Abstract
Objective
Because colorectal cancer (CRC) has a long natural history, estimating the effectiveness of CRC screening programmes requires long-term follow-up. As an alternative, we here demonstrate the use of a temporal multi-state natural history model to predict the effectiveness of CRC screening.
Methods
In the Finnish population-based biennial CRC screening programme using faecal occult blood tests (FOBT), which was conducted in a randomised health services study, we estimated the pre-clinical incidence, the mean sojourn time (MST), and the sensitivity of FOBT using a Markov model to analyse data from 2004 to 2007. These estimates were applied to predict, through simulation, the effects of five rounds of screening on the relative rate of reducing advanced CRC with 6 years of follow-up, and on the reduction in mortality with 10 years of follow-up, in a cohort of 500,000 subjects aged 60 to 69.
Results
For localised and non-localised CRC, respectively, the MST was 2.06 and 1.36 years and the sensitivity estimates were 65.12% and 73.70%. The predicted relative risk of non-localised CRC and death from CRC in the screened compared with the control population was 0.86 (95% CI: 0.79–0.98) and 0.91 (95% CI: 0.85–1.02), respectively.
Conclusion
Based on the preliminary results of the Finnish CRC screening programme, our model predicted a 9% reduction in CRC mortality and a 14% reduction in advanced CRC.
Introduction
The effectiveness of colorectal cancer (CRC) screening using the faecal occult blood test (FOBT) has been demonstrated in randomised controlled trials.1,2 Participation in screening, compliance with colonoscopies, and sensitivity of the FOBT may vary from country to country, even if the natural history of the disease is similar. Additionally, the characteristics of the specific health services may be substantially different in a scientific trial than in routine application. The CRC FOBT screening programme recently incorporated into the routine Finnish health services can thus provide insight into the effectiveness of FOBT CRC screening as a health service. However, determining the effects of this screening practice on mortality, even using surrogate endpoints such as advanced CRC, requires a long follow-up period.
A temporal multi-step natural history of CRC, in which individuals transition from CRC-free to the pre-clinical screen-detectable phase (PCDP) and finally to the clinical phase (CP), can be used to evaluate the efficacy of a population-based screening programme in reducing CRC-related mortality, with a shorter follow-up period. 3 The transition rate from the PCDP to the CP is often represented as the mean sojourn time (MST, the average duration of staying in the PCDP), which is an informative metric that helps to determine the inter-screening interval. However, the estimation of the MST and the transition rates assumes no overdiagnosis, and is affected by the estimated sensitivity of the screening tool. Several previous studies have demonstrated an inverse relationship between the estimates of false negative proportions (sensitivity) and the MST. Therefore, it is important to estimate the MST while accounting for the sensitivity estimate of the screening tool.4,5
We expanded the three-state model (from state of cancer-free to PCDP to CP) to a five-state model, by incorporating a tumour stage, and by accounting for stage-specific sensitivity. 5 The clinical parameters obtained from the natural course of the five-state model could provide insight into the impact of screening on reducing the rates of both advanced cancer and mortality. This relationship in turn could provide model-based information on the efficacy of screening before the long-term follow-up on mortality outcomes can be observed.
Finland’s most recent population-based, randomised screening study of a biennial FOBT using faecal test-kits (Hemoccult®)6–8 provided an opportunity to estimate the transition parameters of the five-state model, while adjusting for sensitivity to project the screening-induced reduction in advanced CRC and mortality. Data from the first two rounds of the programme from 2004 to 2007, and from survival by stage in Finland overall before screening, were used.
The purpose of this article was to estimate the screening-induced reduction in advanced stage CRC and death from CRC that resulted from the Finnish biennial FOBT screening programme in individuals aged 60 to 69. The empirical data were used to estimate the natural history parameters and sensitivities, which were then used to predict the reductions in advanced stage cancer in the 6 years following initial screening and in CRC mortality in the following 10 years.
Methods
We defined five disease states related to the occurrence of colorectal cancer: CRC-free, PCDP localised, PCDP non-localised, clinical localised, and clinical non-localised CRC, which is typically detected because of symptoms or signs related to CRC. The transitions from one state to another were estimated by the incidence rates of PCDP localised (λ1), and the rates of progression from PCDP localised to PCDP non-localised (λ2), to clinical localised (λ3), and from PCDP non-localised to clinical non-localised (λ4) CRC. These transition rates were used as the parameters of the natural history model. The normal CRC-free state could progress to a localised PCDP cancer, which could progress either to a clinically detected localised cancer or to a non-localised PCDP cancer (Figure 1). Clinically detected non-localised cancers were assumed to have progressed via non-localised cancer in the PCDP. The transition rates were denoted as λ1 to λ4. Typical FOBT results consist of both true normal and false negative results among the cases of localised and non-localised cancers in the PCDP. Therefore, we incorporated the stage-specific sensitivities of the FOBT into the model. The false negative cases were observed only after they reached the clinical stages.
Five-state natural history model for colorectal cancer.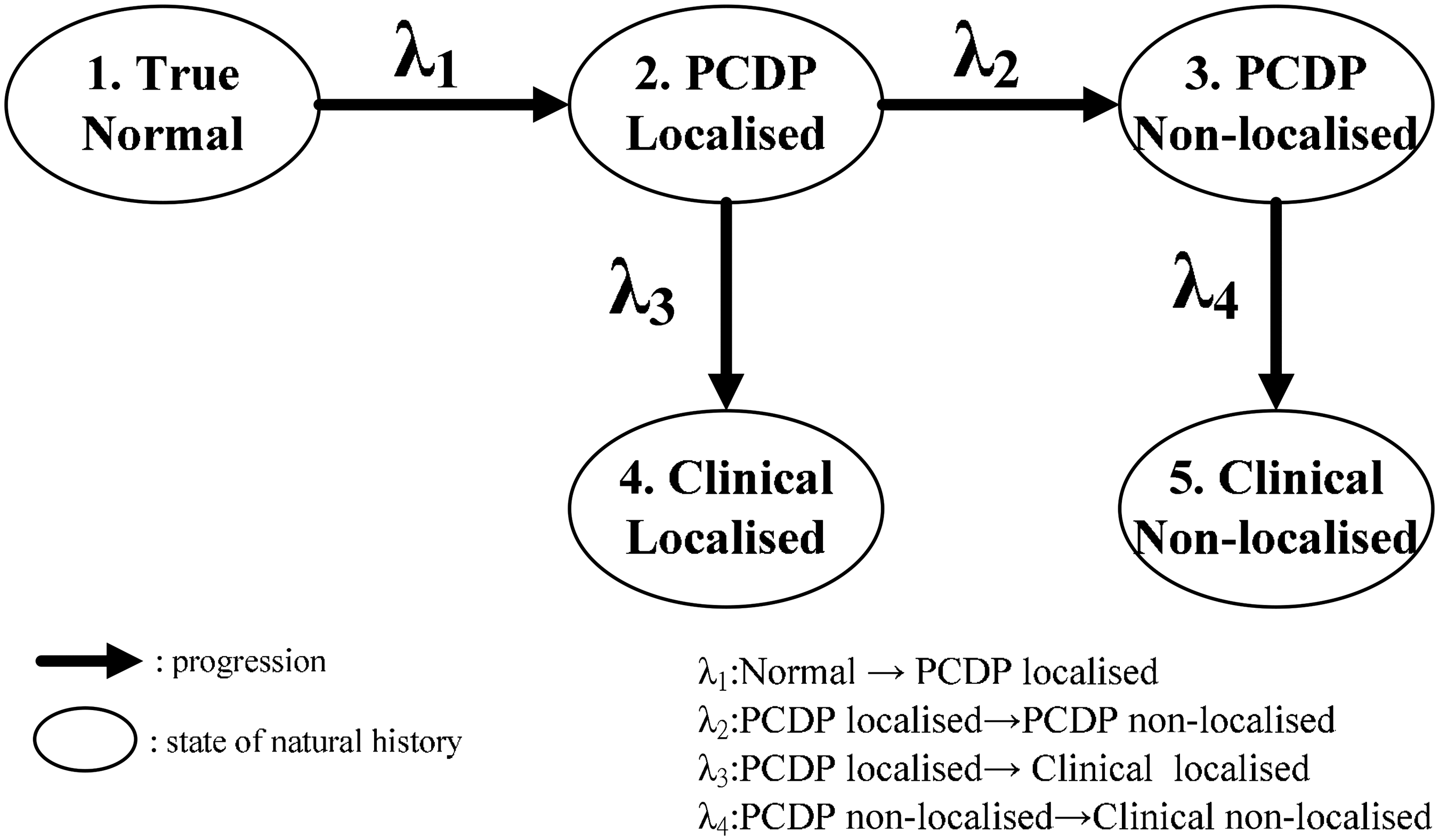
We used data from a Finnish randomised health services study that implemented FOBT screening to obtain estimations of the transition parameters and test sensitivity. The detailed study design and preliminary results have been fully described elsewhere.6,7,9 A total of 106,000 subjects were randomly allocated to the study arm (n = 52,998) or the control arm (n = 53,002) from 2004 to 2006. After excluding CRC cases with a diagnosis before the first screening invitation, 105,489 individuals (52,728 in the screening arm and 52,761 in the control arm) were included. To calculate the person-years and incident cancers in the controls, the mean date of invitation in the screening arm by municipality was used.
The model described in the previous publication with four transitions and two sensitivities 5 was based on the following empirical observations: the number of individuals invited, number screened, number of non-attenders in the first and second rounds of screening, number of screen-detected cancers by stage, number of interval cancers among the attenders, and number of cancers in the non-attenders and controls during the first interval by stage. The stage was classified into localised and non-localised CRC. The terms localised and non-localised were used interchangeably with non-advanced and advanced stage of cancer. Advanced cancer was defined as a CRC with regional or distant metastases or lymph node involvement. 5 The validated data originated from the randomised screening data.
Statistical analysis
The estimation of the transition parameters was based on the temporal model of the natural course of CRC and on the sensitivity of the screening test applied. For the likelihood functions details, we referred to a study by Chen et al.
10
In brief, taking the test sensitivities for localised (Sen(p)) and non-localised (Sen(q)) of CRCs into account, the likelihood function for prevalent screen-detected CRC is
For those who were examined as negative result, the likelihood is
The simultaneous estimation of the transition parameters and sensitivities led to a non-identifiable model. Therefore, we first estimated the sensitivity of the FOBT in identifying localised CRC. The proportionate incidence method (or I/E ratio) originally proposed by Day was applied. 11 The incidence method estimates of sensitivity were then corrected for the transitions from localised PCDP (λ2 and λ3). 10 We plugged this estimate of sensitivity for localised CRC into the likelihood function to calculate the other transition parameters and the sensitivity of non-localised CRC. This resulted in an iterative process because the sensitivity of the localised CRC depended on the transition rates λ2 and λ3. The iterations were repeated until all of the estimates of the two sensitivities and the four transition rates converged (see online Appendix Table A1). The confidence intervals for the estimated parameters were calculated using a maximum likelihood estimation approach with a variance-covariance matrix that was derived from the inverse of a negative Hessian matrix.
Data distribution and goodness-of-fit of the expected data for Finnish colorectal cancer screening using faecal occult blood testing.
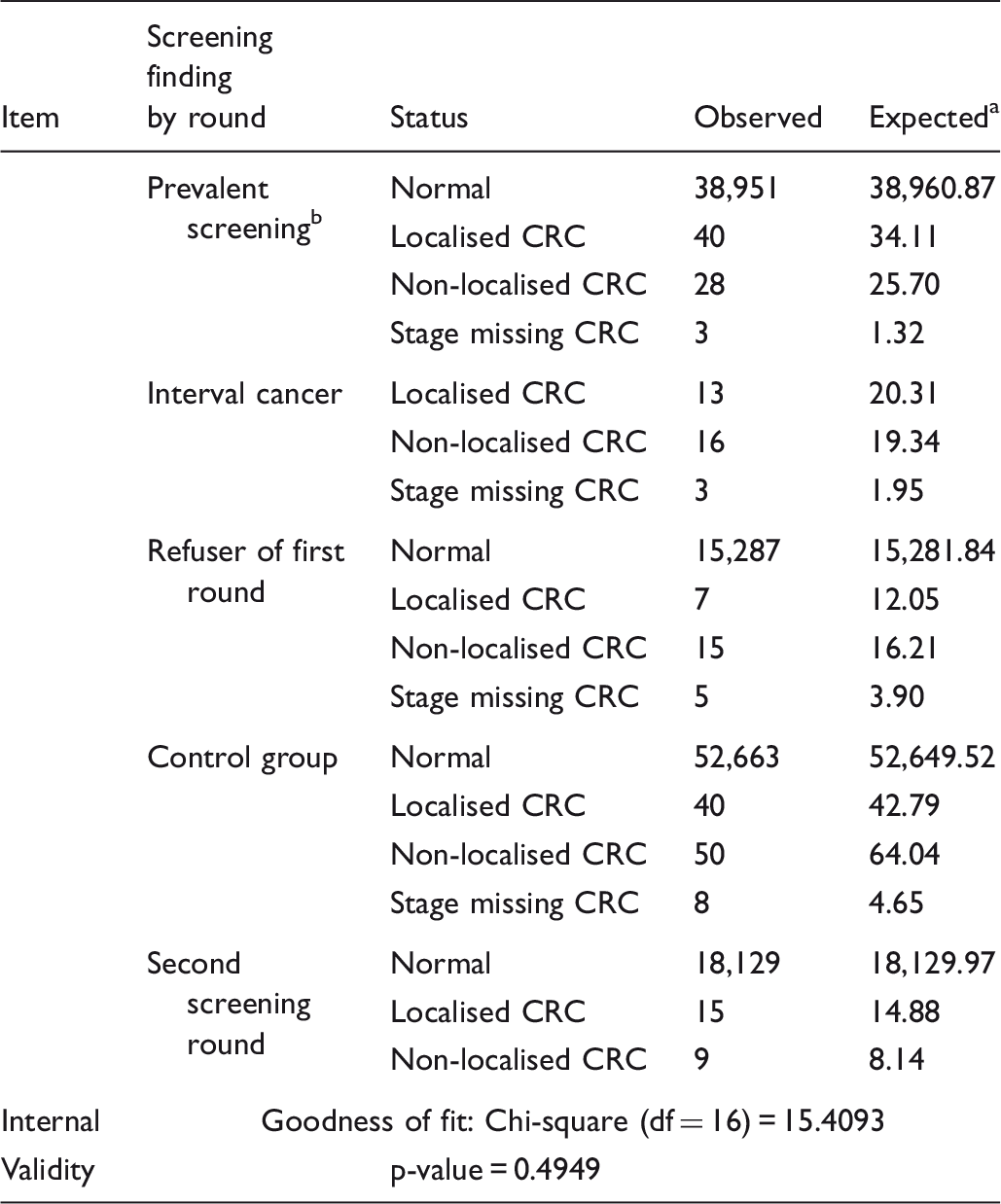
The expected values were computed based on the parameters of transition rates (showed in Table 2), which incorporated with sensitivities during estimation.
Included 1608 subjects who refused first round but attended second round, with 3 CRC cases as screening detected.
Based on the estimates of the transition rates and sensitivities during the first interval and second screening of the Finnish population-based screening programme, we predicted the overall reduction in the number of advanced CRC cancer cases and deaths from CRC. The prediction was determined using a simulation analysis for a population of 2 × 250,000 subjects with a biennial test for those aged 60 to 69, which was identical with the design of the Finnish programme. 6 Because the sojourn time differed between the first round and the subsequent rounds, the simulation was performed with the initial round and four subsequent rounds. The estimates of the transition parameters and sensitivity were obtained by applying the data in Table 1 to the model in Figure 1. The natural history model estimates the effects of screening on incident cancers. The simulation scenario based on the Finnish population-based screening for CRC with FOBT in relation to the five-state model is presented in online Appendix Figure A1. Those who tested positive in the first round of screening were referred for a confirmation of the results by colonoscopy, which resulted in the number of prevalent screen-detected cancers. Those with a negative FOBT followed the same screening protocol at the subsequent biennial screening. 8 The interval cancers and the cancers in the refusers at the initial screening were retrieved from the Finnish Cancer Registry by linkage. Details of the simulation scenario are shown in online Appendix Figure A1.
To simulate the impact of the screening on mortality and advanced CRC in the target population, we used additional empirical information: the attendance rate (70.8%), the positivity rate of the FOBT (2.1%), the compliance rate with colonoscopy (90%) and the 10-year death rates of the localised and non-localised CRC patients, which were allowed to vary by time and were captured by scale and shape parameters according to the Weibull distribution. To account for the variations in simulations, we performed 100 simulations based on those parameters with the initial 250,000 people in each arm. The confidence intervals were estimated in cumulative curves by year.
Results
In the screening arm, 71 cases were detected from initial screening as prevalent CRCs. The clinically detected CRCs, including refusers, interval cases and control group, were ascertained by linking the population from the initial round to the cancer registry until the end of 2007. In total, 27 of the non-attenders were CRC cases, 32 interval CRCs were identified that had negative FOBT results at first round and 98 CRCs from the control group were identified. The median follow-up time following the first negative result was 1.99 years. By the end of 2007, of those who had negative results at the initial screening, 18,153 subjects had completed the second round screening, and 24 CRCs were diagnosed as subsequent screen-detected cases (see observed cases in Table 1). 7
Transition rates estimated by the five-state natural history model of the Finnish colorectal cancer screening programme.
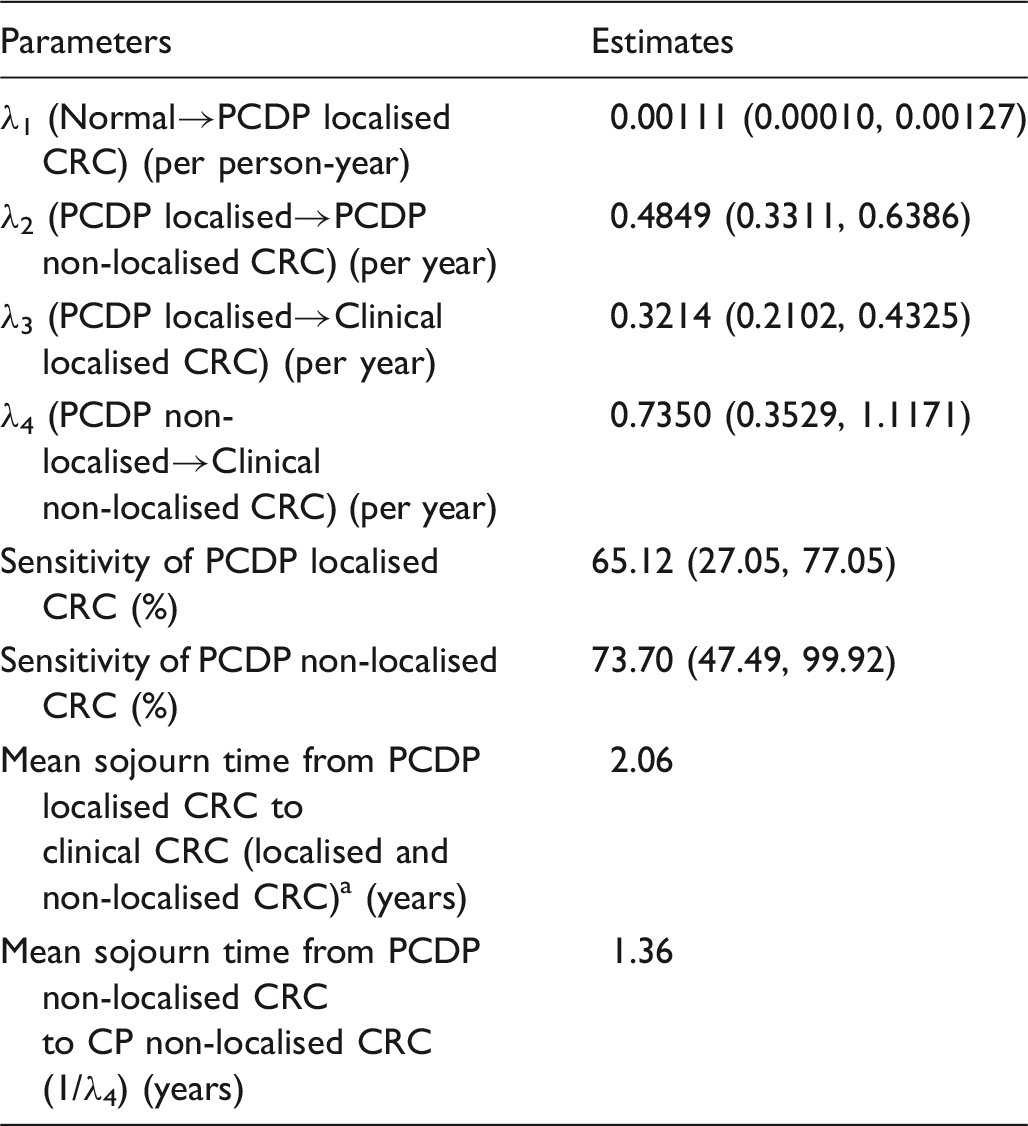
PCDP: pre-clinical detectable phase; CRC: colorectal cancer.
Mean sojourn time of PCDP localised
Figure 2 shows the cumulative risk of advanced CRC and mortality from CRC. The detailed cumulative rates of advanced CRC incidence and mortality by year are shown in online Appendix Table A2. Compared with the control arm, the relative risk of reducing the incidence of advanced CRC from the biennial screening programme was 0.86 (95% confidence interval (CI): 0.79–0.98), and the relative risk of reducing CRC mortality was 0.91 (95% CI: 0.85–1.02). Our study predicted that the CRC screening programme would reduce 14% and 9% of the advanced CRC and CRC mortality cases, respectively.
(a) Cumulative incidence of non-localised colorectal cancer during a 6-year period following randomisation. (b) Cumulative mortality of colorectal cancer with a 10-year follow-up after randomisation.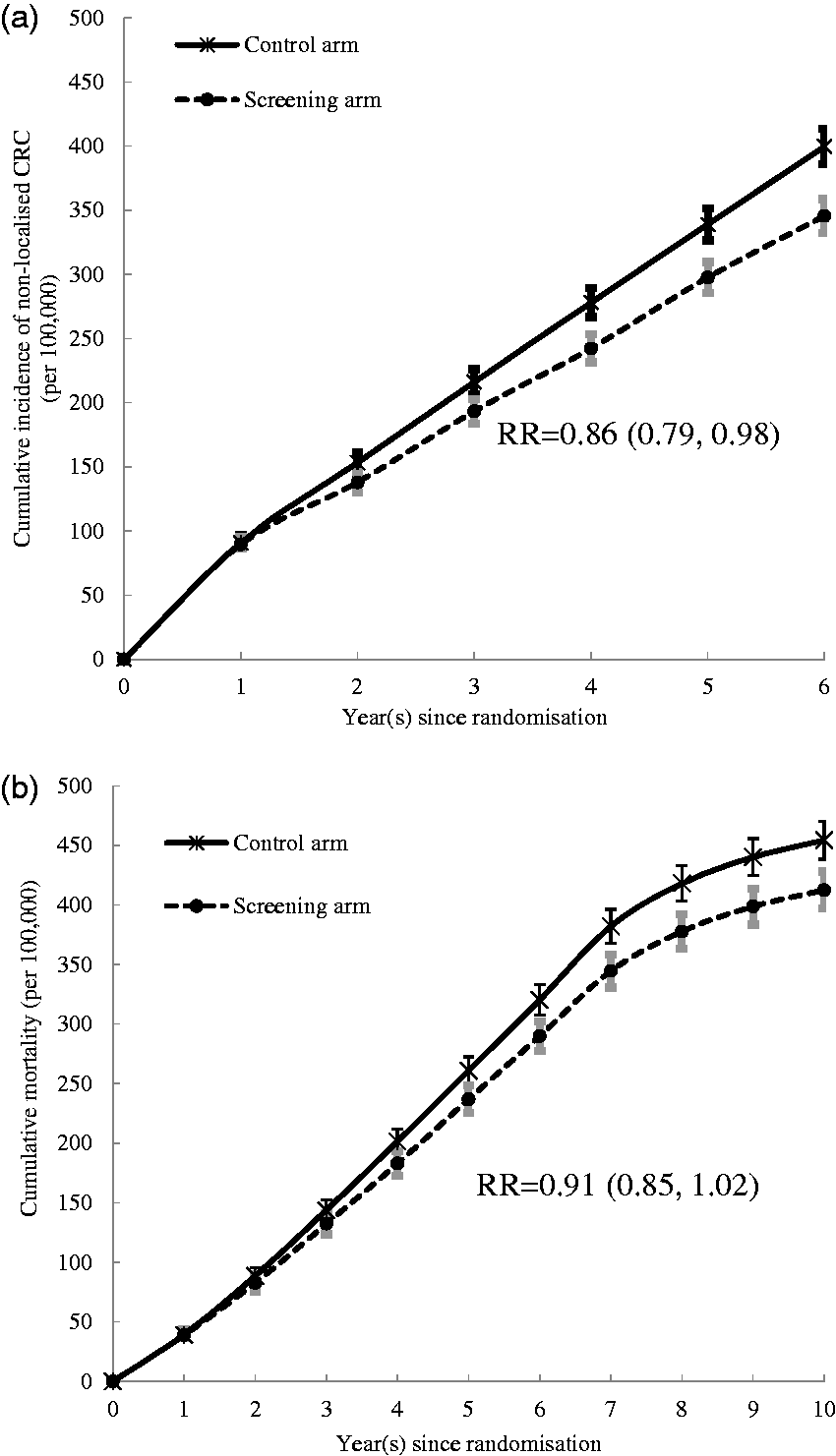
Discussion
Evaluating the effectiveness of screening programmes for diseases with a long natural history via randomised controlled trial demands a long follow-up period and substantial costs, particularly when using the primary end-point of cause-specific mortality. For health policy-makers, evaluating the effectiveness of a study in its early stages is challenging, given the limited information available. Our study applied a five-state Markov model to two screening phases (a first complete round and a second partial round) and the interval between them, from the Finnish randomised health services study, to estimate the transition parameters of CRC progression, distribution of advanced tumour stage and sensitivity of the FOBT. From these estimates, we projected a 14% reduction in advanced colorectal cancers and a 9% reduction in mortality from CRC. A recent analysis of the Finnish programme showed no significant mortality reduction with a median follow-up time of 4.5 years, 12 however, a longer follow-up period should be considered, based on the natural history of CRC.
The pre-clinical incidence rate was 1.11 per 1000 person-years, which was slightly lower than the estimate of 1.48 obtained in the two previous randomised controlled trials.1,2,13 The sensitivity estimates were 65.12% (95% CI: 27.05%–77.05%) and 73.70% (95% CI: 47.49%–99.92%) for localised and non-localised PCDP, respectively. Both the transition rates and the sensitivities indicate that the Finnish routine programme was less effective when compared with the rates found in randomised trials. In fact, the average effectiveness for mortality rates was 12% in the trials after 14–20 years follow-up, 14 which was higher than the predicted 9% in our study. However, the observational results from the Finnish trial require a longer follow-up time to better evaluate the empirical results.
The preclinical incidence rate (0.00111) provides insight into the disease burden of CRC in relation to its biological features. A comparison of this parameter with the incidence rate in the control group reveals the possible over-detection of CRC that results from screening. As the pre-clinical incidence rate estimated in our study was fairly close to the incidence rate of the control group as corrected by the Hakama method 15 (0.00102), the over-detection of CRC ((0.00111–0.00102)/0.00111) was negligible (less than 10%).
The mean sojourn time (MST), an important indicator for screening purposes, is defined as the period between cancers of asymptomatic tumour(s) detected by screening and clinical findings of tumour(s) due to symptoms. These periods vary by population characteristics, screening tools used, interscreening interval, test performance, etc. However, as this metric cannot ethically be directly observed, it is best estimated by modelling based on empirical data. 16 Therefore, the MST also depends on the modelled parameters and the data used. For example, Launoy used results from the first round of a mass screening programme using FOBT in the Calvados region of France and found an MST of 4.5–5 years. 17 However, Prevost used the same data and the MSTs were estimated to be 2, 3 and 6 years for individuals aged 45–54, 55–64 and 65–74, respectively. 18 Jouve estimated the MST to be 2.56 years based on data from the Burgundy region. 19 Using colonoscopy as a screening tool in Germany, Brenner calculated the MST to be 4.5–5.2 years, based on the German national screening colonoscopy registry system. 20
Other studies have calculated the MST of screening with FOBT using a RCT design. For example, Gyrd-Hansen analysed data from Denmark and found that the MST and sensitivity were 2.1 years and 62.1%, respectively. 21 Wu used data from a RCT in the USA (Minnesota) that employed an annual screening interval and showed the MST to be 4.08 and 2.41 years for men and women, respectively. 22 The MST and sensitivity that we calculated from the Finnish RCT were quite similar to those found in Demark, which may be because the same screening tool was used, and the similar background and culture. The similarity in results also suggests that our estimate of the natural history of CRC was credible. The shorter MST in our study may have been affected by the sensitivity of the screening tool compared with colonoscopy.
In a previous meta-analysis on the effectiveness of biennial CRC screening using FOBT (Hemoccult), the CRC mortality reduction was 12% (RR = 0.88, 95% CI: 0.83, 0.94), 14 however, our study predicted a smaller 9% mortality reduction (RR = 0.91, 95% CI: 0.85, 1.02) for the Finnish trial. There are several reasons. First, we used the initial screening and partial second round results to project screening efficacy. In both previous trials (Denmark and UK), the data were based on five or six completed rounds. Repeated screening outcomes might contribute more information on tuning the transition parameters of natural history and sensitivities for localised and non-localised CRC. Second, by looking at the proportion of early stage screening-detected CRC, the Finnish preliminary result showed a lower proportion of early CRC detection (60%) compared with 77% and 73% found in the Danish and UK trials, respectively.1,2 The early CRC proportion of prevalent screening-detection was markedly lower (44%) than in the UK and Denmark (70%). This distribution would influence the transition rate estimation for CRC natural history. Third, compared with the Finnish trial target population (aged 60–69), both previous trials covered a younger population (aged 45–74) as well as the older population. This might affect the sensitivity, tumour stage and prognosis on mortality. These factors may be reflected in the difference in transition rates (PCDP localised→PCDP non-localised) between this Finnish trial (0.4849) and the combined UK and Denmark rates previously published (0.22), 13 which show more than twice as high transition rates as Finnish trial. In addition, the improvement of medical care may also affect the comparison of effectiveness between the study group and the uninvited group (control group), as the era of the organized service screening programme often comes after the era of the randomized controlled trial. These issues require further investigation and more detailed data. Therefore, such a comparison should be made with caution.
The simultaneous estimation of the MST and sensitivity has several advantages. The application of both parameters provides information on the proportion of interval cancers either due to false negative cases, or CRC cases newly diagnosed in the time since the last negative screening. Our estimates suggest that 28% of interval cancers were false negative cases, and 72% were newly diagnosed CRC in a biennial screening programme. The corresponding figures were 37% and 63% for an annual screening programme, and 22% and 78% for a triennial screening programme. This information provides policy-makers with the interval cancers detected with the different screening intervals used (see online Appendix Table A3).
Two points relating to the application of survival curves to predicting CRC death should be considered. The alternative to the use of the Weibull model is the exponential-cure model, which may be useful for prostate and possibly breast cancer, as it is powered to capture not only subjects cured by early treatment, but also overdiagnosis of early cancer. However, overdiagnosis of early cancer is hardly seen for CRC, so the exponential-cure model may not be needed here. The other reason for not using the exponential-cure model is that the predicted deaths from CRC were divided by localised CRC and non-localised CRC. The localised CRC may have already captured most cured patients. In spite of this, we performed the experiment with the exponential-cure model, by estimating the relevant parameters, and found that the survival curves based on the Weibull model and the exponential-cure model were close. The results of effectiveness on the reduction of advanced CRCs and mortality were also similar between both models.
When the mortality effect of screening is estimated via its effect on stage combined with stage-specific survival, the question of lead time arises; if the tumours in the screened population are diagnosed earlier, they will have a period of lead time in which the individual cannot die from the cancer, and it could be argued that some addition to the observation period post diagnosis should be made for screen-detected cancers. However, division by stage largely addresses this, and use of predicted fatality rates common to cancers of all detection modes, varying only by time and stage, should mean that, the predicted mortality effect is, if anything, conservative.
Conclusion
Using data from an ongoing Finnish population-based screening programme to estimate the natural course parameters of CRC and the sensitivity of the FOBT, our model demonstrated MSTs of 2.06 and 1.36 years and sensitivities of 65.12% and 73.70% for localised and non-localised CRC, respectively. These estimates predicted that the effect would be a significant 14% reduction in cases of advanced CRC during the 6-year study period, and a 9% reduction in mortality due to CRC in the 10 years after randomisation. These estimates, based on the preliminary results, fall short of those found in randomised screening trials. The predictions for the present study emphasise the need to confirm the CRC-related mortality in the Finnish programme by direct follow-up.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported in part from the Academy of Finland (Finland Distinguished Professor (FiDiPro)), the Finnish Cancer Registry, Cancer Society of Finland, the University of Tampere, the Ministry of Science and Technology in Taiwan (NSC102-2314-B-182-028-MY3), Chang Gung Memorial Hospital and Chang Gung University (BMRPB81).
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.


