Abstract
Background
Faecal occult blood tests are often the initial test in population-based screening. We aimed to: 1) compare the results of single sample faecal immunochemical tests (FITs) with colonoscopy, and 2) calculate the sensitivity for proximal vs. distal adenomatous polyps or cancer.
Methods
Individuals scheduled for a colonoscopy were invited to complete a FIT prior to their colonoscopy preparation. FIT results were classified as positive, negative, or invalid. Colonoscopy reports were reviewed and abstracted. Because of product issues, four different FIT manufacturers were used. The test characteristics for each FIT manufacturer were calculated for advanced adenomatous polyps or cancer according to broad reason for colonoscopy (screening or surveillance/diagnostic).
Results
Of those invited, 1,026 individuals (43.9%) completed their colonoscopy and had a valid FIT result. The overall sensitivity of the FITs (95% confidence intervals) was 0.18 (0.10 to 0.28) and specificity was 0.90 (0.87 to 0.91) for advanced adenomas or cancer. The sensitivity for distal lesions was 0.23 (0.11 to 0.38) and for proximal lesions was 0.09 (0.02 to 0.25). The odds ratio of an individual with a distal advanced adenoma or cancer testing positive was 2.68 (1.20 to 5.99). The two individuals with colorectal cancer tested negative, as did one individual with high-grade dysplasia.
Conclusions
The sensitivity of a single-sample FIT for advanced adenomas or cancer was low. Individuals with distal adenomas had a higher odds of testing positive than those with proximal lesions or no lesions.
Keywords
Introduction
Colorectal cancer (CRC), the second leading cause of cancer death 1 , is largely preventable, or very curable if detected early. However, approximately 60% of cases are diagnosed at a late stage when 5-year survival rates are very low. 2
The United States national guidelines promote several tests for screening, including tests that detect occult blood and endoscopic tests that visualize the colon.3–5 Faecal occult blood tests (FOBTs) are recommended annually, while colonoscopy is recommended every 10 years, if no polyps are found. In the English population-based bowel screening programme, individuals aged 60 to 69 are mailed guaiac-based FOBTs every two years; this has recently been extended to age 74. 6 The current European guidelines state that the guaiac FOBT interval should not exceed two years, and the faecal immunochemical test (FIT) interval should not exceed three years. 7 These guidelines reported limited evidence for the efficacy of colonoscopy in reducing CRC incidence and mortality. 7 The newer FITs generally have better sensitivity and slightly lower specificity for CRC and advanced polyps, compared with guaiac tests.8–10
FOBTs are much less expensive compared with colonoscopy, and are often preferred by patients.11,12 In many low income settings, FOBTs are the initial option for patient screening, due to the prohibitive cost and limited availability of colonoscopy.13,14
A decision analysis performed for the United States Preventive Services Task Force found no difference in life-years gained by CRC screening using colonoscopy every 10 years vs. annual testing with a sensitive FOBT or a FIT in individuals aged 50 to 75. 15 Although studies using FIT followed by colonoscopy have been conducted in populations outside the United States16–20, we found none conducted in the United States directly comparing these methods. Quintero et al compared over 50,000 individuals randomized to one-time FIT or colonoscopy in a Spanish population. 21 CRC was found in 0.1% of both groups.
Some studies have reported that the sensitivity of FIT for distal adenomas is higher than for proximal adenomas.16,18,22,23
The purposes of this study were to: 1) compare the results of single sample FITs with colonoscopy, and 2) calculate the sensitivity for FIT for proximal vs. distal adenomatous polyps or cancer.
Methods
The study and methods were approved by the University of Iowa Institutional Review Board.
Subject Recruitment
Individuals aged 40 to 75 who were scheduled for a screening, surveillance or diagnostic colonoscopy at University of Iowa Healthcare were mailed an invitation to participate in the study. Exclusion criteria included: having familial polyposis syndromes, ulcerative colitis, or Crohn’s disease, or active rectal bleeding. Those with no symptoms were included in the screening group. Those with previous polyps or colorectal cancer were included in the surveillance group. Individuals with a change in bowel habits, anaemia, positive FOBT or FIT, or appetite change, or abdominal pain were included in the diagnostic group. We did not collect information on the recency of the previous colonoscopy for those in the surveillance group.
Together with the invitation letter, potential subjects were mailed an informed consent document, a FIT with directions, a form asking for the date the stool sample was obtained, and a postage-paid biohazard mailer for return of the signed informed consent and the FIT sample. A toll-free telephone number was provided to enable potential subjects to speak with a research team member. Within 7 to 10 days, non-responders were telephoned, up to three times, to confirm that the invitation had been received, encourage participation, and allow the potential subject to ask questions. If a month had passed and there was no response from a potential subject, a second mailing was sent if there was sufficient time for receipt of a stool sample prior to the scheduled colonoscopy. If the colonoscopy was rescheduled, up to another three telephone calls were made. If the subject returned either the FIT or the informed consent, we telephoned them to request the missing item, and duplicate item was sent if needed.
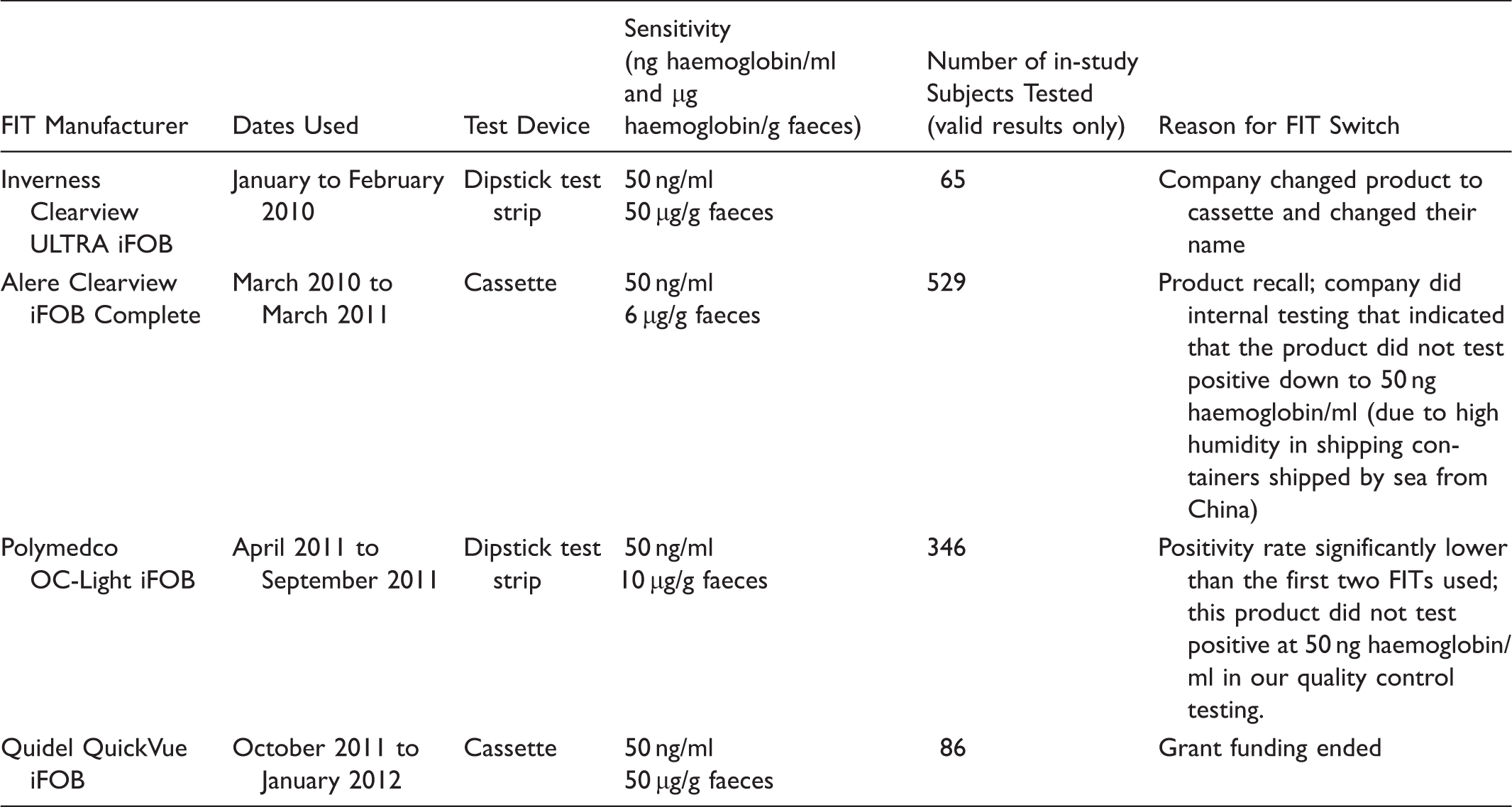
Faecal Immunochemical Tests (FITs) Used in the Study
Faecal immunochemical tests used in this study (n = 1,026).
Collection of Stool Samples and Development of FITs
Subjects were instructed to collect their stool sample prior to the start of their colonoscopy preparation. The sample was mailed back in a supplied pre-paid postage mailer, and developed according to the manufacturer’s directions by two study team members, who witnessed and concurred on the final result. Samples were considered invalid if the liquid had escaped from the vial during transit or if the subject collected too much stool for the buffer to ascend the chromatography paper.
Medical Record Review Form
A detailed analysis of the colonoscopy report was conducted. Portions of a colonoscopy review form (available on request) created for previous CRC studies were used for this study.29–32 Every biopsy sample was classified by size, histology, and location in the colon using the World Health Organization classification system 33 , and those with any villous histology, serrated adenomas, high-grade dysplasia, or any adenomatous polyp 10 mm or greater in diameter were classified as advanced adenomatous polyps.4,34,35 Our size and histology classifications very closely reflect those used by Imperiale et al 36 and Kim et al. 37 Adenomas were classified as “proximal” if in the cecum, ascending colon, hepatic flexure, or transverse colon; they were classified as “distal” if in the splenic flexure or below. There were two individuals with both a proximal and distal adenoma, and they were classified according to the location of the largest adenoma, which was distal in both cases.
Data Analysis
Data were stored in Microsoft Access and imported into SAS 9.2 for Windows (SAS 9.2, SAS Institute, Inc., Cary, NC) for analysis. The data were analyzed by manufacturer using three groups: screening, surveillance or diagnostic, and all combined. Diagnostic and surveillance colonoscopies were combined because these constitute a higher risk group. For each group, contingency tables were constructed, and sensitivity, specificity, positive predictive value, negative predictive value, likelihood ratio positive, and likelihood ratio negative were calculated for each of the four FIT manufacturers. Colonoscopy results were used as the gold standard and were considered positive if an adenocarcinoma or advanced adenoma was found. Invalid FIT results were excluded from these test characteristic calculations, so a FIT could only be positive or negative. To calculate confidence intervals (CIs) for the test characteristics that were proportions (sensitivity, specificity, positive predictive value, and negative predictive value), 95% exact binomial CIs were used. The 95% CIs for the diagnostic likelihood ratios were calculated using the method described by Simel et al. 38 In the surveillance and diagnostic colonoscopy group, Polymedco OC-Light and Quidel QuickVue had 0.5 added to all of the cell counts of their contingency tables before calculation of test characteristics, as described by Altman et al. 39 This was done as some cells had zero counts that prevented the calculation of finite likelihood ratio CIs.
A binary logistic regression model was constructed with FIT result (positive/negative) as the outcome and a predictor defining the location of advanced adenoma, while controlling for FIT manufacturer, age (continuous), and gender if necessary (adding to the model one at a time and determining if they altered the main effect of interest by at least 5%). For this analysis, subjects with both a proximal and distal adenoma were classified according to location of the largest polyp, because this would be the one likely to bleed (n = 2 subjects).
A second binary logistic regression model assessed the relationship between most concerning polyp type (advanced adenoma or adenocarcinoma, other adenoma, or nothing concerning on biopsy/normal colonoscopy) and FIT results. This model was assessed for confounding with the same set of potential confounders and in the same manner as described above.
All models were assessed for multicollinearity, a sufficient number of events/non-events in the response, and the impact of influential observations (standardized Pearson residuals and DFBetas). Age, the only continuous potential confounder, was examined with polynomial terms to check for non-linear relationships with the log odds. Hypothesis testing was performed at a significance level of 0.05 and two-tailed.
Results
Between 22 January 2010 and 22 November 2011, invitations were mailed to 2,336 potential subjects, of whom 1,040 (44.5%) completed a FIT and a colonoscopy. Fourteen individuals had an invalid FIT result (1.4%), leaving 1,026 (43.9%) for analysis of test characteristics. A flow chart depicting how the final sample population was derived is shown in Figure 1.
Flow Diagram of Study Subjects.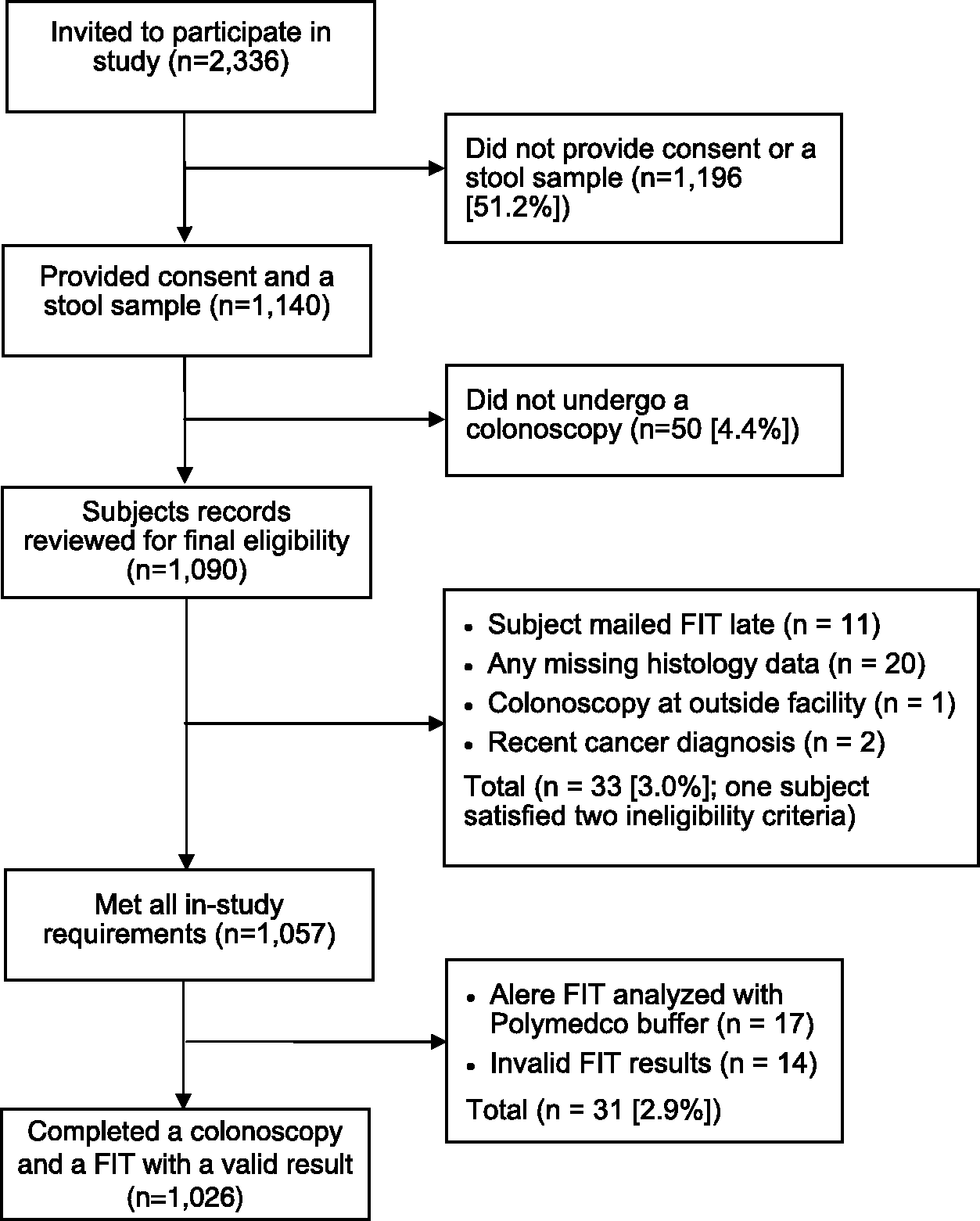
The mean time from collection to testing of the FIT sample for the 606 subjects for whom data is available was 4.9 days (median 5.0 days; standard deviation 2.9 days) and right skewed with a range from 1 to 36 (90th percentile 8.0 days), suggesting that 90% of individuals returned their FIT within 8 days of sample collection.
Demographics (n = 1,040)*.

Includes those who enrolled who had invalid FIT results
63 missing responses; all other demographic variables listed above had less than 26 missing responses
Adenoma Characteristics (n = 1,040 subjects).
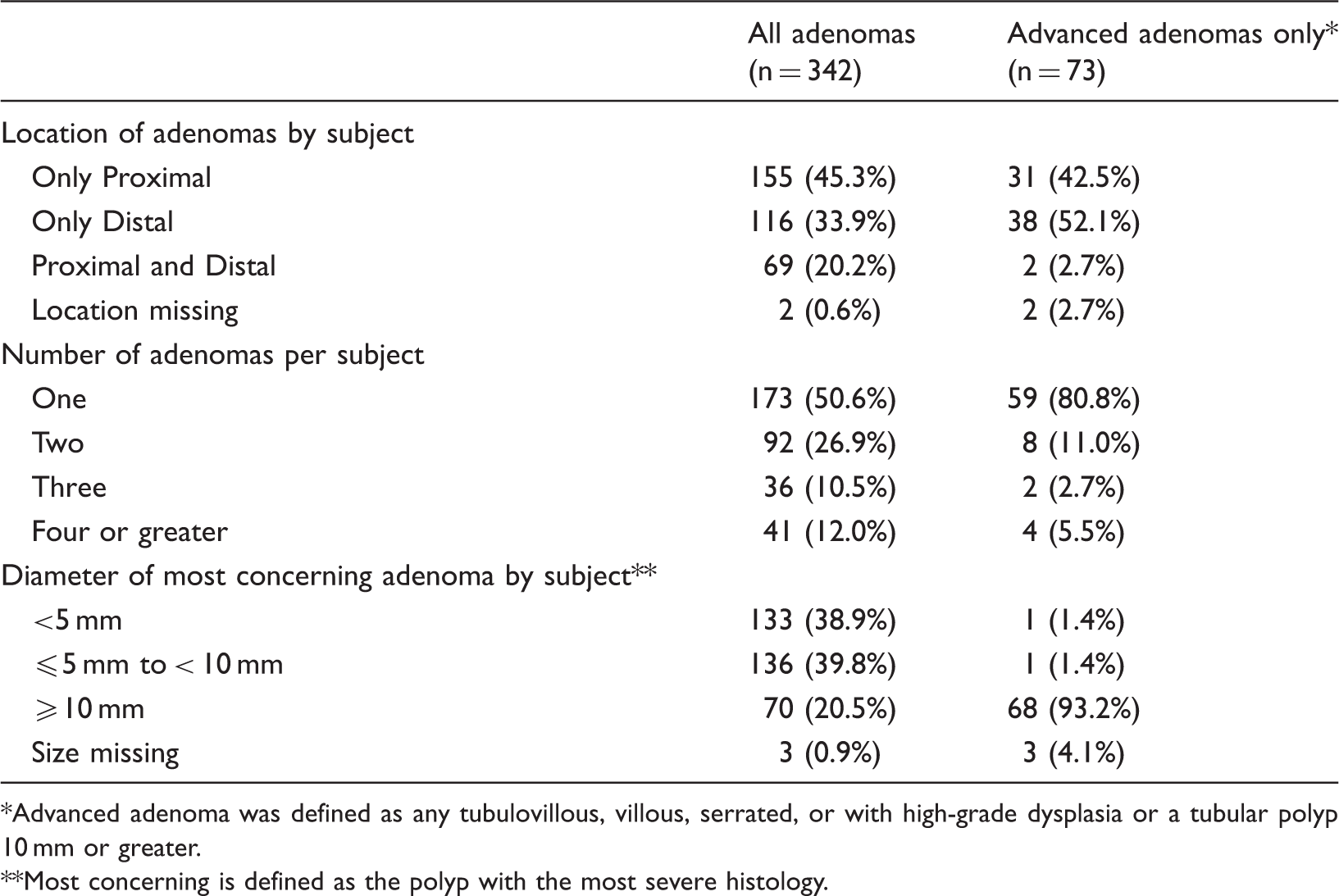
Advanced adenoma was defined as any tubulovillous, villous, serrated, or with high-grade dysplasia or a tubular polyp 10 mm or greater.
Most concerning is defined as the polyp with the most severe histology.
Colonoscopy and FIT Results.
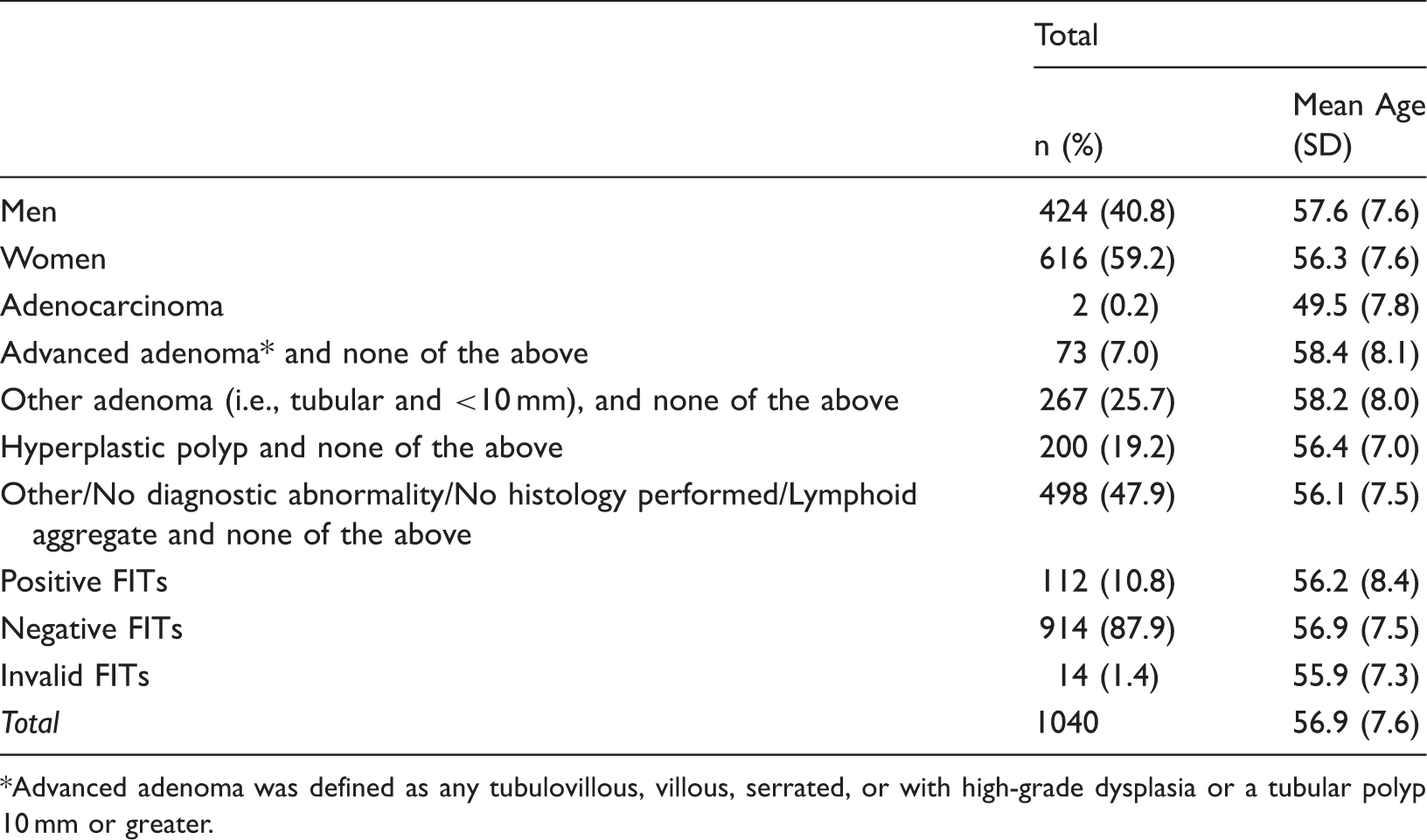
Advanced adenoma was defined as any tubulovillous, villous, serrated, or with high-grade dysplasia or a tubular polyp 10 mm or greater.
Test Characteristics for Advanced Adenomas or Cancer for Four FIT Manufacturers*.
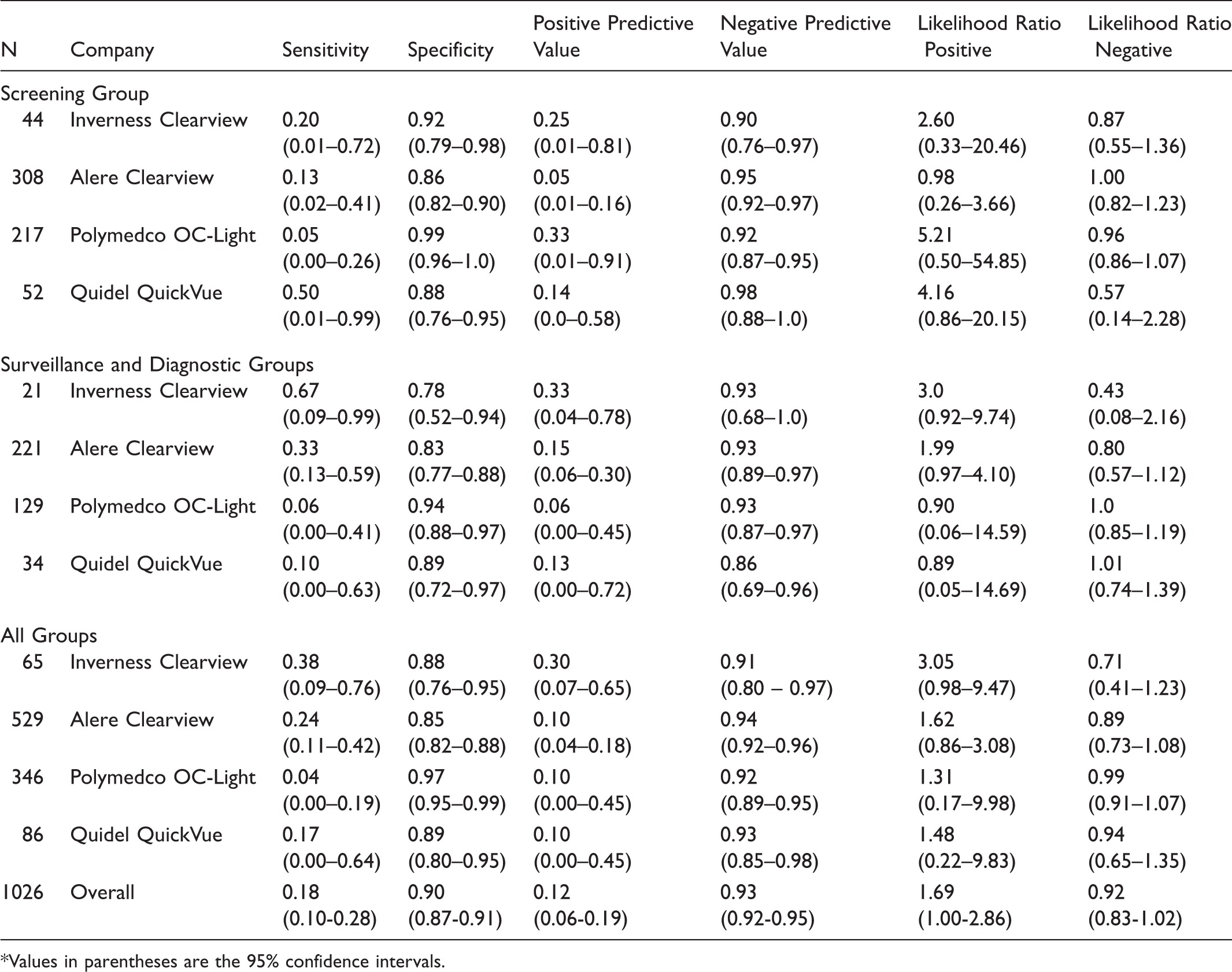
Values in parentheses are the 95% confidence intervals.
Table 6 shows a logistic regression model assessing variables which predicted positive FIT results. Model 1 shows that the presence of distal advanced adenomas or adenocarcinomas led to significantly greater odds (Odds Ratio [OR] = 2.68; 95% CI 1.20 to 5.99) for having a positive FIT result relative to no advanced adenoma or adenocarcinoma. Model 2 shows a nearly statistically significant result for individuals with advanced adenomas having increased odds of having a positive FIT relative to having no concerning polyps (OR = 1.96; 95% CI 1.00 to 3.84).
The Polymedco product was not testing positively as often as the other FIT manufacturers, and we determined that it was not testing at the lower limit of sensitivity stated. We examined the relationship of Polymedco vs. all other FIT manufacturers pooled together for testing positive. Pooling was justified as the positivity rates for each of the other three FIT manufacturers were similar. This produced an OR of 5.93 (95% CI, 3.06 to 11.51) for Polymedco testing negative compared with the other manufacturers. Confounding was explored as an explanation of this relationship as with the previous models, but no evidence of this was found.
Discussion
This study assessed the test characteristics of a single-testing round of liquid-based FITs with optical colonoscopy as the gold standard. Several findings are notable: 1) the overall sensitivity of single-sample FITs for advanced adenomas and adenocarcinoma was low (18%), with a specificity of 90%, 2) Polymedco OC-Light had a statistically significantly lower test positivity rate than all of the other manufacturers, and 3) the sensitivity of the FIT for distal advanced lesions was higher than for proximal advanced lesions; those with advanced distal adenomas had higher odds of testing positive. It is likely that many primary care physicians are not aware of the limitations of FITs. We chose CLIA-waived FITs because many resource-poor settings are not able to provide universal access to colonoscopy, due to lack of colonoscopy providers, prohibitive costs, or both.14,40,41 The FIT is, therefore, potentially a very useful test for resource-poor settings, and has also been recommended for other settings.40,42,43
During this two-year study, we had to use four different liquid-based FITs, when we originally intended to use a single manufacturer. One test was discontinued (Inverness Clearview ULTRA iFOB), one was withdrawn from the market (Alere Clearview ULTRA iFOB), but another with very low positivity compared with the other three manufacturers (Polymedco OC-Light) remains on the market. We discontinued use of Polymedco OC-Light, after we conducted more detailed quality control tests which indicated that it was not testing positive at the lower limit of haemoglobin concentration stated by the manufacturer.
When calculating test characteristics for specific groups and manufacturers, the subsets of data required were often small, resulting in limited precision, as seen through wide CIs. However, there is evidence that the specificity and negative predictive value were generally high across FIT products, and that the sensitivity and positive predictive value were generally low. It is important to note that this was only a single-sample testing round in a population that generally had access to CRC screening, and that when using FIT for screening, annual or biennial testing is recommended.4–7 The pooled estimates for FIT sensitivity for advanced adenomatous polyps or cancer was 0.18 (95% CI, 0.10 to 0.28) and specificity was 0.90 (0.87 to 0.91).
A Cochrane review has shown that FOBT, either annually or biennially, can reduce the risk of CRC mortality by about 15%. 44 Among those attending at least one round, there was a 25% relative risk reduction, with no difference in all-cause mortality. 44 The decision analysis by Zauber et al 15 showed no difference in life-years gained when a strategy of annual FOBT was compared with a colonoscopy every 10 years. Zauber et al conducted sensitivity analyses assuming differing levels of adherence, and for a given level of adherence, the life-years gained were very similar between the two methods. 15
Given the substantial numbers of false positive FITs that were not indicative of advanced adenomas, it would be useful to have a triage test for those with positive results. Despite research showing reasonable sensitivity and specificity for CT colonography in controlled settings37,45, CT colonography would probably not be a useful triage method in those with a positive FIT. Two studies have found relatively high rates of advanced adenoma or cancer (defined as a lesion 10 mm or greater in diameter or cancer) among those with a positive FIT.46,47 Regge et al reported a positive predictive value of 38.5% among FIT positive individuals and Liedenbaum reported a positive predictive value of 82% among those with positive FITs, using a definition of advanced lesion of adenomatous polyps 10 mm or larger or cancer.46,47 CT colonography therefore would not be a useful or cost-effective strategy, due to the fact that 40-80% of individuals undergoing CT colonography would need to go on to colonoscopy.
We found that the sensitivity of FIT for distal advanced lesions was higher than for proximal advanced lesions. This was confirmed by higher odds of patients with distal advanced adenomas testing positive in multivariable modelling. Others have also found higher sensitivity of the FIT for distal vs. proximal adenomas and adenomas greater than or equal to 10 mm in diameter, but did not report ORs for these.18,19,22
A good return rate of FITs was achieved for this study. Our original goal of obtaining 700 subjects was met. Numerous studies have shown that the FIT has higher sensitivity and is more acceptable to patients than the guaiac-based tests.10,48–50 Periodic FITs have been suggested as an alternative to the use of frequent surveillance colonoscopies. 20
Studies outside the United States have investigated the use of automated FITs.8,9,22,25,51 Automated tests can be set to a lower limit of sensitivity depending on resources available for colonoscopy. In a study conducted in the early 1990s with 27,860 subjects, using the automated FIT, OC-Hemodia by Eiken, the manufacturer’s suggested cutoff of 50 ng/ml gave a sensitivity of 86.5% and specificity of 94.9% for CRC. 52 In Taiwan, researchers used the same FIT and compared its findings with colonoscopic examination in 7,411 individuals. 53 The FIT detected 48% of advanced neoplasms and 88% of cancers. 53 Chiu et al 22 reported on 18,296 individuals and found a sensitivity for advanced adenoma of 28.5% for the FIT and 78.6% for CRC. 22 Our study results cannot be directly compared with these studies, because we used a non-automated CLIA-waived FIT. In addition, the populations tested in these studies were asymptomatic adults in an initial screening round, where it would be expected that higher sensitivities would result due to more individuals having a cancer or advanced lesion as yet undetected. 54
Performance characteristics vary widely across FIT manufacturers when analyzing the same stool specimen.18,55 Our study differed from these in that we used a single FIT manufacturer in a given person, but it also provides evidence for variable performance characteristics across FIT manufacturers.
To assess the usefulness of FIT in comparison with colonoscopy, it must be used annually over a span of ten years. Therefore, longitudinal studies are needed to compare strategies of annual FIT vs. colonoscopy every ten years. Such studies have been conducted in Italy, where they have adopted a biennial programme with the use of an automated FIT. Crotta et al 56 invited 2,959 individuals to participate and followed them for 8 years. They found that 60% of invited participants completed each round of FIT screening, but just under half (48%) attended all four rounds of screening. The rate of advanced adenoma detection was similar in each of the four rounds: 0.8 to 1.7%. 56
Strengths of our study were that all the FITs were liquid-based and that we had a high return rate of FITs. Limitations include quality issues with the FITs, companies discontinued making their product (Inverness Clearview), removed their product from the market (Alere), and had very low sensitivity (Polymedco). These issues with the FITs were totally unexpected and could not have been anticipated at the start of the study. By the time we got to our fourth kit (Quidel), funding for the study ran out. Another limitation is that although the FITs were to be collected before patients started their colonoscopy preparation, we did not clearly specify how soon the samples were to be mailed after FIT collection. Other work has shown that delayed mailing and warmer weather leads to breakdown in haemoglobin (thus potentially more false negative samples). 57 However, this was not a large factor in our study; the median time from collection to testing was 5.0 days and the 90th percentile was 8.0 days. Van Roon et al 58 showed stability of FIT for up to 10 days before development, using an automated FIT, but as we used non-automated tests, which may have differing stabilities, this finding may not apply to our situation.
The accuracy for detecting advanced adenomas and CRC in this single round of testing was low, although the overall sensitivity for advanced adenomas or cancer (18%) was very close to the sensitivity of 22% estimated by Zauber et al
15
for adenomas 10 mm or greater. FITs marketed in the United States are likely not to have undergone standard rigorous follow-up testing, because of the expense in having individuals undergo simultaneous testing with FIT and a colonoscopy, and in testing single samples across FIT brands. Although pathology laboratories participate in proficiency testing in order to remain accredited, they are probably not testing the FITs at the lower limit of positivity claimed in product inserts when using the manufacturer’s quality control solution. In addition, proficiency testing centres in the United States will not share how much haemoglobin is in their spiked samples
Given our experience, it is clear that at least one FIT manufacturer does not test at the lower limit of sensitivity claimed. There are 8 FDA-approved and widely used FITs on the market.59,60 Manufacturers of CLIA-waived FITs have a responsibility to ensure that their products perform reliably at the level of sensitivity claimed. It is not reasonable to expect individual health centres to verify that these tests give positive results at the lower limit claimed. Ninety-three percent of samples tested by proficiency centres over a two-year period were CLIA-waived tests, indicating that the overwhelming majority of testing in the United States is conducted with CLIA-waived FITs. 59
Predictors of Positive Faecal Immunochemical Test Results.
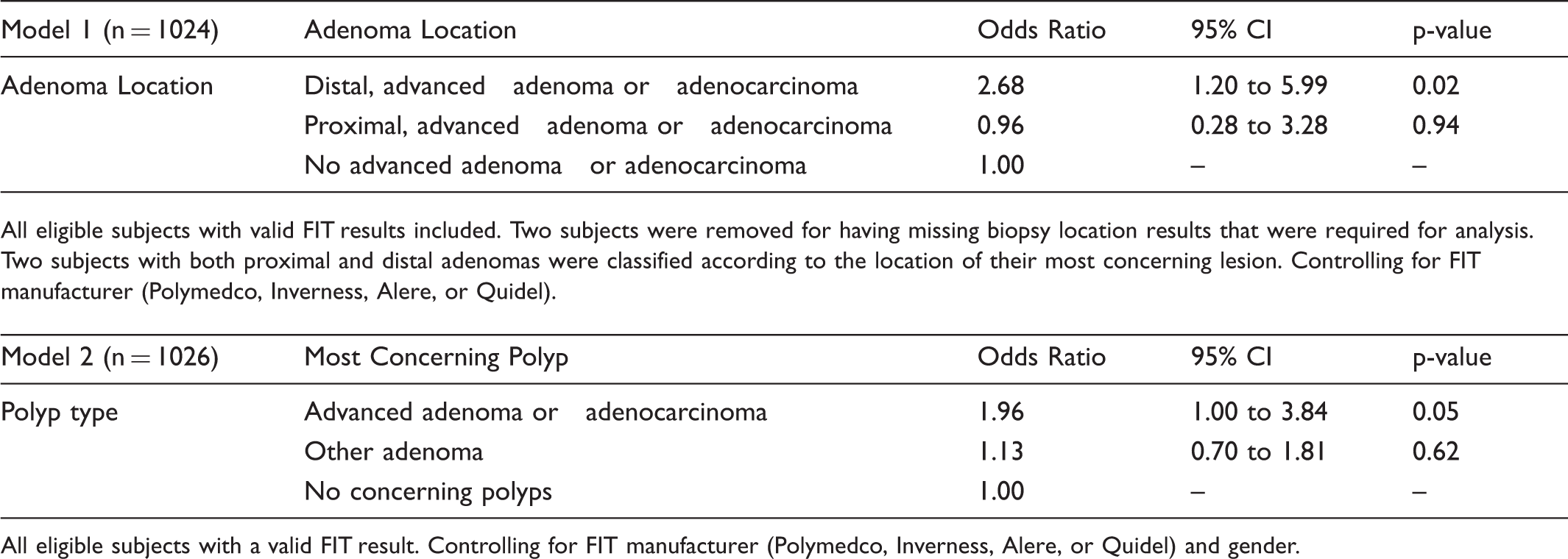
All eligible subjects with a valid FIT result. Controlling for FIT manufacturer (Polymedco, Inverness, Alere, or Quidel) and gender
Footnotes
Funding
This study was funded by NIH 1 RC1 CA144907-01 and the University of Iowa, Department of Family Medicine, Iowa City, Iowa.
All authors had full access to the data used. None of the authors has any disclosures.
The study sponsor, the National Cancer Institute, had no role in study design or in collection, analysis, or interpretation of the data.
