Abstract
Objective
To evaluate interval cancers in the population-based colorectal cancer screening programme of Stockholm/Gotland, Sweden.
Methods
From 2008, individuals aged 60–69 were invited to colorectal cancer screening using biennial guaiac-based faecal occult blood test (Hemoccult®). Interval cancers, defined as colorectal cancer among participants not diagnosed by the screening programme but registered in the Swedish cancer register, were evaluated by cross-checking the screening histories for all cancers in the region 2008–2012.
Results
Of 203,848 individuals from nine different birth cohorts who participated (∼60%), 4530 (2.2%) tested positive. All invited individuals were followed up for 24 months after invitation. The cancer register reported 557 colorectal cancer, 219 (39.3%) screen-detected cancers and 338 (60.7%) interval cancers, generating both test- and episode sensitivities of approximately 40% and an interval cancer-rate of 17.1/10,000 tests. Among individuals with positive tests without colorectal cancer diagnosed at work-up colonoscopy, 37 interval cancers (10.9%) occurred. There was statistically significant lower sensitivity in women, ranging 22.4–32.2%, compared with 43.2–52.0% in men. Age-group and tumour location were not strongly correlated to screen-detected cancer rates. The programme sensitivity increased by year (20.3–25.0%), with successively more colorectal cancers diagnosed within the expanding programme (11.6–16.2%).
Conclusion
Interval cancer is a quality indicator of a screening programme. As the interval cancer-rate determined in a well-organized population-based screening programme was actually higher than the screen-detected cancer rate, a change to a more sensitive screening test is indicated. The lower screen-detected cancers among women, and compliance and quality of work-up colonoscopies also need attention.
Introduction
Following European Union recommendations to screen for colorectal cancer (CRC) with faecal occult blood test (FOBT) in 2003, 1 many European countries introduced national and regional programmes. 2 The recommendations were based on the results from four randomized controlled trials with guaiac-based FOBT (gFOBT), initiated in the 1980s, demonstrating a CRC-mortality reduction of around 16%.3–6 The 2010 European Guidelines for Quality Assurance in Colorectal Cancer Screening and Diagnosis (EG) provide further details. 7
The Stockholm/Gotland (Sweden) CRC screening programme with gFOBT using Hemoccult® started in 2008. To evaluate effectiveness, the implementation followed a model with random invitation of different birth cohorts aged between 60 and 69. 8 Over five years, more than 200,000 individuals from nine different birth cohorts were invited, with an approximate 60% compliance rate, that increased with age, female gender and subsequent screening round. 9
We used the EG definition of interval cancer (IC), i.e. a CRC that occurs in a screening participant after a negative FOBT-test result or after a positive test with a negative work-up colonoscopy, before the next invitation round, 7 which in the Stockholm/Gotland screening programme is fixed 24 months after the latest invitation. Hence, an IC is a CRC not diagnosed by the screening programme. This definition is also in line with that used in breast cancer screening by European guidelines for quality assurance in breast cancer screening and diagnosis. 10
The number of ICs is an intermediate measure of the effectiveness of the programme in decreasing CRC mortality. The frequency of ICs, together with the participation rate, determines the ability of the screening programme to detect cancer in the target population (programme sensitivity) and has to be monitored regularly. Hence, the programme sensitivity is used to evaluate the effect of the screening programme in the population. In a CRC screening programme with gFOBT, the programme sensitivity depends on the number of cancers not detected by the test (test sensitivity), but also on the number of cancers missed at the work-up colonoscopies after a positive test (episode sensitivity).11,12 For simplicity, we have not used the proportional incidence method, but defined test sensitivity as the screen-detected cancers (SDC)/(SDC + IC with negative gFOBT) and the episode sensitivity (screening process sensitivity) as SDC/(SDC + all IC). Similar simple measurements have been validated by Andersen et al
This study aimed to prospectively evaluate the sensitivities of the Stockholm/Gotland CRC screening programme using gFOBT, by identifying the ICs, defined as the number of IC/10,000 negative tests and negative outcome of assessing a positive test, and to retrospectively evaluate the effect of the programme on the cancers occurring in the target population. Secondarily, we evaluated the differences in ICs and SDCs with respect to age-group, gender, and localization in the colorectum.
Methods
Screening performance indicators of the guaiac-based programme of Stockholm/Gotland.
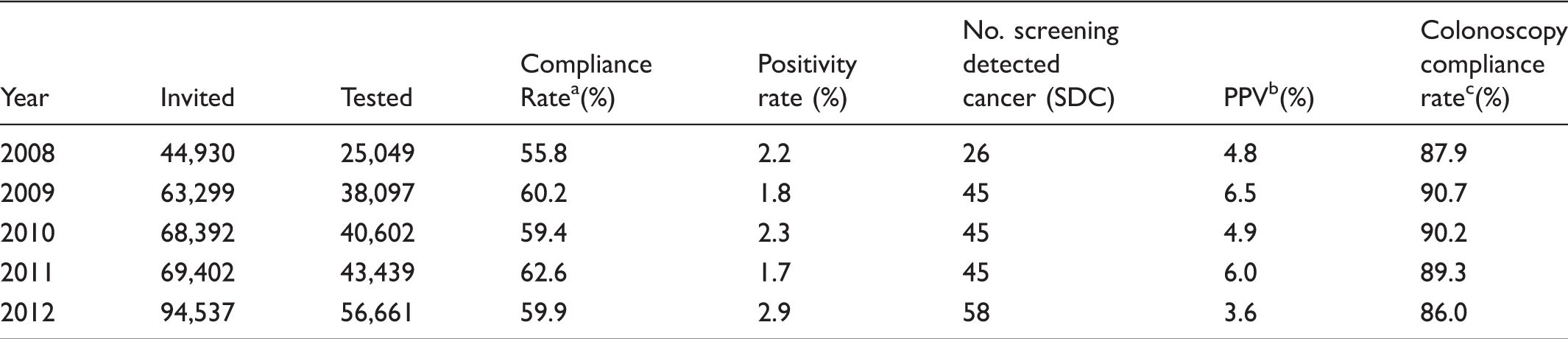
No. of individuals invited and tested that particular year. Participation occurs up to six months after invitation.
Positive predicted value.
Individuals invited in that year having had a colonoscopy 9 .
Each individual in Sweden is assigned a unique identification number that is used in all population statistics. The number allows for matching with relevant health registers. The screening register was matched with the Swedish cancer register maintained by the National Board of Health and Welfare. 15 The cancer register has a 98% coverage of all cancers diagnosed in Sweden. 16 Information on CRC diagnosed during 2008–2012 in all individuals aged 60–69 living in the Stockholm/Gotland region was retrieved and stratified into CRC among screening participants, non-participants and non-invited.
Prospective analysis
The aim of the prospective study was to evaluate the sensitivity of the gFOBT (test sensitivity), i.e Prospective and retrospective analyses with time line and events.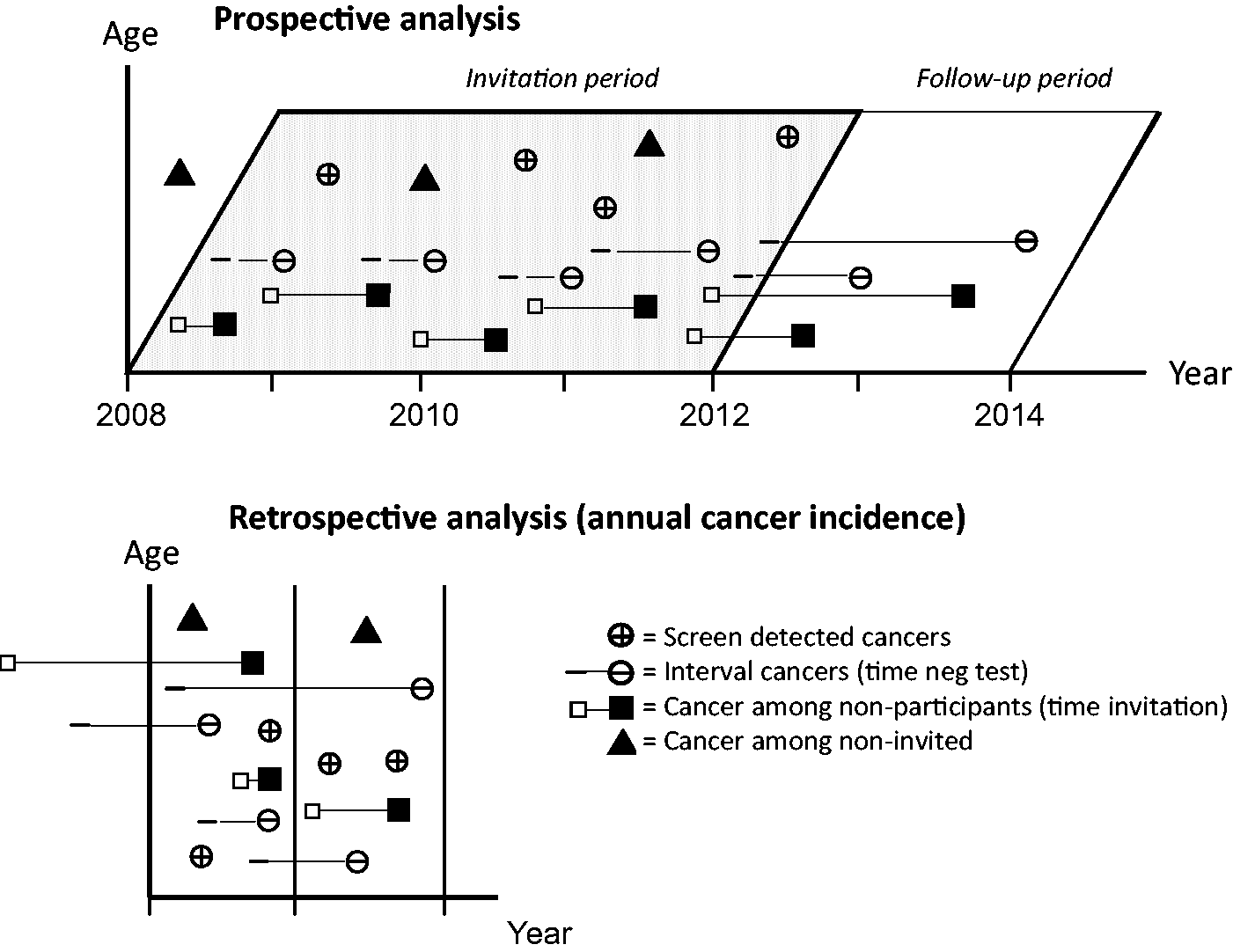
Retrospective analysis – Mode of detection
To study the effectiveness of the screening programme to detect CRC in the target population (programme sensitivity), 12 we retrieved all CRC diagnoses between 2010 and 2012 from the Swedish cancer register. The rational for the time interval was that one screening interval of 24 months must have passed from start of the programme in 2008. We cross-checked with the screening register to evaluate all the CRC in the birth cohorts invited to the screening programme. There is a steady state of invitations from start of the programme in 2008, but due to the successive implementation, four cohorts had had prevalence screening rounds and five cohorts had undergone incidence rounds as well. 9 For each individual with a CRC, we then retrospectively evaluated if they, within a screening interval (24 months), were invited, participated or did not participate, were diagnosed with a SDC, or participated and had a negative screening result (i.e. IC (negative test or positive test without CRC diagnosed with work-up colonoscopy)). The CRCs were categorized according to the mode by which they were detected (mode of detection); CRCs among non-invited (not yet covered by the programme), CRCs among invitees detected by the programme (SDCs), or not detected by the screening programme (ICs), CRCs among non-participants, or CRC among individuals having an incorrect test.
Statistical analysis
Each participant was prospectively cross-checked between the years 2008 and 2012 in the Swedish cancer register to analyse SDC-rate, IC-rate, and sensitivities. The screening episode sensitivity was defined as the number of SDCs divided by the sum of SDCs plus all ICs among participants (episode sensitivity = SDC/(SDC + all IC)). The test sensitivity only considers the IC after a false negative test (test sensitivity = SDC/(SDC + IC with negative gFOBT)). IC-rate has been presented as number of IC/10,000 negative tests, or a positive test without an assessment colonoscopy diagnosing the CRC. The IC-rate by year of screening invitation was then compared with the predicted CRC incidence per 10,000 individuals in the absence of screening (expected incidence rate (EIR)) according to the proportional incidence method. 17 The EIR is estimated by a 10-year extrapolation of the actual age-standardized incidence of CRC in the region between 1998 and 2007. The difference between the IC-rate and the predicted CRC incidence without screening is an indirect estimate of the ability of gFOBT to detect CRC in the population targeted for screening.
Comparative analyses of differences in SDC, IC, and episode sensitivity by age-groups 60–64 vs. 65–71, and by gender, and tumour localization in colorectum (right- vs
Results
Prospective analysis
Prospective analysis of test result, screen-detected cancer (SDC) and interval cancer (IC).
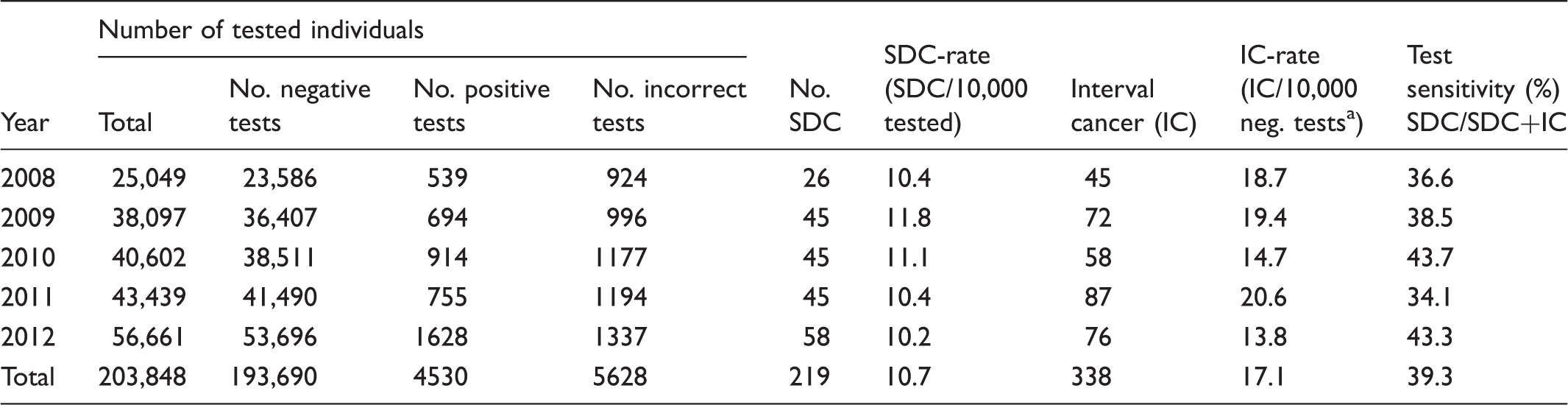
IC/10,000 tested negative or tested positive with a negative colonoscopy.
Prospective analysis of interval cancer (IC) by negative test result, colonoscopy outcome after a positive test, and time period when the IC was diagnosed outside the Hemoccult® CRC screening programme. The rate of IC is correlated to the experienced incidence of Stockholm/Gotland, Sweden, by year.
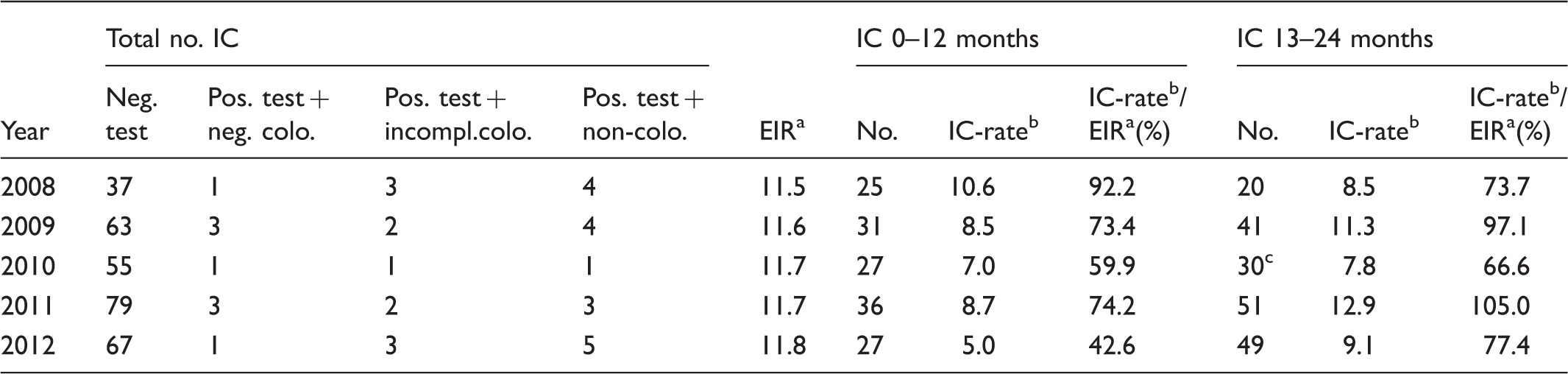
Expected mean incidence per 10,000 individuals for the period 2008–2012 in absence of screening estimated by extrapolating the incidence 10 years prior to screening start (1998–2007).
The number of IC/10,000 negative tests or a positive test with a negative colonoscopy.
One IC > 24 months.
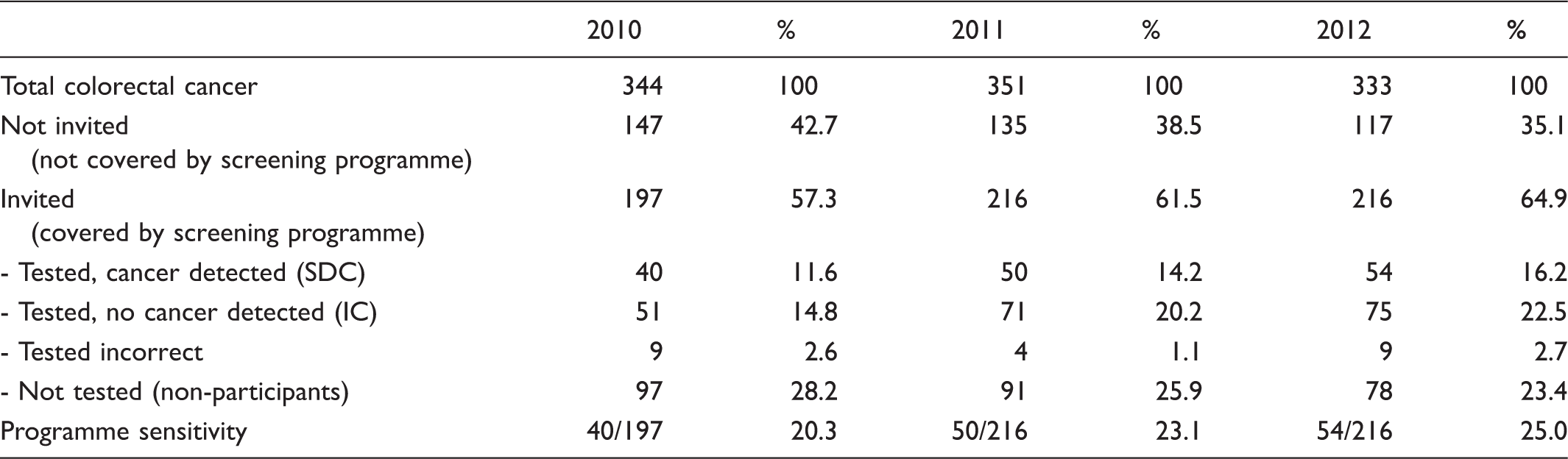
Approximately 11% (37/338) of the ICs were among individuals having had a positive test but negative follow-up colonoscopy (9/37, 24%), incomplete colonoscopy (11/37, 30%), or not complying with the colonoscopy work-up (17/37, 46%) (Table 3) The screening test sensitivity, only considering the CRC missed due to a negative gFOBT (false negative test), was 42% (219/[219 + 338−37]) and the episode sensitivity was just above 39% (219/557).
Participating individuals followed for 24 months after a test with screening-detected cancer (SDC), and interval cancer (IC) by age-group, gender and tumour site stratified as right (dx) or left (sin) colorectum, estimation of differences in sensitivity, and by year of invitation.
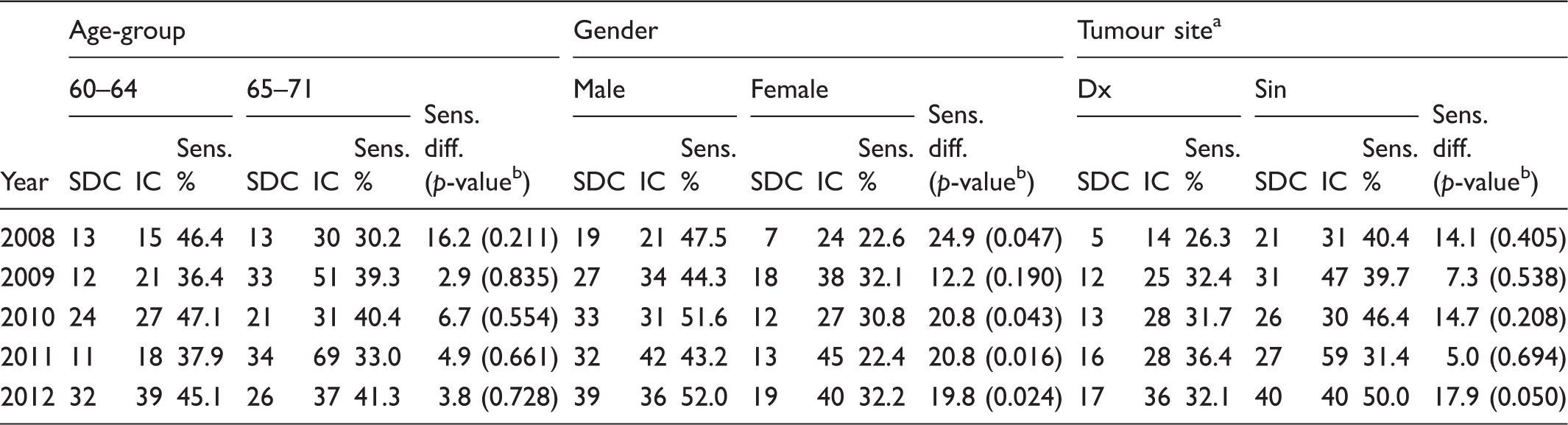
SDCs not specified as dx or sin; 2 in 2009, 6 in 2010, 2 in 2011 and 1 in 2012.
Fisher’s exact test.
Retrospective analysis – Mode of detection
Mode of colorectal cancer detection. Retrospective analysis of screening history for all cases of CRC of the screening-population registered in the Swedish cancer register for the years 2010, 2011 and 2012.
Discussion
In the Stockholm/Gotland population-based gFOBT CRC screening programme, the number of ICs was actually larger than the number of SDCs between 2008 and 2012, generating a test- and episode sensitivity of just above and below 40%, respectively. The episode sensitivity was in the lower limit compared with other population-based gFOBT screening programmes, predominantly in Europe, reporting sensitivities around 40–60%.18–22 In Scotland and in the Catalonian region of Spain, increasing frequencies of IC were reported over the screening rounds from 31–59% to 32–46%, respectively.19,22 The lower sensitivity in the last round in Scotland was explained by a decrease of SDCs rather than an increase in ICs. There could be many explanations for the IC differences.
First, compared with the Spanish programme using hospital data to identify CRC, 22 we used Swedish cancer registry data. The completeness of the cancer register is around 98%, so close to all of IC-cases in Stockholm/Gotland are registered and detected in retrospective analysis of mode of detection. 16 Underestimation of the IC-frequency could generate a falsely high sensitivity.
Second, the sensitivity of the gFOBT seems to increase with lower age, and the target groups of both the Scottish and Spanish programmes were aged 50–69,19,22 compared with age 60–69 in the Stockholm/Gotland programme. Older people have a higher incidence of adenomas and CRC, but the unspecific bleeding of other causes dominates as the positive predictive value for cancer of the positive test in the cohort aged 60–69 in the Stockholm/Gotland programme is only 5.6%. 9
Third, 11% of ICs in Stockholm/Gotland occurred among individuals with positive tests but without CRC diagnosed at work-up colonoscopy. The episode sensitivity, combining the ability of the screening test and the work-up colonoscopy to detect CRC, should be addressed. 12 The programme sensitivity could then be increased through higher compliance with work-up colonoscopies after a positive test, and higher quality examinations. High quality work-up colonoscopies are essential both to ensure that screening has a positive effect for the individual, and also for the overall incidence- and mortality reduction potential of the screening programme.23–27 A programme intervention is required when close to half of individuals with IC after a positive test simply do not comply with the diagnostic work-up colonoscopy. The intervention could be more thorough information on the necessity of a work-up colonoscopy following a positive test, and frequent re-calls as reminders.
Fourth, intra-observer differences in the subjective assessment of a positive Hemoccult®-test result in a variation of the positivity rate that could be avoided by changing gFOBT to the quantitative faecal immunochemistry test (FIT). As FIT analyses the quantity of human occult blood, all that is required is the quantity of blood that would be considered a positive test, i.e
We did not find any statistically significant differences in sensitivity between age-groups nor, as had been reported previously, more ICs on the right side of colon.19,36 The proportion of IC was the same in both the right and left sides of the colon, but because the expected incidence is higher on the left side (approximately two thirds of all CRC 37 ), the denominator is higher, and test sensitivity is also higher for left-sided CRC.
On the other hand, even though there were similar numbers of ICs between men and women, we found more than twice as many SDCs in absolute numbers diagnosed among men during the time period. Women have an up to 10% higher compliance rate in the Stockholm/Gotland screening programme, 9 so the rate of IC in this programme is actually relatively higher among women, as has been reported elsewhere.19,21,36,38 To equalize the SDCs and ICs between genders, a change to FIT screening could also be adequate. The amount of blood in the stool in screening is associated with CRC and adenomas, but also with male gender.39–41 In changing to FIT, the cut-off for a positive test could be set to a more sensitive lower level for women, to generate the same positivity rate and volume of assessment colonoscopies as in men, although, because more women comply with the programme, the potential decrease in ICs among women with FIT at lower cut-off would be at the expense of an increased number of negative work-up colonoscopies after a positive test.
In general, all IC-rates except one were lower then the predicted incidence rates of CRC in the absence of screening. We could not adjust for potential selection bias from individuals with lower risk of CRC participating in the screening programme, which could generate an overestimation of the effect of gFOBT in detecting CRC (low IC due to few CRC in the population participating). On the other hand, by definition, the non-attenders could not have had an IC.
As the screening programme expanded, both the SDC and IC-rates increased by year, compared with CRCs among the non-invited. The CRC-rate among non-participants decreased by year, possibly due to screening invitations to younger age-groups, with both a lower participation-rate and lower prevalence of CRC, compared with the older age-groups. 9
The four randomized studies that constitute the basis for the evidence that screening reduces CRC mortality were all guaiac-based.3–6 It is reasonable to believe that, despite the low sensitivity in the Stockholm/Gotland routine screening programme, a similar effect is achievable. Changing to a more sensitive test (e.g
Conclusion
The IC-rate indicates the sensitivity of a screening programme to detect CRC and is a surrogate measure of programme effectiveness. In the organized Stockholm/Gotland gFOBT screening programme, there are actually more ICs than SDCs. A change to a more sensitive screening test with the potential to alter cut-off levels to equalize work-up colonoscopies between genders may increase programme sensitivity. Moreover, interventions to increase compliance with and quality of work-up colonoscopies have to be evaluated.
Footnotes
Acknowledgement
We thank Sini Kilpeläinen, Regional Cancer Centre Stockholm Gotland, Sweden for statistical work.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
