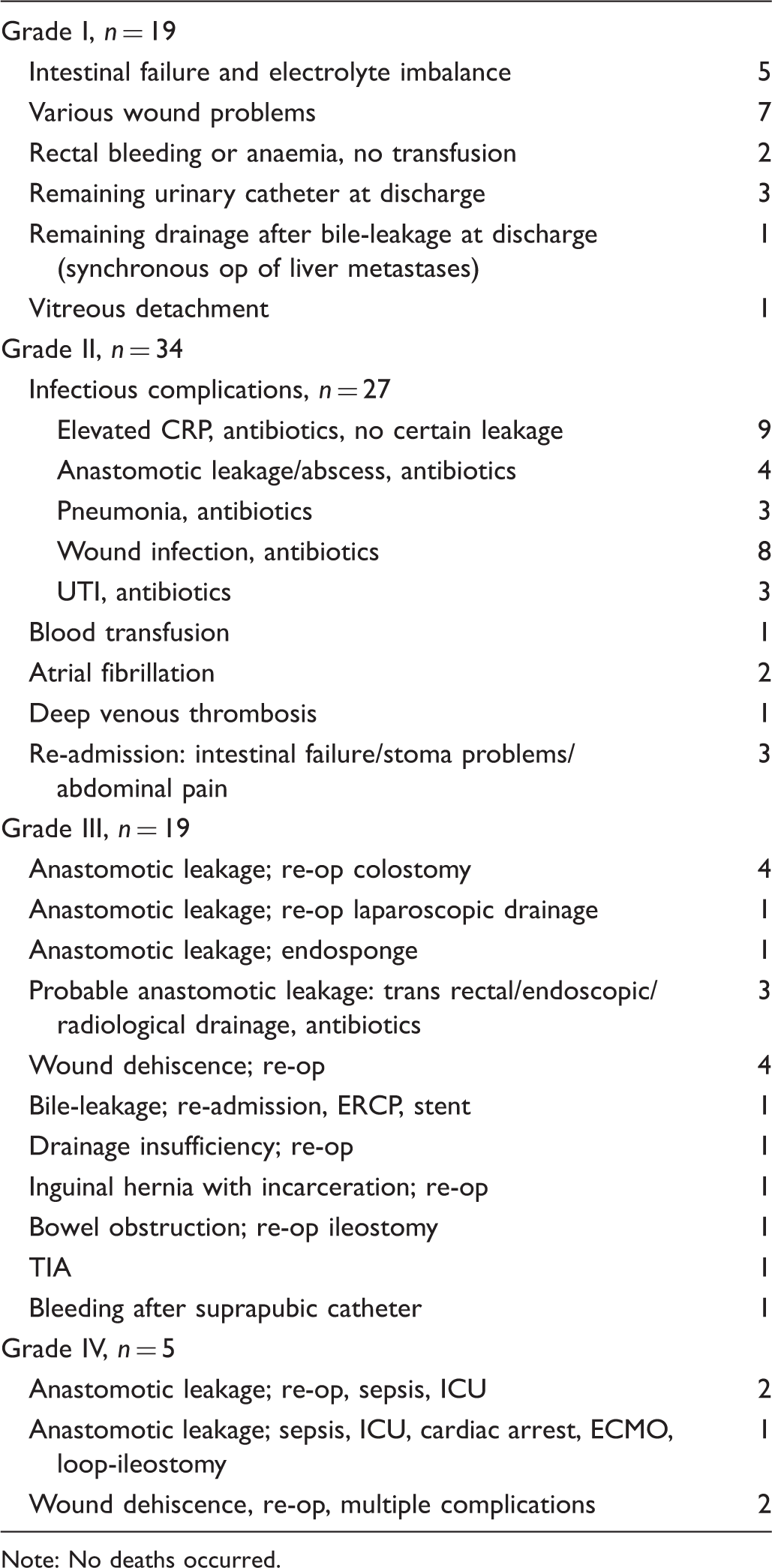
Note: No deaths occurred.
Discussion
The present study assessed the collected complication rates following colonoscopy work-up and subsequent surgery in a population based CRC screening programme with FOBT. The complication rates were 1% after colonoscopy, 27% following surgery for pre-malignant lesions, and 50% following surgery for CRC. Although significant, these rates are considered to be within reasonable limits compared with the literature. However, the rate of anastomotic leakage was relatively high in both the adenoma- and CRC-surgery group.
The detected rates of post-polypectomy bleeding, and the risk of perforation in colonoscopies, with and without polypectomy, were found to be in concordance with the quality indicator guidelines for endoscopic procedures according to the American Society for Gastrointestinal Endoscopy. 17 There was one death within 30 days after colonoscopy, but review of the medical charts found no obvious correlation to the index endoscopy itself. The incidence of stroke following colonoscopy was 1.3/1000 in this screening series. Ko et al. 10 studied almost 19,000 patients (age ≥ 40) and reported a 30-day post colonoscopy incidence of 0.33/1000 in stroke or transient ischaemic attack, and an expected adjusted rate of stroke 0.48 per 1000 over 30 days. The present study is smaller and includes older patients, which makes direct comparison difficult, but data highlight that a risk of severe morbidity appears to be associated with the screening colonoscopy itself. Apart from post-polypectomy bleeding and perforation, it is, however, difficult to establish a definitive relationship between the colonoscopy and any subsequent morbidity although it may appear plausible.
The total complication rate after resection surgery for adenomas was less than that after surgery for CRC, but the rate of anastomotic leakage was 13%, which was higher than expected. According to Swedish national quality data (2009–2013), anastomotic leakage rates after CRC were 8% and 4%, and overall postoperative complication rates (Clavien–Dindo ≥ grade 2) were around 40% and 30% after surgery for rectal and colon cancer, respectively.13,14 In benign surgery, the rate could be expected to be in the same range, or lower. In the present study, anastomotic leakage rates were equivalent in the adenoma and cancer surgery groups. However, the number of patients with anastomotic leakage after adenoma surgery was small, and a larger study group would be needed before drawing firm conclusions on prevalence or underlying causes of anastomotic leakage in this group of patients. A high adenoma detection rate (ADR) is considered a quality measure of colonoscopy, and has been shown to reduce the rate of interval cancers. 18 However, a high ADR will generate complications imposed by the endoscopic and surgical efforts taken to remove these adenomas, which have an unknown natural history if left without intervention. Removal of adenomas without obvious risk of malignancy could increase the morbidity imposed by screening, and hence the risk of overtreatment within screening for CRC should be considered.
The overall complication rate was 50% after CRC surgery, and 37% if Grade I was excluded. Rates and assessment of complications vary in the literature, but in rectal cancer surgery total complication rates of 50% have been reported.19,20 Previous studies have demonstrated an anastomotic leakage frequency of 7–12% for the rectum and 3% in the colon.20,21 The frequency of anastomotic leakage for both colon and rectum in the present study was 12%, without correlation between serious complications and tumour stage. The validity of data in the present study (i.e. operation procedure and diagnosis codes) relies on the reporting accuracy to the IPR, which is high due to economic reimbursement systems, and one of the strengths of the study is that all complications were also assessed through in-detail review of medical charts, generating robust data. The definition of anastomotic leakage was very broad, including all events with any suspicion of this condition.
The benefit of screening for colorectal cancer has long been proven, however, screening investigations (i.e. colonoscopy) as well as endoscopical and surgical treatment of screening detected disease will generate morbidity, and even mortality. Standards for screening colonoscopy are well established, but the chain of events after a positive screening test does not end with colonoscopy. Therefore, monitoring and assessment of morbidity and mortality after surgery for screening detected adenomas and cancer is of equal importance, to assure high-quality medical service through all stages of screening investigations and treatment. In our study, the total complication rate after colonoscopy and surgery for adenomas and cancer was 4%. The complication rates after colonoscopy and surgery were largely in concordance with findings from previous studies. Accordingly, these results confirm that the total complication rate after investigations and treatment of screening detected disease is acceptable, and mortality is low.
Conclusion
This study addresses the total morbidity and mortality after colonoscopy and surgery for adenomas and cancer imposed by CRC screening. Overall complications rates were acceptable, and mortality low. However, the rate of anastomotic leakage after surgery for both adenomas and CRC was higher than expected. The risk of complications associated with investigations and treatment of screening detected disease should be considered, monitored, and continuously evaluated.
Footnotes
Acknowledgements
Contributing with extraction of register data: Robert Johansson, MSc, Regional Cancer Centre North, Department of Radiation Science, Oncology, Umeå University, Sweden. Sini Kilpeläinen, Statistician, Regional Cancer Centre Stockholm Gotland, Sweden. Tongplaew Singnomklao, Data manager, Swedish Colorectal Cancer Registry, Regional Cancer Centre Stockholm Gotland, Sweden.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Financial support was provided through the Regional Agreement on Medical Training and Clinical Research (ALF) between the Stockholm Community Council and Karolinska Institutet, and also by the Swedish Cancer Society and the Stockholm Cancer Society.
