Abstract
Objectives
Type 2 diabetes has been identified as a risk factor for colorectal cancer, but little is known about whether it influences participation in colorectal cancer screening programmes. This study tested the extent to which Type 2 diabetes is negatively associated with colorectal cancer screening uptake.
Methods
We analysed individual data of screening eligible men and women aged 60–75 without cancer diagnosis from wave 6 of the English Longitudinal Study of Ageing (collected 2012–2013), to investigate whether Type 2 Diabetes influences colorectal cancer screening behaviour independently of demographic characteristics, body mass index, socio-economic status and other chronic diseases.
Results
Individuals who reported to have Type 2 diabetes or had glycated haemoglobin (HbA1c) levels of 48 mmol/mol or higher were less likely to have ever completed a screening test (faecal occult blood test; 62.8% vs. 75.8%, p < 0.01) or to be up-to-date with their biennial screening invitation (60.2% vs. 72.0%, p < 0.05). The negative associations of Type 2 diabetes on colorectal cancer screening were found both in unadjusted and adjusted regression models.
Conclusions
Future qualitative and quantitative research should identify reasons for this discrepancy, to inform interventions to increase screening uptake in this high-risk population.
Keywords
Introduction
Individuals with type 2 diabetes (T2D) are at higher risk of developing cancer, including kidney, non-small cell lung, pancreas, early gastric, breast, ovarian and colorectal cancer (CRC),1–7 potentially due to insulin resistance and compensatory hyperinsulinemia. 8 As the number of T2D patients worldwide has consistently increased 9 and CRC is among the most frequently diagnosed cancers, 10 it is important to investigate the role of T2D in CRC-preventive behaviours, such as CRC screening uptake. Because individuals with T2D are at higher CRC risk, regular participation in CRC screening programmes is particularly recommended. 11 Studies have shown that women with T2D are less likely to undergo cervical12–15 and breast cancer screening,13,14,16 although support for this has not been completely consistent. 12
Findings for CRC screening are equivocal. Two US studies looking at samples of women aged 40 or older demonstrated that those with T2D were more likely to be screened for CRC than those without.12,17 Similarly, a recent US study looking at men and women aged 50 or older showed that those with T2D were more likely to be up-to-date with the recommended CRC screening. 18 In contrast, a US study of women aged 67 or older found a negative association. 15 All these studies were based in the US and looked at opportunistic rather than organized CRC screening programmes. The only non-US study is an English prospective survey of 55-year olds intending to have flexible sigmoidoscopy screening. 19 which found an independent negative association between reporting T2D and screening attendance, however, the study was limited to people who had already expressed an intention to attend. Currently, there is a lack of studies examining the association for men and women in the context of an organized population-based CRC screening programme.
To address this limitation, the current study used data from the English Longitudinal Study of Ageing (ELSA; a nationally representative study of community-dwelling English adults aged ≥50 that started in 2002–2003 (wave 1) 20 ) to examine the role of T2D in CRC screening in a screening-eligible sample of adults in England aged 60–75. In England, the National Health Service (NHS) invites people aged 60–75 to complete a home-based stool test (faecal occult blood test, FOBt) every two years. We tested the hypothesis that people who reported having been diagnosed with diabetes or who had elevated HbA1c levels at wave 6 were less likely to have ever been screened, and less likely to be up to date with CRC screening.
Methods
We used individual data of screening-eligible men and women aged 60–75 without cancer diagnosis from ELSA’s wave 6 questionnaire and nurse visit, which were collected in 2012 and 2013. All participants gave informed consent at each wave of data collection. ELSA was approved by the London Multicentre Research Ethics Committee (MREC/01/2/91), and informed consent was obtained from all participants. All study methods were performed in accordance with the Helsinki Declaration and good clinical and scientific practice.
Ever screening was measured from the individual questionnaire in wave 6 of ELSA ‘Have you ever completed the NHS bowel cancer screening test using the home test kit?’ with response options being ‘yes’ or ‘no’. Individuals who reported having completed the screening test were then asked to indicate the date of their most recent bowel cancer screening test. The response to this follow-up question was used to determine whether individuals followed the recommended biennial screening interval. Of the 5160 participants in the screening-eligible age range of 60–75 without cancer diagnosis, 4925 (95.4%) responded to the date of the screening question.
T2D was measured using the question ‘Has a doctor ever told you that you have diabetes or high blood sugar?’ from the individual wave 6 questionnaire, and from the glycated haemoglobin (HbA1c) level measured in the blood sample taken by the nurse during the wave 6 nurse visit. HbA1c values ranging from 15 to 137 mmol/mol were dichotomized into two categories: those below 48 mmol/mol and those with values of 48 mmol/mol or higher. 21 The question about having a diabetes diagnosis was answered by 4882 participants in the eligible age range, while blood samples were obtained from 3270 participants. For the purpose of this study, we only considered respondents who answered the self-reported question and had a blood sample result recorded. Individuals were classified as having diabetes if they either reported a diabetes diagnosis or had an HbA1c value of 48 mmol/mol or higher.
Objective body mass index (BMI) measurements (height in m2/weight in kg) were taken from the wave 6 nurse visit and categorized into normal weight (BMI<25), overweight (BMI 25–29.9), class I obesity (BMI 30–34.9), class II obesity (BMI 35–39.9) and class III obesity (BMI ≥ 40). 22 Assessments of coronary heart diseases such as angina or myocardial infarction, depression, respiratory diseases like asthma and lung disease and stroke were based on the questions from the individual questionnaires ‘What type of emotional, nervous or psychiatric problems do you have?’ and “Has a doctor ever told you that you have any of the conditions on this card?”
We included gender, age, living arrangement, education and household non-pension wealth as socio-demographic covariates from the individual questionnaire. Living arrangement was coded as in binary as either living alone or with somebody. Education was assessed in four categories ranging from 0 = ‘no formal education’ to 3 = ‘university degree or higher’. Non-pension wealth is an indicator of socio-economic status (SES) in older people. 23 Non-pension wealth was measured at the family level, and this is the sum of net primary housing wealth, net physical wealth (other property wealth, business wealth and other physical assets) and net financial wealth. It included saving accounts, Individual Savings accounts, Tax Exempt Special Savings Accounts, premium bonds, national savings, Personal Equity Plans, shares, trusts, bonds, other savings, minus credit card, private and other debt. Quintiles of non-pension wealth (1 = low, 5 = high) were used for the analysis. 24
Characteristics of the sample were described using mean scores (standard deviations) for continuous variables and numbers (percentages) for categorical variables. We used unadjusted and adjusted multivariate logistic regression models to investigate whether T2D was associated with CRC screening behaviour. The adjusted model controlled for demographic characteristics, BMI, SES, chronic heart disease, depression, respiratory problems and stroke. We did not adjust for ethnicity, as the vast majority (97.2%) of the analytic sample reported a White ethnic background. All statistical analyses were conducted with Stata/SE version 15.1 (StataCorp LP, College Station, TX).
Results
The characteristics of the analytic sample of 3270 participants are presented in Table 1. The mean age of the participants was 66.7, and the majority fell into the overweight and obese categories. There were 422 participants (12.9%) classified as having T2D. Most of these reported a diabetes diagnosis and had an elevated HbA1c level of 48 mmol/mol or higher (N = 235; 55.7%). There were 104 (24.6%) of the 422 participants in the diabetic group who did not report a diagnosis, but were classified as having T2D because of their HbA1c level, and 83 (19.7%) who self-reported a diagnosis but had an HbA1c level below 48 mmol/mol. Using the alternative classification of diabetes, 108 were classified as having undiagnosed diabetes, 83 as diabetes patients who control their diabetes well and 235 as diabetes patients who control their diabetes poorly.
Participant characteristics (N = 3270).
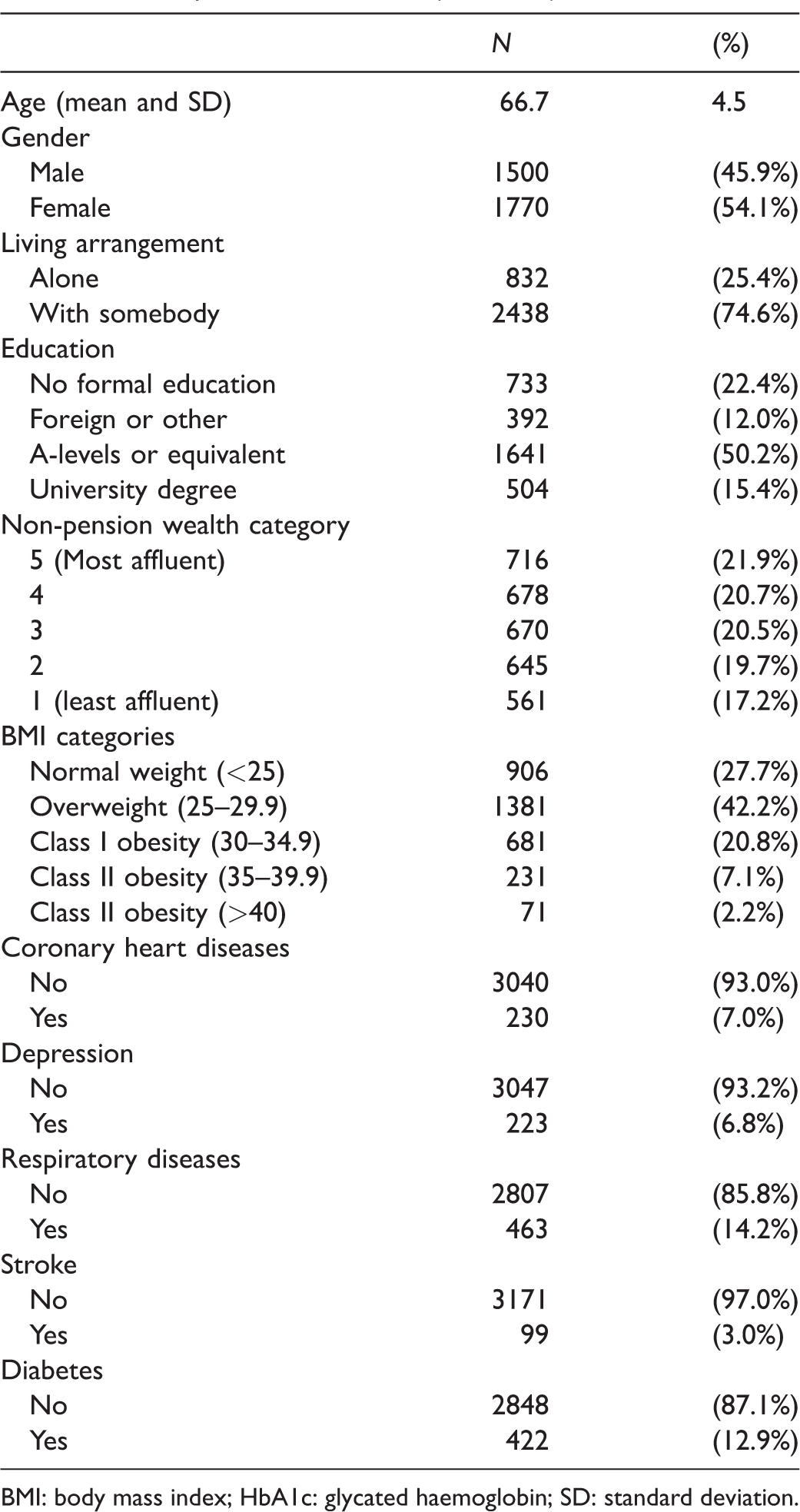
BMI: body mass index; HbA1c: glycated haemoglobin; SD: standard deviation.
Table 2 shows that individuals were less likely to have ever undergone CRC screening if they had T2D in both the unadjusted and the adjusted regression models (62.8% vs. 75.8%, Odds Ratio (OR) 0.54, 95% confidence interval (CI) 0.43–0.67, p < 0.01 and adjusted Odds Ratio (aOR) 0.70, 95% CI 0.56–0.89, p < 0.01. Table 3 shows full results of the univariate and multivariate analysis. Class III obesity and increasing age were negatively associated with self-reported ever uptakes. Reporting a coronary heart disease or respiratory disease was also negatively associated with CRC screening in the unadjusted, but not the adjusted regression. In contrast, women and participants who were cohabiting, respondents with formal education, and those from a higher income group were more likely to have done the stool test.
Screening uptake according to demographics (N = 3270).
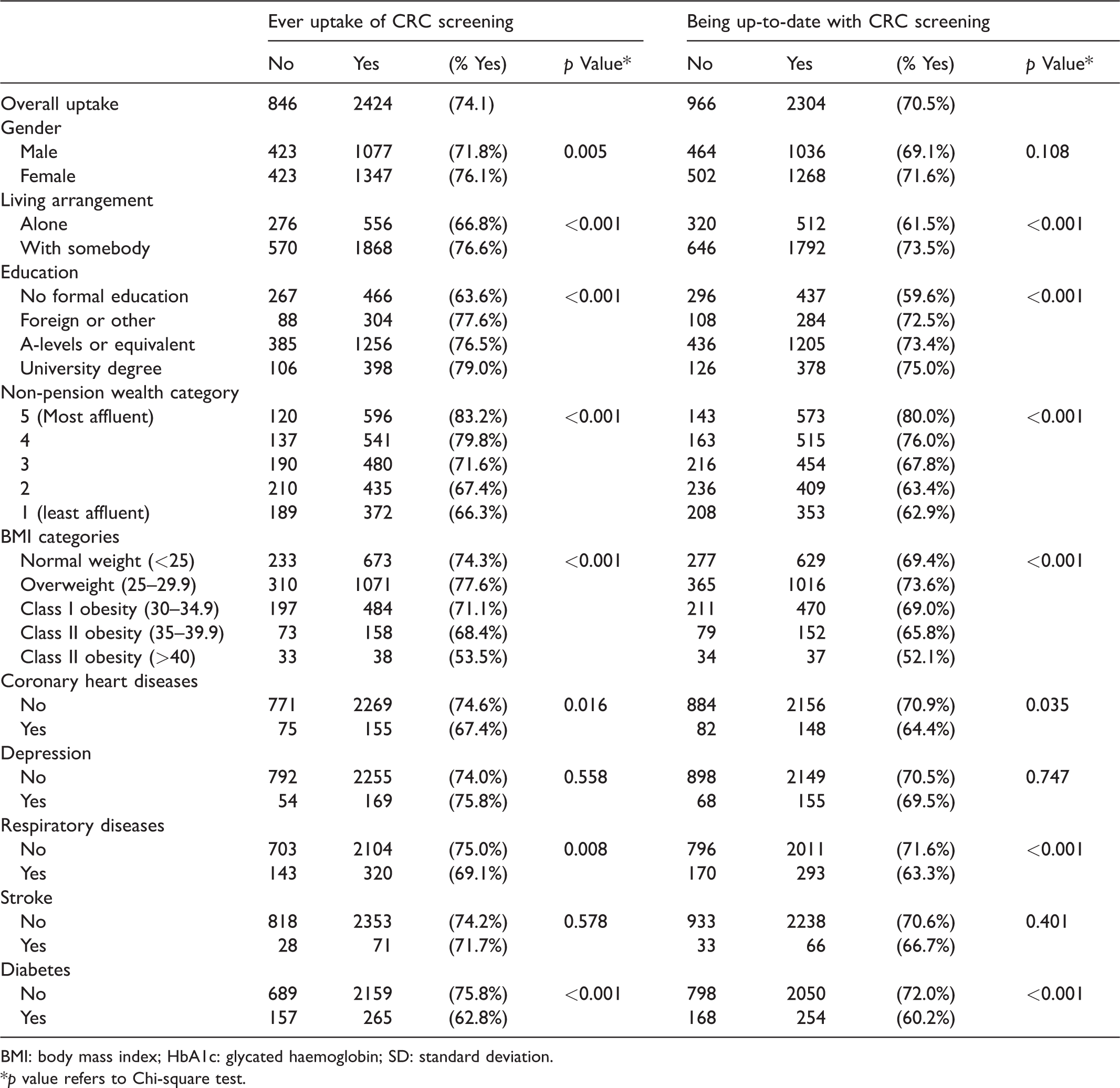
BMI: body mass index; HbA1c: glycated haemoglobin; SD: standard deviation.
p value refers to Chi-square test.
Predictors of CRC screening.
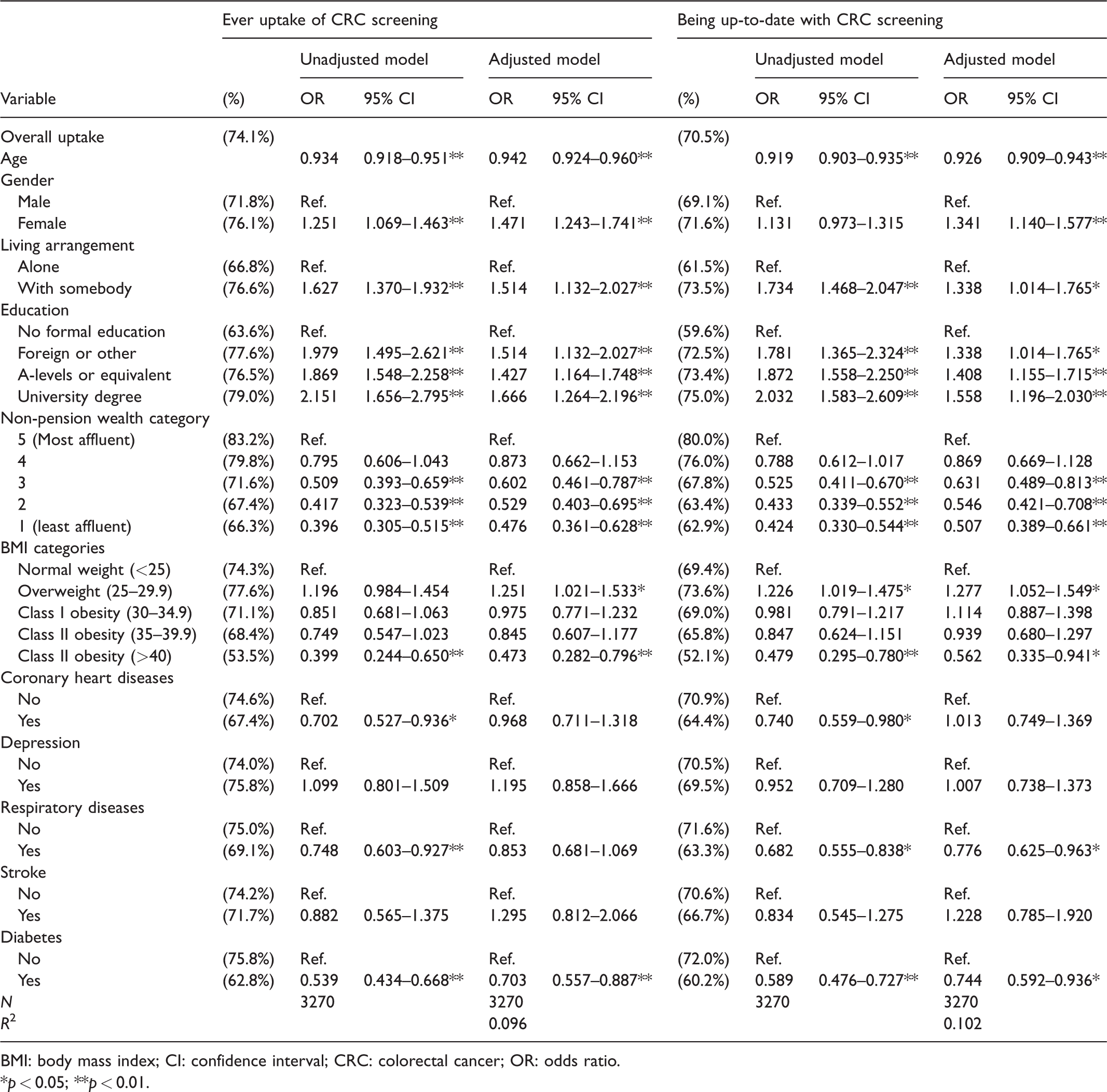
BMI: body mass index; CI: confidence interval; CRC: colorectal cancer; OR: odds ratio.
p < 0.05; **p < 0.01.
T2D was also negatively associated with being up-to-date with the recommended CRC screening interval in both the unadjusted and adjusted regression models (60.2% vs. 72.0%, OR 0.59, 95% CI 0.48–0.73, p < 0.01 and aOR 0.74, 95% CI 0.59–0.94, p < 0.05). Similar to the results for ever uptake, age and Class III obesity were negatively associated with being up-to-date with CRC screening. While diagnosis of chronic heart diseases and respiratory diseases was, again, negatively associated with CRC screening in the unadjusted regressions, only the association for respiratory diseases remained statistically significant in the fully adjusted model. In contrast, relative affluence, being female, and having A-levels or a university degree were positively associated with screening.
Discussion
In this study diabetes was negatively associated with CRC screening behaviour, independently of whether it was defined by self-reported diabetes alone or in combination with HbA1c. Individuals with diabetes were less likely to have ever undertaken CRC screening or to be up-to-date with their biennial CRC screening invitation. These findings are particularly concerning as people with diabetes are at a moderately increased risk of developing CRC. 24
Our results are in line with a recent US study looking at individuals with diabetes, which found that those who had an HbA1c level of 48 mmol/mol or higher were less likely to have been screened for CRC. 25 The present study adds to the literature by looking at the link between diabetes and CRC screening behaviour in an organized population-based CRC programme with routine call and recall. Previous research has focused mainly on the US.12,15,17,18 One previous study of men and women in the UK included only those who had already expressed an intention to attend flexible sigmoidoscopy screening, limiting the generalizability of the findings to the population at large, 19 but the results of this study are in keeping with our analysis, whereby those with diabetes were less likely to report being up to date with the recommended biennial screenings. Our study findings are also consistent with studies showing lower cervical12–15 and breast cancer screening uptake13,14,16 in people with diabetes. The association we found was independent of several potential confounders, particularly the level of obesity, SES and other comorbidities, which indicates that there is a need to explore the diabetes specific barriers to screening.
Diabetes treatment guidelines highlight the importance of engaging in positive health behaviours, 26 and attending CRC and other population-based cancer screenings are a part of engaging in proactive behaviours to benefit long-term health. Awareness of a chronic condition such as T2D could be considered a trigger for positive lifestyle change, 27 however, the findings of this study support research suggesting that lower levels of engagement with positive health behaviour may be habitual and difficult to modify in this population. 28 There could be a common underlying mechanism in how individuals engage in an unhealthy lifestyle, leading to increased risk of diabetes, cancer or other chronic conditions. 29 These mechanisms could extend to the perceived susceptibility of subsequent associated risks and cues to action (e.g. CRC screening).30,31 Low SES could exert a strong influence on these complex mediating pathways, via low literacy, healthcare access, healthy food options etc. and our results for education and wealth seem to support this mechanism. A recent study, using the ELSA cohort, demonstrated that limited positive behaviour change appears to occur following the diagnosis of T2D, with no changes in physical activity, fruit and vegetable intake or alcohol consumption detected. 32
A recent conceptual framework describing the potential influence of comorbidities on the timely diagnosis of cancer described mechanisms through which comorbid conditions could either facilitate help-seeking and screening, or be associated with delays. 33 In the case of cancer screening, individuals with diabetes might have competing demands, as the management of diabetes can take priority (for both patients and healthcare providers), and this might interfere with participating in screening.33,34 The competing demands mechanism has been reported to interfere with help-seeking for symptomatic patients with serious comorbidities. 35 It can be even more relevant in the context of screening, where individuals are not experiencing cancer symptoms, and instead have other more urgent healthcare needs.
Our study had some limitations. First, we used self-reported measures for CRC screening participation and comorbidities, which may be subject to recall bias. However, a recent study comparing self-reported CRC screening behaviour with participation recorded by the programme found that more than 90% of the study participants were able to accurately report whether they had ever completed a FOBt. 36 Secondly, this cross-sectional study does not consider the date of diabetes diagnosis, making the direction of the association between diabetes and CRC screening unclear.
Conclusion
This study found evidence that people with T2D are less likely to undergo CRC screening in England. Further research is required to understand how to motivate and facilitate CRC screening in this and other groups at moderately increased CRC risk.
Footnotes
Authors’ note
Sandro T Stoffel is now associated with Business School, University of Aberdeen, Aberdeen, UK.
Authors' contributions
STS, CvW, DC, RAH and PD developed the study concept and design. STS and CvW performed the data analysis and interpretation. STS drafted the manuscript, and all authors provided critical revisions. All authors approved the final version of the manuscript for submission.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article. However, Stacy Bailey has served as a consultant to Merck, Sharp & Dohme Corp, Northwestern University/Gordon and Betty Moore Foundation, Pfizer, Inc and Luto LLC for work unrelated to this manuscript. She has also received funding support via her institution from Merck, Sharp & Dohme Corp and Eli Lilly and Company. Michael Wolf has served as a consultant to Merck, Sharp & Dohme Corp, Abbvie, Vivus, Inc., Luto LLC, Pfizer, Inc, Anheuser Busch Imbev, DenverHealth, and Teva Pharmaceuticals for work unrelated to this manuscript. He also has received funding support via his institution from Merck, Sharp and Dohme Corp, Eli Lilly and Company, Abbvie, and UnitedHealthcare.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Financial support for this study was provided entirely by a Cancer Research UK (C1418/A14134). The funding agreement ensured the authors’ independence in designing the study, interpreting the data, writing and publishing the report. RJB is supported by Yorkshire Cancer Research University Academic Funding.
