Abstract
Objectives
In Italy, regional governments organize cervical, breast and colorectal cancer screening programmes, but there are difficulties in regularly inviting all the target populations and participation remains low. We analysed the determinants associated with invitation coverage of and participation in these programmes.
Methods
We used data on screening programmes from annual Ministry of Health surveys, 1999–2012 for cervical, 1999–2011 for breast and 2005–2011 for colorectal cancer. For recent years, we linked these data to the results of the national routine survey on preventive behaviours to evaluate the effect of spontaneous screening at Province level. Invitation and participation relative risk were calculated using Generalized Linear Models.
Results
There is a strong decreasing trend in invitation coverage and participation in screening programmes from North to South Italy. In metropolitan areas, both invitation coverage (rate ratio 0.35–0.96) and participation (rate ratio 0.63–0.88) are lower. An inverse association exists between spontaneous screening and both screening invitation coverage (1–3% decrease in invitation coverage per 1% spontaneous coverage increase) and participation (2% decrease in participation per 1% spontaneous coverage increase) for the three programmes. High recall rate has a negative effect on invitation coverage in the next round for breast cancer (1% decrease in invitation per 1% recall increase).
Conclusions
Organizational and cultural changes are needed to better implement cancer screening in southern Italy.
Keywords
Introduction
The European Commission recommends population-based organized screening programmes for cervical, breast and colorectal cancer. 1 In Italy, the major barriers to effective screening are the incomplete rollout or missed implementation of such programmes and, where implemented, the poor participation of the target populations. To date, no more than one in three people in the target population actually regularly participates in the proposed test in an organized programme.2–4
Italian organized screening programmes had a slow scale-up phase, starting from the late 1990s and still not completed. For cervical and breast cancer screening, all Italian Regions have now at least partially implemented programmes, although in some areas (particularly in the South) there are difficulties in regularly inviting all the target populations. Invitation coverage (i.e. the proportion of the target population regularly invited) in 2014 was 70, 75 and 66% for breast, cervical and colorectal cancer, respectively (Figure 1).5–8 There is a dramatic difference in the spread of organized programmes between the North, Centre and South of Italy for breast and colorectal cancer screening.
5
Trends of (a) invitation coverage and (b) participation in the screening programme by geographic macro-area: diagonal lines: North; horizontal lines: Centre; vertical lines: South; black: all Italy.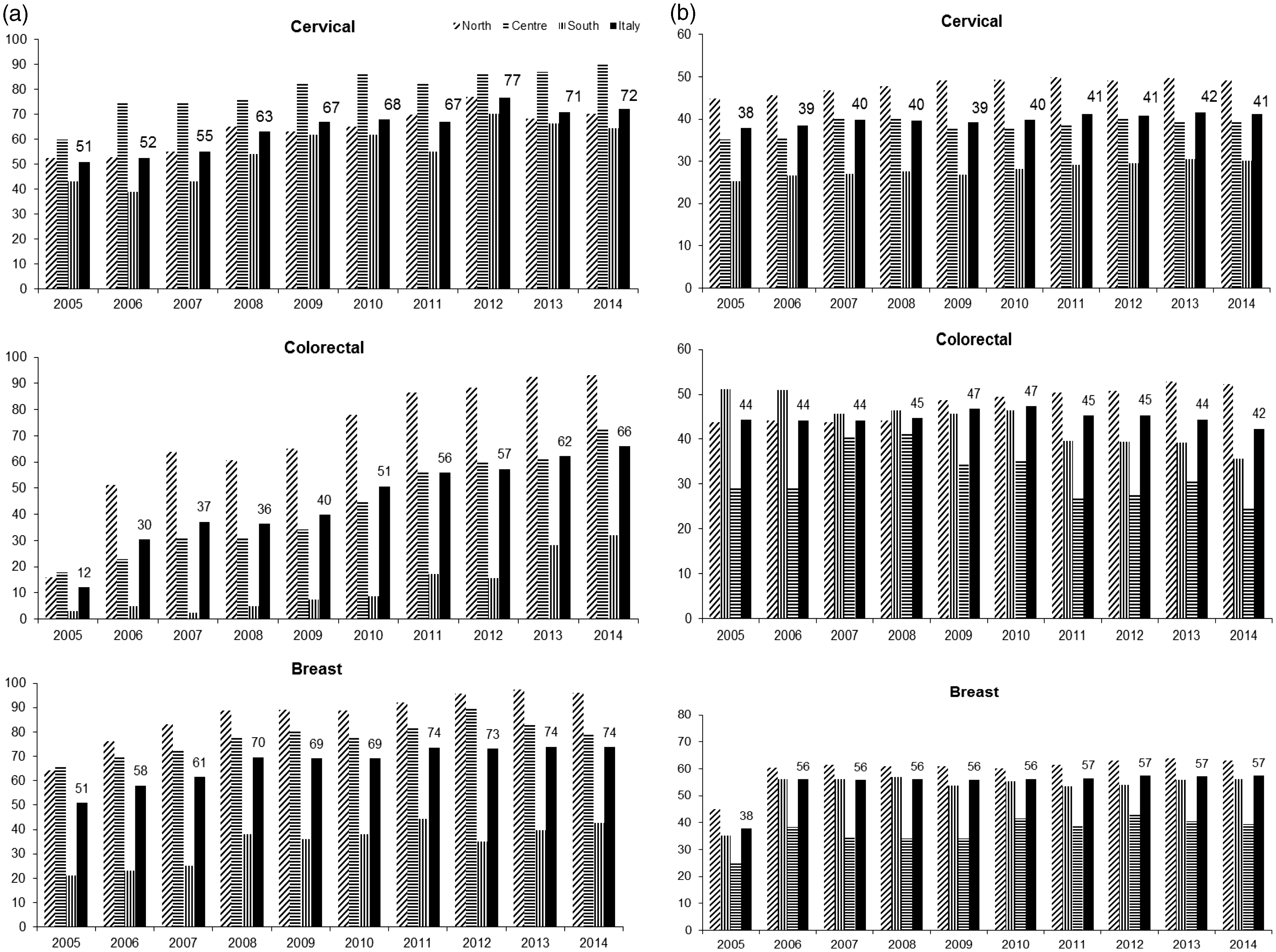
Participation on average is low for all the three programmes, with lowest levels in the South, which is also the most deprived area (Figure 1). Low participation does not equate to low test coverage because, for cervical and breast screening, parallel opportunistic screening is widespread in Italy, pushing up the test coverage. However, coverage due to opportunistic screening is very similar in the South and the North, and so spontaneous (mostly private) testing activity does not level the differences. 2
Many studies9–16 have identified individual characteristics and organizational barriers affecting participation, but most identified determinants are not modifiable (e.g. gender or age) or are not easily modifiable (e.g. socio-economic status). To implement more effective programmes, modifiable factors must be identified and addressed. Furthermore, despite considerable debate on how the programmes should be implemented,17–20 determinants of the delay in implementation have not been effectively studied. Some barriers to extending invitations to all populations, other than lack of resources, may be modifiable. The theoretical background of this research is the PRECEDE–PROCEED model 21 applied to the screening programmes. 22 We propose part of the epidemiological, educational and ecological assessment to identify enabling factors and modifiable barriers that could be addressed by national, regional or local health policy and actions. The aim of this paper is to analyse factors associated with invitation coverage of and participation in screening programmes.
Methods
In Italy, in accordance with European Commission recommendations 1 , Regional Health Systems are committed to organizing screening programmes that actively invite the target population. Programme implementation is at Local Health Authority level, which generally corresponds to the administrative area of a province. Since 1999, Italian guidelines 23 have recommended inviting women aged 25–64 for a Pap test every three years (since 2013 for women over 30, the human papillomavirus (HPV)-DNA test has been recommended every five years) 24 and women aged 50–69 for a mammogram every two years. Colorectal cancer screening has been included since 2006 with two strategies: men and women aged 50–69 for faecal occult blood test (FOBT) every two years, or (in the Piedmont region) a once only sigmoidoscopy at age 58–60, plus FOBT for those not responding to sigmoidoscopy. 23 All target population residents must be invited, and all screening tests, ascertainment and treatments are free of charge. The Ministry of Health monitors if the Regions actually guarantee this essential level of assistance through a system of indicators. The achievement of standards of invitation coverage and participation is linked to the funding of the Regional Health Services by the National Health Fund.
This study is an analysis of repeated cross-sectional surveys. Data from annual surveys on screening programme activity are collected by the National Screening Monitoring Centre (Osservatorio Nazionale Screening, ONS). The characteristics of the programmes have been analysed as possible determinants of the level of the invitation coverage of the target population of the programme and of participation among invited people. For recent years, it has been possible to link data from ONS surveys to the results of sample surveys based on the telephone interviews collecting self-reported information on spontaneous screening uptake, not included in the screening programme activity. 2
The National Screening Monitoring Centre, on behalf of the Italian Ministry of Health, annually collects aggregated data from all Italian screening programmes.5–8 Surveys have been available since 1999 for cervical and breast cancer and since 2005 for colorectal cancer. The last year of activity available at the time of the analyses was 2012 for cervical and colorectal cancer and 2011 for breast cancer. The surveys collect data on the programme target population, invitations, test attendance and results, ascertainment and final findings for all invitations sent in a given year and tests performed for these people until 30 April in the following year.
We considered as active the programmes that invited at least 1000 people in a given year. For each programme that participated in the survey at least once in the study period, we generated a complete historical series, with zero invitations for the missing years. Each record, reporting indicators summarizing the activity of a programme during a given year and the characteristics of the programme in that period, is considered as the primary statistical unit ‘programme/year’. Some programmes did not participate in the survey for some years, but declared that they did not stop their activity; for these cases (21 for cervical, 10 for breast and 7 for colorectal screening), we arbitrarily generated a record with the number of invitations and participants equal to the mean of the previous and following year.
Progressi delle Aziende Sanitarie per la Salute in Italia (PASSI) is a national surveillance system that continuously collects information via phone calls about behavioural health risk factors and the spread of preventive health care services. From 2010 to 2013, more than 151,000 people aged 18–69 were interviewed. The sampling and survey methodologies are described in detail elsewhere.25–27 Information on screening includes test uptake (Pap smear, HPV, mammography, FOBT and colonoscopy), date of the last test, provider of the last test (paying or free, proxy of the organized screening programme), reason for not participating in screening and screening promotion/recommendation received. A putative determinant of both the implementation and participation in the screening programme was the proportion of the general population that had paid for a test (i.e. in the private sector outside the organized screening programmes). This variable is ecological and was considered at provincial level. For cervical cancer, the proportion was computed including women aged 25–64 who had a Pap test in the three years before the interview or an HPV test in the five years before the interview; for breast cancer, women aged 50–69 who had a bilateral mammogram in the two previous years; and for colorectal cancer, an FOBT in the previous two years for men and women aged 50–69 or a colonoscopy or flexo-sigmodoscopy in the previous five years for those aged 50–59 and one in a lifetime for those aged over 60–69.
The outcomes included in this study are:
Invitation coverage of the target population: the number of invitations divided by the annual target population; this outcome has been considered as a proxy of the programme implementation, because active invitation cannot be sustained by the health providers if at least the basic resources and requirements to perform first-level mammograms and assessment are not present and organized. Participation in the screening programme: number of participating invitees over the total number of invited people minus those excluded from screening after invitation.
The statistical unit used in the analyses was the ‘programme/year’ (i.e. data about the activity of each programme in a given year).
For invitation coverage, we considered the following possible determinants: calendar year, geographic macro-area (North, Centre and South), metropolitan area (programmes covering the areas of Rome, Milan, Naples, Turin, Genoa and Palermo) or other cities, age of the programme (first round, second round or further rounds), referral rate in the previous year and diffusion of spontaneous screening in the province. For this outcome, to study the effect of the geographic variable and calendar year, we also included in the database the years in which the programme was not active, considering the level of implementation as 0. For all the other variables, only the years in which the programmes were active are included. For participation, we considered as possible determinants: calendar year, geographic macro-area (North, Centre and South), metropolitan area or other cities, age of the programme (first round, second round or further rounds), invitation coverage in the previous round and spread of spontaneous screening in the province. For calendar year, geographic macro-area and metropolitan area, the models included all the available years. The effect of spontaneous screening was only considered for the last two years of the survey as the PASSI interviews were collected from 2008 to 2013 asking about tests performed in the previous two or three years.
All the results are adjusted for the decrease in invitation coverage in the previous round. When programmes are forced to reduce their activity and invite only a part of the target population, they usually give priority to participants from a previous round as these have a much higher propensity to participate than the general population.
We used the STATA 13 Generalized Linear Model command with a binomial error distribution for estimating and participation relative risk.
Results
Up to 2012, 123 cervical cancer screening programmes and 117 colorectal cancer screening programmes had been active for at least one year. By 2011, there had been 114 breast cancer screening programmes. The resulting activities of screening programmes reported to the ONS for cervical, breast and colorectal cancer screening, respectively, were 1271 programmes/year, 1089 programmes/year and 646 programmes/year. Some programmes were interrupted for one or more years: 17.1, 8.8 and 6.0% for cervical, breast and colorectal cancer, respectively. Interruptions were more common among the programmes in southern Italy (5.1 vs. 9.8% overall).
Determinants of invitation coverage
Determinants of the invitation coverage of the target population by screening programme. a
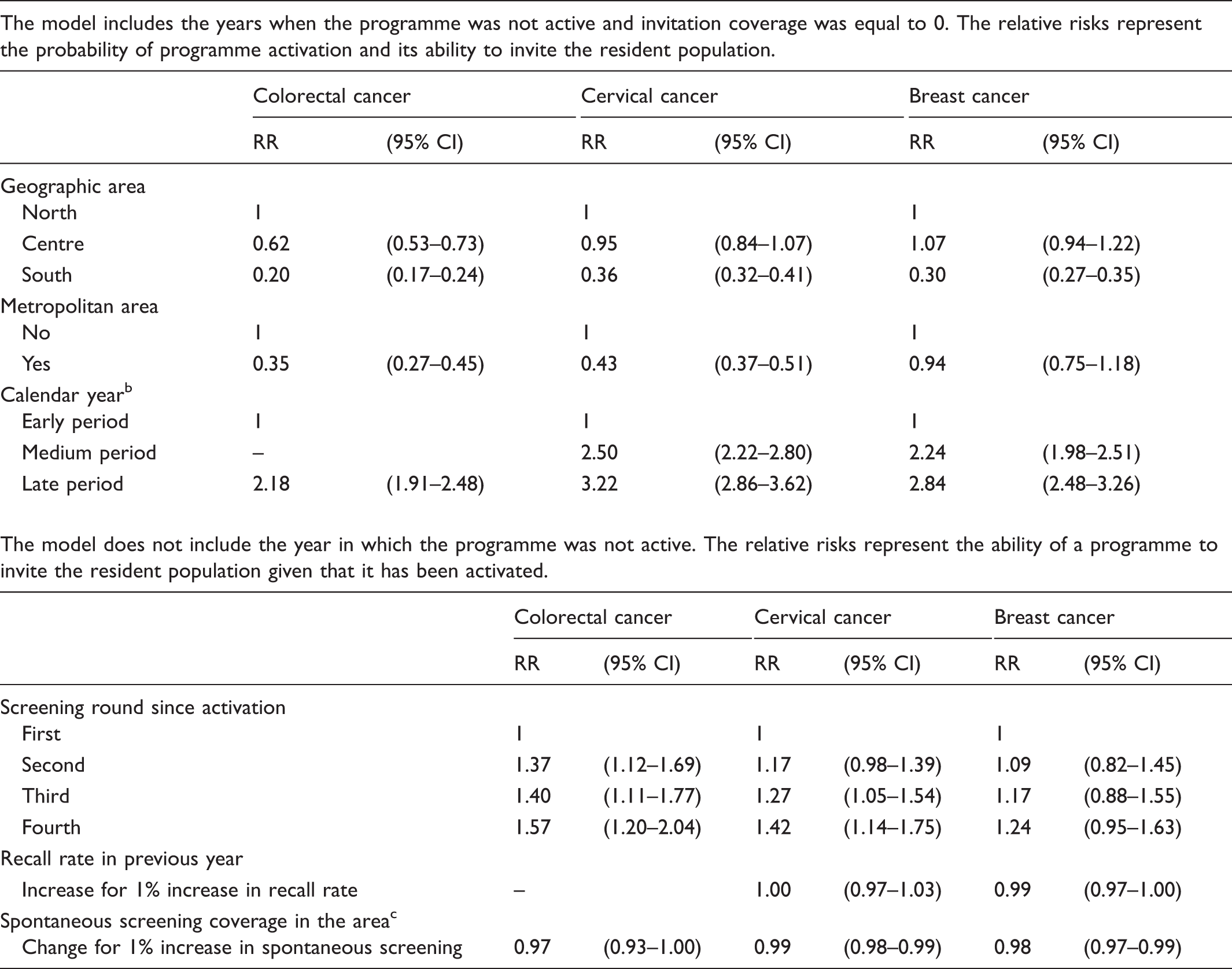
CI: confidence interval; RR: relative risk.
All the relative risk (RR) estimates are adjusted for the other variables.
For colorectal cancer: early = 2005–2009; late = 2009–2011. Cervical: early = 1999–2003; medium = 2004–2008; late = 2009–2012. Breast: early = 1999–2003; medium = 2004–2008; late = 2009–2011.
Estimated only for the last two years of the dataset, i.e. 2011–2012 for cervical and colorectal cancer screening, 2010–2011 for breast cancer screening.
Determinants of participation
Determinants of the participation in screening programmes. a
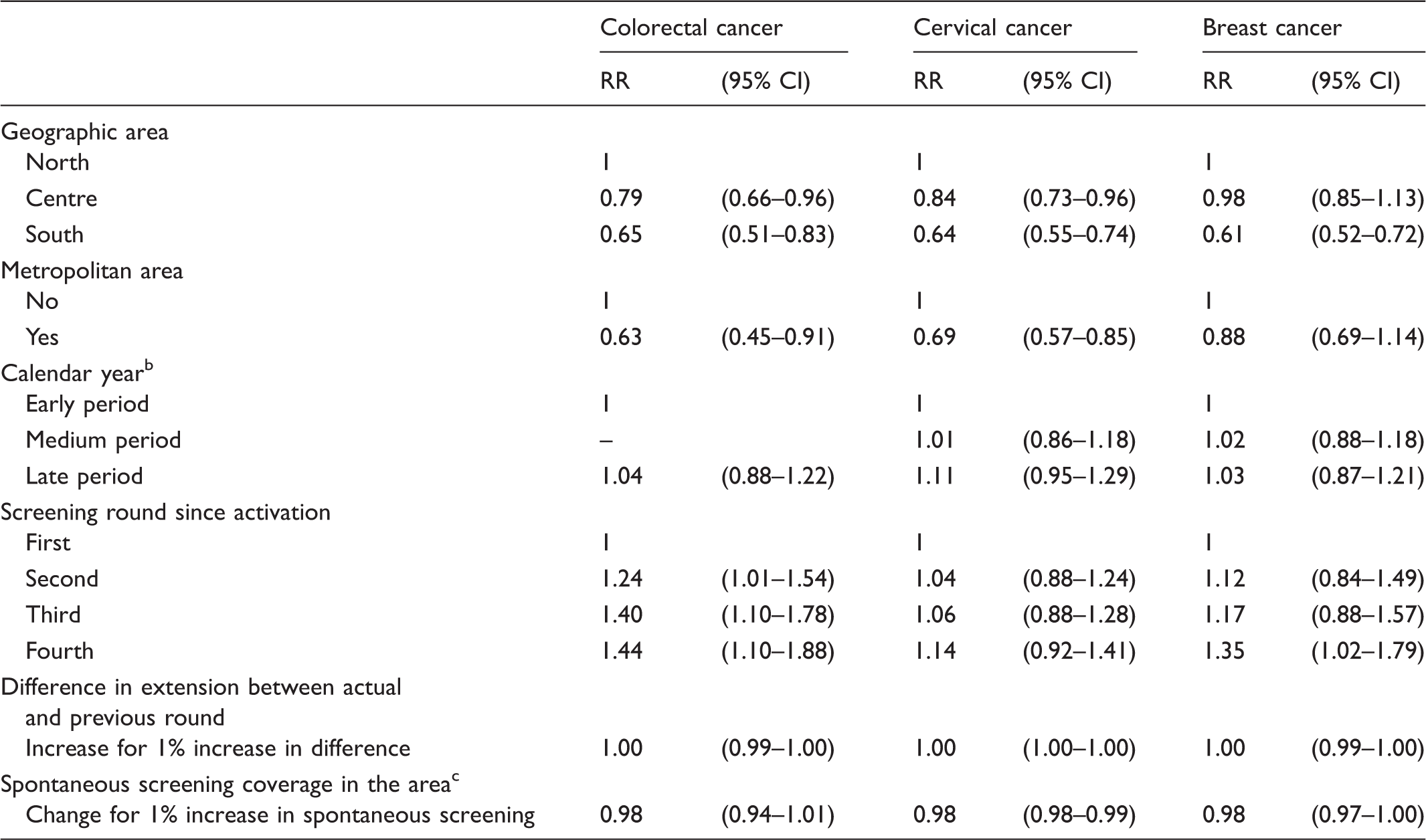
CI: confidence interval; RR: relative risk.
All the relative risk (RR) estimates are adjusted for the other variables.
For colorectal cancer: early = 2005–2009; late = 2009–2011. Cervical: early = 1999–2003; medium = 2004–2008; late = 2009–2012. Breast: early = 1999–2003; medium = 2004–2008; late = 2009–2011.
Estimated only for the last two years of the dataset, i.e. 2011–2012 for cervical and colorectal cancer screening, 2010–2011 for breast cancer screening.
Discussion
Invitation coverage of the target population and participation in screening programmes differ between geographic macro-areas, with the Southern regions of Italy having the worst outcomes, as has been previously reported.2,5 Programmes in metropolitan areas have more difficulties in both invitation coverage and participation, as do programmes at their first screening round compared with those at second or third round. It is difficult to make comparisons on this with other literature, particularly regarding the factors linked to complete rollout. For participation, the urban/rural area showed an inconsistent trend in different European countries.28,29 In recent years, the programmes experienced fewer difficulties in covering the population, but participation decreased for colorectal and breast cancer and increased only for cervical cancer screening. For this positive trend in cervical cancer screening participation, it is possible to imagine an effect of the economic crisis affecting Italy since 2008 30 that may have forced women to shift from private gynaecologists to public programmes for their routine Pap test. 2
A high recall rate for mammographic screening had a negative impact on the ability to invite the target population of the target in the next year. This was expected, as second-level ascertainment absorbs many resources in the programme in general and radiologists in particular. One ascertainment needs about 5–10 times the time needed by the radiologist for a first-level test. 31 Thus, high levels of referral rate are unsustainable for many programmes. When waiting times for mammography responses and for assessment appointments become too long, the only choice for the screening coordinating centre is to reduce the number of invitations in the following months. This effect was not noticeable for cervical cancer screening, but in this case, the colposcopy workload does not require the same human resources used in the first-level examination, probably making the effect of recall on invitations less direct. This finding provides further evidence to support the fact that preventive mammography needs a different approach from clinical mammography. In prevention, a balance between sensitivity and specificity of the first-level test is necessary, for both balancing benefits and damage at the individual level, and to make programmes sustainable at the organizational level. 32
The most interesting result of these analyses is the negative effect of spontaneous screening on both participation and implementation. The negative association with screening implementation is clear for all three screenings, even though the level of spontaneous screening uptake is 9% for colorectal cancer, 19% for breast cancer and 37% for cervical cancer screening. Nevertheless, this association can be easily interpreted as reverse causality, because high spontaneous screening uptake could be a consequence of low level of organized screening implementation. The negative association with participation is relevant for both breast and cervical cancer screening, but is not present for colorectal cancer. In this case, reverse causality is not a plausible explanation, as most of the spontaneous screening tests included in this survey were performed before the invitation to organized screening. It is also notable that the effect was not present for colorectal cancer, where spontaneous screening does not have a private market in Italy, and is not influenced by conflicts of interests of the professionals, while these kinds of interests are strong for breast and particularly for cervical cancer screening.
Importantly, our analyses only include areas in which a screening programme has been in place for at least one year during the study period. Consequently, we will have underestimated the differences between North and South as, at least for breast and colorectal cancer, the areas where the screening programmes have never been active are predominantly in the South.
Our study has some limitations. The cross-sectional approach does not allow causes and effects to be distinguished when an association is found. This limitation is even stronger as we only have data on spontaneous screening uptake for the most recent years. Nevertheless, for some possible determinants, our study design can partially overcome this problem. For screening activity-related variables (i.e. recall rate in the previous year and invitation coverage in the previous round), we could analyse the determinants and the outcomes in a correct time sequence. The problem does not have a simple solution for studying the effect of spontaneous screening uptake on implementation and, in to a lesser extent, on participation as explained above.
Our estimate of the spontaneous screening is based on a question about having paid for the test. There could be a substantial misclassification because in many regions, the screening programmes are not completely activated. Also, it is possible to have the recommended tests free outside organized screening, so all the tests not free are not performed within organized programmes, but not all the free tests are really part of the organized screening. In addition, the questions asked in the PASSI survey do not allow the overlap of organized and spontaneous screening to be observed, as only the last test is requested. This overlap in Italy was estimated in local studies at the beginning of cervical screening programme implementation to account for 15–25% of the target population.33,34 Nevertheless, the comparison of the estimates of organized screening test coverage obtained by the Screening Monitoring Centre with those obtained from the PASSI survey are quite similar for the regions where the programme activation is high, and the misclassification of free tests out of screening is expected to be small. 2
As in most previous analyses of the determinants of screening uptake in Italy2,5, the geographic macro-area still accounts for the largest part of the differences, and this is not a modifiable characteristic. Northern, Central, and Southern Italy have different cultural background, income and behaviour, but the regions in the three areas also have different organizations of screening services and different reputations of public services. Some of these characteristics may be modifiable determinants. In particular, how the local health service manages opportunistic screening seems to be a critical factor in the success of the screening programmes. This is an actionable point, especially considering that part of opportunistic screening is provided by the Public Health Service itself. Furthermore, reducing competition and overlapping organized and opportunistic screening increase the efficiency of the system, releasing a large amount of resources with minimal or no impact on health benefits. 18 The effectiveness of proposed managerial tools should be tested in studies with the appropriate design.
Conclusion
Screening programmes need a strong public service able to contrast the interests of public and private providers of opportunistic screening. The health system should organize a multidisciplinary pathway, in which the responsibilities are shared between several units but also the individual units must forfeit part of their autonomy. To provide this kind of organization, the needs of the health system (and of society) must be placed before those of individual professionals. Where the public service is weaker, in terms of technological resources, managerial expertise and reputation, as is the case in southern Italy, it is more difficult to organize multidisciplinary pathways and to reduce or completely close opportunistic access to screening within public services. The attitudes and competence of the professionals, and particularly radiologists, in preventive medicine are determining factors in making mammographic screening sustainable and equitable. High recall rates inhibit the implementation of the programme.
Footnotes
Acknowledgements
We thank Guglielmo Ronco, Pamela Giubilato, Manuel Zorzi and Leonardo Ventura for their work in collecting and improving the quality of the screening survey data. We also thank the National Centre of Epidemiology and Public Health (CNESP) of the National Health Institute (ISS) for its support to the PASSI survey and all National Health Service professionals for collecting interviews, managing and analysing data for the PASSI survey.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship and/or publication of this article: The study was funded by the Italian Ministry of Health through a grant from the Centre for Disease Control and Prevention (CCM-2012) coordinated by the AgeNaS (National Agency for Health Services).
