Abstract
Objectives
Four randomized controlled trials have demonstrated the efficacy of screening using a guaiac faecal occult blood test (gFOBT) on colorectal cancer (CRC) mortality. Whether their results are transposable to the real world is unknown. This study aimed to assess the determinants of participation in the first four rounds of the CRC screening programme using a gFOBT implemented since 2003 in the Haut-Rhin (Alsace) part of the French national programme.
Methods
We performed a population-based open cohort study of all residents aged 50–74, around 200,000 people. They were invited by mail to participate every other year. The gFOBT kits (Hemoccult II) were first provided by general practitioners, and then directly mailed to persons who failed to comply.
Results
The uptake decreased significantly across all rounds, from 54.3% to 47.1% (p < 0.0001), mainly in people younger than 60. The proportion of people screened by general practitioners increased significantly from 77.0% in the first round to 84.2% in the fourth (p < 0.01). Overall, 61.3% of the invited population participated at least once, and 14.3% had completed all the four tests. The colonoscopy uptake was around 91%, among the highest ever reported.
Conclusions
Despite the involvement of general practitioners, the uptake and adherence to repeat testing are modest and deteriorate with time, so that the reduction in CRC mortality in reality will be significantly lower than that in the trials. The benefit-risk balance of the French programme is, at present, less favourable than that shown in the trials.
Keywords
Introduction
Colorectal cancer (CRC) is the second leading cause of cancer death in Europe and the United States.1,2 Four randomized controlled trials (RCTs) have demonstrated the efficacy of screening using a guaiac-based faecal occult blood test (gFOBT) on CRC mortality,3–6 and many countries have launched gFOBT-based CRC screening programmes. The French programme started in 2002 in a few pilot areas, and progressed into a nationwide programme by 2009.7,8 The benefit of CRC screening, however, has been modest: an estimated 1.2 to 1.6 deaths were avoided per 1000 persons invited in the three trials with biennial non-rehydrated gFOBT, without significant impact on CRC incidence.4,5,9 Moreover, these trials were conducted by highly motivated research teams. Whether these results are transposable to the real world is unknown. It has been demonstrated that short-term outcomes of the first rounds of the European trials could be reproduced in the real world through organized population-based programmes,8,10 but it is unknown what will occur in the following rounds, especially with regard to uptake. CRC screening with gFOBT has several drawbacks, including the requirement for frequent testing, which may limit compliance and thereby effectiveness. Uptake remained roughly stable round after round in the French and Swedish trials.6,9 Real-world data on programme adherence and performance over time of FOBT screening are scarce, available only from the British pilot studies,11–13 in which uptake decreased in the second round and increased in the third.11,12 No data have yet been published on participation in four rounds of FOBT-based CRC screening in a large population-based open cohort.
The aim of this paper is to report the determinants of participation in the four first rounds of the organized gFOBT CRC screening programme conducted in the Haut-Rhin, as part of the French national programme.
Methods
We performed a population-based open cohort study of the determinants of participation in the first four rounds of the biennial gFOBT CRC screening programme implemented since 2003 in the Haut-Rhin, an administrative area situated in Alsace, eastern France, with a population of around 750,000. Together, the four rounds lasted eight years, starting between September 2003 and September 2004, depending on the district, and ending between September 2011 and September 2012, respectively. In accordance with French law, this study was exempt from ethics committee review as it was an evaluation of an existing programme.
Screening programme organization
The programme has been previously described in detail. 8 Participants were residents of Haut-Rhin, aged 50--74, beneficiaries of the French statutory health insurance schemes, that is, approximately 99.6% of the age-range population. They were identified using health insurance schemes database files (updated twice a year), and invited by mail every other year to participate. A first letter invited them to visit their general practitioner (GP) for CRC screening. Three additional letters were mailed to all those who had not complied. The second mailed reminder letter included the gFOBT kit. People with serious illness, those who had undergone recent CRC screening or had high CRC risk (symptoms, personal or family history of colorectal neoplasia, inflammatory bowel disease) were excluded. The exclusions were identified by the GPs, the gastroenterologists, or the invited individuals themselves. Depending on the reason, the duration of exclusion was two or five years, or indefinite. The gFOBT (Hemoccult II, Beckman Coulter, Brea, CA, USA) was used without dietary restriction and processed without rehydration. Faecal material was assessed from two samples from each of three consecutive stools. The test was defined as positive if at least one window was positive. People with a positive gFOBT were referred for colonoscopy. The test result was defined as inadequate in case of inadequate sample, identification problems, or excessive delay (>14 days) before analysis. People with an inadequate result were requested to complete another test.
Colonoscopies
Colonoscopies were performed by certified community endoscopists practicing in the area. As usual in France, all colonoscopies were performed by gastroenterologists and most (95%) participants were under sedation or general anaesthesia provided by an anaesthetist.
Main outcome measures
Main outcome measures and indicators, their definitions and calculation methods.

CRC: colorectal cancer; gFOBT: guaiac faecal occult blood test.
Statistical methods
All crude rates were calculated with a 95% confidence interval (CI). Differences in uptake according to round, age, and sex were tested by Chi2 tests. A two-tailed p < 0.05 was considered statistically significant.
Results
Uptake
Population, exclusions and uptake by round.
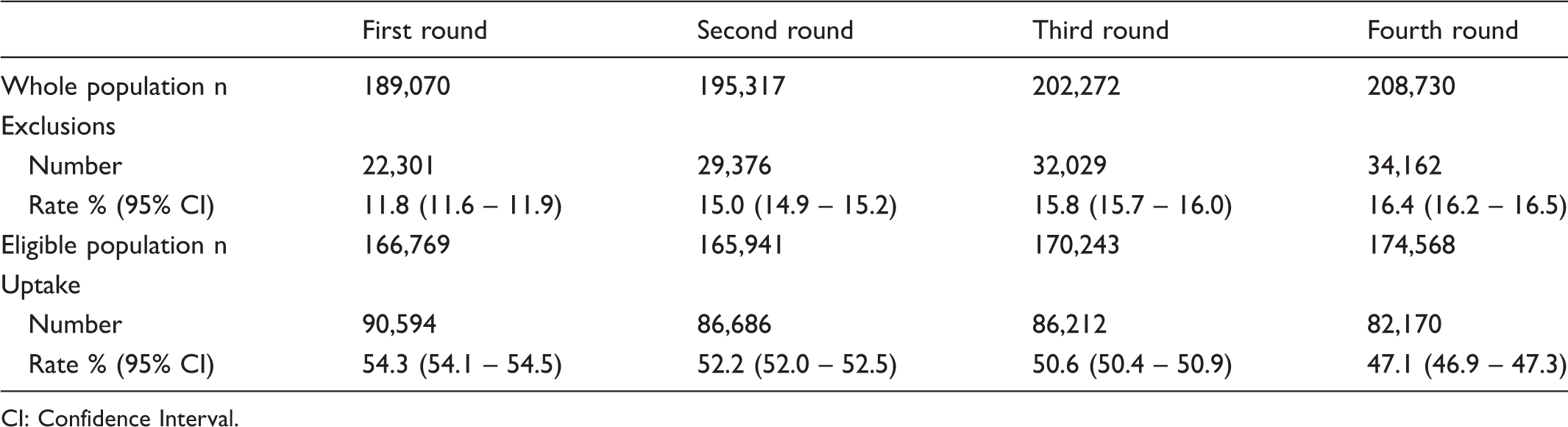
CI: Confidence Interval.
Uptake by round according to sex, age and screening history.
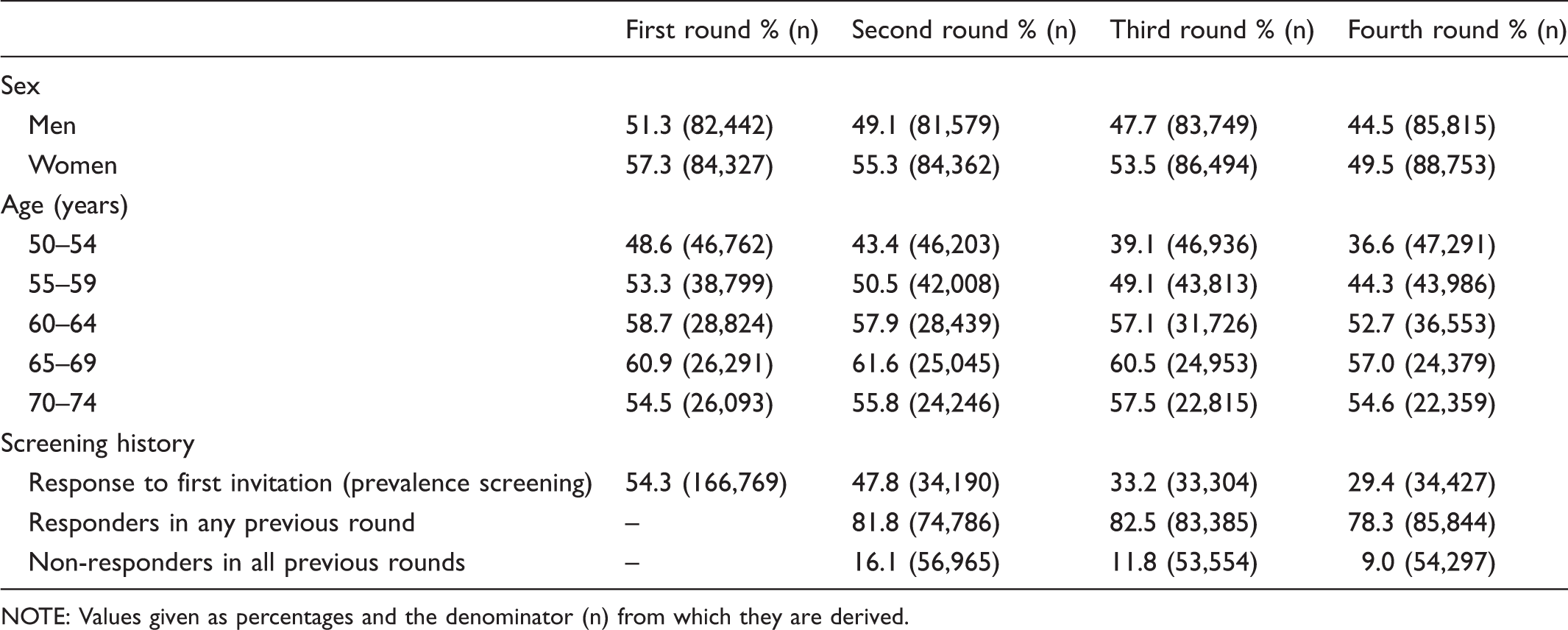
NOTE: Values given as percentages and the denominator (n) from which they are derived.
GP involvement
Depending on the round, 636 to 662 GPs (98.5 to 99.2% of all GPs) participated, each providing a median of 97 to 104 gFOBTs by round (range 1 to 525). The proportion of people screened by GPs increased significantly from 77.0% in the first round to 84.2% in the fourth (p < 0.01), while that of people screened by direct mailing of a gFOBT kit decreased significantly from 15.6% in the first round to 12.1% in the fourth (p < 0.01). A minority had their gFOBT provided during a health check up or by occupational medicine physicians. The uptake by source providing the gFOBT was not different between men and women, but varied with age. The proportion of people screened by GPs increased with increasing age (from 78.0% in age group 50–54 to 89.6% in age group 70–74 in the fourth round). The proportion of uptake following the first two invitation letters increased from 77.9% to 85.8% between the first and fourth rounds. Depending on the round, the gFOBT kit was directly mailed to a little more than the half of the eligible population (85,000--95,000 persons) and the rate of tests that were actually completed decreased significantly from 15.5% to 7.6% from the first to the fourth round (p < 0.001).
gFOBT results
Positivity rate by round according to sex, age and screening history.
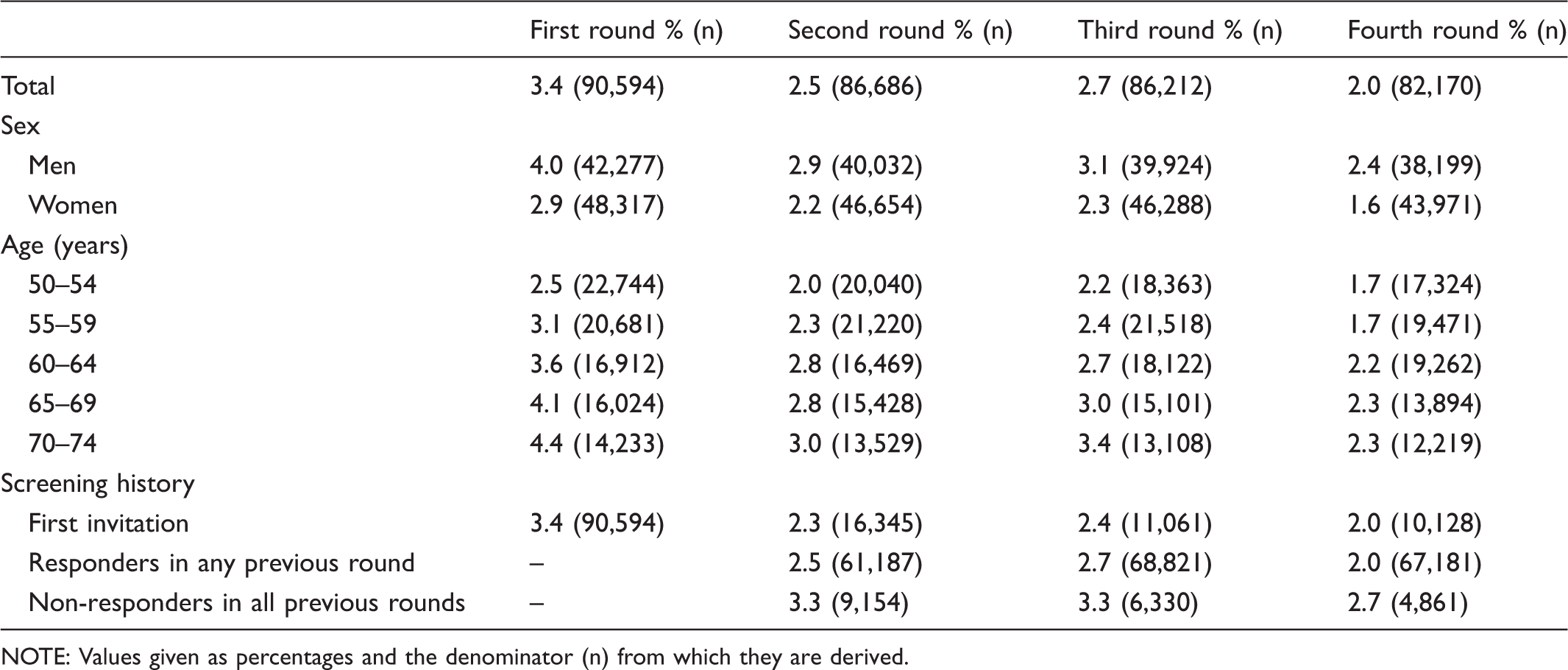
NOTE: Values given as percentages and the denominator (n) from which they are derived.
Colonoscopy
The uptake of colonoscopy did not differ between rounds: 89.1%, 91.5%, 90.8%, 90.8% respectively (at six months, it was 80.7%, 84.8%, 84.4%, and 84.4%, respectively). Uptake did not differ by sex and age. It was significantly higher in those who had received their gFOBT from GPs than in those who received the kit by direct mailing (92.2% vs 87.9%, p < 0.0001). After four rounds, 8,417 (5.7%) persons had a colonoscopy.
Discussion
Main findings
The main finding of this real-world study is the significant decrease of uptake round after round. The decrease was age-dependent, almost entirely attributable to people younger than 60 who represented more than half of the eligible population. More than 80% of the uptake was obtained by GPs, but their involvement varied greatly. Some GPs had almost all their eligible patients screened, whereas others had almost none.
Comparison with other studies
The comparison of the results of real-world screening programmes with those of RCTs is compromised by the fact that the first involve open cohorts, whereas the latter have closed cohorts. Uptake in this programme was 54.3% in the first round, at the lower end of rates achieved by the European trials (52.8%--67.2%).4–6,9 It was intermediate when compared with those of other European programmes (17%--71%).11,12,14–17 Uptake decreased over time to 47.1% in the fourth round. A decrease was also observed in the second round in 13 out of 18 French pilot areas, 18 and the whole national uptake decreased slightly, year after year, from 34.3% in 2008--2009 to 31.0% in 2012--2013. 19 The British pilot studies also observed a decrease in uptake in the second round, but an increase in the third round.11,12 By contrast, an increase was observed in the second rounds of Finnish and Spanish pilot programmes, and in the second and third rounds of the Swedish programme.14,15,17 Little is known about adherence to repeat testing, which is a critical component of FOBT-based screening programmes. Adherence to yearly repeated FOBT is low. 20 Adherence to rescreening is higher with longer intervals between tests.3,21 Overall, after four rounds, 61.3% of our invited population participated at least once. This rate is at the lower end of rates achieved by the European trials (59.6%-70.0%).5,6,9 It was 64.8% after three rounds in the Scottish pilot. 22 Compliance with repeated testing was 14.3% in our programme, much lower than the rates observed in the European trials (38.1%--47.2%),4–6,9 and in the three first rounds of the Scottish pilot (39.2%). 22
Uptake following direct mailing of FOBT kits is insufficient in France, estimated at between 26 and 34%, much lower than that observed in more northern European countries.11,12,15,17,23 In contrast with most programmes, GP involvement is the cornerstone of the French programme. The GP role is pivotal, not only to increase participation in gFOBT screening, but also to assess CRC risk, recommend and explain the most appropriate screening test, and ensure adherence with repeat testing for people who have negative gFOBT results, and colonoscopies for those with positive results. Thanks to the strong involvement of most GPs in the Haut-Rhin, our uptake is among the highest achieved in France, estimated at 46.9% in 2012–2013, varying from 7.3% to 50.1% depending on regions. 19 Overall, the involvement of GPs is highly variable, so that the national uptake remains insufficient in France, estimated at 31.0% in 2012–2013. 19 This modest uptake might be partly explained by cultural reasons, as there seems to be a north-south gradient in Europe and in France, participation in gFOBT CRC screening programmes being lowest in southern regions.11,12,14–17,19 This uptake will probably limit the reduction in CRC mortality to only 8% in our country, markedly lower than the 15% obtained in the RCTs.24,25 It could be even less when considering that the Scottish pilot reported a 10% reduction despite a 60.6% participation rate. 13
Our positivity rates (from 3.4 to 2.0%) were higher than the British rates (2.1 to 1.2%).11,12 This is probably the consequence of the difference in positivity criteria between programmes and because CRC incidence in the Haut-Rhin is one of the highest in Europe. 26
Because of GP involvement, our colonoscopy uptake was very high (around 91%), among the highest ever reported in RCTs and real-world programmes.3–6,9,11,12,14–17 GPs were regularly able to persuade patients who had initially refused to undergo colonoscopy.
Strengths and limitations of the study
The real-world population-based setting is the main strength of our study. Other strengths include its large scale and long duration. Moreover, we specified the definitions and calculation methods of all our outcome measures to enable comparison between programmes. Like others, we excluded people with an inadequate gFOBT result from the numerator for the calculation of uptake.11,12,15 Doing this, we did not follow previous recommendations because we felt that people with an inadequate screening result cannot be considered as participants.27,28 The rate of inadequate results can vary greatly between screening tools and programmes, and hamper any comparison of the rates of people actually screened.
The main limitation of our study is that the low-sensitivity gFOBTs we used are now considered obsolete by many experts, and no longer recommended by many guidelines.29–31 However, gFOBTs are less expensive and sensitive than faecal immunochemical tests (FITs), and are still used in many countries as they remain well adapted to environments with low resources and/or colonoscopy capacity. Moreover, as the screening process is the same, our data on longitudinal adherence to gFOBT screening and its determinants can be extrapolated to any FOBT-based programme, including FIT. Another limitation is that we did not report the results of short-term outcomes other than uptake, because they were similar to those of other gFOBT programmes and RCTs.
Future research
Future research should be directed at enhancing uptake and adherence to repeat testing, especially in people younger than 60. The deterioration of uptake with time was observed despite several interventions directed at raising the awareness of the target population and the GPs, including annual awareness campaigns during March, education and training of GPs, financial rewards for GPs, triennial mail sent to the GPs with a feedback on their patients who were overdue for gFOBT screening. GP involvement and recommendation are strong predictors of patient participation in CRC screening.23,32,33 The reason(s) why some GPs had only a minority of their patients screened also deserves further investigation. A possible explanation is that some GPs promote CRC screening with colonoscopy, owing to the low sensitivity of gFOBT screening. Another explanation might be that the French health care system is organized such that gastroenterologists might have a financial interest to promote CRC screening with colonoscopy, rather than with gFOBT. Additional research is needed to identify effective interventions that enhance GP involvement, such as reminder systems, practice visits, audit and feedback, or pay-for-performance initiatives. Several client-directed interventions that proved to be efficient in other settings, such as patient navigator, phone counselling, or involvement of pharmacists, are currently under evaluation in France.
The replacement of gFOBT by a quantitative FIT should help increase uptake.31,34 However, FIT is evolution, not revolution. In other words, an FIT remains an FOBT, and whether the use of an FIT will stop the continuous decline in uptake observed in our country is not established. We lack data about the long-term use of FITs in large population-based real-world programmes. A small Dutch pilot found a significant decrease in participation in the second round, 35 whereas a larger population-based Italian programme found an increase. 36 Compliance with repeated testing was estimated at 38.3% in an Italian pilot involving a small closed cohort and using an FIT performed on a single sample. 37 Whether FIT screening with longer intervals (eg. Triennial) could help to increase uptake without loss of effectiveness deserves further investigation. 21
Considering the low uptake rate (12 to 16%) related to direct mailing of gFOBT kits in our programme, and the higher cost of FIT over gFOBT, further research is needed to assess the cost-effectiveness of this type of invitation in our country. 38 The cost savings could be allocated to increase GP involvement, which might have a greater impact on compliance.
Conclusions
The uptake and adherence to repeat testing in this programme are modest, at the lower end of rates achieved by the European trials, and they deteriorate significantly with time, mainly in people younger than 60. As our uptake appears to be among the best achieved in France, the reduction in CRC mortality will be markedly lower in our country than that reported in RCTs. The present results, together with those of our previous study demonstrating that the harms of colonoscopy were underestimated in all RCTs and are greater in our real-world programme, suggest that the benefit-risk balance of the French programme is currently less favourable than that stated in RCTs. 39 Our findings, together with those from the Scottish pilot, indicate that the benefit-risk balance demonstrated in research is not reproduced in practice. 13 A substantial effort is needed to enhance uptake and adherence to repeat testing. The replacement of the gFOBT by a quantitative FIT, which is scheduled for the beginning of 2015 at the very earliest in France, will help to increase uptake, but will not be the universal answer to all these concerns.
Footnotes
Competing interests
The authors have no competing interests
Funding
This study was performed as part of a quality assurance programme within the CRC screening programme in Alsace without dedicated funding. The sources of funding of ADECA Alsace, the association in charge of the programme, include the French statutory health insurance scheme (Assurance Maladie), the French Ministry of Health and the Haut-Rhin and Bas-Rhin Administrations (Conseils Généraux du Haut-Rhin et du Bas-Rhin). They had no role in study design, data collection, analysis, and interpretation, or writing the report.
Acknowledgements
We thank the GPs of the Haut-Rhin for their active participation in this programme, the staff of ADECA Alsace, and the participating gastroenterologists and pathologists for their contributions.
