Abstract
Objective
In Italy, colorectal screening programmes using the faecal immunochemical test from ages 50 to 69 every two years have been in place since 2005. We aimed to measure the incidence of interval cancers in the two years after a negative faecal immunochemical test, and compare this with the pre-screening incidence of colorectal cancer.
Methods
Using data on colorectal cancers diagnosed in Italy from 2000 to 2008 collected by cancer registries in areas with active screening programmes, we identified cases that occurred within 24 months of negative screening tests. We used the number of tests with a negative result as a denominator, grouped by age and sex. Proportional incidence was calculated for the first and second year after screening.
Results
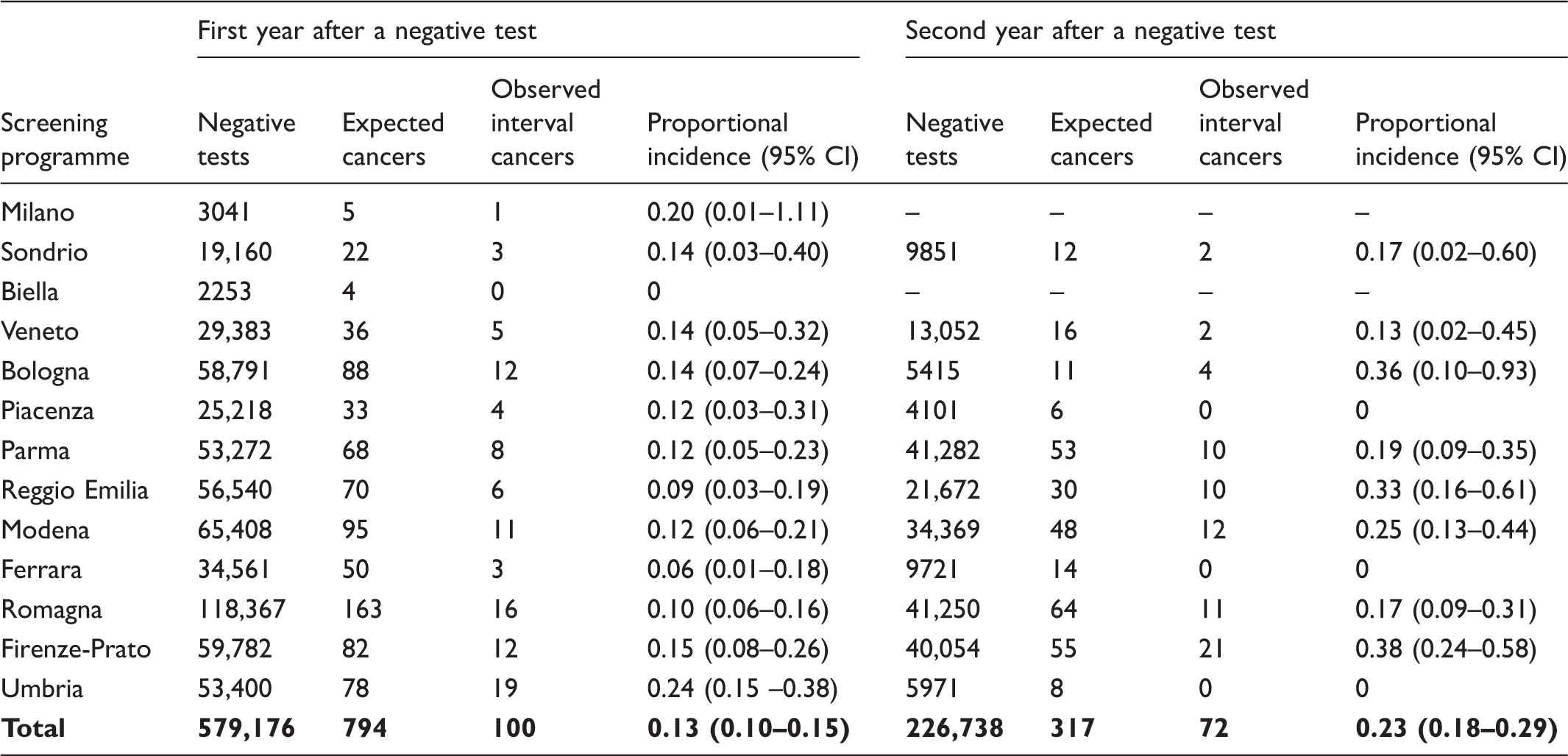
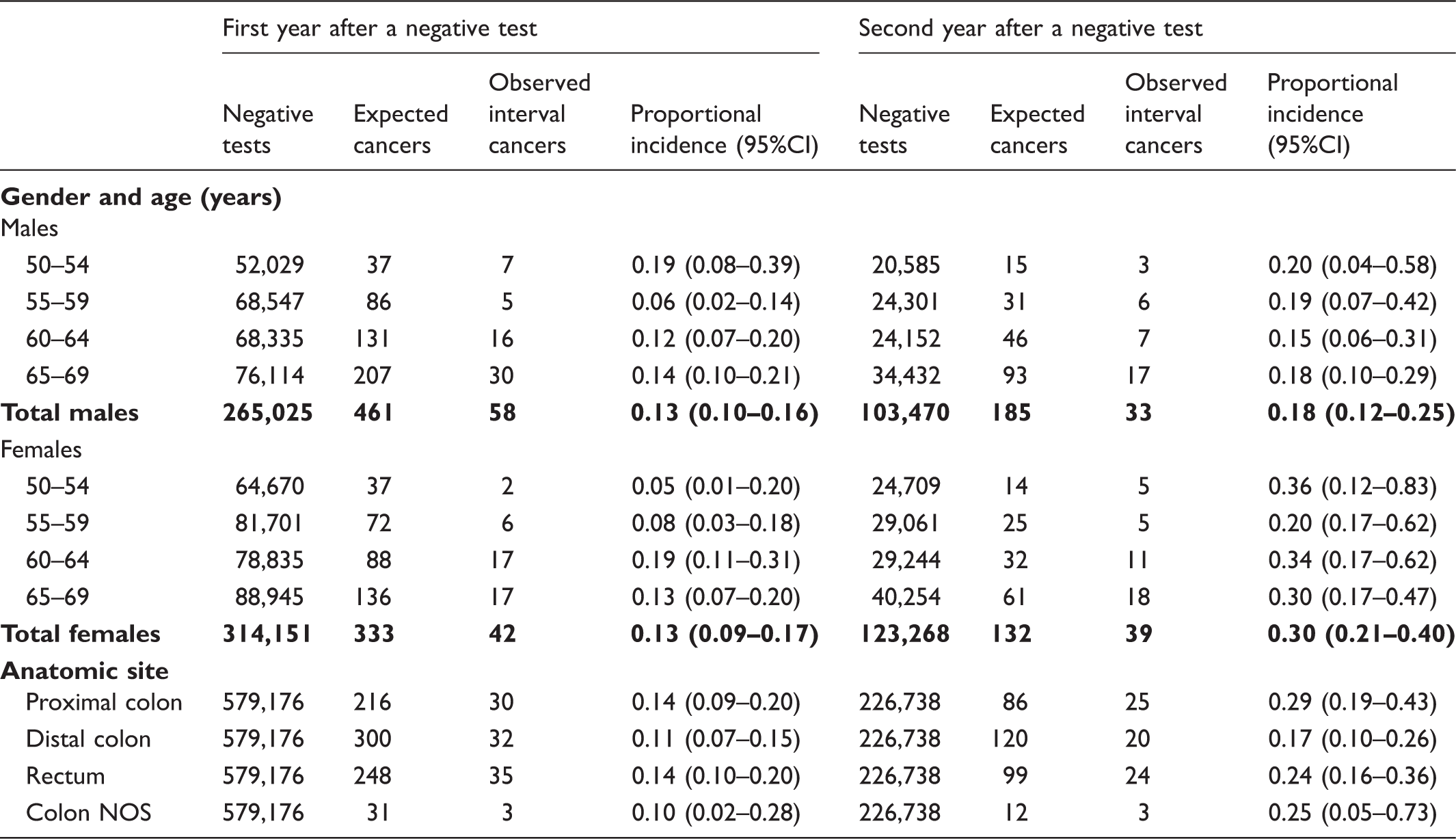
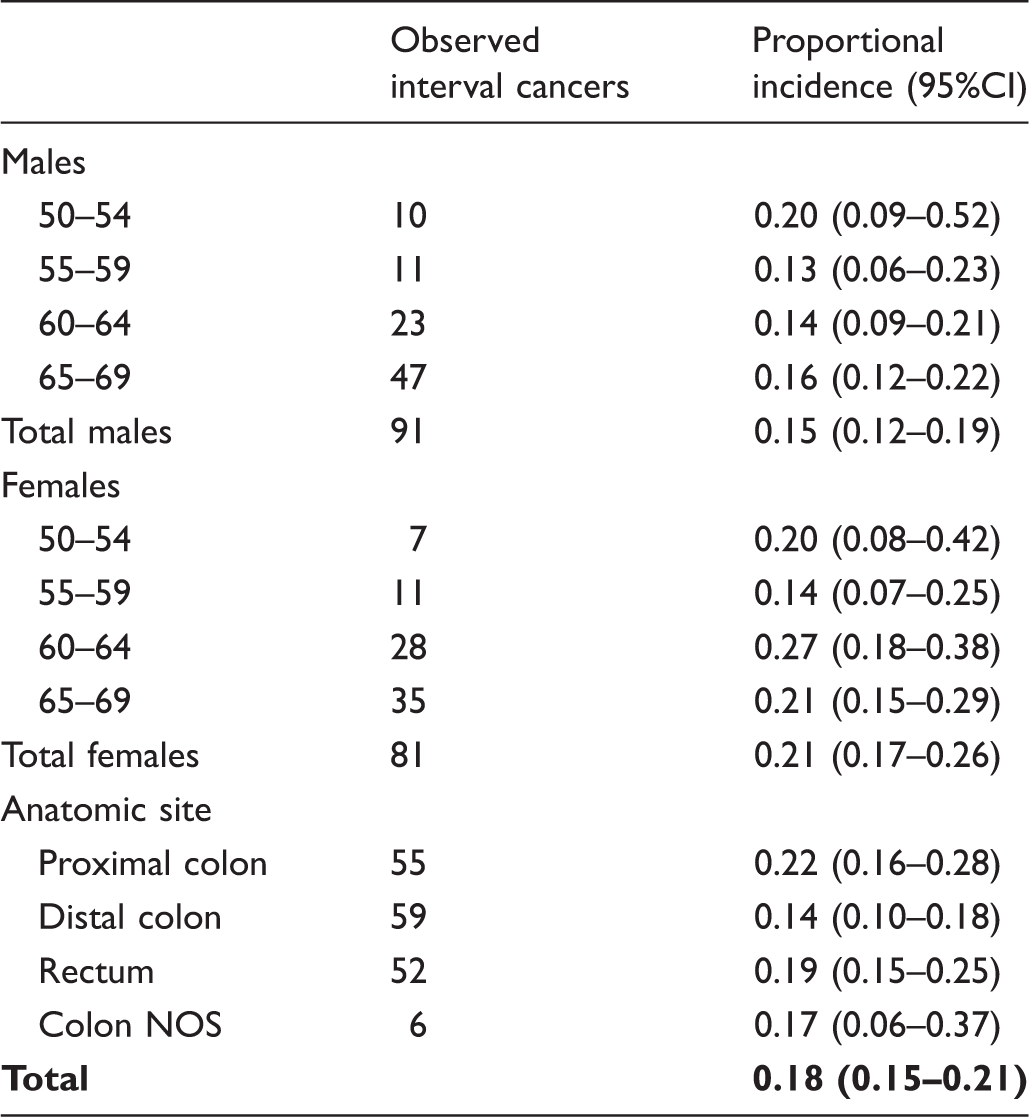
Among 579,176 and 226,738 persons with negative test results followed up at 12 and 24 months, respectively, we identified 100 interval cancers in the first year and 70 in the second year. The proportional incidence was 13% (95% confidence interval 10–15) and 23% (95% confidence interval 18–25), respectively. The estimate for the two-year incidence is 18%, which was slightly higher in females (22%; 95% confidence interval 17–26), and for proximal colon (22%; 95% confidence interval 16–28).
Conclusion
The incidence of interval cancers in the two years after a negative faecal immunochemical test in routine population-based colorectal cancer screening was less than one-fifth of the expected incidence. This is direct evidence that the faecal immunochemical test-based screening programme protocol has high sensitivity for cancers that will become symptomatic.
Introduction
Colorectal cancer (CRC) is the third leading cancer by incidence and the second by mortality in Italy. 1 Large randomised trials showed that screening with the Guaiac faecal occult blood test reduces mortality for CRC by more than 15%, 2 but the more sensitive and specific 3 faecal immunochemical test (FIT) has now been adopted in most population-based screening programmes. The performance and effectiveness of screening programmes in part depend on the sensitivity of the screening algorithm, i.e. on the ability to identify cancers and pre-cancerous lesions that would otherwise produce symptoms in subsequent years. Screening programme sensitivity can be measured by comparing the incidence of interval cancers with the incidence of cancers in the absence of screening. This indicator does not require assumptions on the lead time (i.e. to what extent the diagnosis has been anticipated) and it is not influenced by potentially over-diagnosed cancers. However, it only makes sense when adopting a pre-fixed time frame and it cannot easily take into account the cancers occurring after the recommended interval and before the actual invitation due to delay and other organisational problems related to the programme. 4 Studies on interval cancers give quite consistent estimates of the test sensitivity and proportional incidence in routine screening programmes, ranging from 73% to 82%.5–8
In Italy, the implementation of organised CRC screening programmes started in 2005–2006, but the national scale-up has happened slowly and inconsistently. By 2013, active screening programmes covered about 79% of the Italian population, and about 62% were regularly invited. 9 In 2008, the Ministry of Health funded a project for systematically evaluating the impact of screening programmes by merging screening and cancer registry archives (the IMPATTO study).10,11
We examined the proportional incidence of interval cancers in Italian biennial FIT CRC screening, as estimated from the routine survey of the National Screening Monitoring Center and from cancer registry archives, completed with individual screening history.
Methods
Setting
The Italian guidelines recommend organised population-based screening programmes for CRC. The target population is aged 50–69, but in some regions this has been extended to age 74. Programmes may exclude from invitation people who had an invasive CRC or a recent documented colonoscopy. In this study, 5.5% were excluded from invitation, and 1.4% were excluded after presentation. The interval is two years. The OC-Hemodia latex agglutination test, developed with the OC-Sensor Micro instrument (Eiken, Tokyo, Japan), was used by all included programmes. The FIT positivity threshold is 100 ng Hb/mL. Subjects with a positive FIT are contacted to undergo a total colonoscopy, usually performed at an endoscopic referral centre. Patients with screen-detected neoplasms are treated endoscopically or referred for surgery, and then enrolled in a follow-up programme.
The oldest programme commenced in 1982, but most started after 2005. The implementation of screening programmes is almost complete in Northern and Central Italy, but in the South and Islands there are still large areas where programmes are not active, or invite a small proportion of the target population. 9 Participation in the screening programmes included in this study from 2011 to 2012 ranged from 25% to 72% (average 45%). 9 In Italy, cancer registries are not mandatory, but many local or regional health authorities have set up a register. In 2015, the population coverage was 53%. 1 The IMPATTO study collected data from CRC cases in subjects aged 40–79 that were diagnosed between 2000 and 2008 in the populations covered by 23 population-based cancer registries in 13 Italian regions.10,11 Participation in the IMPATTO study was voluntary. Of 84 screening programmes active in 2008, 43 were in an area covered by a cancer registry. Only 16 pairs of cancer registries and screening programme coordinating centres participated in the IMPATTO study.
Data sources
All CRC cases (International Classification of Diseases, 10th revision: C18–C20) diagnosed during the study period in people aged 50–79 were collected. The inclusion criteria for cancer are described in detail elsewhere.
10
The minimum core dataset included incidence date, morphology and topography, stage at diagnosis (according to Dukes’ classification as modified by Astler and Coller
12
) and grading, surgical intervention, lymph nodes examined and positive lymph nodes. The tumour histological type was recorded according to the International Classification of Diseases for Oncology, 3rd edition. All the participating registries fulfilled quality criteria for inclusion in the IARC database,
13
death certificate only cases for CRC being below 1%. Cancer registries and screening programmes linked the two databases to retrieve individual data on the screening history of patients before the incidence date by collecting the date of the first invitation and the dates of screening tests. Patients were then classified according to the following screening patterns:
screen-detected at the first screening episode; screen-detected at a repeat screening episode; screen-detected at follow-up, i.e. after a positive FIT, but detected after the first colonoscopy; cancer in subjects with a positive screening FIT, but not compliant with diagnostic work-up; interval cancers, i.e. cancer in subjects with at least one previous negative screening test; cancers in subjects who have been invited but never participated (i.e. invited, but not tested within the screening programme); never invited to screening.
Our study only includes interval cancers with an interval between the last negative test and the incidence of ≤24 months.
Denominators
Data on the number of negative tests performed each year by the screening programmes were collected through the survey conducted by the National Screening Monitoring Center. Surveys for CRC screening are available from 2005 and have been integrated by the coordinating centres of the SPs. The surveys collect data on the programmes’ target populations, invitations, test attendance and results, ascertainment, and final findings. The data are checked for consistency and completeness, in collaboration with the regional and local screening coordinating centres.
Reference incidence
Age- and sex-specific data on incidence in the year leading up to screening start were used to estimate the expected cases. For the Florence area, where the cancer registry started registration after screening implementation, we used the incidence estimated for the geographic macro area by the pool of Italian cancer registries at the beginning of the study period.
Data analysis
We classified cancers diagnosed within 12 months after negative screening as interval cancers 0–12, and those between 12 and 24 months as interval cancers 12–24. On the basis of the survey data, follow-ups of 12 and 24 months were defined for each screening programme and cancers with a screening test after the follow-up period were excluded. The cancers expected if screening had not taken place were calculated by applying the sex- and age-specific reference incidence rate to the number of negative tests recorded (person-years at risk) during follow-up. To determine the proportional incidence, the interval cancers were divided by the expected cancers. An overall 0–24 proportional incidence was estimated as the rate between the average incidence of interval cancer 0–12 and 12–24 and the expected incidence in the first year. We calculated 95% confidence intervals (CIs) assuming a Poisson distribution of the observed cases. The proportional incidence calculation is based on the assumption that subjects with a negative test were not re-tested before 24 months, and that screening tests were performed in the same calendar year as the screening invitation. We also assumed that the follow-up fixed at 12 or 24 months, together with the use of whole calendar years, does not require an equal distribution of calls during the calendar year. In addition, a likelihood-ratio test was used to determine whether the proportional incidence varied according to screening programme and anatomic site. After the application of these analysis criteria, three registries were excluded because they did not have one year of complete cancer registry follow-up after the activation of the screening programme.
Results
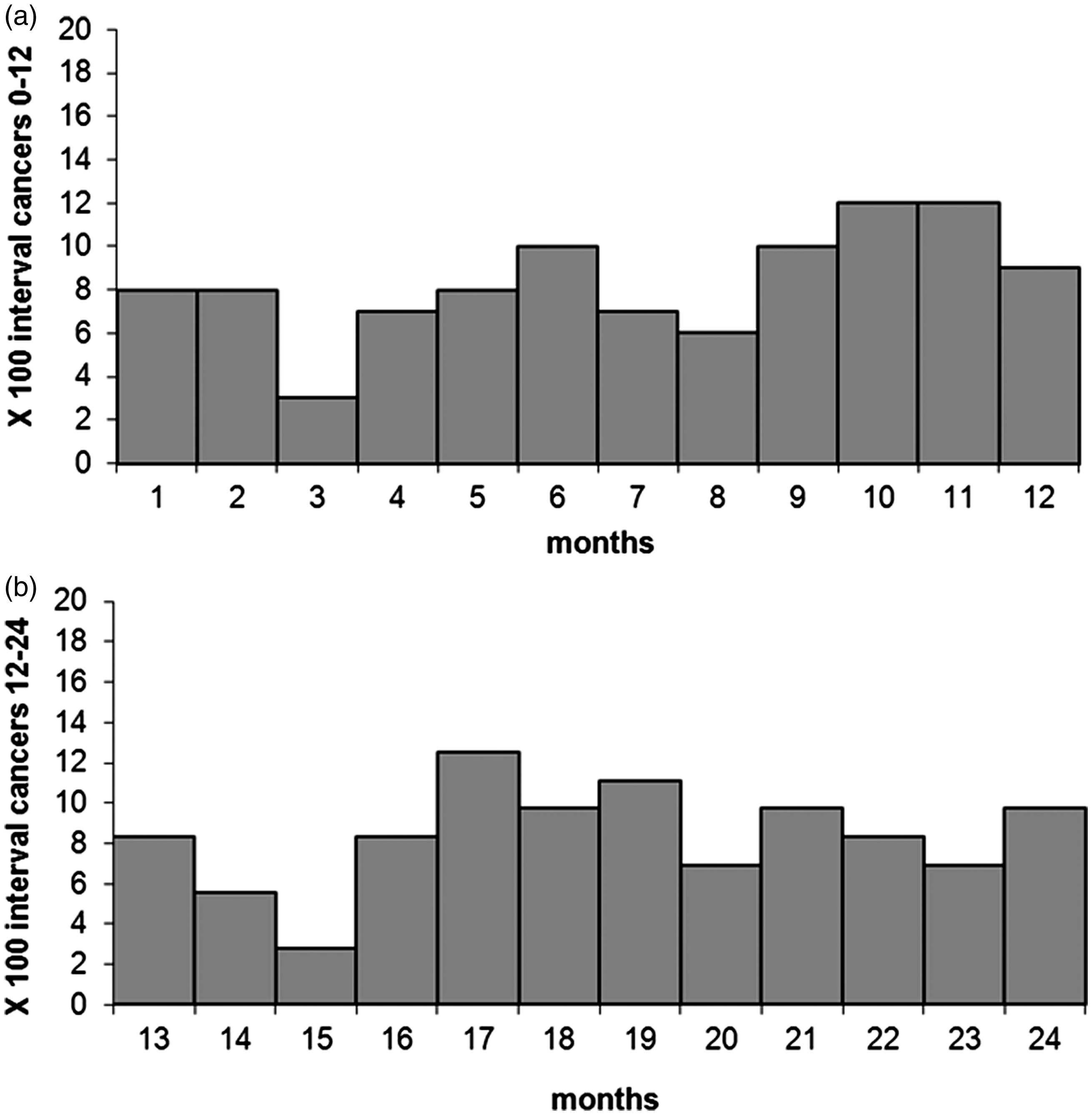
The available data for the IMPATTO study include 13 screening programmes for incidence in the first year after a negative screening test, and 11 for incidence in the second year (Figure 1), for a total of 579,176 and 226,738 screened people, respectively. In these populations, we observed 172 interval cancers, 100 in the first and 72 in the second year. The number of expected cancers was 794 in the first year and 317 in the second year, respectively. The overall proportional incidence was 13% (95% CI 10–15%) and 23% (95% CI 18–29%), respectively. Despite a higher incidence in the second year, the distribution of cancers by month in each year did not show a clear increasing trend as time elapsed following a negative test (Figure 2). The variability among programmes of proportional incidence is that expected due to random fluctuations (likelihood ratio test Years of screening activity and follow-up included in the study and number of yearly negative screening tests, by area. The boxed area represents the period of cancer registry data availability for each area; the dark grey area represents the years of screening activity (with relative numbers of negative tests per year) for which at least 24 months of follow-up are available and that are suitable for analysis of interval cancers in the first and second interval year; the light grey area represents the years of screening activity (with numbers of negative test) for which only 12 months of follow-up are available and are suitable only for the analysis of interval cancers in the first interval year; the striped area represents the last year of cancer registry data. All the screening programmes are continuing screening activity up to 2016. Distribution of interval cancers diagnosed in the first and second year after a negative screening test, by month. Negative screening tests, expected cancers, observed interval cancers, and proportional incidence with 95% confidence interval in the first and second years after a negative test, by screening programme. Negative screening tests, expected cancers, observed interval cancers, and proportional incidence with 95% confidence interval in the first and second years after a negative test, by age, gender and anatomic site. Observed interval cancers and proportional incidence 0–24 months with 95% confidence interval, by age, gender, and anatomic site.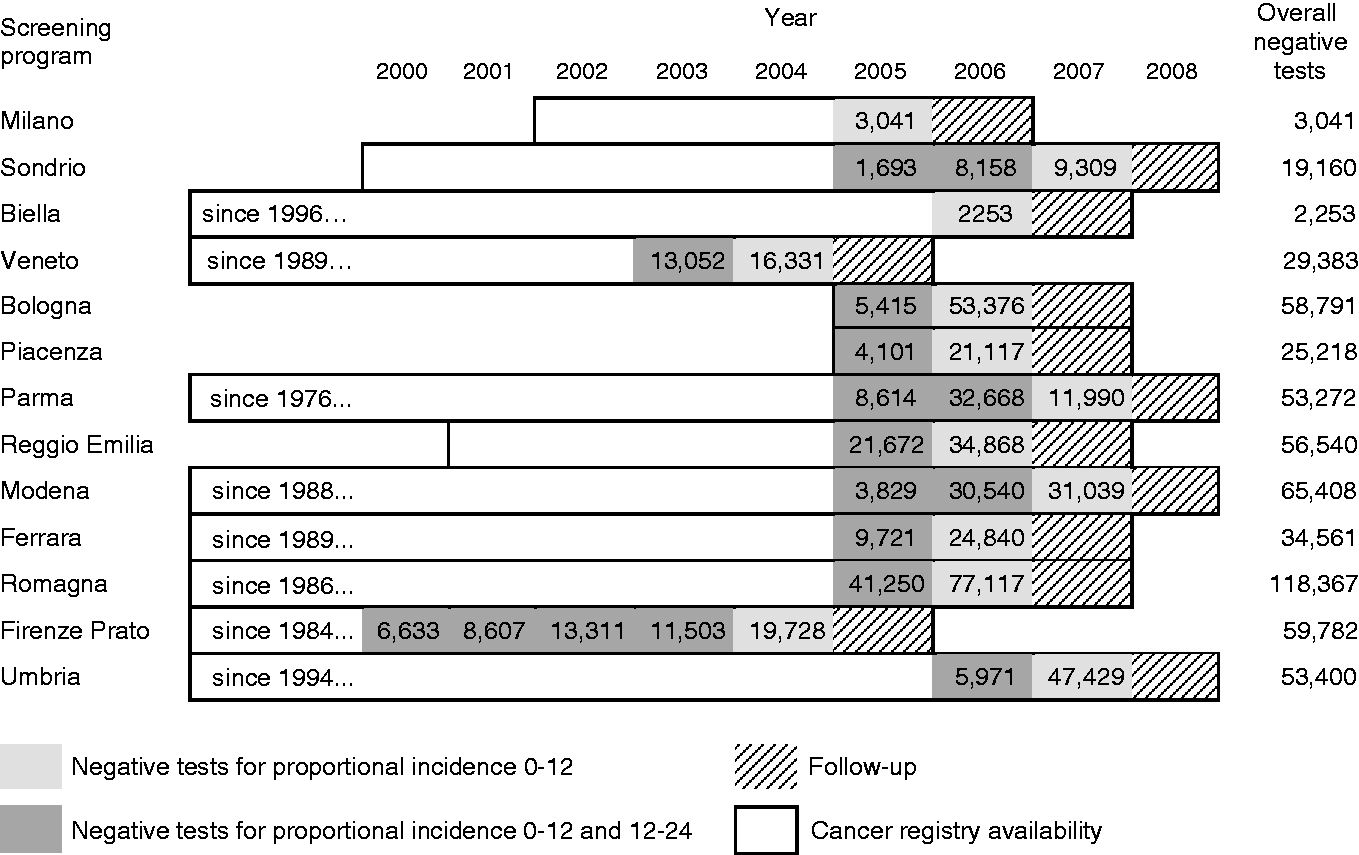
Discussion
We followed up 579,176 and 226,738 people with a negative FIT for 12 and 24 months, respectively, and found a proportional incidence of 18%. Our results are consistent with those of previous studies, while being at the lower limit of the range.5–8 In fact, the estimated proportional incidence ranged between 18% and 27% in other studies on FIT organised screening programmes with a biennial interval. We included in our denominator all the tests performed in people invited in the reference year, which possibly slightly overestimated the denominator and therefore the expected cases. Even if a slight underestimation of the proportional incidence of interval cancers is possible, all those studies showed very high sensitivity of FIT for invasive cancers leading to symptoms in the next two years. The sensitivity observed in our study is much higher than that reported for programmes using the Guaiac test.14–18 These results are in line with the reported figures on the impact of FIT-based CRC screening programmes19–23 that showed a much greater reduction of CRC incidence and mortality than that observed in the trials based on the Guaiac test. 2
We also confirmed a higher sensitivity of FIT in males and a lower proportional incidence of interval cancers in the distal colon. These two observations are likely to be related. It has been reported that patients with proximal CRC are more often females than males24,25 and that proximal CRC is a more aggressive type of tumour compared with distal CRC.24,26 Proximal CRCs are more often flat, while distal tumours are polypoid-type, which is more detectable by FIT 27 and more distinguishable by colonoscopy. 28 This evidence may suggest that CRC screening in women requires greater test sensitivity. However, further evidence is needed to determine the most adequate gender-specific screening guidelines. Simply lowering the positivity threshold to identify a larger proportion of flat cancers (i.e. lesions with no evidence of a long pre-clinical phase and the presence of detectable pre-invasive lesions) would have minimal impact on test sensitivity, and no impact on screening efficacy.
Given the strong increase in CRC incidence with age, it is of note that the absolute risk of interval cancer increased with age, but screening sensitivity was substantially constant. As expected, we also found that interval cancer incidence was higher in the second year than in the first. The real risk of having a symptomatic cancer in the first year is probably lower than the risk we observed. In fact, the number of cancers diagnosed in the first month after the negative FIT was more than twice those observed in months 3–6. Some of these cancers could be due to preventive colonoscopies prescribed just after a negative FIT by some general practitioners or gastroenterologists who do not trust the faecal occult blood test.29,30 Misclassification of the test result was excluded through an individual check of each case by the screening coordinating centres. Screening sensitivity showed little variability among centres, compatible with random fluctuations.
Although there are no international standards for the sensitivity for CRC of FIT-based screening programmes, our results indicate that the Italian programme screening protocol ensures a significant reduction in the risk of interval CRC. We recorded low levels of proportional incidence across all the evaluated subgroups (i.e. by gender, age class, anatomic site, screening programme), suggesting that there are no settings for which the performance of a screening based on biennial FIT is below acceptability levels. A possible exception may be the use of FIT in countries where high ambient temperatures may cause significant drops in haemoglobin concentration.31–33 Our study could not evaluate the seasonal variation of proportional incidence, however, the impact of warming on the frequency of interval cancers has been shown to be limited. 34 Moreover, some manufacturers of FIT recently modified the buffer of their products to prevent haemoglobin degradation at high temperatures. 35
Strengths and limitations
In this, the first attempt to use routine National Screening Monitoring Center and cancer registry data to estimate the incidence of interval cancer in Italy, we did not need any individual data for the denominator, but only a characterisation of the cancers according to their screening history. We tried to define the proportional incidence of interval cancer using routine data that are now available in almost all the screening programmes where a cancer registry is active, a situation common to many European screening programmes.
The lack of data records at individual level for the person/time denominator introduces several limitations. First, we could not be certain that screening participants were not tested again within 24 months of a negative test, but as this would be exceptional, we assume that its effect on our results would be negligible, if any. Second, we could not assess the risk beyond the two-year interval because individual data on the actual re-invitation and related compliance were not available. Thus, we could not calculate the person-time for the denominator beyond the 24th month, when most people are expected to be invited for a new screening round. However, as the screening interval is two years, analysing the interval cancers diagnosed beyond the 24th month would be useful only to include in the proportional incidence interval cancers due to organisational problems of the programmes, i.e. delay in re-inviting the target population, but not to draw any general conclusion on the protocol screening interval. Furthermore, given the timing of the survey conducted by the National Screening Monitoring Centre, we possibly underestimated the proportional incidence of interval cancers, because we included in the denominator all the tests performed in people invited in the reference year, also including those who actually performed the test in the three months of the following year, while we only considered interval cancers with a negative test in the reference year. The effect of this limitation on our results could be particularly relevant for programmes contributing with only one or two years of screening. Because we combined all the study years in a single analysis, including undue subjects in the denominator only affects the last study year and is therefore diluted by the whole number of screened people in the entire study period. The median delay from invitation to test is about two months. 36 As the most recent periods of each year in our study population have higher risk because the tested population increased constantly during the study period, we are probably underestimating the incidence of interval cancers of the last year of observation by at least the cases occurring in 2 months out of 12, i.e. by about 17%. Not having individual data for the denominator also meant we could not take into account emigration and deaths that occurred during the follow-up, but as follow-up was very short (12 or 24 months), and with the relatively low emigration of the Italian population in that age group, 37 this bias should be limited.
This study only evaluated tests performed within organised screening programmes; opportunistic screening was not considered. Spontaneous screening for CRC in Italy is below 20% 38 and over the past 10 years has been almost exclusively based on colonoscopy. The anomalous peak of interval cancers in the first month after a negative FIT could be explained by opportunistic screening (i.e. some individuals who underwent screening and had negative results may be advised by their general practitioner to have a colonoscopy in any case, as a significant proportion of GPs do not trust faecal occult blood testing). 29 In general, the effect of opportunistic screening is to increase the incidence of interval cancers through the diagnosis of asymptomatic cancers that would otherwise be screen-detected at the following round.
Some further possible sources of bias exist. Our estimate of proportional incidence is strongly influenced by what we use as expected incidence in the absence of screening, i.e. the area-specific pre-screening incidence. For Florence, this reference incidence is quite chronologically distant from the study period, so if the underlying incidence has changed with time, there may be bias. We can note that, in the absence of screening, CRC incidence in Italy in the last decades showed a slight increasing trend for colon and no trend for rectum. 39 Another issue is the self-selection bias of screening participants. If participants have a different background risk from the whole population, we calculate the proportional incidence on the basis of the wrong expected incidence. This is possible as people with a recent colonoscopy, who could be at higher risk for familial CRC than the general population, can be excluded from invitation or have lower propensity to participate. Nevertheless, in the study period, very few people were excluded from invitation and at presentation. The last possible bias due to non-random or selective roll-out is that in some programmes people in their 68th or 69th year were invited first. This bias should be controlled by age-adjustment. Even though this was a national study, only 16 out of 42 programmes participated. This self-selection of the participants limits the generalisability of the results.
Conclusion
The incidence of interval cancers in the two years after a negative FIT in routine population-based CRC screening is about 20% of the expected incidence. This is a direct indicator of the high sensitivity of the screening protocol.
Colorectal Cancer Screening IMPATTO Working Group
Marco Zappa (Osservatorio Nazionale Screening; Istituto per lo studio e la prevenzione oncologica ISPO, Firenze); Emanuela Anghinoni (Gruppo italiano screening colorettale (GISCoR); Servizio medicina preventiva nelle comunità, ASL di Mantova); Carlo Senore (Centro per la Prevenzione Oncologica (CPO) Piemonte, Torino)
Footnotes
Acknowledgement
The funder had no involvement in the study design, the collection, analysis and interpretation of data, the writing of the manuscript, or the decision to submit the article for publication.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Ministry of Health – Agenzia di Sanità Regione Abruzzo (grant 2008).
