Abstract
Objective
Comorbidity is associated with adverse outcomes for all lung cancer patients, but its burden is less understood in the context of screening. This review synthesises the prevalence of comorbidities among lung cancer screening (LCS) candidates and summarises the clinical recommendations for screening comorbid individuals.
Methods
We searched MEDLINE, EMBASE, EBM Reviews, and CINAHL databases from January 1990 to February 2021. We included LCS studies that reported a prevalence of comorbidity, as a prevalence of a particular condition, or as a summary score. We also summarised LCS clinical guidelines that addressed comorbidity or frailty for LCS as a secondary objective for this review. Meta-analysis was used with inverse-variance weights obtained from a random-effects model to estimate the prevalence of selected comorbidities.
Results
We included 69 studies in the review; seven reported comorbidity summary scores, two reported performance status, 48 reported individual comorbidities, and 12 were clinical guideline papers. The meta-analysis of individual comorbidities resulted in an estimated prevalence of 35.2% for hypertension, 23.5% for history of chronic obstructive pulmonary disease (COPD) (10.7% for severe COPD), 16.6% for ischaemic heart disease (IHD), 13.1% for peripheral vascular disease (PVD), 12.9% for asthma, 12.5% for diabetes, 4.5% for bronchiectasis, 2.2% for stroke, and 0.5% for pulmonary fibrosis.
Conclusions
Comorbidities were highly prevalent in LCS populations and likely to be more prevalent than in other cancer screening programmes. Further research on the burden of comorbid disease and its impact on screening uptake and outcomes is needed. Identifying individuals with frailty and comorbidities who might not benefit from screening should become a priority in LCS research.
Introduction
Lung cancer remains the leading cause of cancer deaths globally, with 1.8 million estimated deaths in 2020. 1 The five-year survival rate for lung cancer is 16–22% in high-income countries. 2 Patients with localised lung cancers have better five-year survival rates (above 50%) compared to most advanced distant stages (less than 10%).3,4 This large disparity highlights the crucial role of early detection in maximising the survival benefit for high-risk individuals. The National Lung Screening Trial (NLST) was the first large-scale randomised study that reported a mortality benefit, with an estimated 20% reduction in lung cancer mortality using pulmonary low-dose computed tomography (LDCT) compared to X-ray. 5 In addition, the Dutch-Belgian Randomised Lung Cancer Screening Trial (NELSON) demonstrated a 24% reduction in male lung cancer mortality. 6 Following the NLST results, the US Preventive Services Task Force (USPSTF) issued its recommendations in 2013 for annual screening of lung cancer with LDCT for current or former smokers aged 55 to 80 years. However, participation in lung cancer screening (LCS) among eligible smokers in the United States (US) has been limited to 6.6% on average in 2019 and remained the same in 2020. 7 When looking at the US state-level LCS uptake rates; nine states reported to have more than 10% participation rate. 7 In contrast, population-based LCS programmes in several Chinese provinces showed high uptake rates of 34%-52%.8–10 The high uptake in China is believed to be owing mainly to the accessibility and affordability of LDCT scans across many provinces. 11 While LCS in the UK is not yet implemented on the national level, data from population-based targeted screening in Liverpool and Manchester showed high participation rates of 40% and 26%, respectively.12,13 The presence of chronic diseases may positively impact participation in screening programmes because comorbid individuals might have direct contact with healthcare systems and services, leading to more opportunities for referral to or engagement with screening services primarily when LCS is widely implemented such as in the US. 14 However, the presence of comorbidities might limit management options for screen-detected lung cancer (mainly surgical resection), thereby impacting the effectiveness of treatments and patient outcomes. 15 Also, reduced life expectancy related to comorbidity would limit the life-years gained through early detection by screening, especially in people with advanced or life-threatening illnesses. 16 The burden of comorbidity in LCS candidates has not previously been estimated. We systematically reviewed the literature and utilised meta-analysis to obtain an overall estimate of the prevalence of comorbidities among high-risk populations who were selected for LCS. Additionally, LCS guidelines and recommendations were identified and summarised with respect to how they addressed comorbidities and frailty.
Methods
Search strategy and inclusion criteria
A systematic search was conducted in February 2021 to identify evidence regarding the comorbidity and frailty status of LCS (or screen-eligible) participants. The review protocol is registered on the PROSPERO database (Registration number CRD42021237040) and is available from https://www.crd.york.ac.uk/prospero/display_record.php?RecordID=237040. The search was conducted using MEDLINE (OVID), EMBASE (OVID), EBM reviews- Cochrane Database of Systematic Reviews (OVID) and CINAHL (EBSCO) databases. The timeframe was from 1 January 1990 to 8 February 2021 with no language restriction, using a strategy of subject headings and free text words (Appendix 1, see online supplementary files). The timeframe of 1990 was used as a start point to adequately capture studies investigating the utilisation of LDCT for LCS. We excluded case reports, case series, modelling studies, qualitative studies, conference abstracts, reviews, commentary, and editor letters. Titles and abstracts were screened for eligibility using Rayyan software. 17 Studies were considered for inclusion if they were 1) conducted in a LCS setting that included screened or high-risk eligible participants, and 2) reported comorbidity or frailty status; either as a prevalence of a specific condition, proportion, or a summary score (e.g. the Charlson Comorbidity Index (CCI)).18,19 A 20% sample of the total identified titles and abstracts was doubled screened by a second reviewer (OT), and disagreements were resolved through consensus. We have also considered and included clinical guidelines and recommendations that addressed comorbidity or frailty in the context of LCS as a supplementary goal of the review. No specific keywords were added to identify clinical guidelines as we relied on our main keyword strategy to include these articles.
Data extraction and quality assessment
Data were extracted using a standardised data extraction form by one author (AA), and a 20% sample of extracted data was independently checked by another author (OT). Comorbidity prevalence was calculated as the number of people with the condition (numerator) divided by the total sample size (denominator). When studies reported only a proportion of a particular comorbidity, the numerator was converted to absolute numbers. Individual comorbidities were chosen based on their 1) clinical relevance to lung cancer (chronic obstructive pulmonary disease (COPD), chronic bronchitis, bronchiectasis, asthma, and pulmonary fibrosis), 2) competing nature in elevating the risk of death (stroke, peripheral vascular disease (PVD), and ischaemic heart disease (IHD)), or 3) frequent reporting in the included studies (type 2 diabetes and hypertension). We utilised the Cochrane risk of bias tool (RoB 2) to assess the quality of randomised control trials. 20 For observational studies (cohort and case-control studies), the Newcastle-Ottawa Scale (NOS) was used to assess the risk of bias. 21 A modified version of NOS was used for the quality assessment of cross-sectional studies. 22
Meta-analysis
The meta-analysis was conducted by using the metaprop command in STATA (version 16.1) to provide an overall pooled estimate (proportion) with inverse-variance weights obtained from a random-effects model. Confidence intervals were computed using the score method, 23 and heterogeneity across studies was evaluated using I2 statistic. 24
Results
The initial search identified a total of 5439 records. After removing duplicates, screening titles and abstracts, and excluding not retrieved studies, 335 were included for full-text screening. Two hundred and sixty-six studies were further excluded after the full-text screening, leaving 69 studies that met our inclusion criteria and were considered for final qualitative and quantitative synthesis (Figure 1). Of the 69 included studies, seven reported comorbidity using summary measure scores (Table 1), two reported performance status (Table 1), 48 reported individual comorbidities (Table 2), and 12 publications highlighted the recommendations of undergoing LCS for comorbid or frail individuals (Table 3).
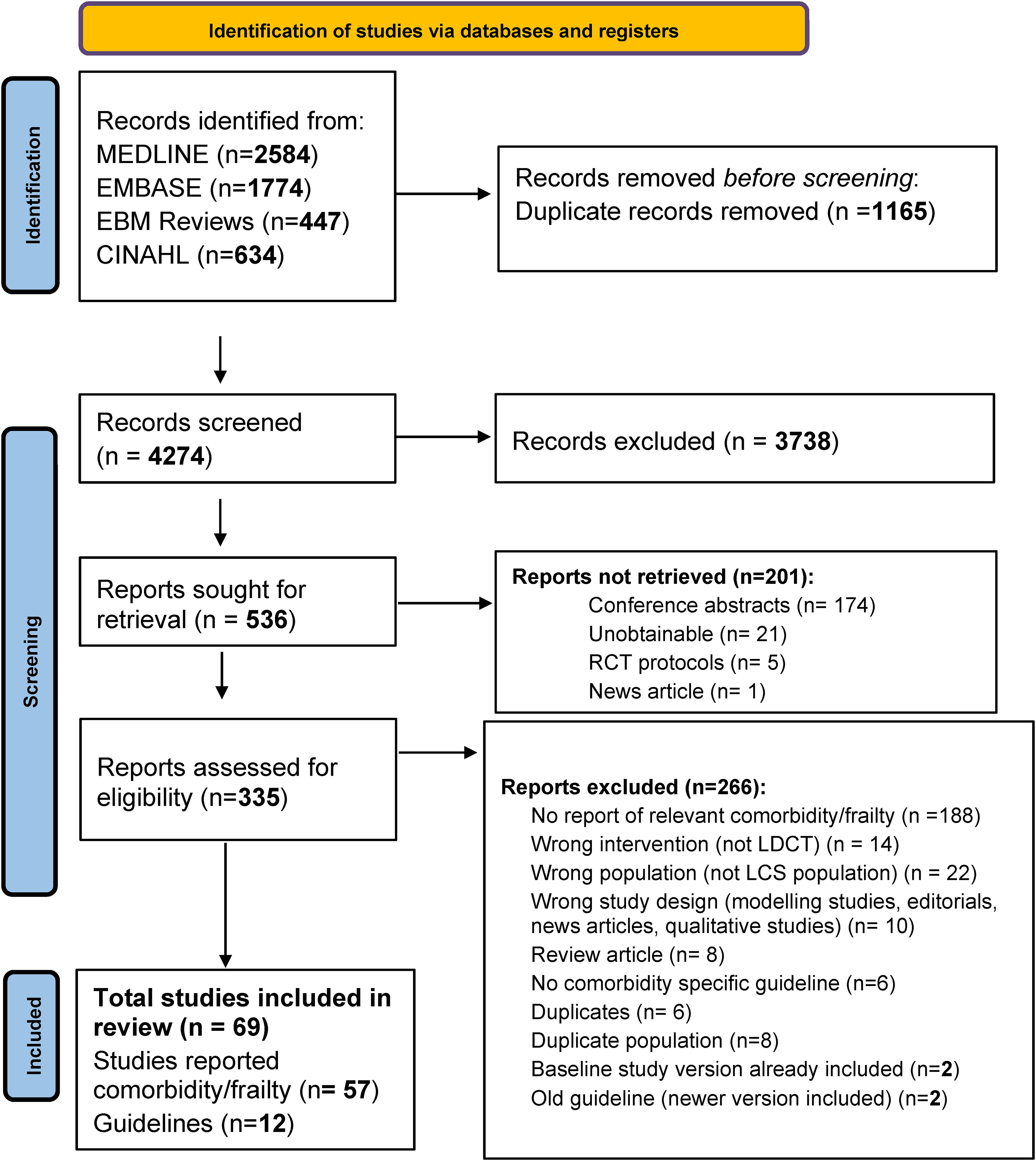
Flow diagram of the selection process according to the PRISMA 2020 statement.
Studies that reported comorbidity/frailty measure score.
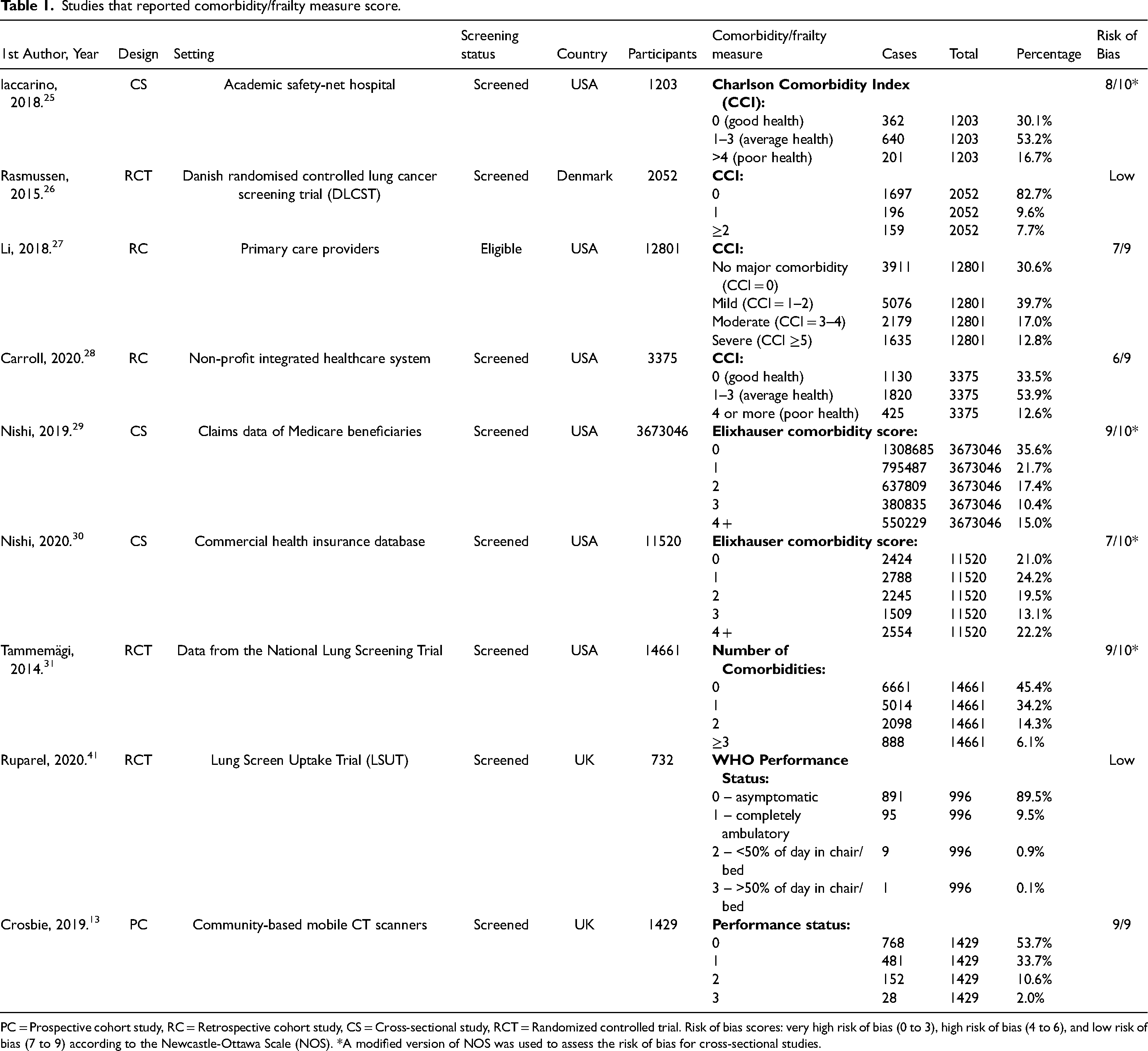
PC = Prospective cohort study, RC = Retrospective cohort study, CS = Cross-sectional study, RCT = Randomized controlled trial. Risk of bias scores: very high risk of bias (0 to 3), high risk of bias (4 to 6), and low risk of bias (7 to 9) according to the Newcastle-Ottawa Scale (NOS). *A modified version of NOS was used to assess the risk of bias for cross-sectional studies.
Studies that reported individual comorbidities.
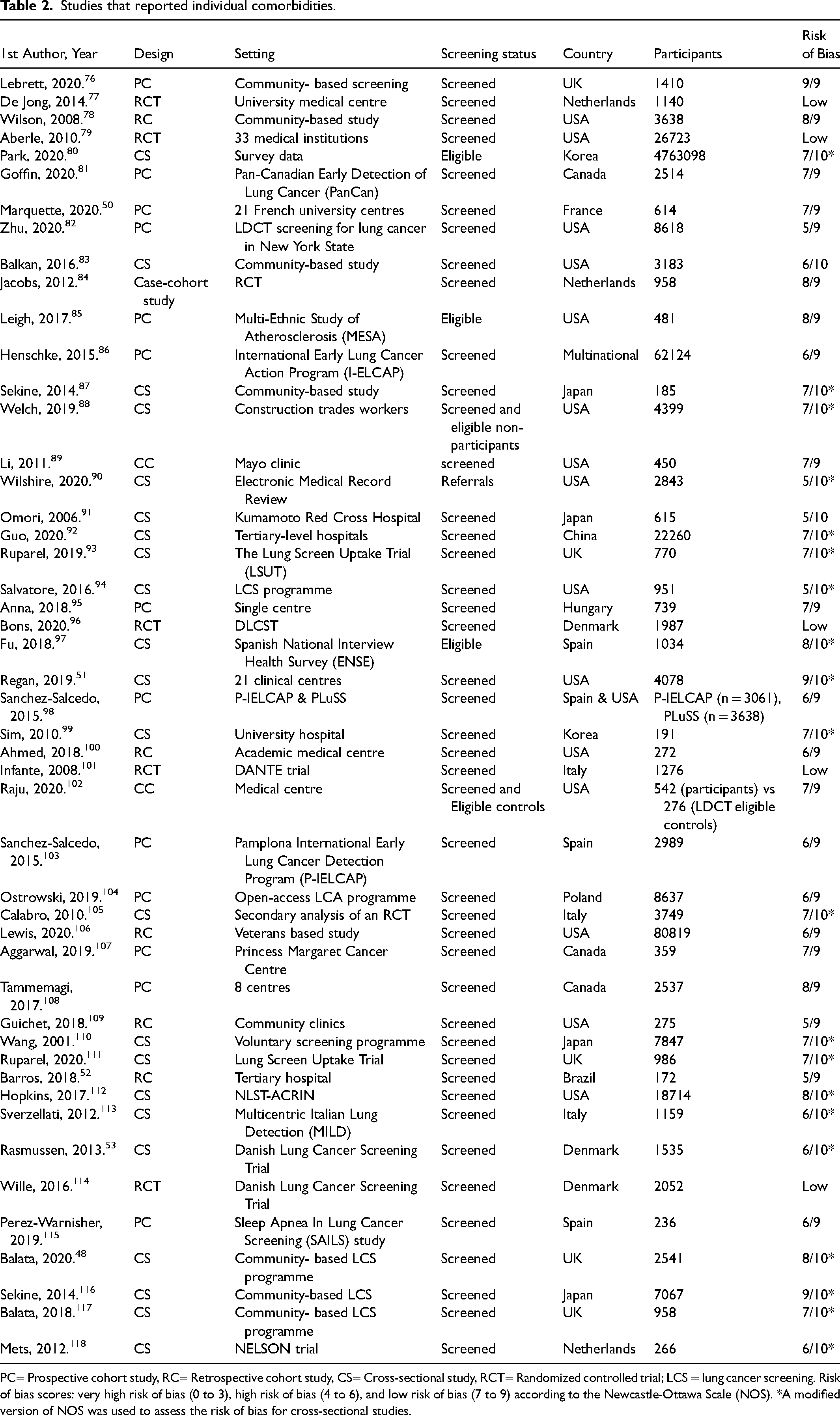
PC= Prospective cohort study, RC= Retrospective cohort study, CS= Cross-sectional study, RCT= Randomized controlled trial; LCS = lung cancer screening. Risk of bias scores: very high risk of bias (0 to 3), high risk of bias (4 to 6), and low risk of bias (7 to 9) according to the Newcastle-Ottawa Scale (NOS). *A modified version of NOS was used to assess the risk of bias for cross-sectional studies.
Clinical guidelines that address the issue of comorbidity among LCS candidates.

When reporting comorbidity using summary measure scores, four studies used the CCI,25–28 two used the Elixhauser Comorbidity Index,29,30 and one study used a simple comorbidity count. 31 We could not perform a meta-analysis of the four studies that utilised CCI as two studies used different sub-categories. The search results did not retrieve any study that directly utilised an established frailty measure, although two studies reported performance status.
Studies that reported individual comorbidities
Most studies were conducted in North America (42%) and Europe (42%), with only seven studies (12%) conducted in Asia, one in Brazil, and one was multinational. The review included studies varied in design with 26 cross-sectional studies, 22 cohort studies, seven randomised controlled trials (RCTs), and two case-control studies. All RCTs were found to have a low risk of bias. The majority of non-randomised studies (32 studies) were rated as having a low risk of bias (scored between 7 and 10) based on the NOS system, and 18 studies were rated as having a high risk of bias (scored 5 and 6).
Pooled estimations of the prevalence of individual comorbidities among LCS populations are presented in forest plots and included in the supplementary files (Appendix 2). The results show that the estimated prevalence of individual comorbidities sequentially ordered by proportion was: hypertension (35.2%, number of reported cases (c) = 1,498,429, total screening population (n) = 4,812,180), history of COPD (23.5%, c = 9868, n = 67,662), chronic bronchitis (17.2%, c = 31,329; n = 92,102), IHD (16.6%, c = 22,236, n = 94,379), PVD (13.1%, c = 327, n = 2648), asthma (12.9%, c = 4043, n = 36,134), diabetes (12.5%, c = 93,6813, n = 4,822,167), advanced COPD (stage 3 and 4) (10.7%, c = 3747, n = 35,778), bronchiectasis (4.5%, c = 13,221, n = 86,530), stroke (2.2%, c = 1068, n = 42,004), and pulmonary fibrosis (0.55%, c = 102, n = 33,072).
Findings from clinical guidelines
Findings regarding recommendations for screening comorbid individuals set by clinical guidelines are summarised and presented in Table 3. The American Association for Thoracic Surgery (AATS) 32 and The National Comprehensive Cancer Network (NCCN) 33 guidelines do not endorse LCS in individuals with limited functional status or comorbidity that might affect potential curative treatment. Other guidelines such as the American College of Chest Physicians (CHEST), 34 the European Society of Radiology (ESR) and European Respiratory Society (ERS), 35 and the Canadian Thoracic Society (CTS) 36 guidelines condition the screening of comorbid people on their ability to undergo curative treatment without considering functional status or frailty. The USPSTF 37 and the Chinese national 38 LCS guidelines specify the ability to undergo lung surgery as a criterion for screening individuals with comorbidity or serious health illness. In addition, a consensus statement from Poland recommends that the decision to undergo LCS should be a shared one between the physician and patients with comorbidities, 37 Unlike other guidelines, the International Association Study Lung Cancer (IASLC) guideline was the only one that recommended performing fitness assessments for high-risk individuals before their enrolment in LCS programmes. 39 Finally, the AATS organised a multidisciplinary panel in 2017 that emphasised the need for future research on incorporating comorbidities and functional status in selecting candidates for LCS. 15
Discussion
This systematic review is the first to estimate the prevalence of comorbidities among LCS populations, evaluating 57 studies and 12 clinical guidelines. Most of the included studies (84%) were from western countries (Europe and North America), and only 12% were conducted in Asia. The total number of included studies in our review is fewer than the total number of LCS studies available in the literature because we find that not all LCS programmes report comorbidity or frailty data. Despite using a comprehensive search strategy, we found only a small fraction of included studies (10%) used validated comorbidity indices, and none utilised a pre-screening validated frailty assessment tool. However, two studies (24, 25) utilised performance status, which might be a proxy for frailty. These findings highlight the underutilisation of comorbidity and frailty measures among LCS population.
Even though only seven studies used comorbidity summary scores, we found that proportions of people without comorbidity (CCI = 0) were higher in clinical trial settings (83% and 45%)26,31 compared to population-based screening settings (range: 30%-33%),25,27,28 indicating a potential role of healthy volunteer effect in LCS trials. 40 The same observation applies to studies that utilised WHO performance status; one RCT 41 reported asymptomatic performance status (score 0) in about 90% of participants compared to 54% from a community-based LDCT screening programme. 13
The comorbidity profile of LCS participants differs from those reported in breast and colorectal cancer screening programmes. The proportion of individuals without comorbidity (CCI = 0) reported previously in large breast cancer (56%, 42 76%, 43 84%, 44 and 93% 45 ) and colorectal cancer (65%, 46 52% 47 ) screening studies is much higher than what we observed in LCS studies included in our review (30%-33%).25,27,28 A possible explanation is that breast and colorectal screening programmes sample from the entire community with age as the primary risk factor. In contrast, lung cancer screening relies more on smoking as the leading risk factor, which is associated with comorbidities. This observation highlights the need for more research and innovations to deliver LCS to those with greater life expectancy, considering the presence of comorbidities.
The prevalence of history of COPD was also considered for meta-analysis, resulting in a pooled estimate of 23.5% (95% CI: 16.5, 31.4) from 11 studies. The sub-group analysis by data collection method indicated significant heterogeneity (p < 0.001). The estimated prevalence from studies that utilised health records to identify the history of COPD was 38.7% (95% CI: 31.5, 46.2, I2 = 95.6%, p < 0.001) compared to 16.9% (95% CI: 10.5, 24.5, I2 = 99.7%, p < 0.001) pooled from studies that relied on self-reporting. This finding suggests that relying on self-reporting of COPD may underestimate the true burden of COPD in a LCS population, potentially misclassifying participants having low risk when using lung cancer risk-prediction methods that incorporate COPD (i.e. PLCOM2012).48,49 We also estimated the prevalence of advanced COPD, stages 3 and 4, to be 10.7% (95% CI: 6.1, 16.4), as reported in 12 studies using the Global initiative for chronic Obstructive Lung Disease (GOLD) criteria (www.GOLD.org). Most studies reported a prevalence of advanced COPD of less than 10%, while four reported a prevalence of advanced COPD above 20% (range: 20.3%-31.0%).50–53 Screening individuals with advanced COPD remains controversial as they may not benefit from screening due to inoperability and an increased risk of respiratory and other competing causes of death.54,55 The utilisation of functional assessment tools, such as the BODE index, 56 is suggested to be a better way of assessing the severity and fitness of patients with advanced COPD by incorporating not only the degree of airflow obstruction but also functional dyspnea, body-mass index, and exercise capacity.15,56
We also estimated the prevalence of four lung diseases: chronic bronchitis, asthma, bronchiectasis, and pulmonary fibrosis. The pooled estimate for chronic bronchitis was 17.2% (95% CI: 5.5, 33.0) among the LCS population and considered higher than what is usually found in the general population, which is around 3%.57,58 The increasing age and smoking habits could explain the higher prevalence of chronic bronchitis in the LCS population. 59 Estimate of asthma prevalence was 12.9% (95% CI: 8.3, 18.3), which is more than what is normally observed in the general population.60,61 A similar observation was reported by Zahnd et al. 62 when they found that individuals with asthma tend to utilise LCS more than those without asthma (22.9% vs. 12.9%, p = 0.006). Bronchiectasis and pulmonary fibrosis were less prevalent with pooled estimates of 4.5% (95% CI: 0.4, 12.4) and 0.55% (95% CI: 0.18, 1.10), respectively. Previous studies, not captured by our search as we did not include bronchiectasis in our search strategy, reported a prevalence of bronchiectasis ranging from 0.2% to 16%.63–66 The higher presence of bronchiectasis in the LCS population is suggested to be associated with a higher incidence of new nodules and false-positive results on both baseline and subsequent screening rounds. 67
In addition to respiratory comorbidities, we estimated the prevalence of ischemic heart disease (IHD), PVD, and strokes as conditions that may have a competing cause of death in the LCS population. We found that IHD was prevalent in 16.6% (95% CI: 11.0, 23.0) of screened candidates across eight studies (9 estimates). The estimated prevalence of IHD in our review is more than twice the prevalence of IHD found in the general population of the United States 68 and the UK. 69 Smoking habits and age might explain the elevated IHD prevalence in the LCS population compared to the general population.
The second part of this systematic review is related to the inclusion of comorbidity and frailty in LCS guidelines. Overall, clinical guidelines were generally vague and did not fully address comorbidity or frailty in their LCS recommendations. The ability to tolerate curative treatment before undertaking LCS was agreed upon as an inclusion criterion among most included guidelines. Only the USPSTF 37 and the Chinese 38 guidelines recommended LCS for people who could withstand lung surgery. The ability to tolerate treatment or withstand surgery is not well defined across clinical guidelines, with little information about how physicians should communicate the benefits and harms of LCS to their patients who have frailty or severe comorbidity. The American Thoracic Society issued a research statement in 2018 acknowledging this dilemma, and outlined future research directions to incorporate the severity of comorbidities and functional status into the selection process. 15
Our review highlighted the current scarcity of pre-screening frailty or functional assessment tools in LCS programmes. Previous studies have demonstrated the association of frailty with poor cancer screening consequences, 70 postoperative complications,71,72 and higher mortality of non-cancer causes.73,74 In addition, the prevalence of frailty among lung cancer patients of different stages was recently estimated to be 45%, with a significant adverse impact on survival. 75 Future LCS programmes and research should invest in this area by examining the feasibility and usefulness of incorporating a pre-screening assessment of frailty and comorbidity severity.
To the best of our knowledge, this study is the first to estimate the burden of comorbidity among LCS candidates to inform researchers and policymakers about the magnitude of this public health problem. The strengths of this review include utilising a comprehensive search strategy and including a large number of studies. The variability of the methods used to report single comorbidities is a potential limitation of this review, as most studies relied on self-reporting of comorbidities, with only a few studies utilising medical records and administrative databases. As a result, the pooled prevalence estimates should be interpreted with caution. Another limitation is the observed high heterogeneity between studies, and accounting for different study locations and designs didn’t explain this observation. In addition, there might be other clinical guidelines not covered by our review as we didn’t incorporate guidelines-specific keywords in our search strategy.
Conclusion
In this study, we reviewed 57 studies and 12 clinical guidelines to estimate the prevalence of comorbidities in LCS populations and summarise the clinical recommendations for screening comorbid individuals. Detailed prevalence of selected comorbidities was reported. LCS is an essential element of early detection and cancer control and will likely become more available to high-risk individuals in the coming years. Before widely implementing LCS, identifying subpopulations with a high burden of comorbidities and frailty who would be less likely to benefit from screening should become a priority. To optimise the benefits of screening and increase its cost-effectiveness, future LCS research needs to incorporate existing comorbidity and frailty measures and develop new approaches for personalising the selection process.
Supplemental Material
sj-docx-1-msc-10.1177_09691413221117685 - Supplemental material for The prevalence of comorbidity in the lung cancer screening population: A systematic review and meta-analysis
Supplemental material, sj-docx-1-msc-10.1177_09691413221117685 for The prevalence of comorbidity in the lung cancer screening population: A systematic review and meta-analysis by Anas Almatrafi, Owen Thomas, Matthew Callister, Rhian Gabe, Rebecca J Beeken and Richard Neal in Journal of Medical Screening
Supplemental Material
sj-docx-2-msc-10.1177_09691413221117685 - Supplemental material for The prevalence of comorbidity in the lung cancer screening population: A systematic review and meta-analysis
Supplemental material, sj-docx-2-msc-10.1177_09691413221117685 for The prevalence of comorbidity in the lung cancer screening population: A systematic review and meta-analysis by Anas Almatrafi, Owen Thomas, Matthew Callister, Rhian Gabe, Rebecca J Beeken and Richard Neal in Journal of Medical Screening
Supplemental Material
sj-xlsx-3-msc-10.1177_09691413221117685 - Supplemental material for The prevalence of comorbidity in the lung cancer screening population: A systematic review and meta-analysis
Supplemental material, sj-xlsx-3-msc-10.1177_09691413221117685 for The prevalence of comorbidity in the lung cancer screening population: A systematic review and meta-analysis by Anas Almatrafi, Owen Thomas, Matthew Callister, Rhian Gabe, Rebecca J Beeken and Richard Neal in Journal of Medical Screening
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Data Availability
Prevalence data extracted from all articles are available as a supplementary file (SR_data.xlsx).
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
