Abstract
Background
Polysomnography remains the diagnostic gold standard for obstructive sleep apnea syndrome (OSAS), but it is time consuming and requires dedicated personnel and setting. It may be more useful to plan a polysomnogram based on a preliminary screening.
Objective
To verify whether a questionnaire of general quality of sleep, the Pittsburgh Sleep Quality Index (PSQI), could outperform a dedicated questionnaire (Epworth Sleep Scale: ESS) in targeting OSAS patients in an at risk population.
Methods
254 consecutive subjects attending the outpatient clinic for respiratory diseases were clinically evaluated for sleep apnea and referred to a 12 channel night-time polysomnography. All patients were administered the ESS and the PSQI before the procedure. The correlation between the Apnoea/Hypopnoea Index (AHI) and the global score of the PSQI was calculated; Sensitivity, specificity, positive and negative predictive values (PPV and NPV, respectively), Diagnostic accuracy and the area under the receiver operating characteristic curve (AUC) were calculated. ESS performance was used as a control reference.
Results
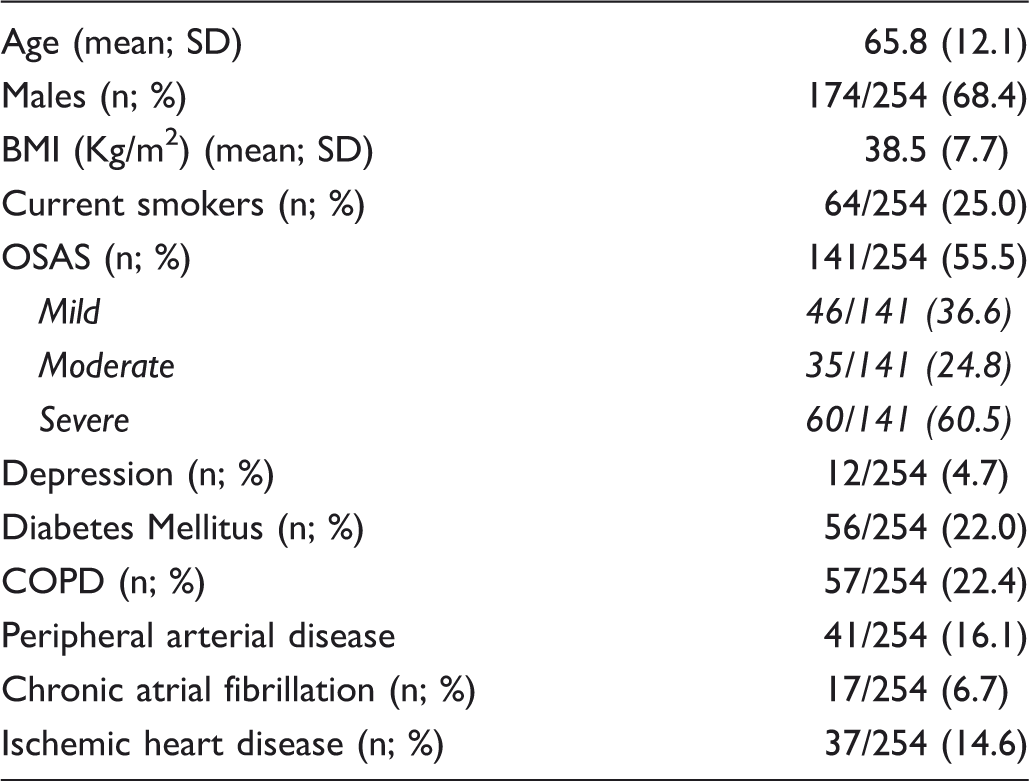
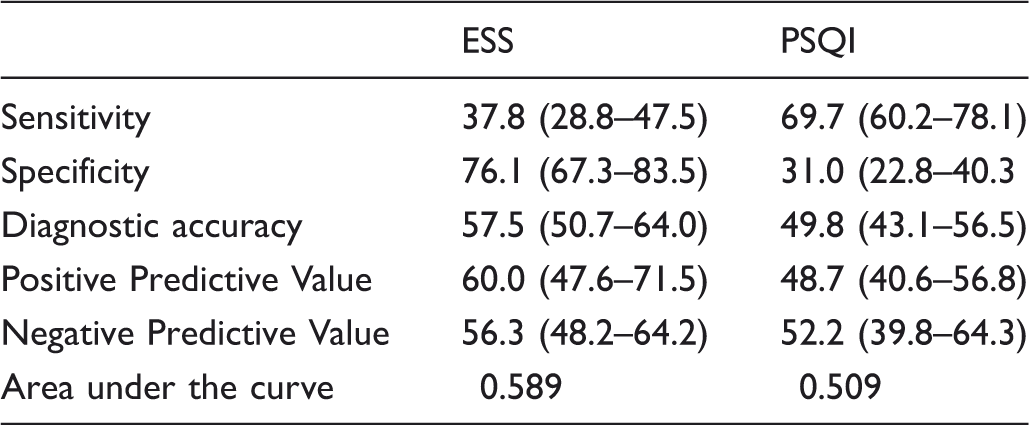
The mean age was 65.8 (standard deviation: 12.1) and the study group was 68.4% male. The mean BMI was 38.5; SD 7.7. Prevalence of OSAS in the study population was 55.5%; OSAS was severe in 60.5% of OSAS patients. ESS was significantly, but weakly, correlated with the AHI (AHI vs ESS: R = 0.308; p < 0.001), whereas PSQI was not (R = 0.037; p = 0.581). Both PSQI and ESS, however, performed unsatisfactorily: sensitivity 37.8% and 69.7%; Specificity 76.1% and 31.0%; Diagnostic Accuracy 57.5% and 49.8%; PPV 60% and 48.7%; NPV 56.3% and 52.2%; AUC 0.589 and 0.509, respectively.
Conclusions
The PSQI score is not helpful in the pre-polysomnographic assessment of people with suspected OSAS. Further studies are required to provide reliable pre-clinical instruments targeting patients amenable to polysomnography.
Background
Obstructive Sleep Apnea syndrome (OSAS) affects about 5% of the population in western countries, 1 but prevalence as high as 20--25% has been reported in people aged over 65. 2 In addition to causing daytime sleepiness and neurologic deficiency, OSAS has untoward metabolic and cardiovascular effects, and is associated with cardiovascular and all causes mortality. 3 Adequate treatment is usually effective at decreasing symptoms and reverses selected cardiovascular and metabolic effects. 4 Early recognition and treatment of OSAS in the general population is therefore a major issue. 5
Polysomnography (PSG) remains the diagnostic gold standard, but it is time consuming and requires dedicated personnel and setting. It may therefore be reasonable to plan a polysomnogram based on a preliminary screening, based on a dedicated questionnaire. 6
In clinical practice, the commonest symptom leading to a PSG is excessive daily sleepiness (EDS). Objective measurement of EDS, however, requires a sleep latency test that is as time consuming as PSG and does not really qualify as a screening procedure. 7 Alternative measures of EDS are self administered questionnaires, the most common of which is the Epworth Sleep Scale (ESS). Despite its widespread use, the accuracy of this questionnaire is far from optimal. In a study conducted on a population of over 1900 people at high risk for OSAS, ESS was weakly correlated with the respiratory disturbance index (r = 0.17, P < 0.0001). 8
The sensitivity and specificity of ESS in the diagnosis of OSAS were 54% and 57% respectively; PPV and NPV were 64% and 47%, respectively. 8 It is unclear whether alternative questionnaires exploring different domains in sleep quality may improve the detection of individuals with OSAS, especially in relation to elderly patients because classic symptoms of OSAS typically decline with age, whereas atypical presentations, mainly with feelings of morning tiredness, nocturia, and number of daily naps, predominate. 9 Excessive daytime sleepiness is also far from a reliable instrument to assess the efficacy of OSAS and to monitor treatment, because non-sleepy patients, representing 30--40% of OSAS sufferers and having a worse prognosis, 10 do not complain of daytime sleepiness.
We therefore proposed testing the screening properties of a questionnaire exploring the average quality of sleep. The Pittsburgh Sleep Quality Index (PSQI) questionnaire is one of the most widely used tools to assess general sleep quality. It is simple, validated, reasonably quick and easy to administrate, and is already recommended in a number of clinical settings, but has never been tested in individuals with OSAS. 11 Moreover, it is the only instrument currently available with psychometric reported data giving information about interpretability of results. 11 It may therefore be of value in assessing people at risk for OSAS. The PSQI is a 19-item self-reporting questionnaire that measures sleep quality over the previous month.12,13,14 It has been proved to reliably assess sleep quality in several medical conditions, 12 and assesses seven clinically derived domains of sleep difficulties (sleep quality, sleep latency, sleep duration, habitual sleep efficiency, sleep disturbances, use of sleeping medications, and daytime dysfunction). Usually a global score higher than 5 is considered an indicator of relevant sleep disturbances in at least two components, or of moderate difficulties in more than three components. 12 More recently, an overlapping of some components has been observed, and three distinct factors have been extracted, 15 sleep efficiency (including sleep duration, and habitual sleep efficiency), perceived sleep quality (including subjective sleep quality, sleep latency, and use of sleeping medication), and daily disturbances (including sleep disturbances and daytime dysfunction).
The aim of our study was to verify whether PSQI can target OSAS patients in a high risk population more accurately than the ESS questionnaire.
Methods
Participants
254 consecutive subjects attending the outpatient clinic for respiratory diseases at Campus Bio-Medico teaching hospital, Rome, for any cause were evaluated for sleep apnea and referred for a 12 channel night-time PSG to investigate respiratory sleep disturbances. Subjects were included if they displayed any of the following symptoms and signs: snoring, abnormal sleep duration and schedule, daytime nap habits, excessive daytime sleepiness, witnessed sleep apnea, increased neck circumference, morbid obesity (BMI > 36), facial dimorphisms (ie. macroglossia, micrognatisms, uvulopalatal hypertrophy etc), otherwise unexplained nocturia, sleep apnea referred by patients or their partners. Subjects were excluded if they were unable to give signed informed consent to participate, or if they had a history of any neurologic condition that might negatively affect sleep quality (eg. parasomnia, epilepsy, extra pyramidal disorders). Subjects with suspected restless leg syndrome or periodic leg movement syndrome at the clinical interview were not excluded because these symptoms often coexist with OSAS. All patients undertook the ESS and the PSQI test on the day before the sleep study. The study protocol was approved by the local Institutional Ethical Committee, and the subjects gave written informed consent, according to the Declaration of Helsinki.
Sleep evaluation
Subjects were tested for OSAS using cardio-respiratory poligraphy or complete PSG according to current international guidelines. 16 PSG was performed in 35 subjects and cardio-respiratory monitoring in 219 recordings by using a dedicated device (Somnomedics; GmbH, Randersacker, Germany). Sleep apnea was defined as a complete or almost complete cessation of airflow, indicated by reduction to 25% or less of baseline amplitude for 10 seconds or more, and hypopnea was defined as a clear decrease in airflow to 50% of baseline for at least 10 seconds. Only apneas and hypopneas that were followed by a 4% or greater decrease in oxygen saturation were counted. OSAS severity was rated by the frequency of apneic and hypopneic events per hour of sleep (apnea-hypopnea index [AHI] 17 :- (mild (10/h ≥ AHI ≥ 20/h), moderate (20/h ≥ AHI ≥ 30/h) and severe (AHI ≥ 30/h).
PSQI scores
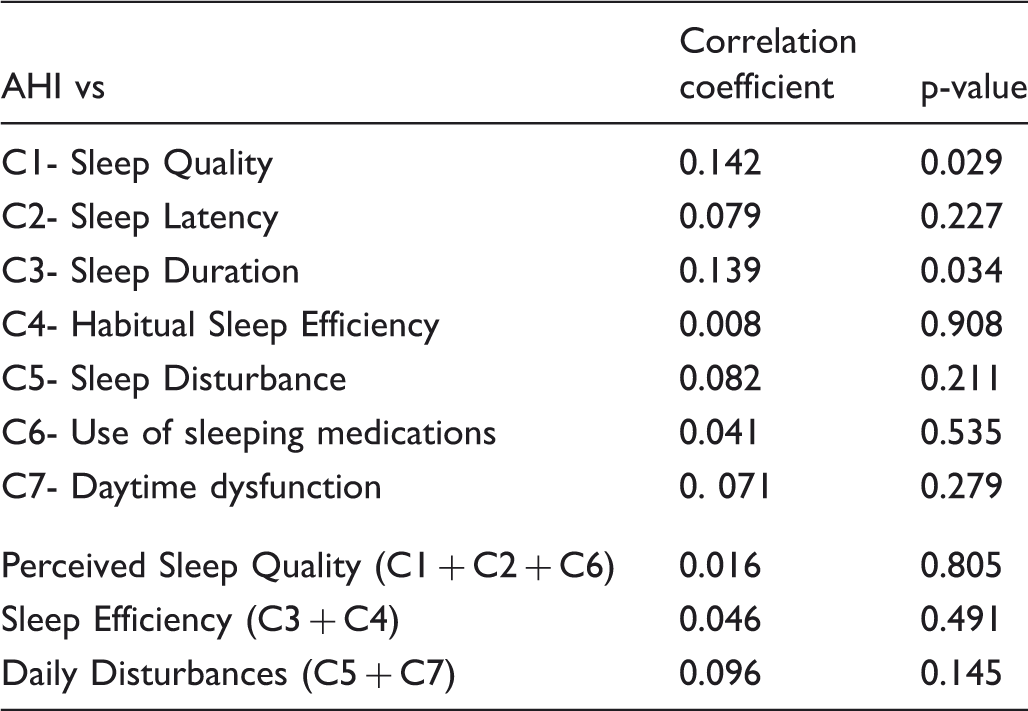
The PSQI was self administered on the day before the PSG recording. Assistance by a trained physician was provided to patients unable to fill the questionnaire. A recently validated Italian language version of the PSQI was used 18 and evaluated following both the original scoring system 12 and the revised 3-factor scoring model as proposed by Cole and colleagues. 16 The dependent variables considered were: Global score; single domains: Subjective sleep quality (C1); Sleep latency (C2); Sleep duration (C3); Habitual sleep efficiency (C4); Sleep disturbances (C5); Use of sleep medications (C6); Daytime dysfunction (C7); aggregated scores: Perceived Sleep Quality (C1 + C2 + C6); Sleep Efficiency (C3 + C4); Daily Disturbances (C5 + C7). Accuracy of individual factors was tested in order to verify whether alternative aggregates of factors had better diagnostic accuracy.
Analytic approach
The concordance between the diagnosis of OSAS and a score ≥ 5 obtained at the PSQI was assessed. Sensitivity, specificity, PPV, NPV, and diagnostic accuracy, expressed as [(true positives + true negatives)/(true positives + true negatives + false positives + false negatives)] were determined. Correlation between the AHI and the PSQI global score was calculated in the whole population and in patients dichotomized according to age (cut off 65 years). To provide an overall index of the ability of the PSQI to identify subjects with OSAS, the area under the receiver operating characteristic (ROC) curve (AUC) was calculated. Individual domains and 3-factor scoring models, reflecting different aspects of diurnal hyper-somnolence and sleep quality, were also evaluated. Indexes of diagnostic accuracy of ESS were computed as standard reference. Statistical analyses were performed using SPSS software (version 19.0; SPSS Inc, Chicago, IL).
Results
Demographics and comorbidity data of the study population.
Polysomnographic respiratory indexes of the study population.

Mean PSQI score differences considering single coexisting disease.
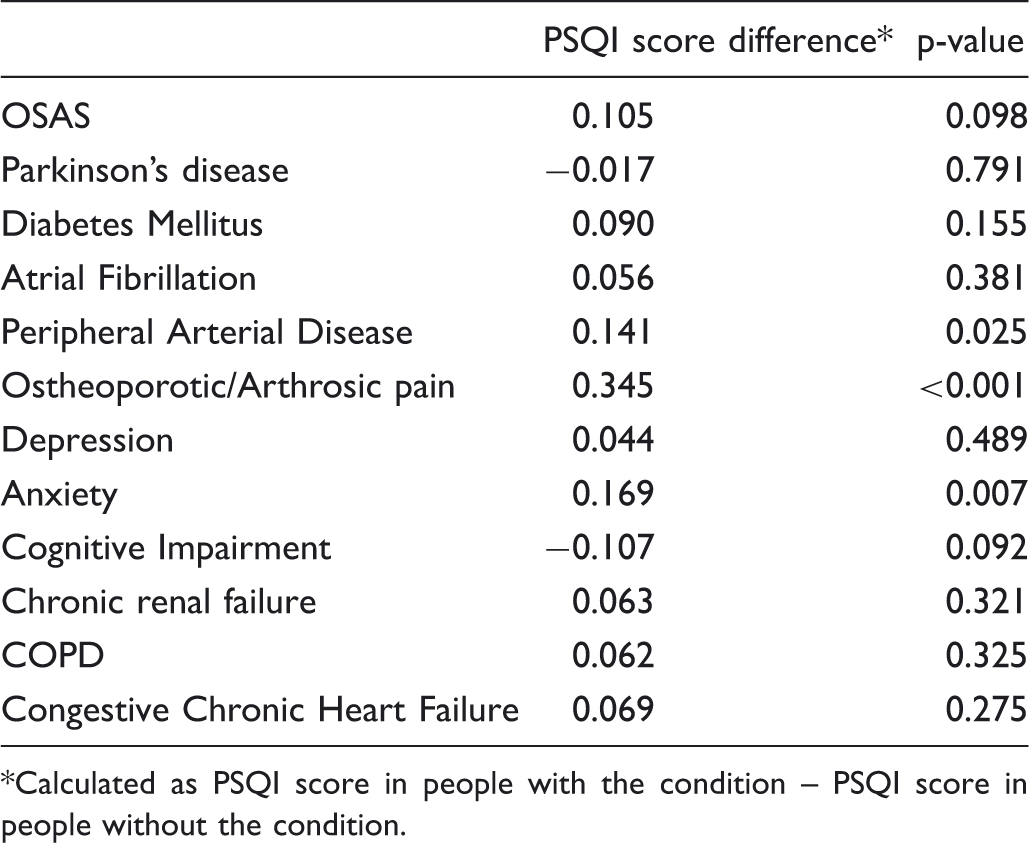
Calculated as PSQI score in people with the condition – PSQI score in people without the condition.
Figure 1 displays scatter plots correlating the scores obtained with ESS and PSQI with the AHI, and showing that only ESS significantly correlated with AHI (AHI vs ESS: R = 0.315; p < 0.001; AHI vs PSQI: R = 0.046; p = 0.491).
Scatter plots correlating the scores obtained with ESS and PSQI with the AHI.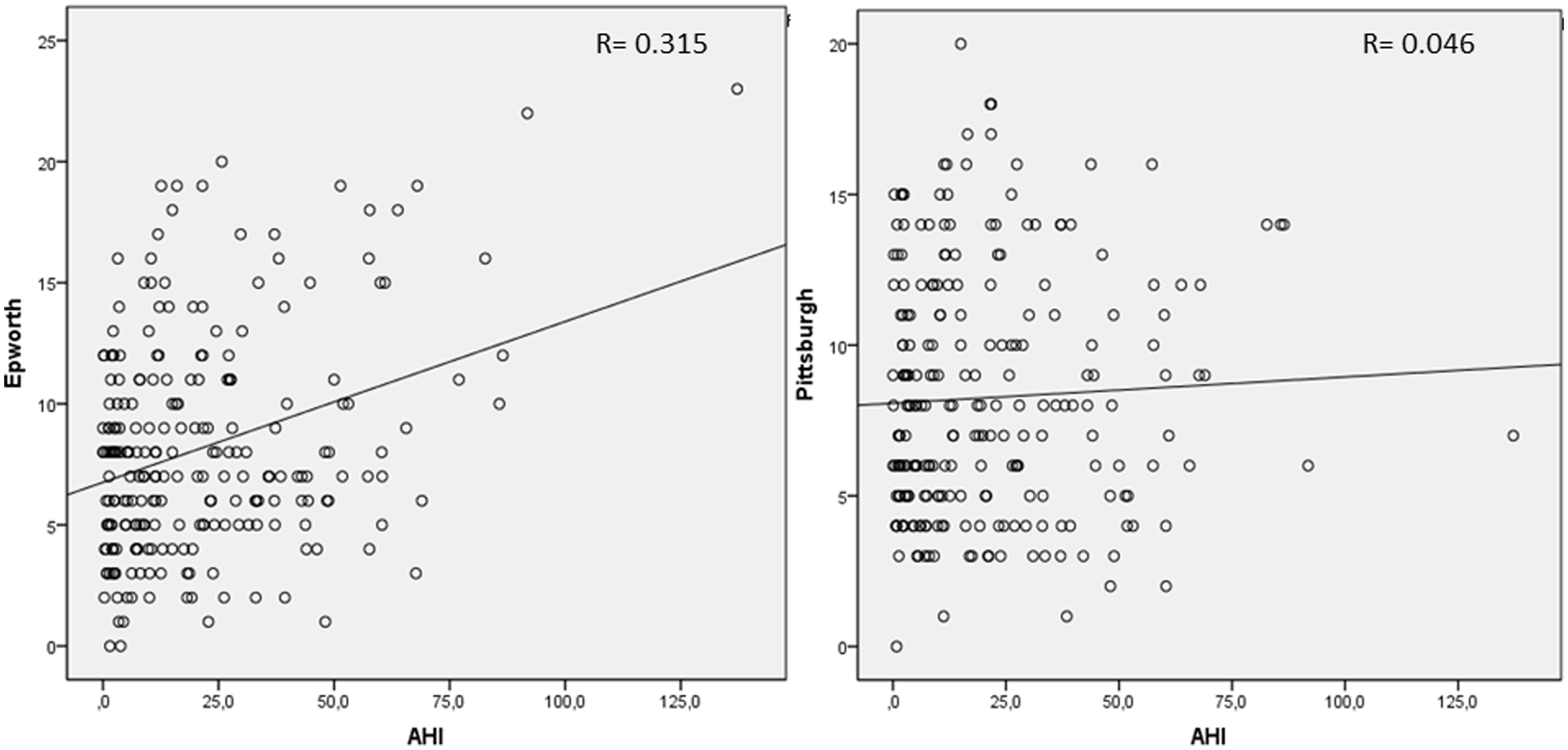
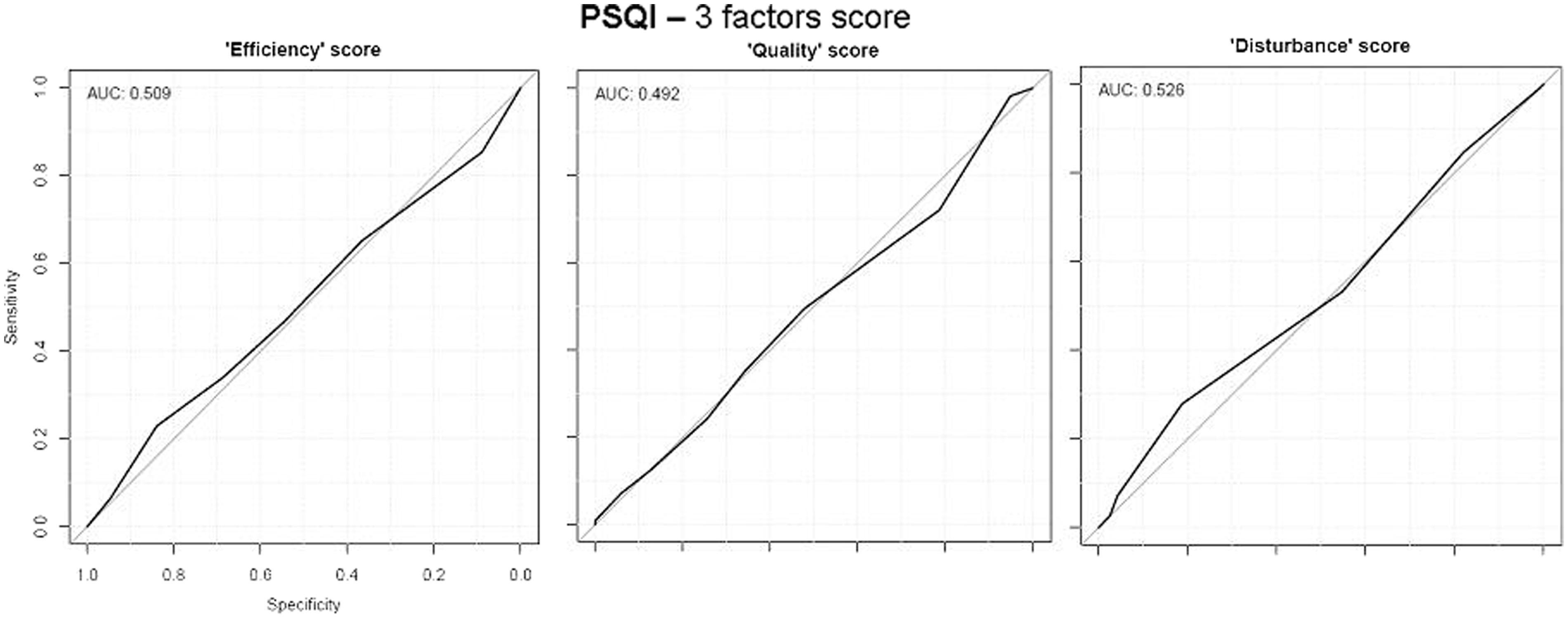
Similarly, figure 2 shows the ROC curves for ESS (upper panel) and PSQI (lower panel) in all subjects (AUC 0.590 and 0.509, respectively), and in those aged under 65 (AUC 0.557 and 0.493, respectively) and over 65 (AUC 0.613 and 0.517, respectively). ROC curves for the 3 factor scoring model are consistent with a poor diagnostic performance (figure 3).
ROC curves for ESS (upper panel) and PSQI (lower panel) in all subjects, those aged under 65 and those aged over 65. ROC curves of the 3-factor scoring model.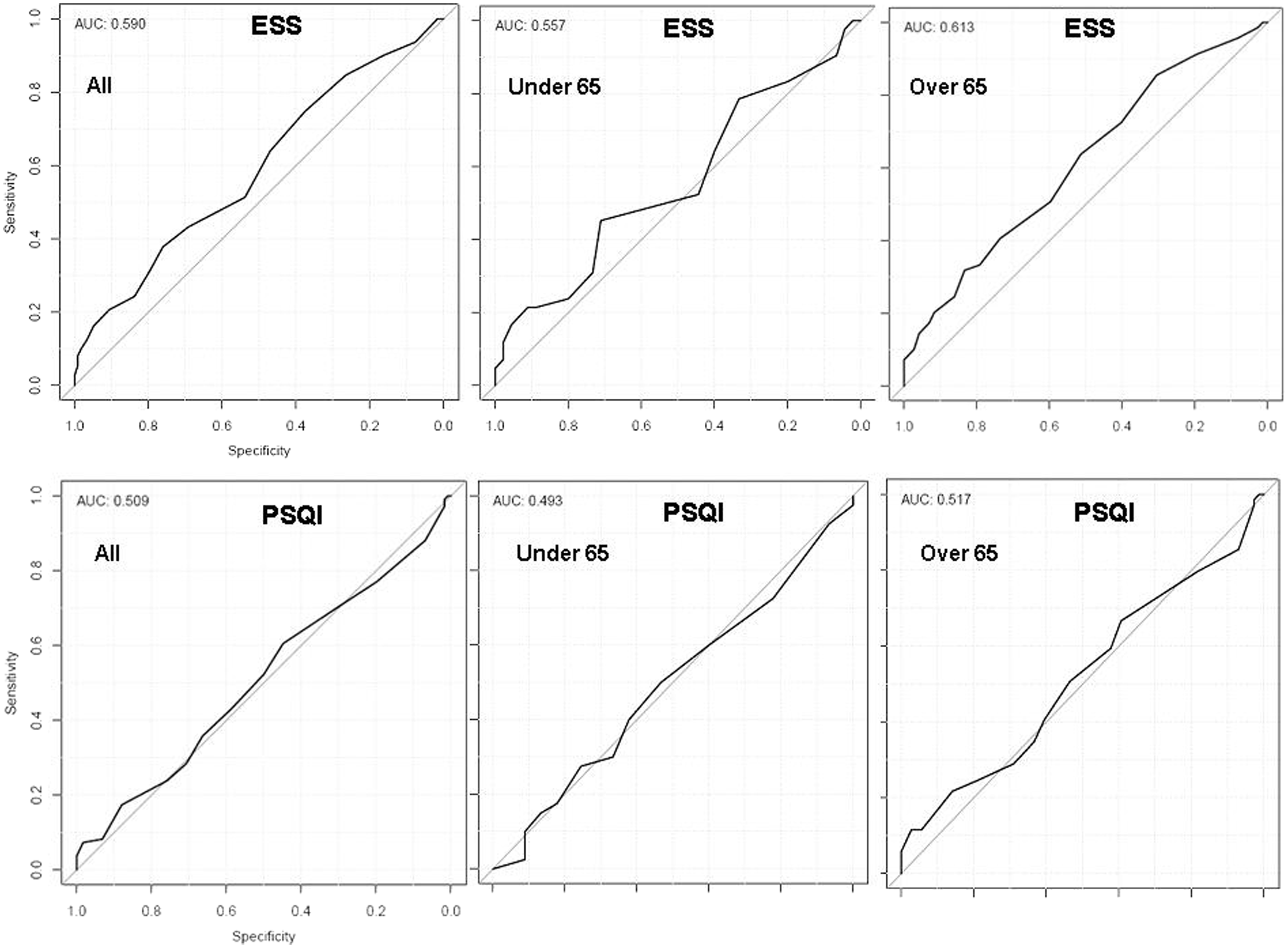
Linear correlation between the AHI score and Pittsburgh’s single and aggregated factors.
ESS and PSQI diagnostic accuracy for OSAS in the study population. Data are expressed as percentages.
Discussion
This is the first study, to our knowledge, assessing whether the PSQI, a screening of global sleep quality questionnaire, could effectively target OSAS patients within a high risk population. We found the PSQI global score to be minimally useful in identifying OSAS patients (AUC for the global score 0.509). The questionnaire lacked satisfactory sensitivity and specificity, and its diagnostic accuracy was poor, regardless of age-related self perceived sleep quality. Even when we considered 3-factor aggregated scores (Perceived Sleep Quality [C1 + C2 + C6]; Sleep Efficiency [C3 + C4]; Daily Disturbances [C5 + C7]), no association was found, probably because these aggregated domains do not efficiently capture self perceived changes in sleep quality occurring in patients with OSAS.
There are several potential reasons for the lack of accuracy. First, PSQI was an important correlate of anxiety and chronic pain in our population. PSQI seems tailored to detect subjective domains of poor sleep quality rather than objective, instrumental features such as cardio-respiratory and polysomnographic ones. Second, the PPV and NPV of a screening instrument depend on the prevalence of the disease in the population. In our population, such prevalence is very high because we deal with outpatients evaluated by PSG for clinical suspicion of sleep disturbances. This instrument may therefore not be ideal as a screening device for a high risk population. PSQI is a questionnaire to assess “normal” sleep quality, although it has been repeatedly used for pathological symptoms (ie. post traumatic stress disorders, chronic fatigue syndrome, and depression). In normal subjects, PSQI was associated, both in adults and elderly, with theoretically related variables (ie, actigraphic variables, depressive symptoms, mobility/instrumental activities of daily living, and health-related quality of life). 20 PSQI effectively captures sudden changes in sleep quality like those experienced after unexpected psychological stresses or traumas 21 but may be less sensitive to sleep problems related to chronic conditions such as sleep apnoea. Finally, many OSAS patients, especially the older ones, do not correctly perceive their sleep problems; they consider them a result and expression of normal senescence. Perceived poor sleep quality is not the major complaint by patients with OSAS; these patients usually seek medical care due to snoring. 22
Despite these limitations, a significant correlation existed between the main measure of apnoea (AHI) and some PSQI components, namely Sleep Quality and Sleep Duration (C1 and C3, respectively) and secondarily Daytime dysfunction (C7). These components also correlate with ESS, clearly indicating that the most involved dysfunctional aspects in OSAS patients are related to sleep quality and quantity, and to daytime (dys)functioning.
We found a significantly increased mean PSQI score in people with conditions known to worsen the quality of sleep and associated with a bad self-reported sleep quality 23 , indicating the value of the PSQI to detect subjective changes in sleep quality despite lack of correlation with objective measurements.
Other instruments and questionnaires have been specifically and successfully used in clinical settings to predict OSAS. A STOP-Bang questionnaire score of 5--8 (or an augmented score at the Berlin Questionnaire) identified, for example, patients with high probability of moderate/severe OSAS.24, 25 This instrument, however, is more suitable in preoperative clinics for a scheduled inpatient surgery, and has not been tested in general medicine settings. 25 On the other hand, on the basis of our study results, assessing general sleep quality to screen for OSAS cannot be definitely considered a first choice strategy.
It could be argued that the use of limited multichannel sleep studies, also known as portable respiratory monitoring (PM), might unify the screening and diagnostic phases of the work up for OSAS, thereby making a screening questionnaire useless. 26 Although promising evidence is emerging, the use of PMs is still under debate. The Medical Expert Panel for Federal Motor Carrier Safety Administration in 2008 reported that PMs should be adequately validated as an alternative to PSG 27 as current data show sensitivity ranging from 75 to 90%, but, at best, 80% specificity. 28 The cost/benefit of PM is also still controversial because the false negative rate is relatively high and current guideline recommend that all negative PM tests in patients with a high risk pretest probability of OSAS should be referred to a full in-laboratory PSG. 29
Our study has some limitations. First, because data were obtained from study visit, results might not apply to the general population and should be considered with caution. Selective attrition of participants due to the onset of specific symptoms and medical morbidity may have produced an over selected sample. In addition we did not use a structured clinical interview to refer patients to PSG. The decision was taken by different physicians on an individual basis. Finally, it should be recognized that sleepiness scores reflect only one aspect of the OSAS syndrome: hypoxic oxidative stress, sympatho-vagal mismatch, and cellular metabolic unbalance might be only weakly related to sleepiness. 30 This makes questionnaire-based screening and, probably more so, the monitoring of ventilated OSAS patients, only partially representative of the OSAS multifaceted biological and clinical expressions.
The lack of a correlation between the PSQI score and the AHI index in obstructive sleep apnoea confirms that PSQI is not helpful in the pre-PSG assessment of this population. However, our study identifies selected sleep dimensions worthy of further assessment in order to develop a more accurate screening instrument.
Footnotes
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
