Abstract
Objective
To identify factors associated with delayed adherence to follow-up in lung cancer screening.
Methods
Utilizing a data warehouse and lung cancer screening registry, variables were collected from a referred sample of 3110 unique participants with follow-up CT during the study period (1 January 2016 to 17 October 2018). Adherence was defined as undergoing chest CT within 90 days and 30 days of the recommended time for follow-up and was determined using proportions and multiple variable logistic regression models across the American College of Radiology Lung Imaging Reporting and Data System (Lung-RADS®) categories.
Results
Of 1954 lung cancer screening participants (51.9% (1014/1954) males, 48.1% (940/1954) female; mean age 65.7 (range 45–87), smoking history median 40 pack-years, 60.2% and 44.5% did not follow-up within 30 and 90 days, respectively. Participants receiving Lung-RADS® category 1 or 2 presented later than those with Lung-RADS® category 3 at 90 days (coefficient −27.24, 95% CI −51.31, −3.16, p = 0.027). Participants with Lung-RADS® category 1 presented later than those with Lung-RADS® category 2 at both 90- and 30-days past due (OR 0.76 95% CI [0.59–0.97], p = 0.029 and OR 0.63 95% CI [0.48–0.83], p = 0.001, respectively).
Conclusions
Adherence to follow-up was higher among participants receiving more suspicious Lung-RADS® results at index screening CT and among those who had undergone more non-lung cancer screening imaging examinations prior to index lung cancer screening CT. These observations may inform strategies aimed at prospectively identifying participants at risk for delayed or nonadherence to prevent potential morbidity and mortality from incident lung cancers.
Introduction
Lung cancer screening (LCS) with low-dose chest computed tomography (LDCT) has been shown to reduce disease-specific mortality in high-risk smokers.1–4 While an estimated 8.7 million persons are eligible for LCS in the United States according to current eligibility criteria, only 2–14.4% of eligible persons have undergone LDCT for LCS since reimbursement became widely available in 2015.5–7 Low enrollment in LCS may be influenced by various barriers8–12 and efforts are underway to mitigate these barriers and increase patient enrollment. 13
Retention and timely follow-up of LCS participants deserve equal attention as patient enrollment. The 20% reduction in lung cancer-related mortality observed in the National Lung Screening Trial included incident lung cancers detected in a cohort that maintained a 95% adherence rate across three annual screening LDCTs. 1 Kavanaugh et al. highlighted the importance of annual LCS, demonstrating a 7% rate of incident lung cancer developing within six years of receiving a negative baseline result. 14 Detection of early lung cancers requires a systematic approach to managing screen-detected findings codified by the American College of Radiology Lung Imaging Reporting and Data System (Lung-RADS®). Currently in version 1.1, Lung-RADS® permits standardized interpretation and issuance of management recommendations according to the level of suspicion and probability of malignancy of screen-detected findings. 15
To better manage patients, LCS programs would benefit from understanding the reasons for delayed follow-up and nonadherence. While researchers have examined adherence rates using various definitions of ‘positive’ screening results, the effect of a Lung-RADS® result and follow-up recommendations on adherence remains understudied.16–20 The purposes of this study were (1) to investigate effect of Lung-RADS® categories 1, 2, and 3 results on adherence to follow-up recommendations, and (2) to examine the relationships of other variables, which might be associated with delayed follow-up.
Materials and methods
The local Institutional Review Board approved this single-center retrospective cohort study, and written informed consent was waived. No funding source was utilized. The Strengthening the Report of Observational Studies in Epidemiology (STROBE) guidelines for reporting observational studies were followed. 21 Patient demographics and Lung-RADS® category were retrieved from each LCS CT that occurred over the study period utilizing a data warehouse 22 to complement a prospectively maintained LCS registry.
Screening population
CT scans from 3110 unique participants in the institutional LCS program from 1 January 2016 through 17 October 2018 were screened for inclusion. Eligibility was determined according to standard LCS criteria. 2 Patient characteristics are detailed in Table 1. All LCS patients participated in shared decision-making visits (SDM) with their referring providers to discuss the logistics, risks, and benefits of LCS according to institutional and current LCS requirements for Medicare reimbursement. All LDCT examinations were interpreted by subspecialty board-certified thoracic radiologists. Reports were standardized using built-in clinical decision support according to the most current version of Lung-RADS® available at the time of the screening (Version 1.0). 15 In the LungRADS® system, nodules are assigned a category based on the probability of malignancy according to morphology and growth patterns. Nodules assigned to category 1 (negative) or 2 (benign) have a less than 1% probability of malignancy, and participants are recommended to return to annual low-dose chest CT. A LungRADS® category 3 result indicates nodules have a 1–2% chance of malignancy with recommendation for interval LDCT in six months. “S-modifiers” may be applied in addition to the LungRADS® category for studies that show potentially clinically significant nonlung cancer findings that require additional diagnostic workup (e.g. aortic aneurysm). Reports were sent directly to the ordering provider via the electronic medical record.
Comparison of demographic variables between participants who received Lung-RADS® 1 and 2 results and those who received Lung-RADS® 3 at index screening.
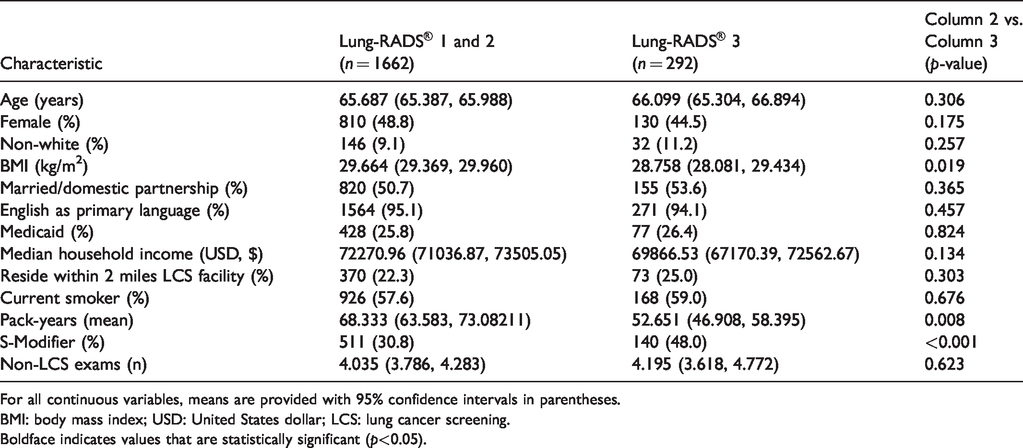
For all continuous variables, means are provided with 95% confidence intervals in parentheses.
BMI: body mass index; USD: United States dollar; LCS: lung cancer screening.Boldface indicates values that are statistically significant (p<0.05).
The index screening exam was defined as the first screening LDCT for each patient in the study period and included both baseline and follow-up exams. Expected follow-up date was calculated with reference to the index screening exam. A “1-year lookback” was performed to capture the total number of imaging exams of any type (not exclusive to thoracic imaging) performed for a patient in the rolling one-year period prior to the index screening exam.
All LCS participants who received Lung-RADS® category 4 results were followed and managed by a dedicated LCS navigator and were excluded from this analysis. Participants who received Lung-RADS® categories 1, 2, or 3 results during the study period were not actively managed and were included in the analysis.
Outcome measures and definitions
The primary outcome was adherence to the recommended timing for the next LCS examination issued by the interpreting radiologist in the final section of the LCS LDCT report. Recommendations followed Lung-RADS(R) guidelines of return for LCS in 1 year for categories 1 and 2, and follow-up CT in six months for category 3. Adherence was defined as undergoing a chest CT within 90 days of the recommended time based on Alshora et al. 16 We also considered a more stringent definition of adherence as follow-up within 30 days of recommended time intervals. Participants were coded as adherent if they returned prior to or on the recommended date or if they had any chest CT during the follow-up period. This included chest CT examinations not specifically performed as a LCS exam, such as a routine chest CT, CT angiography, or pulmonary CT angiography. This strategy avoided failing to capture a follow-up exam due to coding errors and misclassification of exams in cases in which providers cancelled a screening CT because a chest CT had been performed for another reason around the time of the scheduled LDCT. Participants who aged out of screening eligibility, died, or otherwise became ineligible were also considered adherent. Finally, we used a continuous measure of adherence using the number of days a patient was early or overdue for follow-up in relation to their index examination.
Predictors of adherence
Multivariate analysis considered age, sex, race/ethnicity (white vs. non-white), body mass index (BMI), smoking status (current, former), number of pack-years, insurance (private, Medicare, Medicaid, uninsured), relationship status (married, domestic partnership), home address, median income for the county of residence, residence within two miles of a screening facility associated with our institution, number of imaging examinations of any kind in the year prior to the index screening LDCT, Lung-RADS® category of the index LDCT, and potentially significant non-lung cancer findings (S-modifier).8–11, 17 , 18 , 23 , 24 Estimates of median income for county of residence were obtained using web-based zip code demographic analysis tools from CDX Technologies (www.cdxtech.com/). Lung-RADS® category was considered as a binary variable for our primary analysis (Lung-RADS® 1 and 2 vs. Lung-RADS® 3) and considered as an ordinal variable for the secondary analysis (comparing individual Lung-RADS® categories with reference to Lung RADS® category 1). Numbers of imaging examinations in the one-year lookback were tallied using data from the institutional data warehouse.
Statistical methods
Demographic variables and smoking status were compared across Lung-RADS® categories (Lung-RADS® 1 and 2 vs. Lung-RADS® 3) using F-tests and chi-square tests. The proportion of patients who were late for follow-up (6-month or 12-month) was calculated by Lung-RADS® category.
Multiple variable logistic regression analyses were performed to evaluate the effects of Lung-RADS® as a binary variable stratified by management recommendation (1 and 2 vs. 3), S-modifier, sociodemographic variables, and smoking status on timely return for follow-up using both definitions of adherence.
Multiple variable logistic regression analyses using Lung-RADS® were performed to evaluate the effects of Lung-RADS® as an ordinal variable, S-modifier, sociodemographic variables, and smoking status on timely return for follow-up using the primary and secondary definitions of adherence. Adjusted odds ratios were estimated with 95% confidence intervals. A Type-I error of 5% was assumed for all confidence intervals and two-sided hypothesis tests. Analyses were performed using STATA version 11 (StataCorp., College Station, TX).
Results
Over the study period, 4535 screening CTs were performed on 3110 LCS program participants with 2733 having a follow-up CT within the study period, or a period of greater than 30 days had elapsed between recommended follow-up time and the end of the study period. From this group, the first screening examination for each patient within the study period was selected. Patients receiving Lung-RADS® category 4 or 0 were excluded leaving a total of 1954 patients for the analysis (Figure 1).
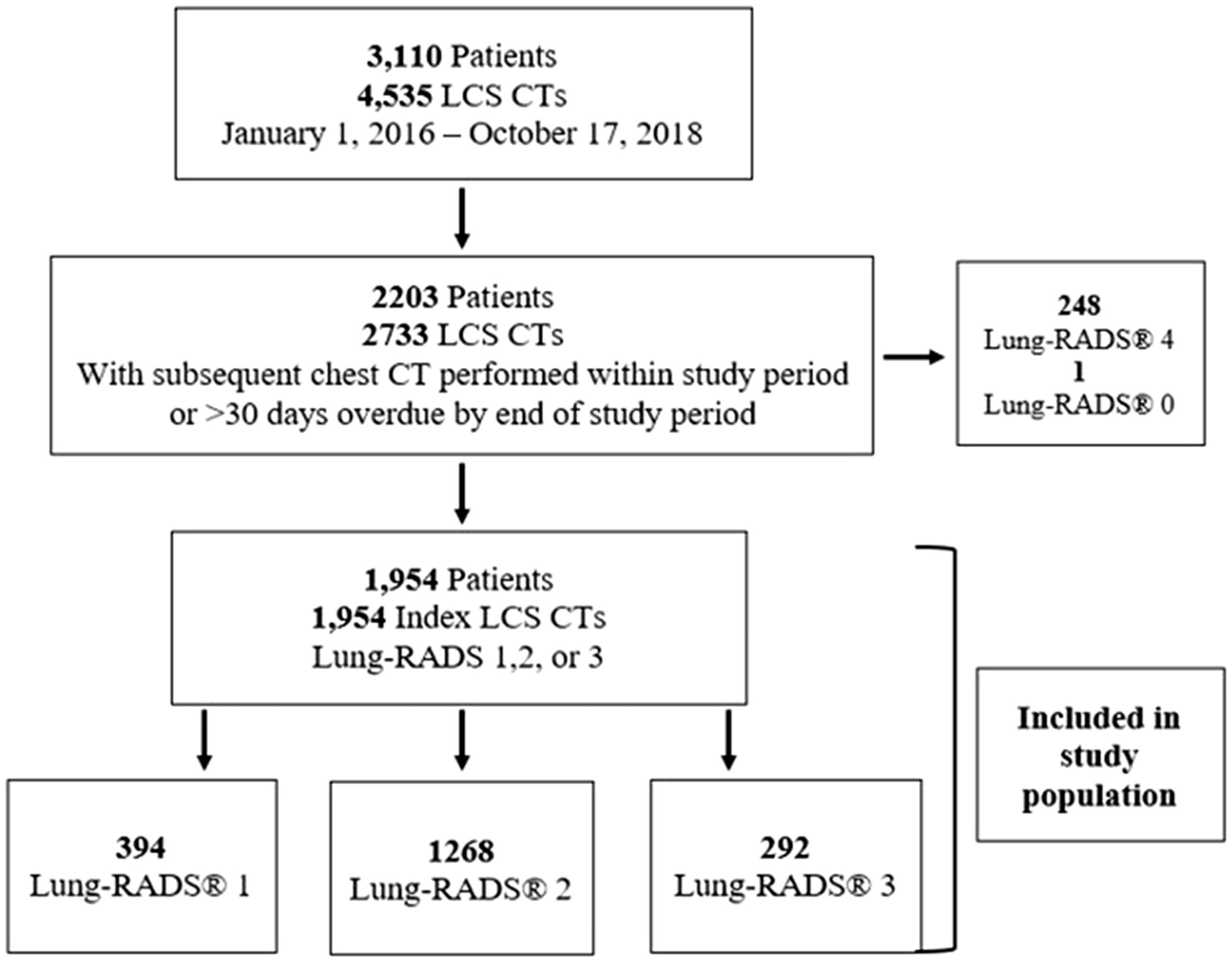
Cohort of patients and examinations included in the study.
Rates of adherence
Of the 1954 patients meeting inclusion criteria, 60.2% did not present for their examinations within 30 days and 44.5% within 90 days of recommended follow-up date. Mean follow-up delay was 115.2 days (95% CI 106.6, 123.8).
Factors associated with timeliness of follow-up
Lung-RADS® categories
As a binary variable (Lung-RADS® 1 and 2 vs. Lung-RADS® 3), Lung-RADS® 3 was associated with a lower rate of delayed follow-up than Lung-RADS® categories 1 and 2 at 90 days (OR 0.59 95% CI [0.44–0.78]) and at 30 days (OR 0.57 95% CI [0.43–0.75], p < 0.001) (Table 2
Variables affecting nonadherence (at 90 days and 30 days) and timeliness to follow-up across study cohort.
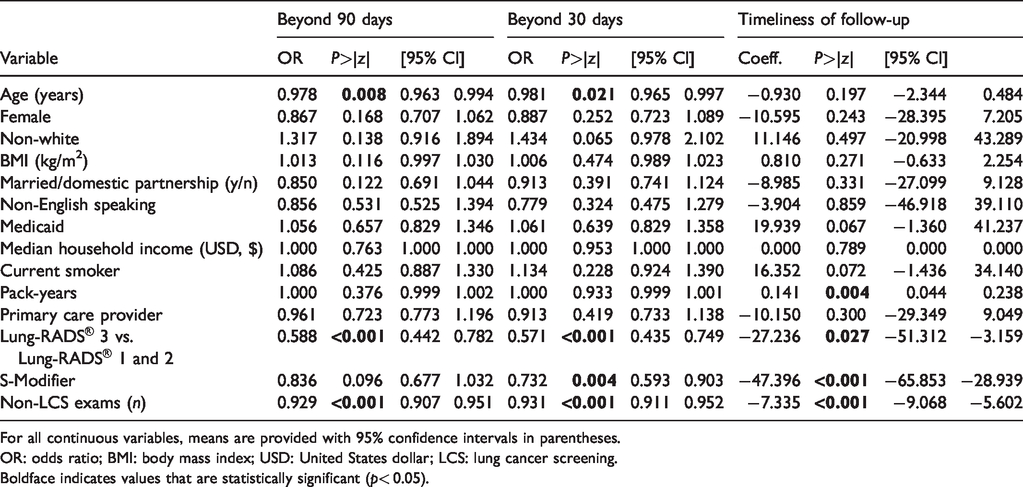
For all continuous variables, means are provided with 95% confidence intervals in parentheses.
OR: odds ratio; BMI: body mass index; USD: United States dollar; LCS: lung cancer screening.Boldface indicates values that are statistically significant (p< 0.05).
As an ordinal variable, we found that patients with Lung-RADS® 2 category had a lower likelihood of delayed follow-up than those with Lung-RADS® 1 at both 90 days and 30 days past due (OR 0.76 95% CI [0.59–0.97], p = 0.029 and OR 0.63 95% CI [0.48–0.83], p = 0.001, respectively) (Figure 2, Table 3). Patients with Lung-RADS® 1 category presented later for follow-up compared with patients with Lung-RADS® 2 and Lung-RADS® 3 categories. S-modifiers were associated with adherence, most significant at 30 days (OR 0.73 95% CI [0.59–0.90], p = 0.004) and less so at 90 days (OR 0.84 95% CI [0.68–1.03], p = 0.10) (Table 2).
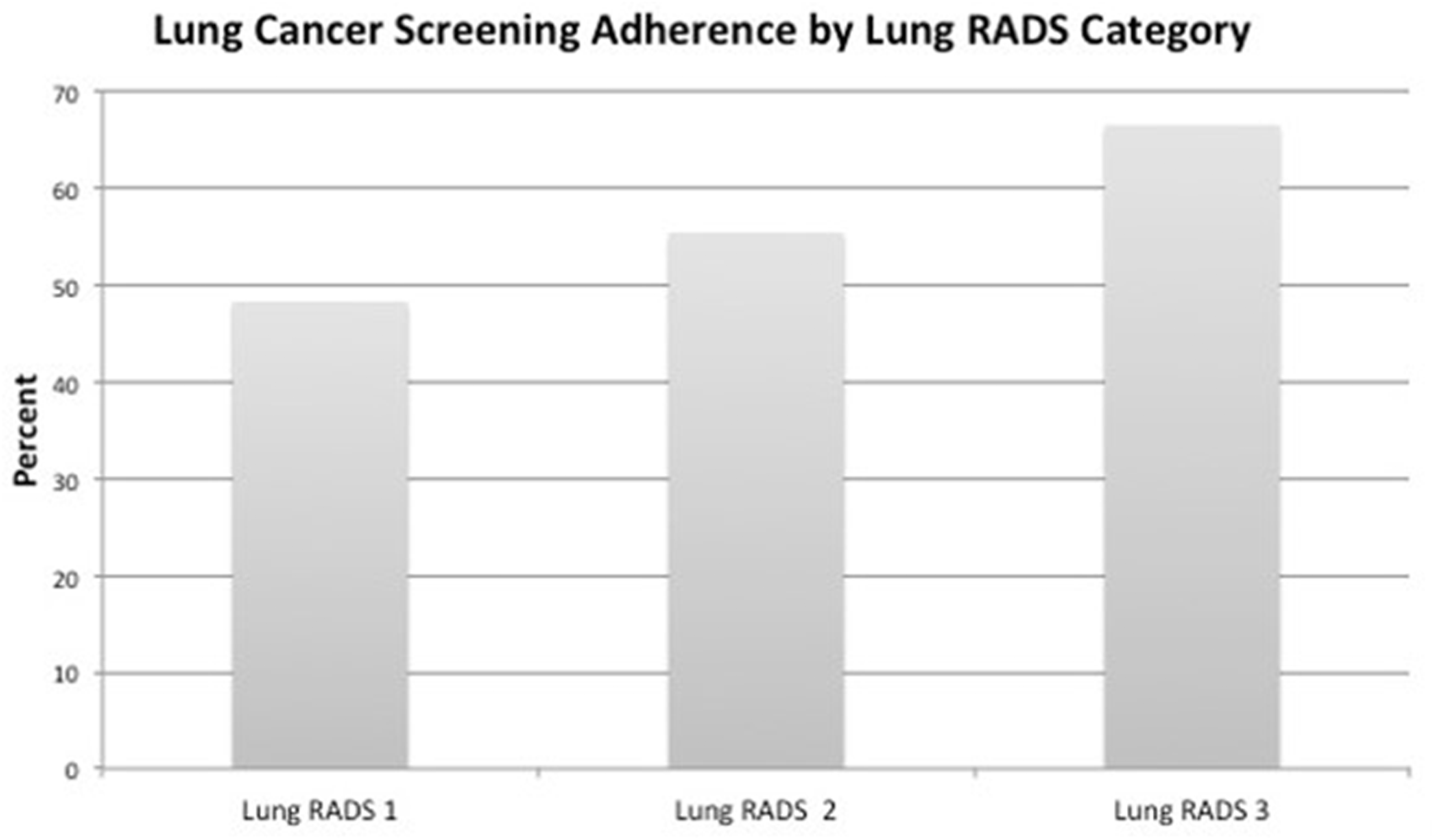
Participants with higher Lung-RADS® category showed greater adherence to recommended follow-up.
Comparison of nonadherence (90 days, 30 days) and timeliness to follow-up by individual Lung-RADS® category results using Lung-RADS® 1 as reference.
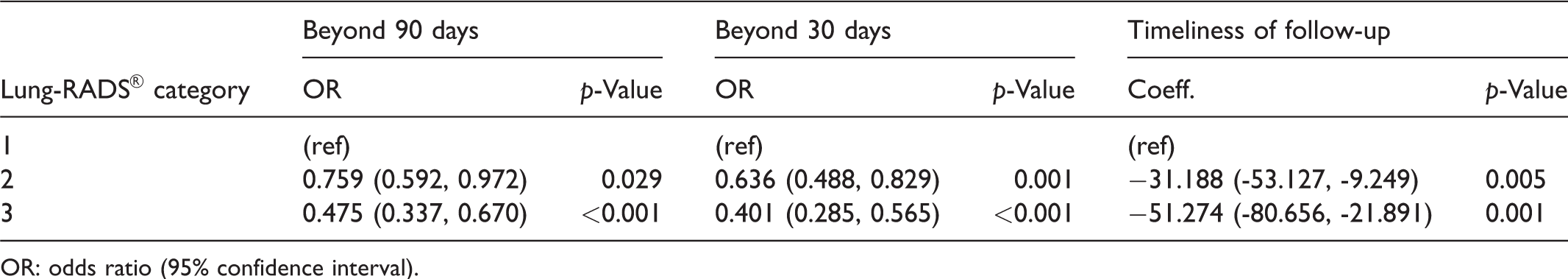
OR: odds ratio (95% confidence interval).
Number of prior imaging examinations
Having prior imaging examinations was associated with lower odds of 90-day (OR 0.92 95% CI [0.91–0.95], p < 0.001) and 30-day nonadherence (OR 0.93 95% CI [0.91–0.95], p < 0.001) and shorter interval between recommended and actual follow-up date (coefficient −7.335, 95% CI −9.068, −5.602, p < 0.001) (Table 2).
Other variables
Older age was associated with lower odds of nonadherence using our 90-day (OR 0.98, 95% CI [0.96–0.99], p = 0.008) and 30-day definitions (OR 0.98, 95% CI [0.97–1.0], p =0.021) but was not associated with timeliness of follow-up (coefficient −0.930, 95% CI −2.344, 0.484, p = 0.197). No significant associations were noted between timeliness of follow-up and gender, race/ethnicity, BMI, smoking status, pack-years, insurance, relationship status, median income, and distance from a screening facility (Table 2).
Discussion
Our study presents variables affecting follow-up adherence in the largest reported LCS cohort in the United States in the era of insurance-supported LCS and standardized reporting with Lung-RADS®. This is the first study to document greater adherence associated with higher Lung-RADS® results on index exams as well as the number of non-LCS imaging examinations one had undergone prior to LCS LDCT.
Lung-RADS® category influences adherence to follow-up
Adherence to annual LCS recommendations and timely follow-up of abnormalities are necessary to ensure a population level effectiveness of clinical LCS programs. Patients and providers should understand that negative baseline LCS results neither exclude a lung cancer diagnosis nor justify cessation of annual screening. Clinical LCS programs have reported nonadherence rates ranging from 11% to more than 50%.16–20, 25 Only three of these programs utilized Lung-RADS® for nodule reporting and management. 16 , 19 , 20 We observed that 44.5% of participants in our cohort failed to return within 90 days of their recommended next LDCT and receiving Lung-RADS® categories 1 and 2 were associated with decreased likelihood of timely adherence.
Adherence tended to be incrementally better among patients who received Lung-RADS® categories 1 (“negative”), 2 (“benign appearance or behavior”), and 3 (“probably benign”), respectively, categories all considered to be low suspicion for lung cancer. The same effect was observed between Lung-RADS® categories 1 and 2, despite both result categories being accompanied by the same follow up recommendation. 15 Perhaps detection of a nodule of any degree of suspicion might incite an acute elevation in anxiety related to the potential of receiving a lung cancer diagnosis and influence one’s motivation to return for follow-up. 24 ,26–28 So, the detection and reporting of very small 1–2 mm nodules on LCS may have important implications for the likelihood of a patient returning to annual screening.
The strength of conviction contained in radiologists’ reports is tied to the level of suspicion of abnormal imaging findings and may influence clinician and patient adherence to follow-up recommendations. 29 , 30 Perhaps the language included in category descriptors provided by Lung-RADS® might also contribute to these observations. For example, the descriptor phrases “no nodules or definitely benign nodules” (category 1) and “nodules with a very low likelihood of becoming a clinically active cancer due to size or lack of growth” (category 2) may convey a false impression to the patient or provider that the low-suspicion screening result means that the patient is unlikely to develop lung cancer. All screened patients at our institution engage in a SDM process with their referring providers prior to each LCS CT. Therefore, in this cohort, SDM alone may not be enough to ensure adherence among patients who receive LungRADS® categories 1 and 2 results. Emphasizing the importance of annual screening at patient office visits, in the LCS report and result letter, and in follow-up reminders may help combat these false impressions.
S-modifiers were associated with improved adherence, which may have reflected a trend towards early return for chest CT for follow-up of acute abnormalities such as infection detected at index CT. A recent analysis showed better adherence to follow-up of incidental findings at LCS (S-modifiers) among patients who had greater than high school education levels. 31 Standard follow-up for many other abnormalities associated with an S-modifier (such as thyroid nodules or indeterminate renal lesions) includes imaging other than chest CT and would not have affected adherence as measured in our study.
In our practice, patients who receive suspicious results (Lung-RADS® 4 A, 4B, and 4X) are actively managed by a nurse navigator, not unlike the approach reported by Lee et al. 20 LCS programs that actively manage patients who receive both positive and negative baseline results report much higher adherence rates.16–19, 25 For example, Alshora et al. failed to show a statistically significant difference in adherence among patients who received either positive (Lung-RADS® 4), “probably benign” (Lung-RADS 3®), or nonpositive (Lung-RADS® 1 and 2) baseline results, likely an effect of written and telephone reminders to patients and ordering providers. 16 Our results may have unmasked a significant effect of assigned Lung-RADS® categories on adherence in the absence of navigation as a confounding variable highlighting the link between active navigation and follow up adherence.
Other non-LCS imaging examinations
To our knowledge, we are the first to demonstrate an association between LCS adherence and the number of non-LCS examinations performed in the year prior to the index screening LDCT. Those with fewer total non-LCS imaging studies were less likely to receive subsequent annual LCS LDCT. Perhaps patients who are already undergoing imaging for other conditions might have a more robust integration into imaging systems and be more accustomed to imaging. It is also possible that socioeconomic, cultural, or geographic factors might have influenced both the timeliness of LCS follow up and access to other imaging services. Radiologists might prospectively identify patients with fewer total imaging studies as at-risk of low adherence and inform the referring clinicians. Systems processes including automated provider reminders and letters also may be incorporated, similar to screening mammography, targeting participants with fewer non-LCS imaging exams. 17 , 32
Other variables
Our analysis did not show statistically significant associations among the multiple other variables for both adherence to and timeliness of follow-up. Some studies have reported differences in LCS uptake, adherence, and rates of late-stage diagnoses among persons of racial and ethnic minority backgrounds, 9 , 18 , 33 , 34 but we observed no such association in our cohort, which included a relatively small proportion of non-whites. Many of our disadvantaged patients receive care in health centers and are managed by patient coordinators to facilitate adherence for appointments and other follow-up, which may have influenced this result. 25 , 35
It was recently shown that insured individuals had higher LCS utilization than those without insurance. 7 Wildstein et al. found higher rates of adherence among “no-pay” patients compared to self-pay patients (88 vs. 62%) in their LCS cohort. 17 In another analysis, persons with noncommercial insurance had greater odds of a missed outpatient appointment for advanced imaging studies. 36 However, we found that having Medicaid coverage had no association with adherence to or timeliness of follow-up.
Smokers living in sparsely populated areas have been shown to have less access to LCS facilities as LCS sites tended to be clustered in urban environments. 10 , 11 , 18 We found no such association in our cohort from a fairly clustered metropolitan environment connected to a robust public transportation system.
We found a relatively high percentage of active smokers among our screened cohort (58–59%) compared to 33–48% reported by others, but less than that reported by Guichet et al. (81%). 1 , 16 , 17 , 37 Although some studies have shown that former smokers are more adherent to LCS than active smokers, 18 , 23 we found no association between smoking status or pack-years of smoking and adherence.
Limitations
The single institution, retrospective study design introduced several limitations to our analysis. First, we did not capture if participants may have received follow-up imaging at facilities outside of our network, and it is possible that the overall adherence rate is higher than our data suggest. Furthermore, we could not directly assess participants’ perceived lung cancer risk and motivation for LCS, both of which have been associated with a greater likelihood both to enroll in and adhere to screening. 17 , 18 A minority of our primary care LCS referral base is family medicine-trained, so the effect of provider specialty training on adherence in our cohort may not be representative of the national referral base.
Conclusions
Adherence to follow-up recommendations is vital to ensure long-term success of LCS programs. In our cohort, patients who received less suspicious Lung-RADS® results at index LDCT and those who had undergone fewer total imaging exams were less adherent to follow-up recommendations. These observations may help prospectively identify LCS participants who may be at risk for nonadherence and design strategies to avoid potential adverse outcomes associated with non-adherence to LCS follow-up.
Footnotes
Acknowledgements and disclosures
We acknowledge Maryann A Tateosian, lung cancer screening program coordinator. This project received no funding support. The local Institutional Review Board approved this single-center retrospective cohort study, and the need for written informed consent was waived.
Brent P Little discloses receipt of royalties as an associated editor for Reed Elsevier, Inc.
Jo-Anne O Shepard receives royalties as a textbook editor for Elsevier.
Amita Sharma is a principle investigator for Hummingbird Diagnostics, Inc. and receives royalties as an associate editor for Elsevier.
Dr Fintelmann reported receiving consulting fees from McKesson, nonfinancial support from BTG plc, and research grants from the Society of Thoracic Radiology, the Society of Interventional Oncology, and the American Roentgen Ray Society.
The remaining authors have no relevant financial disclosures associated with this manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
