Abstract
Introduction
Few population-based studies have been conducted on the efficacy of human papilloma virus (HPV) testing as a test of cure for high grade cervical dysplasia.
Methods
A cohort of women (n = 11,521) with high grade (CIN2+) cervical dysplasia diagnosed during 2006–2010, who also had accompanying HPV testing (n = 19,434 HPV tests), was analyzed with respect to National Health and Medical Research Council (NHMRC) follow-up guidelines. Cure rates, and number of follow-up tests and times to reach a cure were estimated.
Results
In the ≥2-years following high grade dysplasia, 53% of women had a single follow-up HPV test, 33% had two, and 14% had three or more HPV follow-up tests. 75% of women with follow-up HPV and cytology testing according to NHMRC guidelines were cured (2,210 from 2,948). Cure rates were lowest in those aged <30 (71%) and highest in women aged 30–49 (80%). Of those cured, 68% attained cure by the second HPV test, 21% by the third, and 11% at the fourth or later HPV test. The median time to cure was 1,097 days. In non-cured women, 56% of test results originated from all-negative cytology with positive HPV, compared with 9% from all-negative HPV results.
Conclusions
Cure rates in women with follow-up testing according to NHMRC guidelines are high. Further studies are needed of the high proportion of women with negative cytology classed as not cured due to HPV positivity, and of the high proportion of women with high grade dysplasia who had one follow-up HPV test only.
Introduction
Numerous follow-up studies of patients with high grade cervical dysplasia have shown human papilloma virus (HPV) testing to be efficacious for detecting residual or recurrent disease.1–16 The high sensitivity of the HPV test (typically 95–100%) means that a negative HPV test result is a highly certain indicator of effective treatment (ie. high negative predictive value). However, the HPV test’s lower specificity will classify some cured high-grade cervical lesions as not cured. While around 75% of women treated for high grade CIN at initial follow-up (6–9 months after treatment) will have negative cytology, a proportion also test positive to oncogenic HPV, but have negative cytology at 12 months follow-up. A 2006 study of HPV testing as a test of cure found that 15% of women with positive HPV and negative cytology at follow-up had recurrent or residual CIN, significantly lower than the 50% of those with an abnormal Pap test at follow-up. 3 Conversely, a UK study found very small proportions of women with negative HPV and cytology 6 months after treatment to be positive at 12 and 24 months follow-up. 17 Based on these latter results, the authors recommended that negative HPV and cytology 6 months following treatment for high grade dysplasia was a sufficient criterion for cure. It is not known if these figures apply to NSW women. Nor, more broadly, is the extent of compliance with the recommended follow-up test-of-cure protocol or cure rates for high-grade cervical dysplasia known. These questions are the subject of this paper.
In July 2006 HPV testing became subsidized by the Australian universal health insurance scheme, Medicare, for use as a test of cure of CIN2+ cervical dysplasia. The present study is a descriptive-analytic study of HPV testing in NSW, the most populous Australian state, with respect to high-grade (CIN2+) cervical dysplasia. Compliance and cure rates are estimated according to criteria established by the National Health and Medical Research Council (NHMRC) for follow-up HPV test of cure for high-grade cervical dysplasia.
Methods
A cohort of women with their most recent high grade (CIN2+) histology occurring from 1 July 2006 to 31 December 2010, and who had subsequent HPV and cytology testing, was extracted from the NSW Pap Test Register (PTR) along with relevant follow-up cytology and HPV test results, the latter occurring from 1 July 2006 to 31 March 2013. The cohort of 11,521 women represented 53% of all women (n = 21,596) who had their last high grade histology diagnosed following the announcement of HPV testing being eligible for Medicare subsidy as a test of cure.
According to NHMRC follow-up guidelines, a woman treated for histological high grade cervical dysplasia (CIN2+) should be managed as follows:
18
Colposcopy and cervical cytology at 4–6 months after treatment Cervical cytology and HPV DNA ‘test of cure’ at 12 months after treatment and then annually until both tests are negative on two consecutive occasions Then return to the usual two-yearly screening interval
For the purposes of this paper, the NHMRC-compliant follow-up intervals were operationalized according to National Cervical Screening Program Minimum National Standards for Follow-up and Reminder Protocols for Cytology Registers: 19 counting the first HPV test occurring at 365 or more days after the histology test request date resulting in a finding of CIN2+; the second follow-up HPV test is regarded as compliant if it occurred 300 days or more following the first NHMRC-compliant HPV test (ie. at least 665 days after the CIN2+ histology and at least 300 days following the first HPV test). If the second HPV test occurred within 665 days of the CIN2+ histology, this test was taken as the first follow-up HPV test, and the next HPV test as the second test. This was because the test-of-cure criterion mandates two consecutive tests at the stipulated intervals be negative before the woman is considered “cured” and returns to normal screening. The exception to this was if the second test was all negative and was followed by a third test, also all negative, but conducted after 665 days. The first and third tests in this scenario only were regarded as ‘consecutive’. The same criteria were applied to follow-up cytology. Most follow-up HPV testing and accompanying cytology occurred at the same time, but there were numerous instances of cytology and HPV testing not being performed simultaneously. For the purposes of describing test pair results, HPV testing and cytology occurring within one month of one another were considered ‘simultaneous’, and distributions of test pair results of these are shown.
Test-of-cure results are examined in relation to the recommended NHMRC interval cut points, as above. No subsequent high-grade histology results are examined. Only the most recent (last) high-grade histology results were selected and linked to subsequent HPV and cytology tests to estimate rates of cure. The study cohort had 19,434 HPV tests performed along with cytology subsequent to the CIN2+ histology. The cut-off date of 31 December 2010 for index histology was chosen to allow sufficient post-histology follow-up time of at least 27 months, up to 31 March 2013. Distributions of HPV and cytology tests subsequent to the CIN2+ histology, and cure rates by broad age group at histological diagnosis (age <30, 30–49, 50–69) are shown.
Index high-grade histology included squamous-cell histology only, and excluded histology relating to glandular cells. Excluded glandular histology codes were: M62000, Abnormal cell, NOS; M67020, Columnar cell atypia; M67030, Glandular cell atypia; M67031, Mild glandular cell atypia; M67032, Moderate glandular cell atypia; M67033, Severe glandular cell atypia; M80762, Squamous cell carcinoma in situ with questionable stromal invasion/Epidermoid carcinoma in situ with questionable stromal invasion; M80772, Intraepithelial neoplasia, grade III, of cervix, vulva and vagina; M80102, Carcinoma in situ, NOS/Intraepithelial carcinoma, NOS; M81402, Adenocarcinoma in situ, NOS; and M85602, Adenosqua- mous carcinoma in situ/Mixed adenocarcinoma and squamous cell carcinoma in situ/Mixed adenocarcinoma and epidermoid carcinoma in situ.
A positive HPV result was defined as detection of an oncogenic HPV type; an accompanying positive cytology result was defined as a low grade squamous intra epithelial lesion (LSIL) or higher.
Proportions of women cured and consecutive test result pairs were plotted along with their exact (Clopper-Pearson) binomial 95% confidence intervals. These confidence intervals are conservative, meaning that their probability of covering the point estimate is at least 95%. SAS version 9.3 was used for the data manipulation, statistical analyses and graph production. Further graph manipulation was performed with GIMP version 2.6.
The Cancer Institute NSW is the organization with delegated authorization to manage the NSW PTR on behalf of the NSW Ministry of Health. Data on cervical screens are collected by the PTR under the NSW Public Health Act of 2010, and management of the PTR includes assessment, review, maintenance and enhancement of the quality of all data held in the register. The work for this project was undertaken within this ambit, and no identified individual case information was utilized. Therefore formal ethical approval was not required to undertake this work.
Results
Test frequency distributions
In the 2-year period (or longer) following high grade dysplasia, 53% of women had a single HPV follow-up test only, 33% had two HPV tests, and 14% had three or more HPV follow-up tests (Figure 1, top). For HPV tests occurring 12 months or longer following histology, 23% of women had none, 48% had one only, and 29% had two or more HPV tests (Figure 1, bottom).
Distributions (%) of numbers of HPV test episodes per patient, ≥ 12 months and any time following high grade (CIN2+) histology occurring during July 2006–December 2010, NSW women.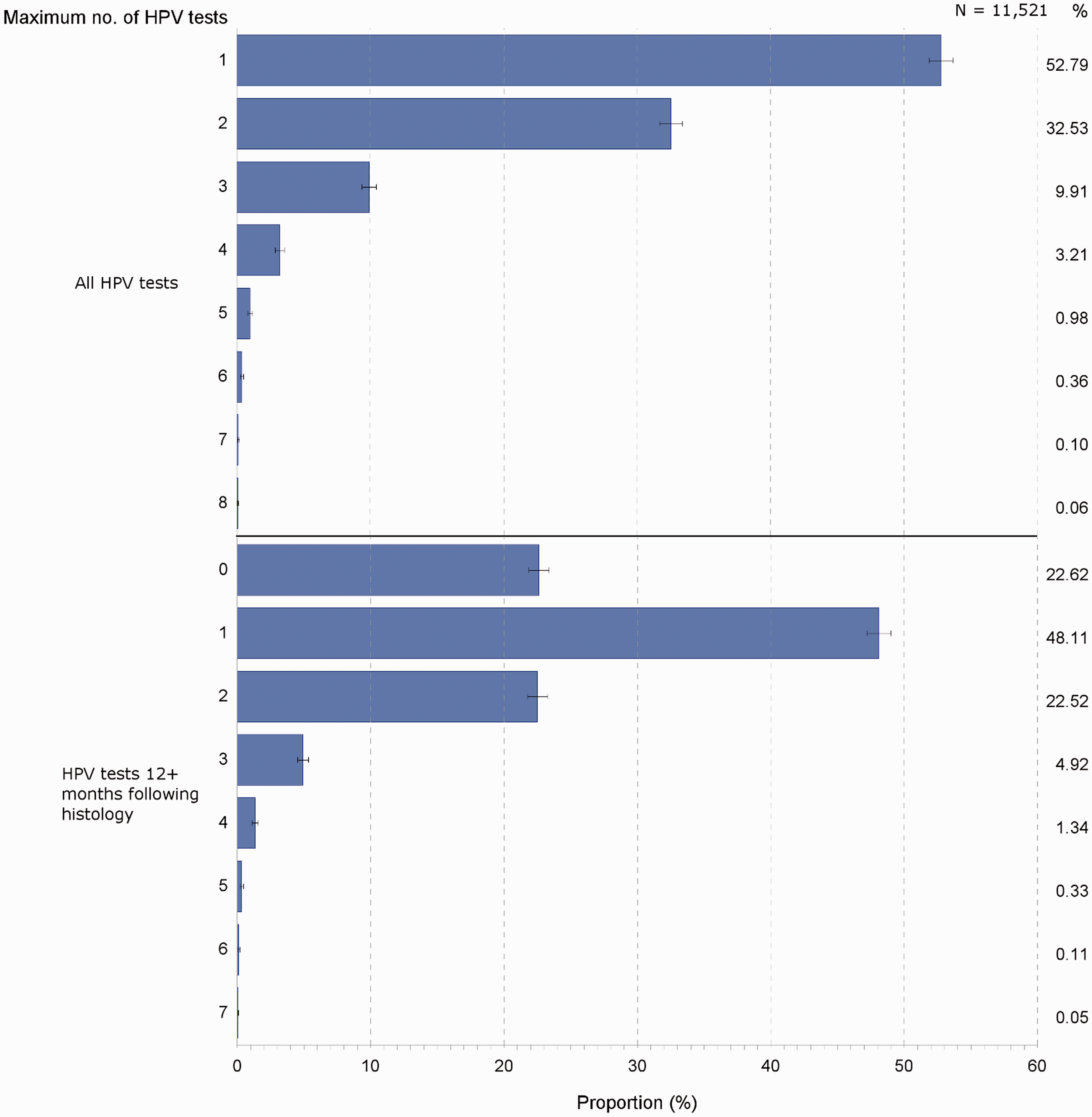
Test result distributions
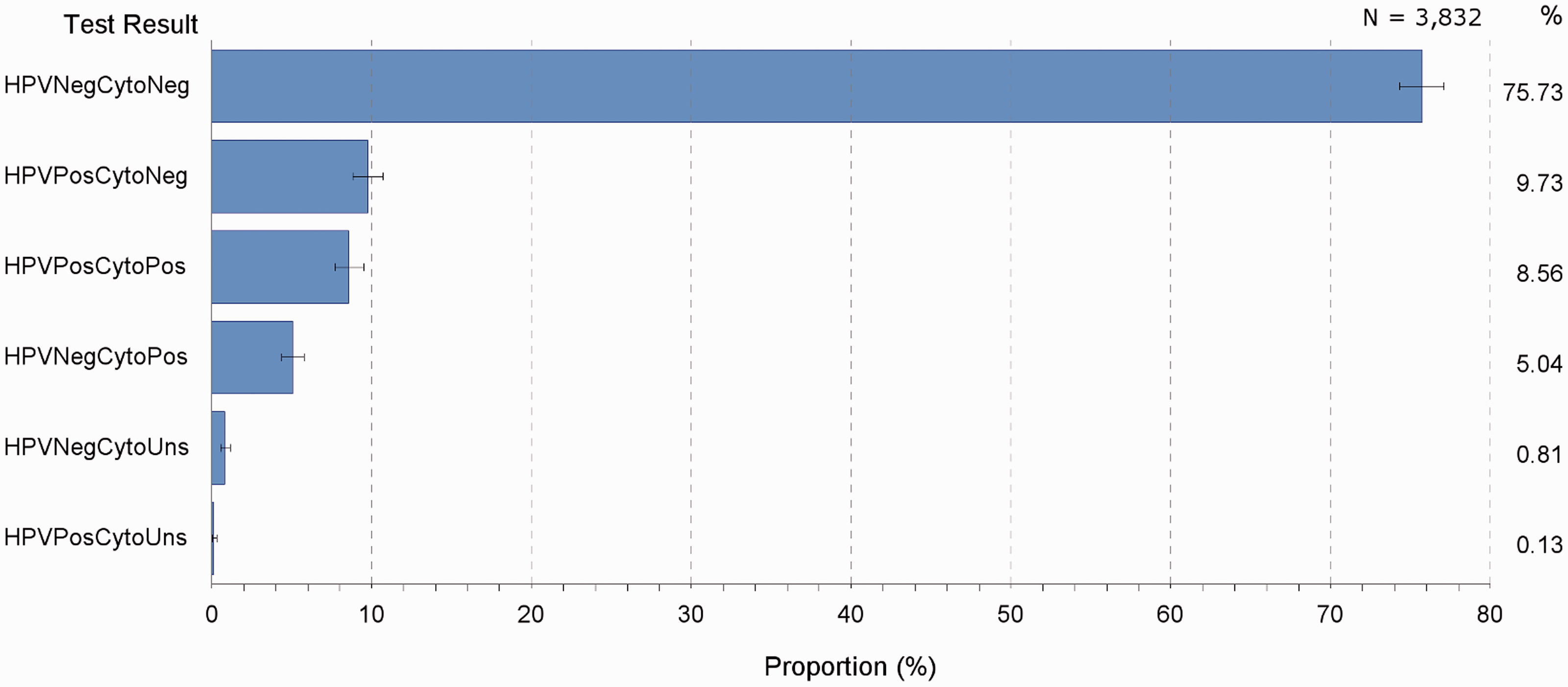
The distribution of HPV and cytology test pair results (HPV and cytology tests conducted within one month of one another) indicates that 73% were both HPV and cytology negative (from 16,860 test pairs); 76% were both negative in the test pairs occurring 12 months or later following histology; and 77% both negative in women who had their follow-up HPV tests exclusively 12 months or later following CIN2+ histology (Figure 2, top, middle, bottom, respectively). Of women who had one follow-up HPV test only, along with cytology occurring within one month of the HPV test, 76% had both negative HPV and cytology (Figure 3). Another 10% had positive HPV accompanying negative cytology, and 5% had negative HPV with positive cytology; 9% tested positive for both HPV and cytology.
Proportions (%) of HPV-cytology test pair results, by all test pairs, test pairs occurring 365 days or later following high grade histology, and test pairs in women whose tests all occurred 365 days or later following high grade histology, NSW, high grade histology diagnosed in 2006–2010. Proportions (%) of testpair results in women who had one follow-up HPV-cytology testpair only, NSW women with last high-grade histology diagnosed during 2006–2010.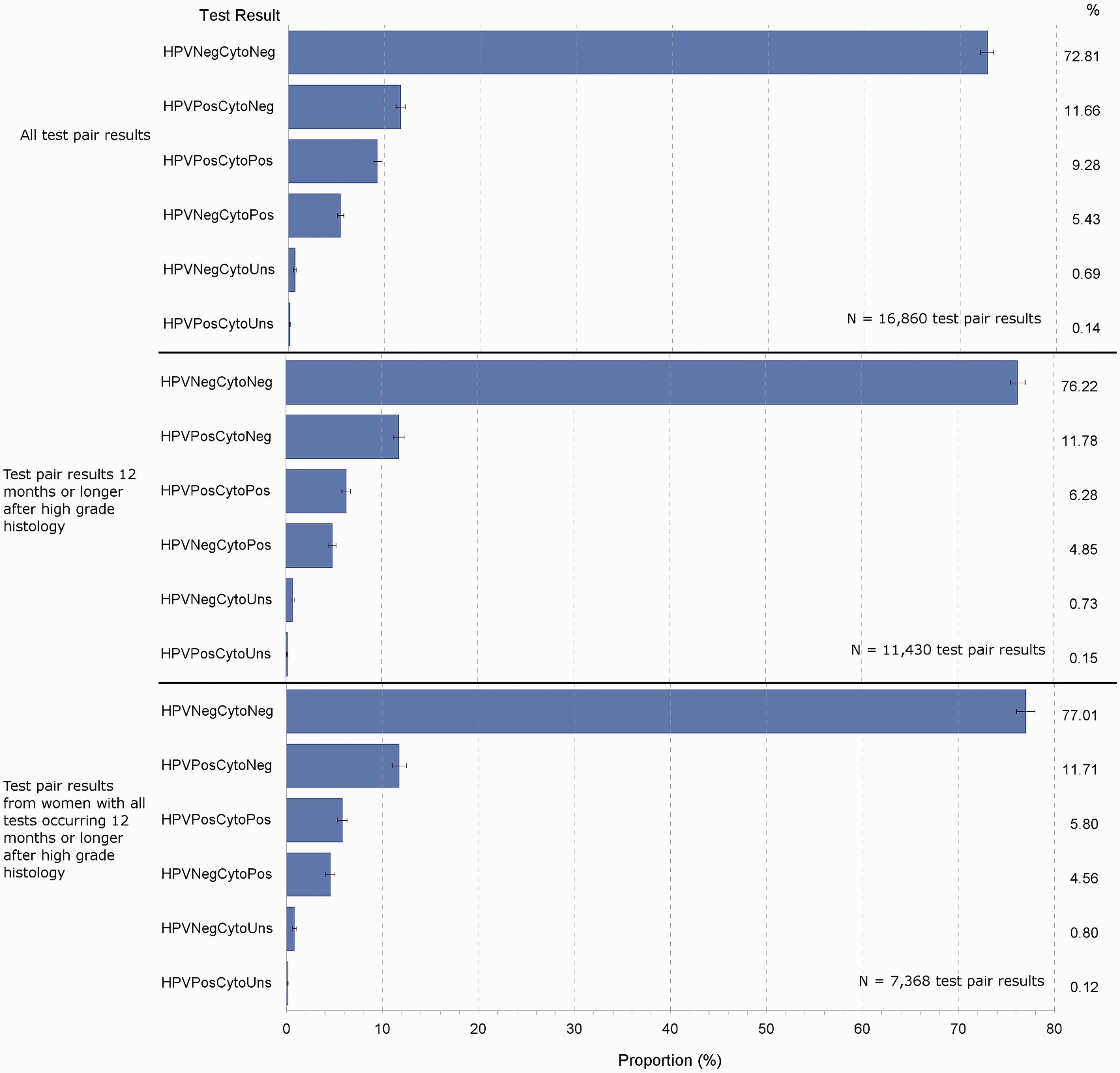
Test proportions and cure rates
Of the women in the cohort, 19% (n = 2,210) were cured according to the NHMRC guidelines (Figure 4), which represented 41% of women who had two or more HPV and cytology tests following high-grade histology (n = 5,371); 66% of women who had two or more HPV tests 12 months or longer after the high grade histology (n = 3,333); and 75% of women who had follow-up HPV and histology testing according to the recommended NHMRC test-of-cure intervals (n = 2,948). This latter proportion would represent an estimate of the rate of cure according to NHMRC guidelines from high-grade cervical dysplasia.
Proportions (%) of women with high-grade histology cured according to NHMRC follow-up guidelines (n = 2,210) within selected test sub-groups, NSW women with last high-grade histology diagnosed in 2006–2010.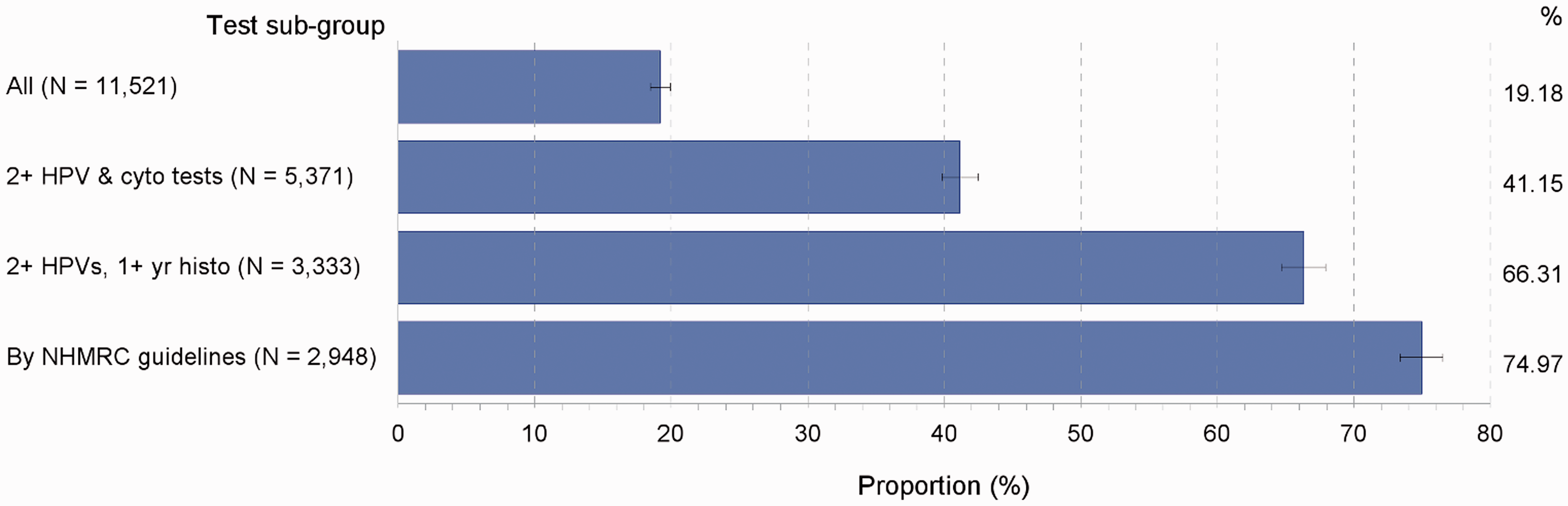
Of women with CIN2/3 histology only (n = 11,388), the above proportions were similar: 53% had one follow-up HPV test only, 33% had two, and 14% had three or more follow-up HPV tests. Overall, 18% were cured, according to NHMRC guidelines, and of women with CIN2/3 and who had follow-up testing according to NHMRC guidelines (n = 2,905), the cure rate was 75% (results not shown).
Number of tests and times to reach cure
Of those cured according to NHMRC guidelines, 68% were cured by the second HPV test, 21% by the third, and the remaining 11% at the fourth or later HPV test (up to seven HPV follow-up tests) (Figure 5, top). Corresponding proportions for cytology follow-up tests were more dispersed: 7% were cured by the second follow-up Pap test, 42% cured by the third, 34% by the fourth, 10% by the fifth, and the remaining 7% by the 6th up to the 19th follow-up Pap test (results not shown). The median time to cure was 1,097 days; the mode was 854 days and the mean 1,190 days; the minimum time to cure was 693 days and the maximum was 2,415 days (ie. >6 years) (Figure 5, bottom).
Distribution of numbers HPV tests (top) and times needed (days, bottom) to attain cure from high grade histology according to NHMRC follow-up criteria, NSW women cured of high grade cervical dysplasia diagnosed in 2006–2010.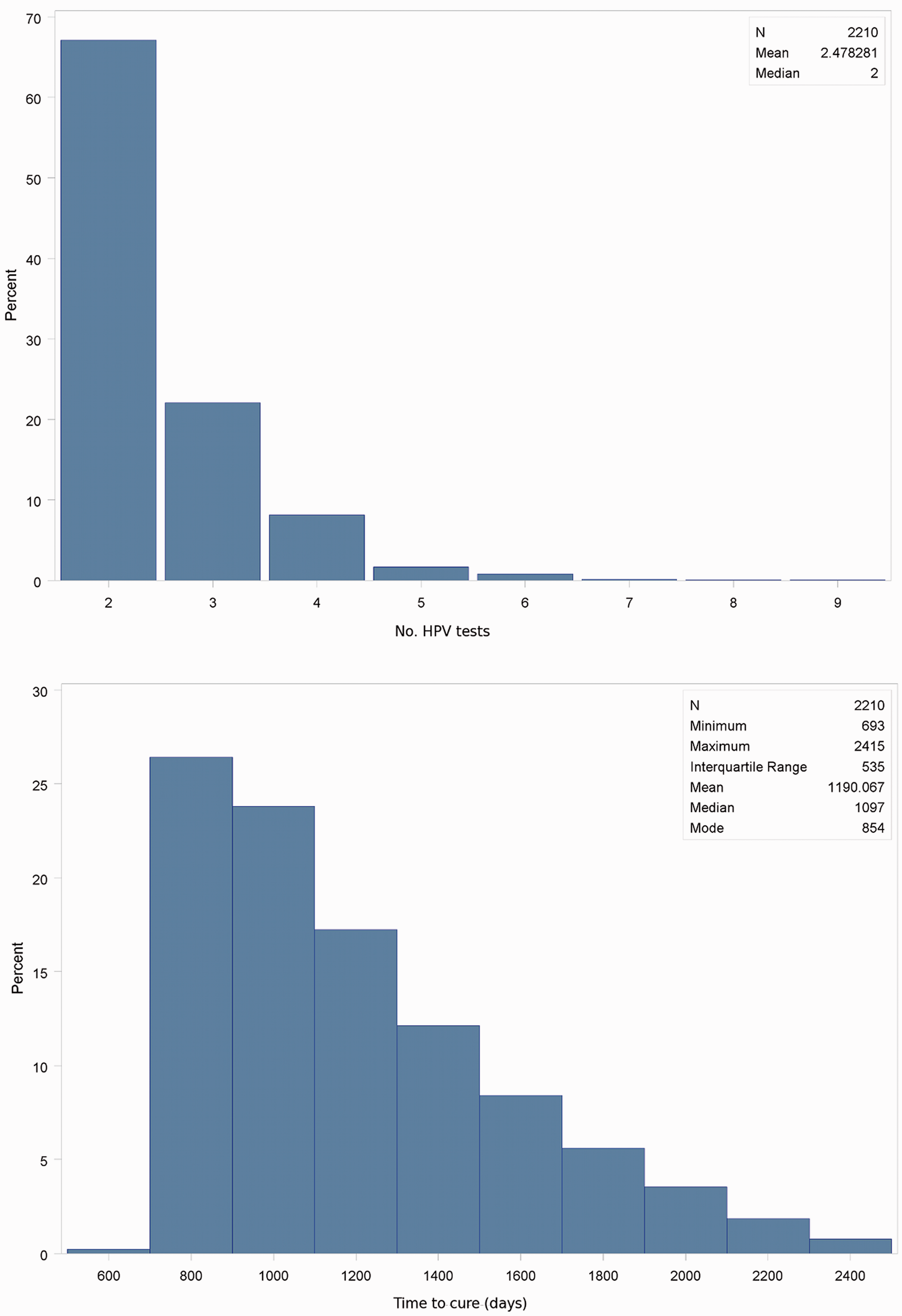
Cure rates by age
In women tested according to NHMRC follow-up guidelines, cure rates were lowest in those aged under 30 (71%), significantly lower than in women aged 30–49 (80%), and non-significantly lower than in those aged 50–69 (73%) (Figure 6).
Proportions (%) of women cured of high-grade cervical dysplasia, NSW women last diagnosed with high grade cervical dysplasia in 2006–2010 and followed up according ot NHMRC follow-up guidelines.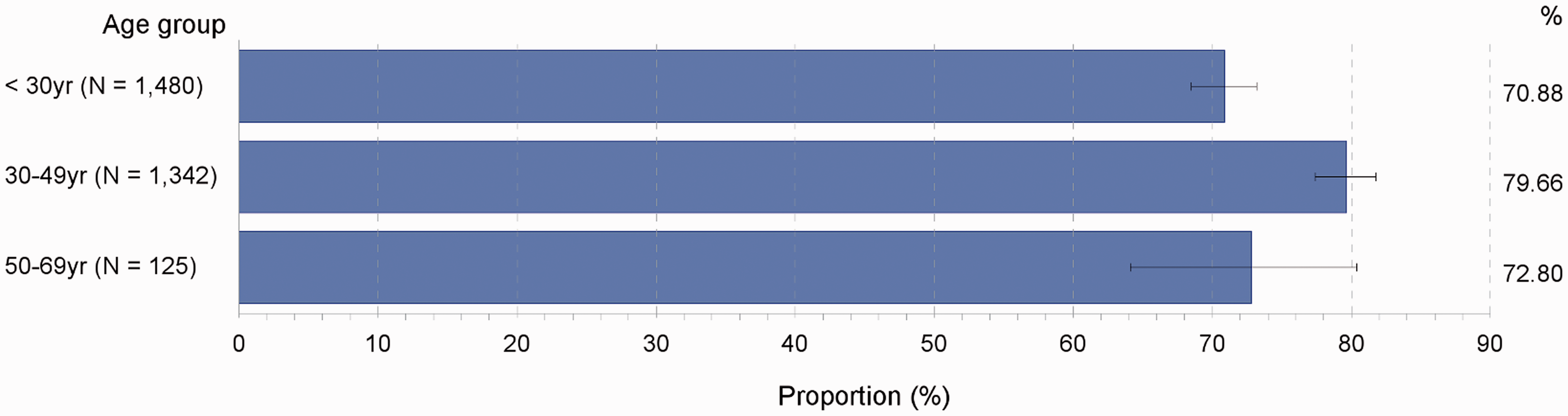
NHMRC-compliant test result profiles
When HPV and cytology tests occurring within one month of each other are regarded as test pairs, a profile of consecutive HPV-cytology test pair results can indicate the efficacy of combined HPV and cytology as a test-of-cure. For clarity, only test pair couplets occurring according to the NHMRC follow-up guidelines are analyzed, and only the most frequent result pair combinations are shown in the figures.
Of all consecutive HPV/cytology test results conducted according to NHMRC follow-up guidelines (n = 3,494 test pair couplets), 65% were all negative; 8% were HPV positive with negative cytology followed by negative HPV and cytology; 6% were both negative followed by positive HPV with negative cytology; and 4% were positive HPV with negative cytology on both occasions (Figure 7). In all, 9.7% of test pair results were both negative at 12 months follow-up but positive on the next occasion, most of which comprised positive HPV in the second round of test follow-up (75%); 1.4% of test pair results were all negative initially and all positive at follow-up.
Proportions (%) of consecutive HPV-cytology test-pair results, NSW women with last high-grade histology diagnosed in 2006–2010 tested according to NHMRC follow-up guidelines.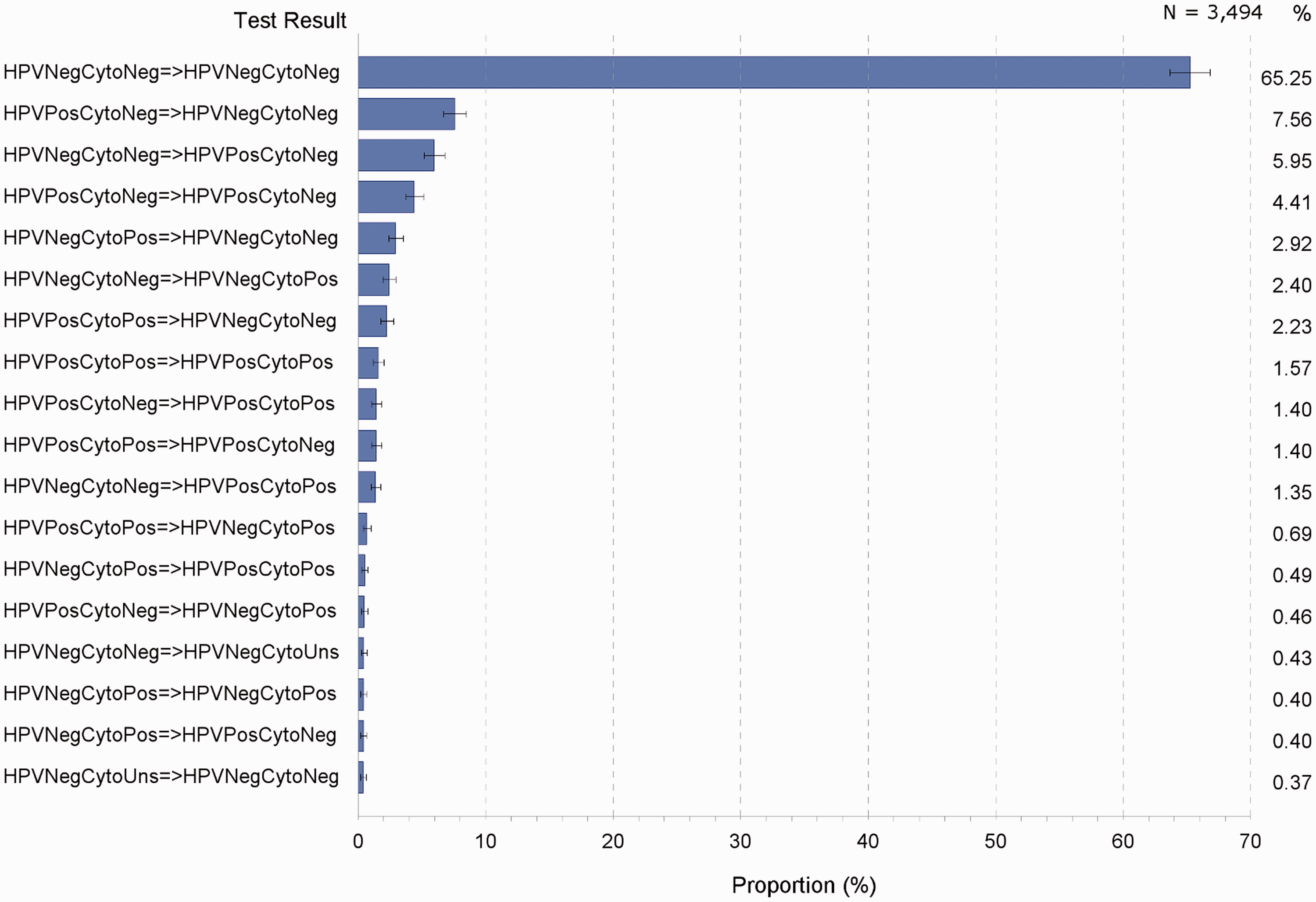
Test result profiles of women classed as cured and not cured
Of consecutive test result pairs in women eventually cured, occurring up to and including the time of cure (2,573 test pair couplets), 89% were all negative on both testing occasions; 2.9% were HPV positive with negative cytology followed by both negative; 2.7% were negative HPV with positive cytology followed by both negative; and 1.9% were both negative followed by negative HPV with positive cytology (Figure 8, top).
Proportions (%) of consecutive HPV-cytology test pair results, women cured (top) and not cured (bottom) of high-grade cervical dysplasia according to NHMRC follow-up guidelines, NSW women with last high grade histology diagnosed in 2006–2010.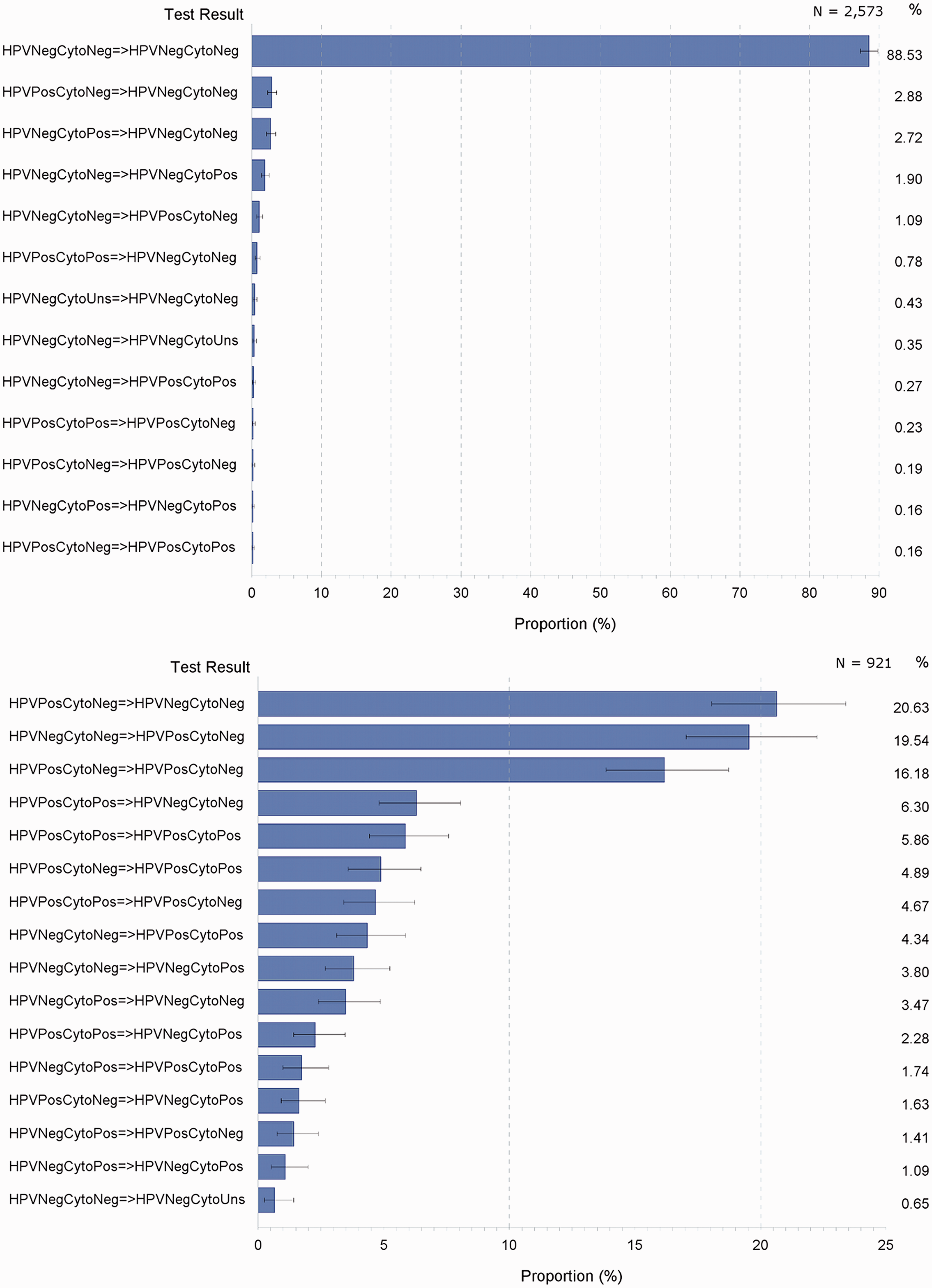
In women not cured, but tested according to NHMRC follow-up guidelines, the most common test result (from 921 test pair couplets) was HPV positive with negative cytology, followed by both negative (21%); 20% had both HPV and cytology negative initially, followed by positive HPV with negative cytology; 16% had positive HPV with negative cytology on both occasions (Figure 8, bottom).
Altogether, 56% of test results in these uncured women originated from positive HPV with all-negative cytology. In contrast, 9% of positive test results emanated from all-negative HPV results in each test pair couplet.
Short follow-up interval test results
The proportion of women testing all negative 3–7 months after treatment (n = 794) who were all negative at the next follow-up was 86% (Figure 9); 8% were HPV positive with negative cytology; 4% were HPV negative with positive cytology; and 2% were all positive on the second round of testing. Some of these results would reflect recurrence/reinfection rather than treatment failure, especially in younger women.
Second follow-up HPV-cytology test pair results in women initially all negative at 3–7 months following treatment for high grade cervical dysplasia, NSW women with last high grade histology diagnosed in 2006–2010.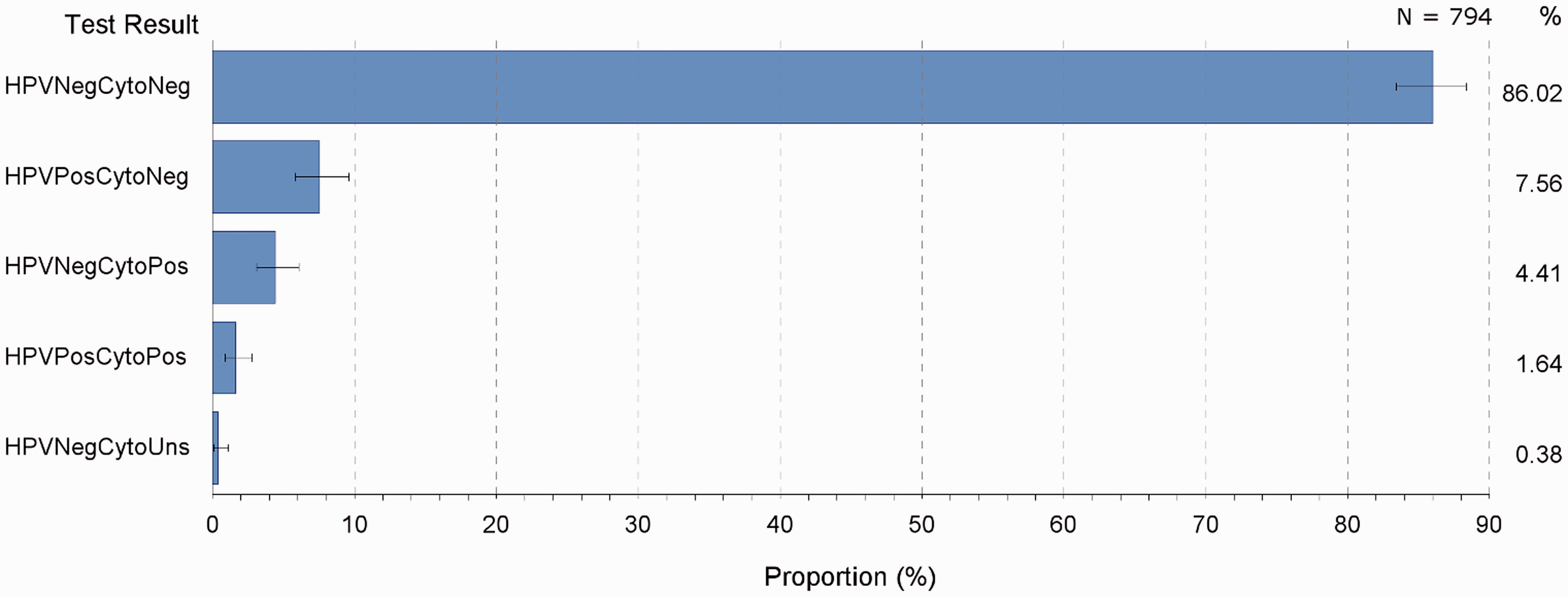
Discussion
HPV testing accompanied by cytology for follow-up of high grade cervical dysplasia has become an established test of the effectiveness of treatment, but until now detailed analyses of test-of-cure results in whole populations have not been conducted. The high sensitivity and consequent high negative predictive value of an HPV test, in conjunction with cytology, would give strong reassurance to those women with negative results that the original lesion has been effectively treated. However, the results shown here also indicate that the majority of NSW women with high grade dysplasia have had only a single follow-up HPV test. It is possible that their general practitioners (GPs) and other treating clinicians are satisfied that a single HPV negative test result, along with successive negative cytology, is sufficient to indicate cure, given the high negative predictive value of the HPV test. Certainly, HPV infection in the presence of negative cytology could validly be interpreted as a risk factor for subsequent cervical dysplasia, rather than a ‘lack of cure’ of prior cervical dysplasia. It may also be possible that GPs are not aware of HPV testing for treatment efficacy of high grade CIN.
A number of studies conducted on recurrence of CIN and HPV reinfection, using HPV testing and cytology as follow-up, have shown a high correlation between outcomes and early HPV/cytology results. A Belgian study of 72 women followed up at six-monthly intervals to 24 months after treatment for CIN2+ found older age to be strongly associated with treatment failure, as determined from colposcopy;3 that all women who had a recurrence (6/72) were HPV positive at the first follow-up occasion 3–6 months after treatment; and no instances of negative HPV at 3–6 months were found with a recurrence. Moreover, 15% of women with positive HPV but negative cytology at 3–6 months had recurrent/residual CIN, compared with 50% of women with abnormal cytology at 3–6 months follow-up. A larger UK study (n = 917), by Kitchener et al. 17 , reported similar results: in initially all-negative women at six months follow-up after treatment (n = 744), 8 women were found to have histological CIN1 and one woman had CIN2. At 12 months, 11 women had histological CIN1 and 2 had CIN2. At 24 months one woman had CIN3 and one woman had cancer. At two years two women altogether (0.25%) had CIN2+. The authors concluded ‘that the cumulative risk of residual disease is so low after 24 months of follow up that a woman who is cytology negative/HPV negative at 6 months can safely be recalled at 3 years…’. From a similar number of women (794), the present study found 108 women (14%) to have a positive test result at the next follow-up, 3–7 months after treatment. While these figures are not based on confirmed histology, and do not necessarily indicate subsequent high grade dysplasia, they represent a relatively high proportion; 14% of these women who had positive cytology at the second follow-up test had cytology suggesting high grade dysplasia. Given these results, the above conclusion from the UK study is difficult to endorse in the NSW context without further investigation.
A positive HPV test following both negative HPV and cytology would more likely indicate re-infection than recurrence or persistence of the original infection, especially in younger women, and would be very unlikely to be a false negative result. This may be a consideration by GPs or treating clinicians, especially with younger patients. However, a relatively high proportion of these women (9%, see Figure 3) had both HPV and cytology positive results without subsequent HPV testing. This is not an artefact of inadequate follow-up time, as the analyses have been confined to high-grade histology up to the end of 2010 to leave sufficient follow-up time to March 2013 (27 months minimum).
In some women eventually cured of the high-grade lesion according to NHMRC guidelines, the time to reach the cure extended to six years, and in approximately 40% of cured women it took around four years or longer to attain a cure. It is questionable whether some, or most of these women, were not effectively cured before this time, and that the test-of-cure guidelines are failing to take into account that some women may have ongoing HPV infection, or are reinfected without cytologic/neoplastic consequences, who nonetheless are at higher risk of cervical cancer. This is the converse of the high proportion of women with a single HPV follow-up test only, in that many GPs and other treating clinicians appear to be adhering to the NHMRC guidelines until a ‘cure’ is attained, regardless of whether the woman is effectively cured. In other words, as is the case with influenza, an HPV infection can clear, with reinfection occurring subsequently. From the timing of follow-up testing, an individual could be classified as ‘not cured’ when, in fact, she was actually clear of HPV for some period of time before being reinfected, and the particular testing intervals failed to capture her short-lived ‘cured’ status. While this scenario may occur in individuals, it is difficult to envisage any population-based testing regime that could cater adequately to each individually varying scenario.
The high proportion of women with a single HPV test only is not a result of hysterectomy subsequent to high-grade dysplasia. Although hysterectomy status is recorded only partially on the NSW PTR, this study is a follow-up of women in relation to their most recent high-grade histology only, and women having a hysterectomy following high-grade dysplasia would be excluded, as they would not have had further cervical cytology. Vault smears are not recorded on the NSW PTR and such women, even with HPV testing, were excluded from the analysis.
It is unclear to what extent negative cytology accompanying a positive HPV test implies the latter to be a false positive result, or whether the positive HPV result is capturing pre-dysplasia infection not detected cytologically, reflecting early stages of re-infection (which in most cases would resolve). The somewhat lower specificity of HPV tests compared with Pap tests may contribute to this. Although the difference in specificity between an HPV and a Pap test is not large, it appears to be greater in younger women aged ≤ 35. 20
For example, let us assume that women whose first NHMRC-compliant test pair (ie. ≥12 months following treatment) was all-negative are nominally “cured”. We also assume that, from these women those with positive result 12 months subsequently, the second test result was not a false positive, as prima facie a false positive HPV result in such women appears less likely. It might then be reasonable to interpret the positive result proportions following all-negative HPV and cytology as upper limits of rates of reinfection and/or recurrence. That is, around 6% of all-negative test result pairs 12 months subsequent to treatment were HPV positive and cytologically negative at 22 months or later. This suggests that up to 6% of women who are negative at 12 months either have a recurrence or acquire a new HPV infection after 22 months (see Figure 8). A smaller proportion (1.4%) of women with test pairs have subsequent all positive HPV and cytology at 22 months or later, suggesting that up to 1.4% of these nominally “cured” women experience either recurrence or rapid conversion of HPV re-infection into a cervical lesion. In other words, even in women testing all negative at 12 months follow-up, rather than at 3–7 months, around 7% are test positive 22 or more months after their CIN2+ histology.
A weakness of the present study is that it does not track the extent of recurrence of high grade dysplasia in women with initial CIN2+ histology. We examined a cohort of women whose high grade histology was their most recent. Thus positive test results subsequent to the CIN2+ histology did not include any histologically confirmed high grade dysplasia, although subsequent positive HPV tests were positive to oncogenic HPV varieties only.
Despite the study population being that of all women in NSW who had HPV and cytological follow-up from high-grade dysplasia, it is also not clear how representative these women are of all NSW women diagnosed with high grade cervical dysplasia in the same period. It is probable that the study sample comprises women who were recommended to have HPV follow-up testing because their treating clinicians thought that they might be at higher risk of treatment failure. Unlike a number of similar studies, this study had no access to data on types of procedures used, treatment margins, or other prognostic indicators to determine how representative the study population was of all women who had high-grade dysplasia.
A strength of the study is that it comprised all women in NSW who had HPV and cytology testing following high grade dysplasia. This produced sufficient statistical power to describe, in some detail, the distributions of test results under different conditions of follow-up, along with times to cure, and number of follow-up testing rounds needed to attain a cure. To our knowledge this information is not available at a whole-population level.
Finally, at least one systematic review has questioned the efficacy of dual HPV and cytology testing following treatment of cervical dysplasia. 21 The review showed HPV testing in a therapeutic setting to have greater sensitivity but similar specificity in detecting recurrent/residual high grade dysplasia compared with repeat cytology. This suggests that negative HPV test results need not be accompanied by cytology, at least for the second consecutive negative HPV test result in NHMRC-compliant follow-up tests, and that cytology might be conducted as a reflex test only when the second HPV follow-up test is positive.
Further avenues of research, some stemming from results reported here, include:
How much does the likelihood of subsequent high grade dysplasia differ in those cured according to NHMRC criteria from those with consecutive negative cytology but without consecutive negative HPV? In particular, outcomes in women with positive HPV and negative cytology could be compared with those in women with negative HPV and negative cytology, to ascertain the extent to which HPV testing is misclassifying women who are cured of high grade cervical dysplasia as not being cured. Is there scope for lowering the threshold for cure in follow-up testing regimes for some women, for example younger women aged <30, given the apparent lower specificity of the HPV test in these women? To what extent would subsequent high grade lesion rates change if negative follow-up HPV test results were not accompanied by confirmatory cytology? What factors predict the relatively high proportion of women who have only one HPV test following high grade histology? In particular, to what extent does this high proportion reflect the apparent prognostic uncertainty GPs may feel, stemming from repeated HPV testing increasing the likelihood of producing HPV positivity that is clinically meaningless or unrelated to the original high grade dysplasia? To what extent do HPV test results vary by the type of HPV test employed? To what extent might a follow-up reminder system reduce the proportion of women with only one follow-up HPV test? Is it necessary? Should it be limited to women with an initially positive follow-up result? If implemented, would such reminders be better directed toward treating clinicians? To what extent would education of GPs and other treating clinicians reduce the preponderance of single HPV tests following high grade cervical dysplasia?
Footnotes
Acknowledgements
The authors acknowledge the assistance of:
Annabelle Farnsworth, of Douglass Hanly Moir Pathology and cytopathology adviser to the NSW Pap Test Advisory Committee, who prompted SM to conduct this study and provided insight into the nature of persistent HPV infection.
Deborah Bateson, of Family Planning Health, NSW who provided input into how service providers might use HPV testing regarding high grade cervical dysplasia.
Neville Hacker, gynaecological oncology adviser to the NSW Pap Test Advisory Committee, who provided insights into service provider considerations of the prognostic role of the HPV test as a test of cure.
