Abstract
Objective
In a randomized controlled trial, the Promoting Early Presentation intervention increased older women’s breast cancer awareness after two years. We investigated whether this increase was sustained at three years, and the effect on breast screening self-referral.
Methods
We randomly allocated 867 women attending their final invited breast screening appointment to the Promoting Early Presentation intervention or usual care. We examined breast cancer awareness after three years and breast screening self-referrals after four years.
Results
Women in the Promoting Early Presentation intervention arm had higher breast cancer awareness at three years than the usual care arm (odds ratio: 10.4; 95% confidence interval: 3.1 to 34.8). There were no differences in proportions self-referring for breast screening between arms, but statistical power was limited.
Conclusion
The Promoting Early Presentation intervention has a sustained effect on breast cancer awareness in older women. The effect on self-referral for breast screening is unclear.
Keywords
Women who delay presentation of breast cancer symptoms have poorer outcomes, probably because they present with more advanced disease.1,2 This is a particular problem in older women, who are more likely to have poor breast cancer awareness, to delay presentation with breast cancer symptoms, and have worse survival from the disease.3,4 Women in the UK are invited for breast screening every three years from age 50 to 70, with some invited from 47 to 73 as part of a trial. 5 Following their final invited appointment, women may request further screening (self-referral), however, only about 20% of women aged 70 and over self-refer. 6
We developed the Promoting Early Presentation (PEP) intervention, a scripted 10-min one-to-one interaction delivered by a health professional and supported by a booklet, to provide older women with the knowledge, motivation, confidence, and skills to present promptly with breast cancer symptoms.7,8 In our trial, 867 women aged 67–70 attending their final routine appointment for breast screening in the NHS Breast Screening Programme were randomly allocated to receive the PEP intervention or usual care. 9 The main outcome was breast cancer awareness, a validated composite score encompassing knowledge of breast cancer symptoms, age-related risk of breast cancer, and frequency of breast checking. 10 Participants were considered breast cancer aware if they recognized five or more non-lump symptoms, identified a 70 year old as most at risk of developing breast cancer (compared with a 30 year old, a 50 year old, or a woman of any age), and reported checking their breasts at least once a month. These items were combined to form an ordinal composite score of breast awareness. Each component was given equal weighting and contributed one point to the total score, ranging from 0 to 3, with a score of 3 being breast cancer aware. The trial data and consort diagram have been previously published. 11 The PEP intervention increased breast cancer awareness compared with usual care at two years follow up. 12
This report summarizes further results of the trial: the effect of the PEP intervention on breast cancer awareness after three years, and self-referral for breast screening in the four years after randomization. Breast cancer awareness was assessed by postal questionnaire three years after receiving the intervention. Four years after every participant had attended their final routine breast screening appointment, we asked NHS Breast Screening Services to tell us which women in the trial had self-referred for further breast screening. We used generalized estimating equations to analyse the change in proportion that was breast cancer aware from baseline to three years, comparing PEP intervention with usual care. The analyses were carried out by intention-to-treat. In subsequent analyses, we adjusted the estimates, controlling for health professional, centre, and demographic characteristics at baseline. We examined whether, four years after recruitment, there were differences in the proportions that had self-referred for breast screening between the PEP intervention and usual care using logistic regression. All analyses were performed using Stata version 11.2 (College Station, TX).
Breast cancer awareness at baseline and three years post-randomization.
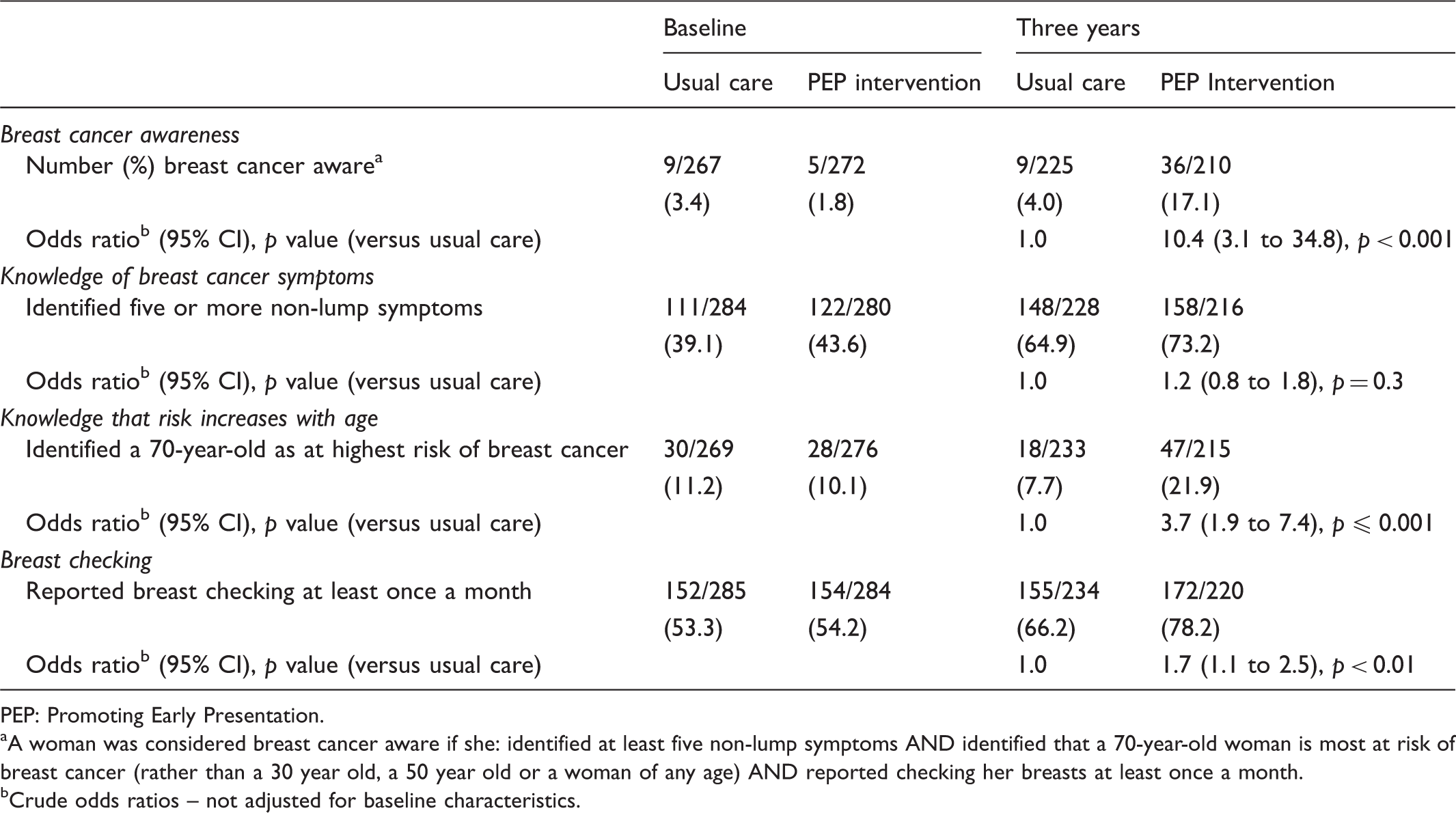
PEP: Promoting Early Presentation.
A woman was considered breast cancer aware if she: identified at least five non-lump symptoms AND identified that a 70-year-old woman is most at risk of breast cancer (rather than a 30 year old, a 50 year old or a woman of any age) AND reported checking her breasts at least once a month.
Crude odds ratios – not adjusted for baseline characteristics.
The analysis of breast screening attendance included 341 women. We excluded 232 women from this analysis because they had died or withdrawn consent (n = 33), were known to be taking part in other studies of breast screening or promoting early presentation of breast cancer (n = 157), or were invited for an additional round of screening by the breast screening service (n = 42). There were no significant differences in the proportions who had self-referred for breast screening between PEP intervention and usual care arms (35/168 versus 31/173; 21% versus 18%; OR 0.8; CI 0.5 to 1.4).
The PEP intervention increased breast cancer awareness in older women; the effect was sustained at three years but diminished over the three years. The effect was most striking for knowledge that the risk of breast cancer increases with age. We found no effect of the PEP intervention on self-referral for breast screening. A strength of the analysis of breast cancer awareness is the very high response rate, and we found no evidence that response bias (i.e. differences in pattern of response by arm) could explain the results. In both arms, knowledge that the risk of breast cancer increases with age was low compared with knowledge of breast cancer symptoms and frequency of breast checking, limiting the overall proportion of participants being able to achieve a score of 3 for breast cancer awareness. Future work should investigate why knowledge that risk of breast cancer increases with age is difficult for participants to retain and to develop better methods of communicating this message. The analysis of self-referrals was limited by low numbers. We would have needed a sample of about 7000 women to find the observed difference in proportions self-referring between PEP intervention and usual care arms statistically significant, with 80% power and significance level of 5%. It is also possible that we collected data too soon after randomization (four years), and that some women may have self-referred for breast screening later.
The PEP intervention has recently been tested in routine clinical practice and has been found to be effective at increasing breast cancer awareness, with sustained effects at one year (Lindsay Forbes/Rachael Dodd, personal communication). 12 In 2013, the All Party Parliamentary Group on Breast Cancer recommended that the PEP intervention should be rolled out in more breast screening services and tested in a variety of community-based health care settings to evaluate the effect on stage at diagnosis and survival. 13 If the PEP intervention were implemented across the whole NHS Breast Screening Programme, it has potential to prevent avoidable deaths from breast cancer resulting from delayed presentation of cancer symptoms in older women.
Footnotes
Acknowledgements
We thank the staff and participating women from the South East London, South West London, and Surrey Breast Screening Services for supporting the trial; Liz Kedge, Rachel Baxter, Ragini Jhalia, and Jo Sippitt for delivering the intervention; and Matthew Hotopf, Rebecca Walwyn, and Gunna Dietrich of the Data Monitoring Committee. Ethical approval was obtained from King’s College Hospital Research Ethics Committee 2007 (Ref. No.: 05/Q0703/51).
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This study was funded by Cancer Research UK (C4047/A12288). Trial registration: National Cancer Research Network and ISRCTN (31994827).
