Abstract
Objective
To determine (i) if electrospray mass spectrometry–mass spectrometry with the SpOtOn Diagnostics Ltd reagent kit for sickle cell screening could be integrated into the English newborn screening programme, under routine screening conditions, and provide mass spectrometry–mass spectrometry results which match existing methods, and (ii) if common action values could be set for all manufacturers in the study, for all assessed haemoglobins, to indicate which samples require further investigation.
Methods
Anonymised residual blood spots were analysed using the SpOtOn reagent kit as per manufacturer’s instructions, in parallel with existing techniques at four laboratories. Mass spectrometry–mass spectrometry instrumentation at Laboratories A and B was AB Sciex (Warrington, UK) AP4000, and at Laboratories C and D, Waters Micromass (Manchester, UK), Xevo TQMS and Premier, respectively.
Results
There were 23,898 results accepted from the four laboratories. Excellent specificity at 100% sensitivity was observed for haemoglobin S, haemoglobin C, haemoglobin E and haemoglobin OArab. A common action value was not possible for Hb C, but action values were set by manufacturer. The two haemoglobin DPunjab cases at Laboratory D were not detected using the common action value. Conversely, false-positive results with haemoglobin DPunjab were a problem at the remaining three laboratories.
Conclusions
This multicentre study demonstrates that it is possible to implement mass spectrometry–mass spectrometry into an established screening programme while maintaining consistency with existing methods for haemoglobinopathy screening. However, one of the instruments investigated cannot be recommended for use with this application.
Introduction
Screening for sickle cell disease (SCD) has been offered to all newborns in England via the heel prick blood spot since 2005. Other clinically significant conditions detected (e.g. beta thalassaemia major (BTM)) are reported to the clinicians to facilitate management of the condition. Techniques currently approved by the National Health Service (NHS) Sickle Cell and Thalassaemia Screening Programme in England are: high-performance liquid chromatography (nine laboratories), iso-electric focusing (two laboratories), and capillary electrophoresis (two laboratories), with standardised interpretation and reporting criteria. 1
Tandem mass spectrometry (MS/MS) is currently adopted in England as the screening technology used for detection of amino acid and other metabolic disorders (e.g. phenylketonuria and medium chain Acyl-CoA dehydrogenase deficiency). The procedure is automated, highly sensitive, quantitative and suited to high-workload volume testing. The sensitivity of MS/MS equipment has increased as the capital costs of these analysers has reduced.
As MS/MS is available in all newborn screening laboratories, using this equipment for newborn SCD screening could optimise its use. This has been previously suggested, 2 and screening at individual laboratories has been reported.3–6 Despite the evidence that MS/MS is an acceptable screening technique, concerns existed that standardised action values had not been tested in different laboratories using a variety of equipment. In addition, reporting strategies needed to be in agreement with existing methods to ensure equality of provision.
To address these issues, a pilot screening project was introduced in England in 2012, with staggered introduction into four newborn screening laboratories, selected for suitability of equipment and diversity of expected cases. MS/MS was run in parallel with standard techniques. The aims were (i) to determine if the SpOtOn Diagnostics Ltd reagent kit for SCD screening could be integrated into the English newborn screening programme under routine screening conditions, and provide MS/MS results which match existing methods, and (ii) to set common action values for all assessed haemoglobins, to determine which samples required further investigation and to review the impact of different manufacturers’ equipment on the proposed action values.
The analytical protocol of the commercially available reagent set incorporates a series of acquisitions performed simultaneously to detect haemoglobins (Hbs): S, C, DPunjab, E and OArab. Results are expressed as ratios of variant Hb signal divided by corresponding wild type signal. Current SCD screening methods all employ a visual assessment of results, with some providing numerical data. MS/MS results are solely numerical. Action values to determine which samples require further investigation therefore needed to be evaluated. Signals for the wild type beta and gamma globin are also assessed, as surrogates for Hb A and Hb F. The gamma/beta wild type ratio represents the relative proportion of Hb F to Hb A. Therefore, as with current procedures, a high ratio suggests low Hb A and prematurity or beta thalassaemia, while a low ratio indicates a low Hb F, which may be due to age, transfusion, gamma thalassaemia or a mutation in the selected gamma peptide.
Methods
Using anonymised residual samples, dried blood spots were analysed as per manufacturer’s instructions (SCD001 kit, SpOtOn Clinical Diagnostics Ltd, London, UK). MS/MS instrumentation in Laboratories A and B was AB Sciex (Warrington, UK) AP4000, and in Laboratories C and D, Waters Micromass (Manchester, UK), Xevo TQMS and Premier, respectively. Instrument settings were optimised at each laboratory. Three laboratories processed raw data through propriety software: Laboratory B: CliquidR Chemoview™ AB Sciex (Warrington, UK), Laboratories C and D: Neo-Lynx™, Waters Micromass (Manchester, UK). Laboratory A reviewed data manually and exported results to an Excel™ spread sheet. Although this project was a direct comparison with results obtained from existing methods, where possible, laboratories utilised internal quality control which had been prepared in-house, and when available, external quality control samples.
Results
In total, 23,898 results were accepted from the four laboratories. Due to technical issues, only 2364 were from Laboratory B. Common action values using numeric ratios of the variant and wild type signal in each acquisition were derived from earlier work, 3 and used to determine which samples required confirmatory testing. There are specific acquisitions for all Hbs, including two-designed to detect BTM. In any given acquisition, absence of wild type can be sufficient to give a result above the action threshold without an increase in the signal of the Hb in question. Results were compared with those reported by existing methods.
Hb S
Numeric values for Hb S ratios at all laboratories.
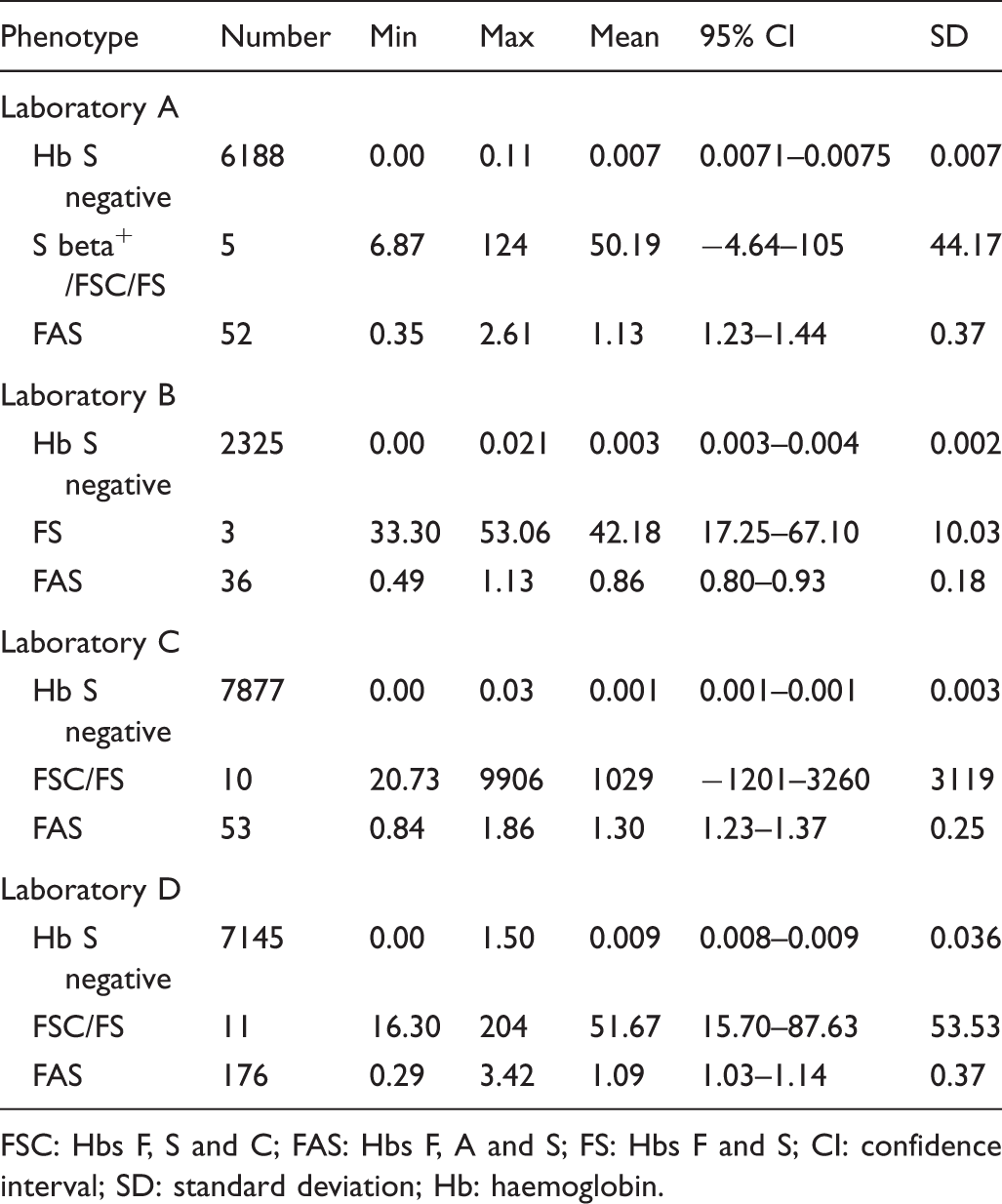
FSC: Hbs F, S and C; FAS: Hbs F, A and S; FS: Hbs F and S; CI: confidence interval; SD: standard deviation; Hb: haemoglobin.
Three laboratories had cases where absence of wild type resulted in a ratio above the Hb S action limits. Two laboratories had BTM cases, Laboratory A (n = 5, range, 0.04–0.66), and Laboratory C (n = 11, range, 0.00–0.30). There were three Hb FC cases at two laboratories, Laboratory A (n = 1, ratio, 0.29) and Laboratory D (n = 2, ratios, 0.06 and 0.1).
Hb C
As it was not possible to have the same action values across all laboratories for Hb C, common values were set for instruments from the two different manufacturers: > 1.00 for Waters Micromass instruments and > 0.5 for ABI Sciex. At these levels, there were no false-negatives, but there were false-positives, at Laboratory A (n = 4, range: 0.62–0.72), Laboratory C (n = 3, range: 1.1–1.28), and Laboratory D (n = 17, range: 1.01–3.58). True-positive numbers were, for Laboratory A: n = 12, lowest ratio, 1.18; Laboratory B: n = 12, lowest ratio, 0.94; Laboratory C: n = 17, lowest ratio, 4.5; and Laboratory D: n = 33, lowest ratio 1.24.
Hb DPunjab
An action value of > 1.0 was set for all laboratories for Hb DPunjab. At this level, at Laboratory D, there were two false-negatives. There were also false-positives at Laboratory A (n = 31, range: 1.01–3.53) and Laboratory C (n = 104, range: 1.01–1.62). True-positive numbers for each laboratory were, for Laboratory A: n = 17, lowest ratio, 1.39; Laboratory B: n = 13, lowest ratio, 1.12; Laboratory C: n = 16, lowest ratio, 1.31; and Laboratory D: n = 2, not detected, below action limits, 0.29 and 0.32.
Hb E
An action value of > 0.19 was set for all laboratories for Hb E. At this level, there were no false-negatives but there were false-positives, at Laboratory A (n = 1, ratio, 0.32), Laboratory B (n = 1, ratio, 0.23) and Laboratory D (n = 2, ratios, 0.23 and 0.36). True-positive numbers for each laboratory were, for Laboratory A: n = 10, lowest ratio, 0.63; Laboratory B: n = 4, lowest ratio, 0.77; Laboratory C: n = 19, lowest ratio, 0.20; and Laboratory D (n = 7, lowest ratio 0.57).
Hb OArab
An action value of > 0.6 was set for all laboratories for Hb OArab. At this level, there were no false-positives or negatives; however, there were only three true-positives, all from Laboratory A (range: 0.72–0.83).
Gamma acquisition
Gamma T2 ratios for all laboratories.
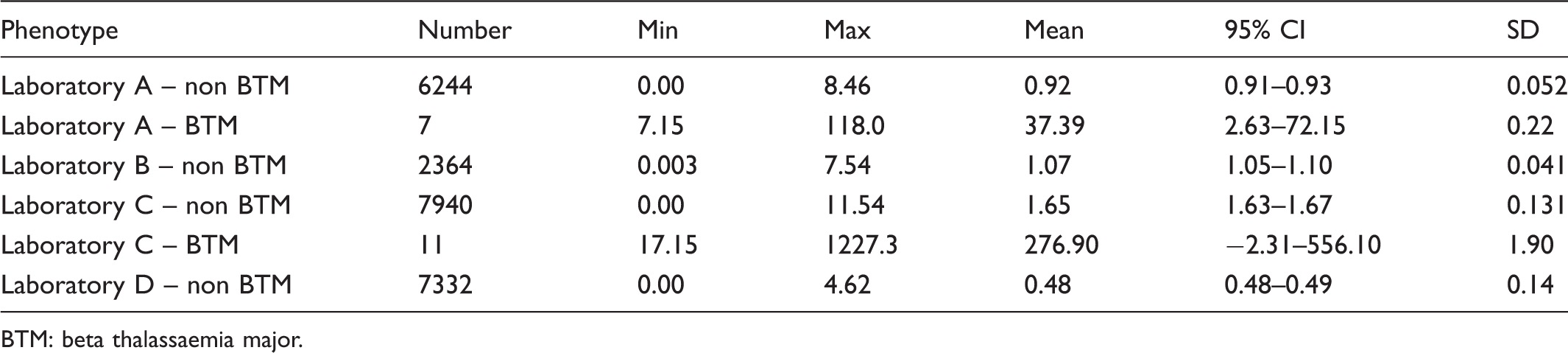
BTM: beta thalassaemia major.
Sensitivity and specificity
Sensitivity and specificity with common action values.
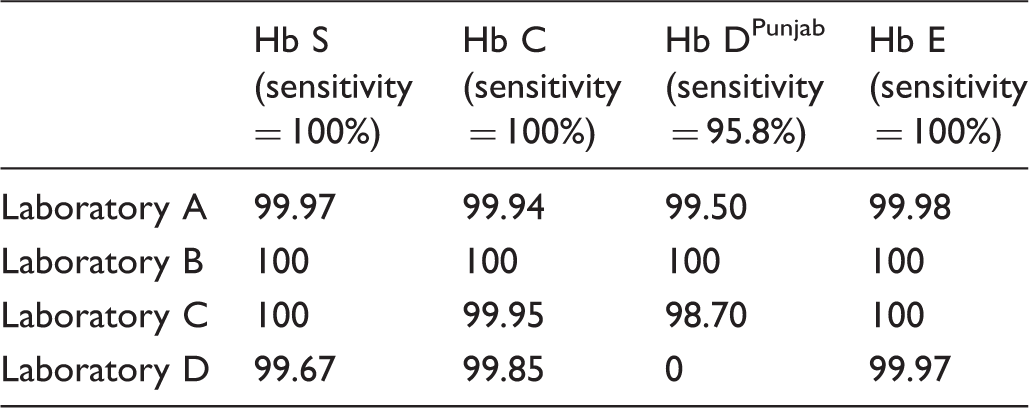
Discussion
The results demonstrate that this method offers an alternative approach for newborn SCD screening. Excellent specificity at 100% sensitivity was observed for Hb S, Hb C, Hb E, and Hb OArab. The superior specificity at Laboratory B is likely to be due to the low sample numbers. In practice, samples that exceed the action value threshold, but do not have the Hb under investigation, will be eliminated by retesting with another method, before the final report is issued. Unlike existing techniques, the method is mutation-specific; therefore, all cases containing the mutation under investigation will be detected, and misclassification of co-eluting variants should not occur. There were challenges in ensuring consistency at multiple laboratories with instruments of varying specification from different manufacturers. A common action value was not possible for Hb C, but action values were set by the manufacturers (>1.00 Waters Micromass and >0.5 ABI Sciex). Individual laboratory values were not consistent with the objectives and would make it difficult to ensure uniformity. The ratios are remarkably consistent with those from recent publications,5,6 although these are from single laboratories, with protocols that differ slightly from those reported in this pilot.
Hb DPunjab presented a number of issues, believed to be due to the small mass difference from wild type, which is exacerbated by any loss of sensitivity in the instrument. The results demonstrate that the Waters Premier instrument, as used at Laboratory D, cannot be recommended for use with this application, as the two cases of Hb DPunjab at this laboratory were not detected and there was reduced specificity with the remaining Hb variants. Conversely, inappropriate results above the action threshold with Hb DPunjab were a problem in the remaining three laboratories; however, it was not possible to raise the action value and maintain sensitivity. We are investigating strategies to reduce this problem and thus minimise the numbers of samples requiring further testing. This includes altering the algorithm to assess both the ratio and the signal obtained for the Hb DPunjab acquisition, as well as working with the manufacturer to improve specificity.
Following this study and an extended pilot at Laboratory A, gamma action values were set at T2 < 0.2 and >3, and T5 > 2.5. Due to the low incidence of beta thalassaemia cases, gamma action values remain under review in one of the pilot laboratories that has chosen to introduce this methodology. Values will also be kept under review in any laboratories that perform evaluations prior to routine use. Current evidence indicates that the action values set will be applicable to all instruments accepted in this study. As with existing protein techniques, low expression of adult Hb due to causes other than BTM, such as prematurity, remains a confounding factor.
Conclusion
This pilot study demonstrates that the SpOtOn Clinical Diagnostics Ltd kit using MS/MS offers a valuable alternative approach for newborn SCD screening. The method shows excellent sensitivity and specificity for Hb S on all instruments used in this study. The objective of this work was to replicate, as far as possible, existing practice in England. However, the excellent performance with Hb S and the flexibility of the approach allow for adaption to fit the requirements of other countries, for example, avoiding the detection of carriers. 6 The results show that it would be possible to implement this method into an established screening programme while maintaining consistency with existing technologies. It was not possible to use the same action values for all Hbs on all MS/MS instruments, although this has been achieved for the majority of Hbs. An advantage of working in multiple laboratories was the ability to obtain greater numbers of positive samples, potentially a problem for individual laboratories. The lack of appropriate commercial control material to monitor the performance of the ratios means that laboratories using this method would need to prepare their own in-house materials. The use of the SpOtOn Clinical Diagnostics kit will be included in the next edition of the NHS Sickle Cell and Thalassaemia Screening Programme Laboratory Handbook, scheduled for publication 2016.
Footnotes
Acknowledgements
We thank the staff in the newborn screening laboratories who took part in this pilot project. Thanks to Prof Neil Dalton, Charles Turner, and Dr Jo Howard for advice and critical reading of the manuscript, and to Waters Micromass for the loan of an instrument.
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Yvonne Daniel is named on the patent for this method but has received no financial benefit.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
