Abstract
Objectives
To evaluate the effectiveness of the NHS breast screening programme (NHSBSP) on breast cancer mortality in England and Wales and to compare findings with a cohort analysis of the same underlying population.
Methods
A nested case-control study within a cohort of 959,738 women in England and Wales aged 49–64 who were eligible for routine NHSBSP screening during 1991–2005. Cases who died from breast cancer in 1991–2005 were matched to controls without breast cancer at the case diagnosis date and alive when the case died. Risk of breast cancer mortality associated with intention to screen (ITS) (7047 cases/28,188 controls) and screening attendance (4707 cases/9413 controls) was examined. Bias was minimised in accordance with currently advocated best practice. Odds ratios (ORs) were calculated using conditional logistic regression. Results were compared with findings from an incidence-based breast cancer mortality cohort analysis.
Results
ITS was associated with a 21% breast cancer mortality reduction (OR = 0.79, 95% confidence interval [CI]: 0.71–0.88,
Conclusions
Case-control studies designed and analysed according to current best practice guidelines offer an effective means of evaluating population breast screening.
Introduction
Following demonstration of the effectiveness of mammographic screening for breast cancer by randomised clinical trials, 1 the UK introduced a National Health Service Breast Screening Programme (NHSBSP) in 1988, offering three-yearly mammographic screening for the early detection of breast cancer. This is now one of the largest nationally organised programmes in the world and currently invites around three million women annually for screening. 2 In the light of improvements in breast cancer treatment, there is ongoing debate over the effectiveness of the NHSBSP and similar programmes in reducing breast cancer mortality.3–5
Evaluation of the effectiveness of population screening relies predominantly on observational studies. Strategies used to evaluate the effect of population breast screening on breast cancer mortality include cohort, case-control, and secular trend analyses. 6 In a screening setting where all eligible women are invited, it is difficult to identify a contemporary uninvited comparison group, so case-control designs have been widely implemented to assess effectiveness.7–12 While case-control evaluations offer a statistically and economically efficient approach,13,14 they are susceptible to bias and are generally considered less robust than cohort studies.15–18 Variation in the design of case-control studies may have contributed to differences in reported estimated effect sizes, which have frequently been larger than those reported from cohort studies. 6 In case-control breast screening evaluation studies, the definition and selection of cases and controls, the definition of screening exposure, and the issue of self-selection can all result in bias, both in nested and un-nested studies. Studies of these issues16,19,20 have led to a raft of recommendations to optimise study design and analysis.14,20
A case-control screening evaluation of a population which has also been the subject of a cohort evaluation offers a means of gaining insight into possible sources of variation between different case-control methodologies and the validity of the current guidance on their design. Nested case-control studies have been conducted within a number of the early comparison studies and randomised controlled trials (RCTs) of mammographic breast screening.21–24 The Health Insurance Plan (HIP) RCT nested case-control study estimated a breast cancer mortality reduction associated with intention to screen of 25% (odds ratio [OR] = 0.75, 95% confidence interval [CI]: 0.60–0.93), compatible with the 20% mortality reduction shown by the parent RCT.21,22 A case-control study nested within the UK Age Trial, which offered annual mammographic screening to women in their forties, reported a breast cancer mortality reduction of 17% associated with screening invitation, compared with the 12–15% reduction estimated by the RCT.25,26 The impact of screening attendance in their study varied according to the study design and analysis methodology used. 25 To date, few studies have directly compared the magnitude of effect from case-control studies and cohort analyses, using the same population, within routine population breast screening. A recent case-control evaluation of population breast screening in Finland 27 investigated the impact of study design options and demonstrated breast cancer mortality reductions (OR = 0.70, 95% CI: 0.49–1.00) similar to a cohort evaluation of the same population (rate ratio [RR] = 0.72, 95% CI: 0.56–0.88). 28
We here use the study population of a cohort evaluation of the NHS breast screening programme (the BRCOH study), 29 over a million women with individual-level screening exposure information, breast cancer incidence, and mortality outcome data, to conduct a nested case-control study to assess the validity of the case-control approach.
Methods
Cases and controls were selected from the previously reported BRCOH cohort. 29 The BRCOH study area covered one-third of England and the whole of Wales. Screening histories were extracted from regional screening call/recall registers (‘Exeter’ system databases). Information on deaths and diagnoses of breast cancer in the cohort was obtained from the Office for National Statistics national death and cancer registers, using individual-level linkage. Socio-economic status was estimated based on postcode of residence at study entry using the Townsend Index, 30 based on 1991 census information, and was available for 96% of the BRCOH population. Ethical approval was obtained from the South East MREC (MREC 02/01/64). The BRCOH population comprised 1,426,379 women who entered the study on 1 January 1991 aged 49–64. We excluded from the analyses women lost to follow-up before entry (14,157), women with breast cancer diagnosed before entry (28,870), and women invited by the NHSBSP before entry (395,262). Women whose first NHSBSP attendance was a self- or General Practitioner-referral were also excluded from the case-control analysis to ensure attendance was in response to routine NHSBSP invitation, providing a source population of 959,738 women.
Cases were women in the cohort who died between 1 January 1991 and 31 December 2005 with breast cancer recorded as the underlying cause of death on the national death register. Controls, retrospectively matched to cases, were cohort members with no diagnosis of breast cancer at the date of diagnosis of the case and who were alive at the date of death of the case. Each control was allocated a pseudo-diagnosis date equal to the date of diagnosis of the case. Women who left the study area were eligible as controls, provided they exited after the date of death of the case.
Investigating the effects of both intention to screen (ITS) and screening attendance on breast cancer mortality required two analysis sets, each comprising cases and controls matched using different pre-defined criteria. For the ITS analysis, four controls were matched per case on date of birth ±≤3 months and Townsend deprivation quintile (Analysis Set-1). For the attendance analysis, two controls per case were matched on date of birth ±≤3 months, Townsend deprivation quintile, and index-invitation date ±≤6 months (Analysis Set-2), index invitation being defined as the most recent invitation to routine NHSBSP screening preceding the date of breast cancer diagnosis/pseudo-diagnosis. 31 Two, rather than four, controls were used for Analysis Set-2 as fewer eligible controls were available after adding the requirement for the index invitation to match within six months. Random selection was used when the number of suitable individuals exceeded the requisite number of controls for a case. Controls for each analysis set were selected independently.
In the NHSBSP, women were scheduled for invitation on the basis of demographic information, but further eligibility, on the basis of health, was checked by their General Practice before invitation. Ill-health could therefore result in a scheduled woman not being invited and so using invitation as the measure of exposure in an ITS analysis could bias the estimate in favour of screening, because of a healthy invitee effect. 29 To exclude this effect, scheduling for invitation was used as the measure of exposure in ITS analyses (subsequently referred to as ‘invitation’). In our primary analysis of screening attendance, exposure was measured as ever/never screened within a defined time period preceding diagnosis/pseudo-diagnosis. The impact of attending the first routine NHSBSP screen was also investigated, to compare with findings from the cohort analysis of the BRCOH study population. 29 Analyses comparing attenders and non-attenders were conducted with the stipulation that cases and controls were invited prior to diagnosis/pseudo-diagnosis.14,16,20
Only the screening history up to and including the date of diagnosis/pseudo-diagnosis was considered. Exposure opportunity bias 13 resulting from inclusion of the screen at which screen-detected cases are identified can exert a substantial bias against screening at the start of a programme when prevalent screening is dominant. Prevalent NHSBSP screening predominated in the BRCOH study population between 1991 and 1993, when 95% of screens were first attendances. To minimise exposure opportunity bias, we excluded the earliest years of the screening programme, 17 as follows. For the analysis of mortality in attenders and non-attenders within a defined exposure window, the whole of the period 1991–1993 was excluded. In analyses comparing risk in invited and uninvited women, however, exclusion of the entire prevalent period would have excluded a large proportion of eligible uninvited women. ITS analyses therefore excluded only the first two years of the prevalent screening period (1991–1992). Sensitivity analysis was conducted to assess the impact of excluding a prevalent period of varying length.
Participation in screening can only influence breast cancer mortality if performed in the period when the breast cancer may be detectable by mammography, but is not symptomatic.16,20,32,33 While this preclinical detectable period is not known at the individual level, published studies suggest an average lead-time of 4–6 years.14,34–38 For primary analysis of mortality in relation to attendance, we therefore used an exposure period of five years prior to diagnosis/pseudo-diagnosis. For completeness, we also considered four- and six-year exposure periods.
The relationship between invitation or screening and breast cancer mortality was assessed using ORs and associated 95% CIs, calculated using conditional logistic regression. Attendance analyses were adjusted for self-selection bias using the methodology developed by Duffy and Cuzick to estimate the effect of offering screening to those who would participate if invited (OR2).
39
The OR in attenders compared with non-attenders is adjusted to take account of the different, often increased, mortality risk in non-compliers, and is calculated as: OR2 = (P.ORA.RRN)/(1-(1-P).RRN) where ORA is the OR for breast cancer mortality in attenders vs. non-attenders, RRN is the rate ratio for breast cancer mortality in non-attenders vs. uninvited women: a population-specific correction factor of 1.19 calculated using data from the BRCOH cohort analysis.
29
P is the proportion attending in response to invitation, 74% for first invitations and 75% for the average uptake over all rounds, and was derived from the BRCOH population. For comparison purposes, the form of the Duffy and Cuzick correction for self-selection bias that approximates an ITS estimate of effect, OR1, was also derived: OR1 = RRN.((P.ORA) + (1-P)). CIs and
Results
There were 7047 cases identified from the BRCOH study population who had died from breast cancer and for whom Townsend deprivation information could be derived. The cases were born during 1926–1942, were diagnosed with breast cancer during 1991–2005 aged 49–79, and died from breast cancer during 1991–2005.
Case-control analysis sets.

DOB: date of birth; SES: socio-economic status, measured by Townsend deprivation quintile; DXI: date of index-invitation.
4706 cases have two matched controls, one case has one matched control
Risk of death from breast cancer, by type of breast screening exposure, case-control and cohort analyses.
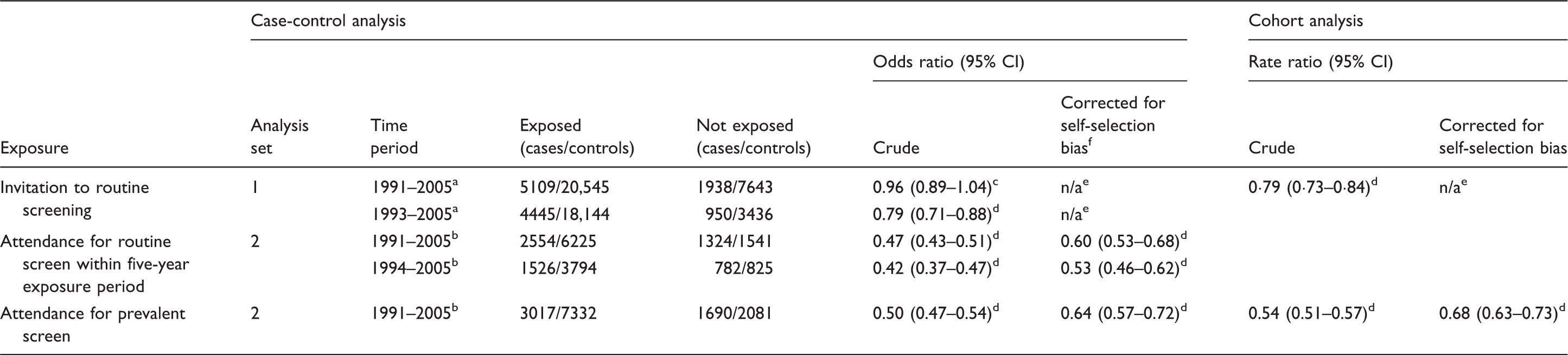
Time period determined by date of diagnosis/pseudo-diagnosis.
Time period determined by date of index-invitation.
self-selection bias does not operate in intention-to-screen analysis.
Compliance rate for bias adjustment: 75% for a five-year exposure period and 74% for prevalent screen.
Of the 14,120 women in Analysis Set-2, 11,644 (3878 cases/7766 controls) were invited for routine NHSBSP screening within the five years preceding diagnosis/pseudo-diagnosis, and 75% attended. The crude risk of death from breast cancer was 53% lower among women who attended during 1991–2005 (OR = 0.47, 95% CI: 0.43–0.51,
Based on Analysis Set-2, the uncorrected breast cancer mortality reduction associated with accepting a prevalent screen invitation between 1991 and 2005 was 50% (OR = 0.50, 95% CI: 0.47–0.54,
Discussion
In this case-control analysis nested within the BRCOH cohort, invitation to screening was associated with a 21% reduction in breast cancer mortality. Attendance in the five years prior to diagnosis/pseudo-diagnosis was associated with a 47% reduction in breast cancer mortality. These findings are consistent with the cohort analysis of the BRCOH population, and with recently reported estimates from a meta-analysis of case-control studies of European breast screening programmes, 6 which reported an estimated 31% reduction in breast cancer mortality associated with ITS (OR = 0.69, 0.57–0.83) and a 48% reduction associated with screening after correction for self-selection (OR = 0.52, 95% CI: 0.42–0.65).
To mitigate against potential biases for which case-control studies for screening evaluation have been criticised, in this study we have adopted best practice as advocated by Verbeek and Broeders 14 and Paap et al. 20 We restricted analyses to women invited before diagnosis/pseudo-diagnosis, excluded the prevalent screening period, used an exposure period based on the preclinical detectable period, and adjusted for self-selection bias using a contemporary setting-specific correction factor.
The screening period on which the BRCOH study is based includes the early years of the NHSBSP. Inclusion of this phase of a programme, when prevalent screens predominate, tends to bias estimates of effectiveness against screening. This is partly due to a higher proportion of cases detected at prevalent screen being of advanced stage. There is also potential exposure opportunity bias, in which cases that were screen-detected are more likely to have been screened than their matched controls, an effect which tends to be minimal outside the prevalent period.10,13,40 The most commonly advocated method to address these sources of bias is exclusion of the prevalent screening period.11,20,41,42 Screening units in England and Wales completed their first round of NHSBSP screening by 1995. 43 Excluding a prevalent period conservatively restricted to 1991–1993 resulted in an additional 5% reduction in breast cancer mortality associated with screening attendance in a five-year exposure window, after correction for self-selection bias. Estimating the impact of ITS on breast cancer mortality relied on including the NHSBSP implementation period, when a suitable unexposed group of women could be identified. However, by definition, the implementation period was when prevalent screens predominated. The compromise we adopted was to exclude cases diagnosed in the first two years of the exposure period, during which 99% of attendances were for prevalent screening, still leaving a sufficient time in the implementation period to detect an effect.
Whether a woman has ever or never been screened has been the most common measure of exposure in published case-control evaluations of breast screening. However, measuring screening exposure in this way, irrespective of how long in the past the screen occurred, has the potential to underestimate a positive effect of breast screening. The importance of using an etiologically relevant exposure window based on the preclinical detectable period is therefore widely advocated,16, 20,32,33 and a 4–6-year period prior to diagnosis/pseudo-diagnosis is considered appropriate.25,32,33
Matching cases and controls on the date of their most recent routine NHSBSP invitation prior to diagnosis/pseudo-diagnosis in our study afforded the advantage of comparing exposure between cases and controls that was contemporaneous and comparable in terms of screening policy and social behaviour. Estimates of effect are therefore less likely to suffer from confounding by changes in non-screening factors over time, although we acknowledge that geographical differences (e.g. in treatment), could still operate. Tamoxifen and adjuvant therapy were generally used in clinical practice throughout our evaluation period,44–46 so changes in patient management are unlikely to have affected study findings substantially.
Few case-control evaluations of population-based screening have specifically estimated the impact of invitation on breast cancer mortality.11,12 Our ITS finding of 21% is similar to the 25% reduction reported in Italy by Puliti et al., 12 but is lower than the 35% reported from East Anglia in the UK. 11 The East Anglian study made use of a form of the Duffy and Cuzick correction for self-selection bias which, in settings where there is no uninvited control group available, can be applied to a comparison of attenders and non-attenders to derive an estimate of the effect of ITS. 39 For comparison purposes, this estimate (OR1) was calculated from the BRCOH case-control attender/non-attender analysis. The value of OR1 derived from the case-control estimate suggested a mortality reduction 7–12% greater than the 21% estimated directly from the ITS analysis. The magnitude of the difference we observed between OR1 and the ITS estimate should aid interpretation of other case-control evaluations of screening in settings where OR1 is the only metric that can be derived for estimating the impact of invitation.
The case-control analysis of breast cancer mortality in relation to attendance for first routine screen was conducted for comparison with the BRCOH cohort analysis, 29 and indicated a mortality reduction 4% greater than the equivalent analysis based on a cohort approach (Table 2). This could support the assertion that the magnitude of the effect estimated by case-control analyses is less accurate than in cohort analyses. However, the difference may, in part, be due to aspects of the cohort analysis, specifically the restriction of attenders to women who attended within six months of their first invitation, and to dilution by later attendance in initial non-attenders.
In our study, the magnitude of the breast cancer mortality reduction estimate associated with ITS was 21%, for both the nested case-control approach and incidence-based mortality cohort analyses of the BRCOH study population 29 (Table 2). A case-control study nested within the HIP RCT, initiated in 1963 in Greater New York, was unusual in investigating the effect of ITS as well as the effect of being screened. The estimated breast cancer mortality reduction associated with allocation to screening in HIP was similar to, though slightly greater than, the mortality reduction shown by the RCT.21,22 Case-control studies within a UK comparison study 24 and a UK trial of breast screening 25 have also shown similar ITS results to those from the studies in which they were nested, though again, the impact estimated by the Age Trial nested case-control study was slightly greater than the estimate from the RCT.25,26 In the context of population screening, Heinavaara et al. 27 performed a case-control analysis of breast screening in Finland and compared findings with a cohort study of the same population. 28 Very similar estimates for the effectiveness of breast screening were obtained from their case-control study and the earlier cohort study. Guidelines for best practice regarding the design and analysis of case-control studies for screening evaluation advocate the importance of appropriate selection of cases and controls, relevant screening exposure definition, and contemporary, population-specific correction for self-selection bias.14,20,24,25,27 These guidelines aim to address the main criticisms of case-control studies for screening evaluation, and the findings of this, and other studies directly comparing case-control and cohort analyses of the same population, provide evidence to support this view.
Conclusion
Nesting a case-control study within the BRCOH population offered the opportunity to implement analysis methodology aimed at minimising the biases specific to case-control evaluations of screening, and to compare results with the cohort analysis. The nature of the biases addressed means our findings have implications for free-standing, as well as nested, case-control evaluations. The magnitude of the mortality reduction associated with ITS estimated by case-control analysis and primary cohort analysis of the BRCOH study population were in agreement. The estimated benefit associated with screening attendance was, however, marginally higher in the case-control analysis. By applying current recommendations on appropriate study design and analysis, case-control screening evaluation studies can provide estimates of effect that are comparable with those obtained from cohort analyses.
Footnotes
Acknowledgements
The authors thank the staff at the former Medical Research Division of ONS, and their colleagues at Titchfield, Hampshire for their work on the linkage of the cohort with mortality and cancer incidence databases at ONS; the breast team at Connecting for Health for development of software to extract relevant data for the study from the screening call/recall system; the call/recall system sites that contributed data to this study; within the CSEU Derek Coleman for his contribution to the cohort analysis of the BRCOH study, and Jane Hemming and Sarah Addou for their contribution to data management; and the Census Dissemination Unit at the University of Manchester for the provision of pre-calculated enumeration-level Townsend scores based on 1991 census area statistics.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was funded by the Policy Research Programme of the Department of Health for England (grant no. 0040064).
