Abstract
Objective
The results of recent studies have shown that using low-dose computed tomography (LDCT) for screening of lung cancer (LC) improves cancer outcomes. The objective of the current study was to evaluate the cost-effectiveness of LDCT in an Iranian high-risk population.
Methods
A Markov cohort simulation model with four health states was used to evaluate the cost-effectiveness of LDCT from a healthcare system perspective in the people aged 55–74 who smoked 25 or more cigarettes per day for 10–30 years. Cost data were collected, reviewing 324 medical records of patients with LC, and utilities and transition probabilities were extracted from the literature. The Monte Carlo simulation method was applied to run the model. Probabilistic sensitivity analysis and one-way analysis were also performed.
Results
LC screening in comparison to a no-screening strategy was costly and effective. The incremental cost-effectiveness ratio of screening versus no-screening was IRR (Iranian rials) 98,515,014.04 which falls below the Iranian threshold of three times GDP (gross domestic product) per capita. One-way and probabilistic sensitivity analyses demonstrated that the results of the economic analysis were robust to variations in the key inputs for both.
Conclusions
Using LDCT for screening of LC patients in a high-risk population is a cost-effective strategy.
Background
Cancer is a world-wide health problem, not least in developing countries.1 –3 Lung cancer (LC), causing 18.4% of total cancer deaths, is the most deadly form, for both genders. It has also the highest incidence across the globe with 2.1 million new cases each year. 4 Cancer registries from the different countries in the Middle East region show that LC is one of the most common cancers in the region. 5 The Iranian National Population-Based Cancer Registry reported that LC was one of the most common cancers among men, accounting for 12.7% of all cancers.6,7 Unfortunately, LC, like many other cancers, is often diagnosed at an advanced stage, and the proportion of patients treated at first stage is less than 50% in the majority of thoracic surgical centers around the world. Moreover, only 15% of patients are still alive five years after diagnosis. 8 Diagnosing LC at an early stage has implications for better treatment prognosis. Thus, screening to achieve early diagnosis of asymptomatic cases is of great importance. Theoretically, it is expected that the screening of LC will lead to a reduction in the incidence of the advanced stages and mortality.
Some aspects associated with LC, such as being an important health problem, gradual growth, being diagnosable at early stages and availability of effective interventions at early stages, 8 make this disease a suitable candidate for a screening program. 9 Several strategies have been proposed for LC screening including: chest radiography (CXR), sputum cytology and low-dose computed tomography (LDCT). Studies that have examined the benefits of screening using different strategies have demonstrated the superiority of LDCT over the other two methods.10,11
The sensitivity and specificity of diagnostic strategies play a major role in the acceptability of these methods in cost-effectiveness analysis (CEA). In the National Lung Screening Trial (NLST), the performance of LDCT was compared with CXR in reducing lung cancer mortality in the United States in 2012. LDCT screening was proven to detect more early stage lung cancers and led to fewer pneumonectomy procedures. Also, the NLST revealed a mortality rate reduction of 20% in patients who had undergone LDCT screening. 9 Another study, the non-randomized international Early Lung Cancer Action Program (I-ELCAP) in 2006, showed that it was possible to detect early stage IA LCs using LDCT with a predicted 10-year survival rate of 88%. 12 The Nederlands–Leuvens Longkanker Screenings Onderzoek (NELSON) trial showed that LC mortality was significantly lower among those who underwent LDCT screening than among those who underwent no screening. 13
The high costs of delivering population screening programs necessitate the conducting of CEAs. Several studies have been conducted to evaluate the cost-effectiveness of LDCT screening around the world. In performing a CEA, some context-specific parameters play an essential role in making a conclusion about an intervention. This study aimed to evaluate the cost-effectiveness of LDCT screening as a method to detect early stage LC within the healthcare system of Iran.
Methods
Intervention, target population and perspective
This is a model-based cost-utility analysis that compares the screening of lung cancer using LDCT with a no-screening strategy in terms of the costs and effectiveness. Only patients diagnosed with non-small cell lung cancers (NSCLC) (ICD-O-3 codes 8010, 8012, 8070, 8071, 8072, 8140, 8481 and 8560) were included in the analysis. The study was conducted from a health system perspective. The target population was people aged 55–74 who smoked 25 or more cigarettes per day for 10–30 years. Screening was assumed to be implemented every three years for this target population.
Summary of data
Cost
Three steps were taken to estimate costs: identifying, measuring and valuation of resources. Two types of costs were obtained for this analysis. Firstly, the cost of performing a screening test with LDCT per person was estimated. The paths for positive and negative cases were followed based on the protocols, and subsequent confirmation tests for positive cases were also included. Secondly, the average treatment cost for each LC state (Figure 1) was estimated by reviewing the medical records of a sample of 324 patients with lung cancer who were admitted to the Ghazi Hospital in Tabriz, Iran. This is a public, educational and specialized hospital for treatment of patients with all types of cancers. These costs were differentiated for a period of one year from the beginning of each state and for the second and following years. All valuations of resources were estimated using hospital bills based on the public hospital tariffs (in Iranian rials, IRR). The treatment costs included outpatient visits, hospitalization, chemotherapy, radiotherapy and supportive care.
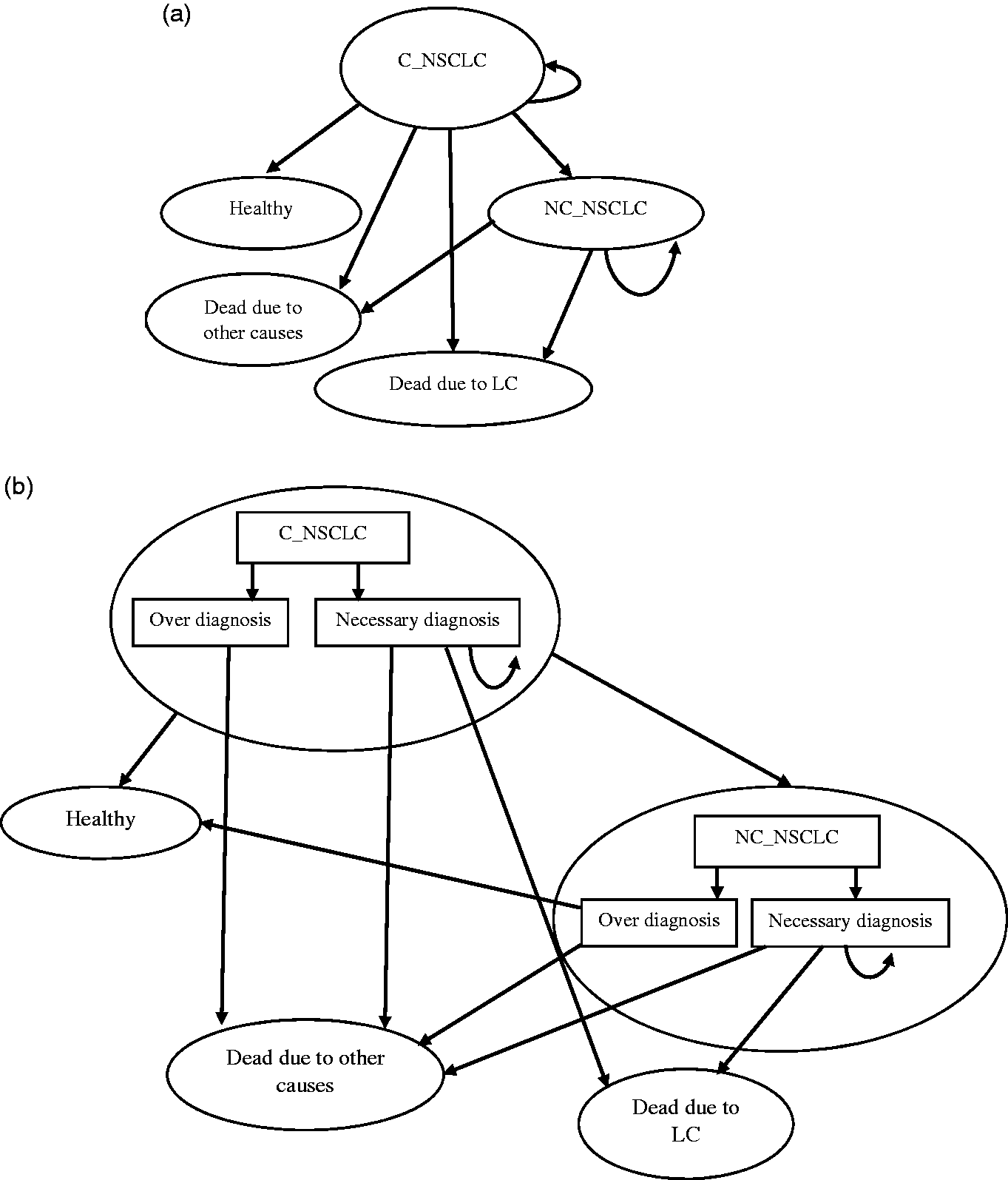
Markov state transition model of NSCLC: (a) no-screening arm; (b) screening arm (including overdiagnosis).
Accuracy
The accuracy of LDCT was assessed according to the true-positive rate (sensitivity) and true-negative rate (specificity) for cancer detection. We carried out a systematic review, the results of which will be published elsewhere, using the value of these parameters obtained from previous studies.14,15
Utility weights
Quality of life was measured in utility terms, which is a measure of preference for a given health state rated on a scale, where 0 equals death and 1 equals perfect health. 16 Quality-adjusted life years (QALYs) were calculated using the utility weights associated with each state. These utility weights were obtained from published literature. 17
Transition probabilities
Transition probabilities represent the likelihood of transition of patients among the different states of the model during a cycle. These parameters were obtained from published studies and by expert opinion in accordance with the natural progression of LC. 8
Basic model
The Markov model was designed based on the following states: curative non-small cell lung cancer (C-NSCLC), non-curative non-small cell lung cancer (NC-NSCLC), death due to lung cancer, death due to other causes and healthy. These states were defined based on the earlier model developed by Cressman et al. 4 Patients with stage IA to IIB LC, and those with stage IIIA LC who undergo surgery as part of treatment are assigned to C-NSCLC. Patients with stage IIIB to stage IV LC, and those with stage IIIA LC without undergoing surgery constitute the NC-NSCLC state.
The comparative advantage of screening over no screening has been shown in the distribution of patients as severe (NC-NSCLC) or mild (C-NSCLC). In the screening arm, the proportion of patients in the mild state is higher. In the false negative branch, it is assumed that the distribution of patients in the Markov states is the same as in the no-screening arm. After identifying the patients, the rate of disease progression to other states in the screening and no-screening arms is assumed to be the same. The diagnostic pathway for false positives involves performing a pathology test immediately after being found positive with LDCT. Therefore, for people who falsely tested positive, in addition to the cost of screening, we included the cost of the final pathology test. The overdiagnosis of cases, as one of the main concerns of screening, has been well documented for LDCT. 18 We have accounted for this within the model by incorporating the overdiagnosis rate to drive out the effect of overdiagnosis on the clinical and economic path. The parameter of overdiagnosis rate for this study was obtained from previous studies.18,19
A one-year cycle length was considered for running the model. A discount rate of 3.5% was used for both costs and QALY per year. A Markov state transition model and tree are displayed in Figures 1 and 2, respectively.
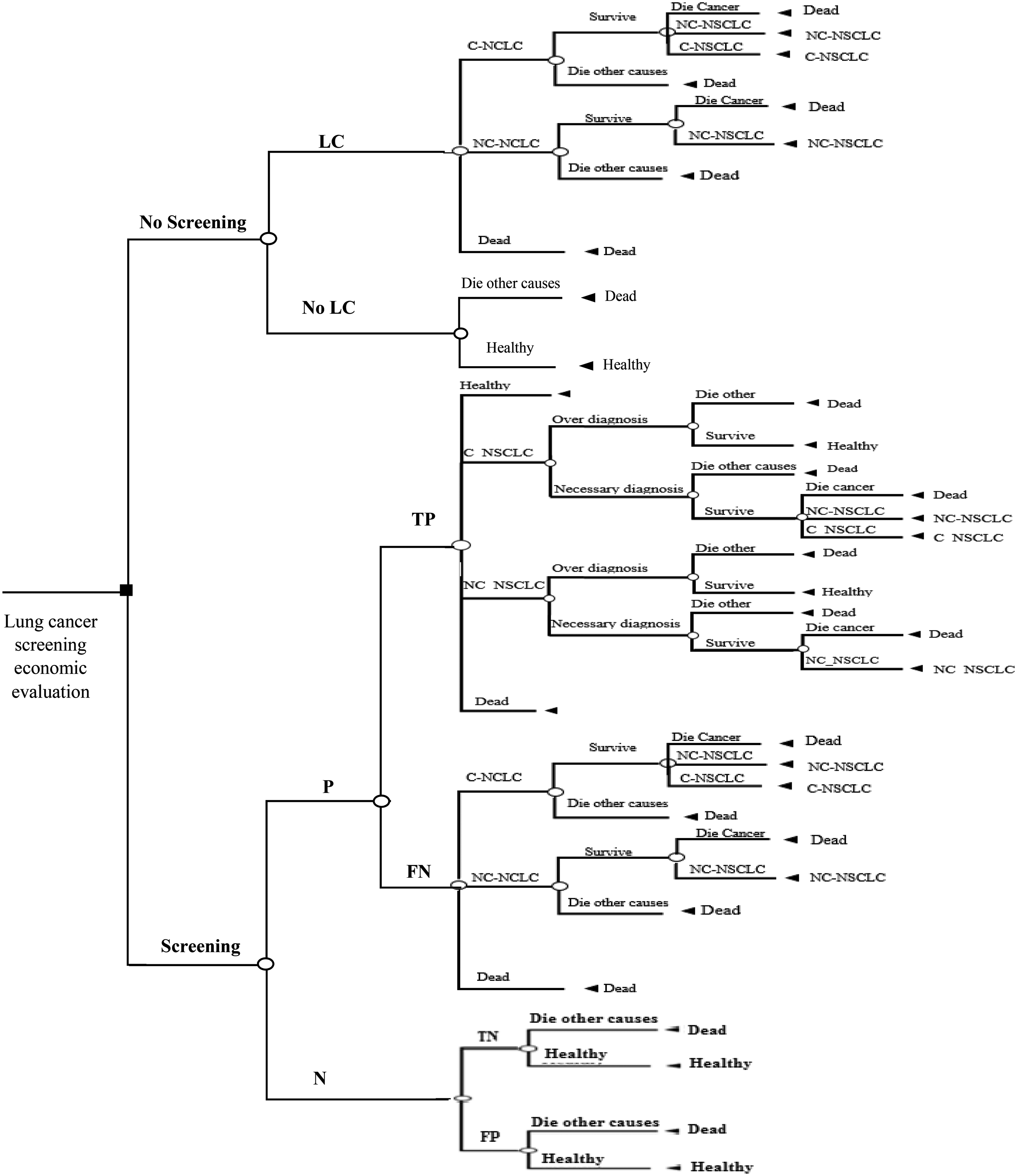
Decision tree for screening versus no-screening. P: positive; N: negative; T: true; F: false.
Uncertainty
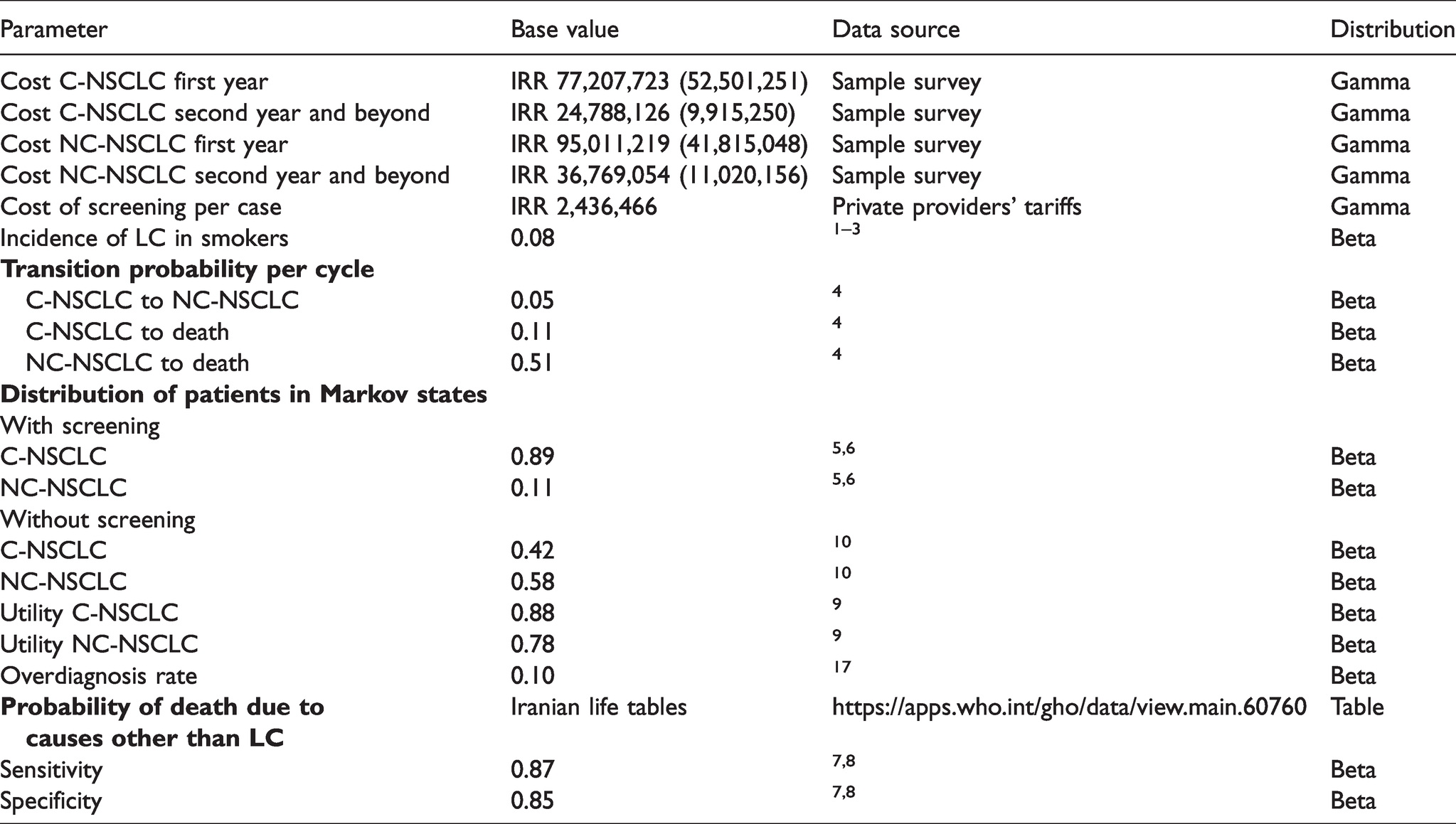
Probabilistic sensitivity analyses were carried out by Monte Carlo simulation to examine the impact of variation for all key variables (Table 1). To reflect the uncertainty in each parameter, distributions were specified based on the mean values and standard errors per parameter. Beta distribution was used for utility values, discounting rates, and transition probabilities and incidence of lung cancer. The Gamma distribution was used for the costs. The probabilistic sensitivity analysis simulation was conducted with 500 reiterations. We used the TreeAge Pro Healthcare software for data modeling. In addition, a one-way sensitivity analysis was performed for the parameters of the initial distribution of the patients in different states for both screening and no-screening strategies within the plausible variation range (±20%) of those parameters. Furthermore, due to variation in the reported rates for overdiagnosis, a one-way sensitivity analysis was performed for this parameter in the range of 5% to 38%.
Health state costs, transition probabilities between health states, distribution of patients in Markov model and probability of death.
Results
Table 2 shows the costs associated with surgery, chemotherapy and radiotherapy services, as well as diagnosis tests. Total costs for NC-NSCLC are higher than those for C-NSCLC.
Cost of interventions.

Cost-effectiveness analysis
The results of the CEA are summarized in Table 3. It illustrates the overall costs and QALYs obtained for the LDCT strategy every three years in comparison with no-screening strategy. The calculations indicate a positive incremental cost of IRR 2,681,590.821 and a positive incremental QALY of 0.02722 leading to an incremental cost-effectiveness ratio (ICER) of IRR 98,515,014.04 per QALY.
Comparative cost-effectiveness results of Monte Carlo simulation (with overdiagnosis).

Cost-effectiveness acceptability curve
Figure 3 illustrates the likelihood of being cost-effective for the two strategies within the given range of willingness-to-pay threshold values on the horizontal axis. At lower threshold values, the no-screening strategy was cost effective but as the threshold increases the probability of this strategy being cost-effective decreases, and vice versa for the LDCT screening strategy.
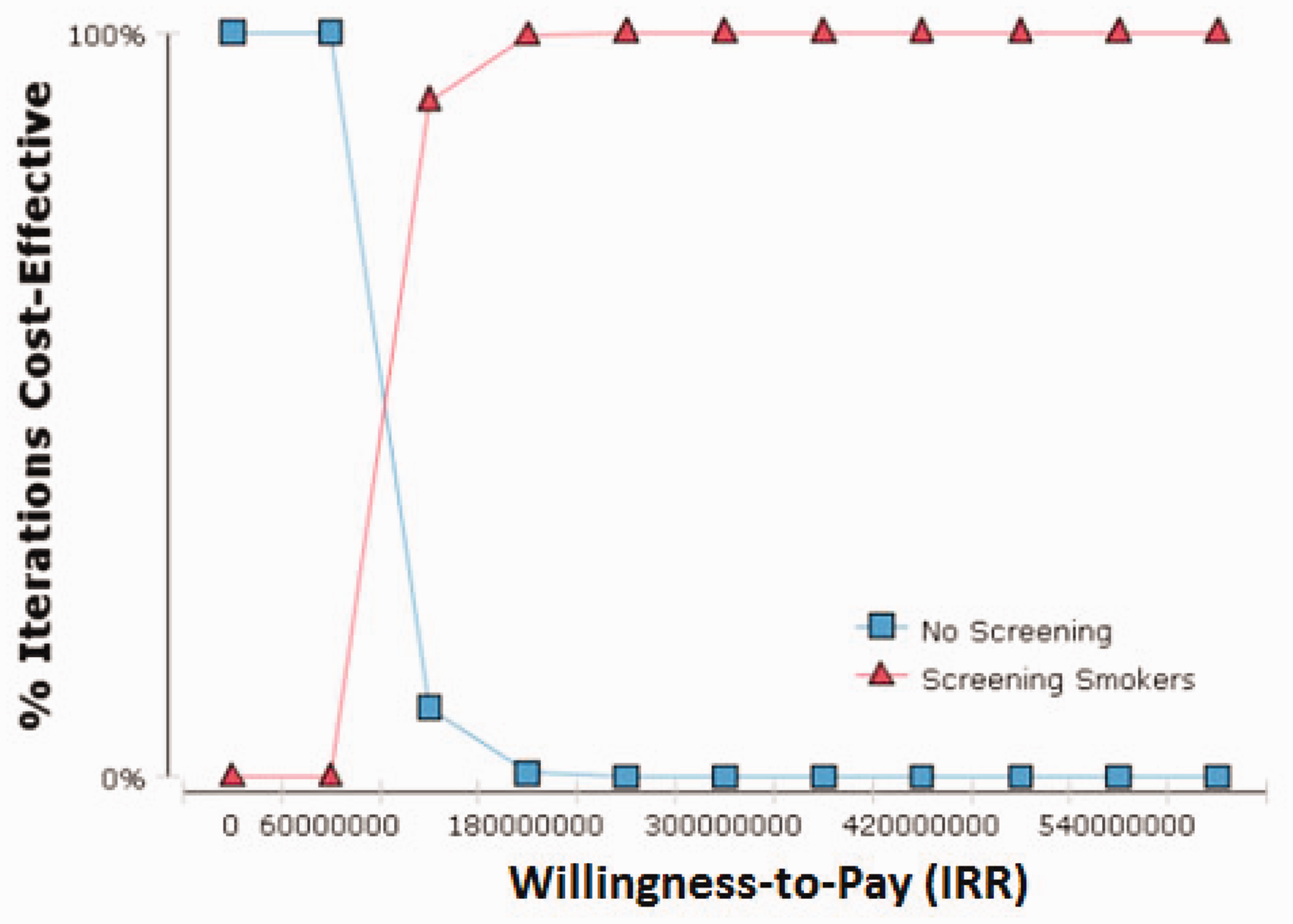
Cost-effectiveness acceptability curves.
Figure 4 illustrates probabilistic sensitivity analysis using the Monte Carlo simulation. This analysis showed that at the identified willingness-to-pay threshold, almost all ICERs are lower than the presumed threshold. In all cases, the results of one-way sensitivity analysis of the parameters of the initial distribution of the patients in different states for both screening and no-screening arms remained robust under the defined threshold (Appendix 1, see Online Supplemental Material).
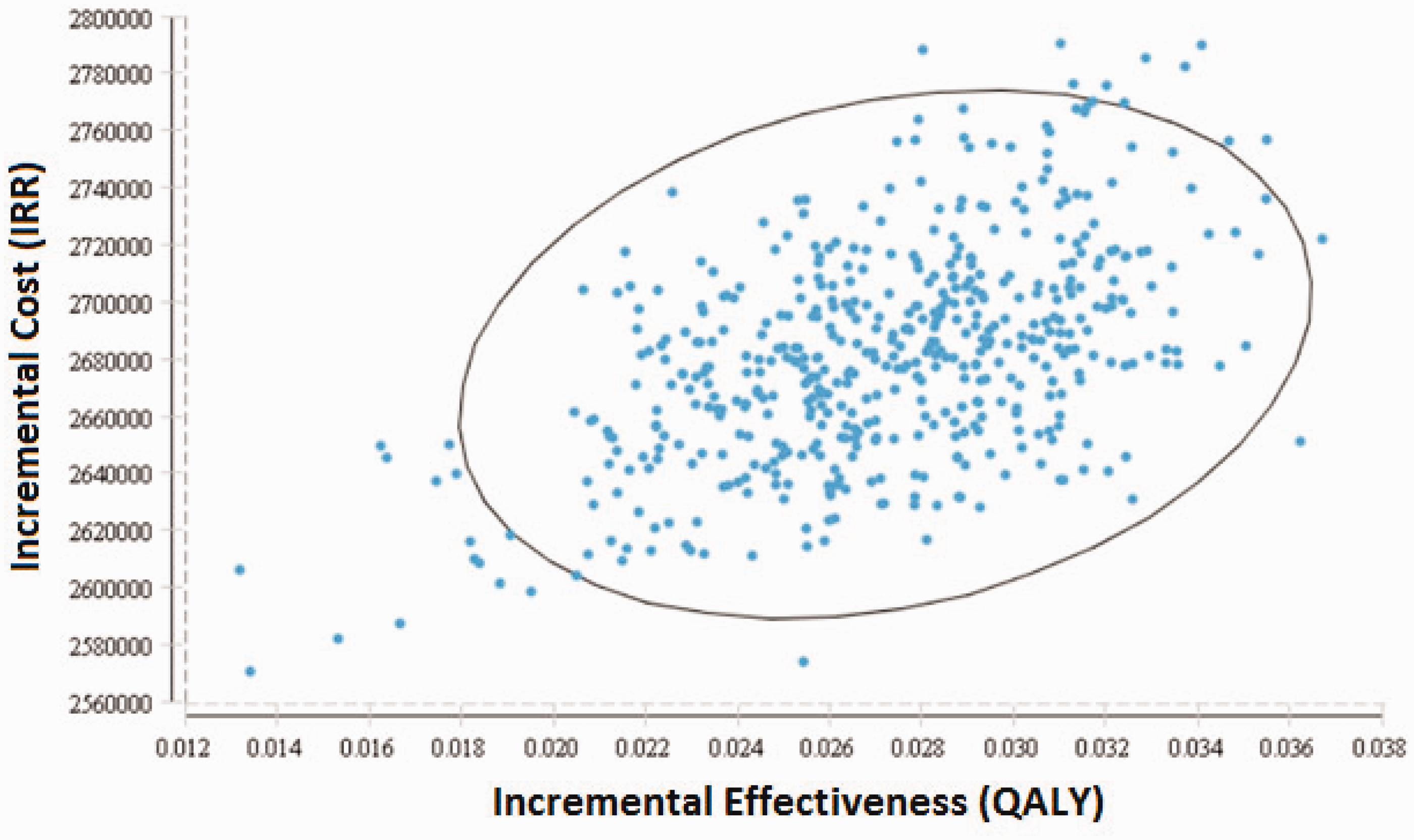
Cost-effectiveness sensitivity analysis for both strategies (LDCT screening and no screening).
Discussion
LC imposes a great economic burden worldwide and is one of the leading causes of cancer deaths in Iran. Screening using LDCT is one of the promising interventions for early diagnosis of LC among high-risk smokers. Scarcity of resources means that the economic analysis of LC screening must be an integral part of the decision-making process. This is, to the best of our knowledge, the first cost-utility analysis conducted to examine the cost-effectiveness of LDCT in patients with LC in Iran.
The comparison of screening, using LDCT, with a no-screening strategy shows that the detection of LC by means of screening is both effective and expensive. For LDCT, in comparison with no screening, an ICER of IRR 98,515,014.04 per QALY was obtained. The criterion for assessing cost-effectiveness was based on the World Health Organization (WHO) recommended threshold for CEA. According to this criterion, if the ICER for an intervention falls below the level of three times the GDP per capita for the given context and in the given time period, then it will be considered a cost-effective intervention. Although, the WHO threshold has been criticized for being too high, 20 the ICER obtained for this comparison was far lower than this target, which indicates that LDCT screening is a cost-effective strategy.
The results of previous studies conducted to assess the cost-effectiveness of screening for LC differ widely from very favorable21 –23 to unfavorable.24,25 The results of this study are consistent with those recommending LDCT as a favorable strategy. The ICERs from the Early in Lung Cancer Action Project (ELCAP) that was conducted in the US were below the acceptability threshold. 21 A study by Marshal et al. demonstrated that one-time screening of high-risk patients for LC was cost-effective. 22 Likewise, screening with single baseline LDCT for LC in a high-risk population in Poland was highly cost-effective. 23 There are published studies that have used data from the NLST and reached controversial results;26,27 the reason for this discrepancy mainly stems from variation in the methods used for analysis. When comparing studies, various parameters in the economic evaluation of LDCT should be taken into account, including: the target population, prevalence of the risk factor and disease, perspective of the study, time horizon, and assumption on the accuracy of LDCT. In the present study, the base-case assumptions on the prevalence of smokers in the community and accuracy rates of LDCT were key parameters affecting the ICER results.
From an individual perspective, conducting screening may not seem a costly option, but when intended to be implemented as a collective service for a target population of smokers, the cost of scale-up would be noticeable, depending directly on the prevalence of smoking in the given population. The higher the proportion of smokers, the higher will be the cost of screening, but depending on the impact of LDCT in reducing mortality, the benefits may outweigh the cost of the implementation.
The findings with regard to QALY show that the utility of each state was different; that is, for severe states the utility was lower and vice versa. This shows that diagnosis of LC at an early stage leads to higher utility and longer survival, as reflected in the number of QALYs gained.
When comparing the results of the model that accounted for the effect of overdiagnosis with those of the model that did not (Appendix 2, see Online Supplemental Material), the ICER of the model without overdiagnosis (IRR 315,589,047) was less than that of the model with overdiagnosis (IRR 98,515,014.04). The reason for this significant difference is that in the case of screening, due to the overdiagnosis of cases, the revealed prevalence of the cancer will be higher than the true (asymptomatically based) prevalence. As a result, since overdiagnosed cases follow a normal mortality rate, the screening arm will end up with a high rate of surviving cases. This model assumes that all the overdiagnosed cases in the first year are treated in the same way as the patients for whom the treatment is necessary, but their progression to death is assumed to be like the normal pattern of mortality in the general population. Developing the methods that can identify overdiagnosed patients before starting treatment and consequently incurring costs will greatly reduce the costs of screening strategies and the ICER.
Using the probabilistic sensitivity analysis, we were able to examine the impact of possible uncertainty of parameters on the ICER. As a result, we found that almost 99% of the ranges of changes occurring in the ICER were below the threshold of acceptance area. Also, the acceptability curve showed that for thresholds beyond IRR 100,000,000, the probability of LC screening being cost-effective was increased.
The results of this study provide useful data on the cost-effectiveness of LDCT that can be used to optimize policy-making action in the treatment of LC. There are some limitations, and so the results should be used cautiously. Some of the parameters used in this study are of the secondary type obtained from settings other than Iran, and this may raise concern over the generalizability of the results. Using LDCT for screening of LC may in the long term have harmful effects on participants, which will need to be quantified and integrated into the model. On the contrary, if LC screening using LDCT can help to detect other diseases in the thorax and adjacent areas,10,23 then this is required to be included in the model to estimate the extra benefits of screening. Considering the method used for the determination of threshold and the existence of documented evidence that shows that the conclusion on cost-effectiveness of LC screening using LDCT depends, to a large extent, on the targeted threshold level, 28 the results of the present study should be used with an awareness of threshold issues.
Conclusion
The findings of this study suggest that implementing screening by LDTC is cost-effective in patients with LC, and is recommended for inclusion in Iranian treatment guidelines.
Supplemental Material
sj-pdf-1-msc-10.1177_09691413211018253 - Supplemental material for Cost-effectiveness analysis of lung cancer screening with low-dose computed tomography in an Iranian high-risk population
Supplemental material, sj-pdf-1-msc-10.1177_09691413211018253 for Cost-effectiveness analysis of lung cancer screening with low-dose computed tomography in an Iranian high-risk population by Mansur Haji Esmaeili, Farshad Seyednejad, Alireza Mahboub-Ahari, Hossein Ameri, Hadi Abdollahzad, Naser Safaei, Farbod Alinezhad and Mahmood Yousefi in Journal of Medical Screening
Supplemental Material
sj-pdf-2-msc-10.1177_09691413211018253 - Supplemental material for Cost-effectiveness analysis of lung cancer screening with low-dose computed tomography in an Iranian high-risk population
Supplemental material, sj-pdf-2-msc-10.1177_09691413211018253 for Cost-effectiveness analysis of lung cancer screening with low-dose computed tomography in an Iranian high-risk population by Mansur Haji Esmaeili, Farshad Seyednejad, Alireza Mahboub-Ahari, Hossein Ameri, Hadi Abdollahzad, Naser Safaei, Farbod Alinezhad and Mahmood Yousefi in Journal of Medical Screening
Footnotes
Availability of data and materials
The datasets used and analyzed during the current study are available from the corresponding author on reasonable request.
Ethics approval and consent to participate
All procedures performed in studies involving human participants were in accordance with the ethical standards of the national research committee (+IR.TBZMED.REC.1396.878.). Before participating, all participants gave written informed consent.
Authors’ contribution
MY, MH-E, and FS conceptualized the study and the design of the study. MY, HAb, FA, and AM-A participated in data analysis and data interpretation. NS conducted the interviews and collaborated in the data entry. HAm and FA wrote the original draft of the manuscript. All authors read and approved the final draft.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by the Tabriz University of Medical Science.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
