Abstract
Background
The NHS Bowel Cancer Screening Programme in England offers biennial screening to those aged 60-74 using a faecal occult blood test (FOBt) sent by post. Data from this national clinical programme can also be used for research. Awareness of the impact of such studies on screening participation is important.
Aims
To investigate the effect on screening uptake of adding a research questionnaire to the postal screening invitation.
Methods
People invited for screening in 2008–10 in two areas of England were randomized to receive or not to receive an additional research study questionnaire, consent form and study information, either with their test kit or 2-3 days later. Uptake of screening was examined in relation to study mailings.
Results
Among 11,579 people invited for screening by the Midlands and North West Bowel Cancer Screening Hub, screening uptake was significantly lower in those who received study documents with their FOBt kit than in those who did not (48.6% vs 53.5% respectively: p < 0.001). The reduction in uptake was similar in men and women, and was greater in people living in more deprived areas. Among a further 36,195 people invited for screening by the Midlands and North West and Southern Hubs, sending study documents by separate mailing 2–3 days after the FOBt kit did not affect screening uptake (uptake with and without additional study mailing: Midlands and North West, 56.7% and 56.2% respectively, p = 0.6; Southern, 52.0% and 51.4% respectively, p = 0.5).
Conclusions
Researchers planning studies that include contact with potential participants within the NHS Bowel Cancer and similar screening programmes should be aware of the potential impact on uptake.
Introduction
The NHS Bowel Cancer Screening Programme uses biennial guaiac faecal occult blood testing (FOBt) to screen both men and women for colorectal cancer. The programme in England began inviting people aged 60–69 to take part in screening in July 2006, and achieved national roll-out in January 2010. Extension of the age range to include men and women up to the age of 75 began in September 2008 and is expected to be completed nationally during 2013. The programme currently sends some 3.8 million invitations a year, and over two million participants complete the FOBt.
The screening programme is an attractive setting for research on bowel cancer screening and epidemiology, as the programme is unique in having a single national database that holds comprehensive call/recall, laboratory and clinical investigation records. A large number of projects are planned or in progress, and it is important to ensure that this research is conducted in a way that is not detrimental to screening uptake. There is limited evidence on the impact of research studies on uptake from other UK screening programmes, and past experience may not be relevant to the Bowel Cancer Screening Programme, which is the first screening programme in the UK to include men, and the first to use a test kit sent by post, completed at home and returned without the direct involvement of a primary healthcare provider.
As part of a planned research study, we investigated the impact on uptake of FOBt of sending a research questionnaire and related study documents, either with the test kit or separately 2–3 days later.
Methods
The NHS Bowel Cancer Screening Programme in England
Five regional Hubs are responsible for providing all services relating to the FOBt (invitations, kit mailing and return, kit processing, sending FOBt results to participants and arranging a clinical appointment for those with a positive test result). Follow-up services for participants with abnormal FOBt results are provided by clinical staff at 59 local screening centres. The initial contact between the Hub and potential participants is an invitation letter and information booklet. After receiving the invitation letter, potential participants can contact the Hub to opt-out of screening; on average fewer than 1% do so. About two weeks after sending the invitation packs, the Hubs send test kits to potential participants, with written instructions describing how to complete the kit. Participants then mail the completed kits back to the Hub for processing. Test results (and, where appropriate, information on subsequent repeat screening or diagnostic tests) are returned to participants by letter.
Study participants
The study was conducted in two of the five regional Hubs - Midlands & North West and Southern - over several months in 2008, 2009 and 2010. All men and women sent an invitation to take part in the NHS Bowel Cancer Screening Programme during the study period and who did not contact the programme to opt out of receiving an FOBt kit were eligible for inclusion (this included people invited for a first or a subsequent screen).
Study documents
The study pack included a four-page study questionnaire with demographic, medical and lifestyle questions, and was designed (and described to potential participants as being) for use in a study investigating the characteristics of people with a false-positive FOBt result. In addition to the request for questionnaire responses, potential participants were asked to give permission for follow-up through medical records. Each questionnaire was accompanied by a study information sheet and two consent forms (one for the participant to keep and one to be filled in and returned with the questionnaire), and a postage-paid return envelope. Study documents explained that the research was being carried out by the Director of the NHS Cancer Screening Programmes, with researchers from the University of Oxford, and was funded by Cancer Research UK. The questionnaires that were sent 2–3 days after the kit were sent with an additional covering letter about the study from the Hub director.
The study information sheet and consent forms were produced in accordance with guidance from the National Research Ethics Service [http://www.nres.nhs.uk/applications/guidance/consent-guidance-and-forms/].. Copies of the questionnaires and study documents are available on the study website (www.ceu.ox.ac.uk/bsfs).
Randomization and mailing
Part 1: Research documents included in screening kit mailing
Between May and August 2008, 11,579 participants invited for screening by the Midlands & North West Bowel Cancer Screening Hub were selected to receive (5,857 people) or not to receive (5,722 people) a study questionnaire pack (questionnaire, patient information sheet, consent forms and reply-paid envelope) included in the same envelope as their screening test kit. For logistical reasons, individual randomization was not feasible and so participants were pseudo-randomized, with every 25th FOBt kit recipient chosen to receive the additional study pack, and every 26th recipient chosen as a control. Invitations for screening within each region are automatically generated from NHS records by the Bowel Cancer Screening System based on date of birth, and sorted for mailing by NHS number, a unique identifier which is not based on any of the personal characteristics of the person to whom it is allocated. The combination of the two 'pseudo-random' elements of invitation generation, by birth date and NHS number, was thought to be sufficient to produce pseudo-randomization of the questionnaires by allocating them evenly through the potential study population.
Part 2: Research documents mailed separately 2–3 days after kit mailing
A total of 36,195 participants invited for screening by the Midlands and North West and Southern Bowel Cancer Screening Hubs were selected to receive or not to receive the study pack, mailed separately from the Hub 2–3 days after their screening kit mailing. To minimize the workload for Hub staff, pseudo-randomization was achieved by allocating recipients in mailing batches, the mailing lists being pseudo-randomly generated as described above. In September/October 2009, at the Midlands and North West screening hub, two daily batches of kit recipients, in all 6170 people, were later sent the additional study mailing, and four batches (13,163 people) were selected as controls. In May 2010, at the Southern screening hub, one day’s mailing batch of 5,784 people were sent the additional study mailing, and two batches mailed on the same day of the week, a week earlier and a week later, were chosen as controls (11,078 people).
Data sources and Statistical methods
Screening uptake was defined for this study as receipt of a completed FOBt kit by the Hub within 12 weeks of the initial invitation to take part (ie.10 weeks after the kit was mailed). Uptake was compared between people sent and not sent a questionnaire and study information, using a two sample test for proportions (z-test) with a 5% significance level. Uptake was also compared by age, sex, and level of deprivation. Screening uptake, age, and sex were ascertained from screening programme records (the Bowel Cancer Screening System [BCSS] database). Participants’ postcodes were used to assign a measure of area-level socioeconomic deprivation (Index of Multiple Deprivation 2007 1 ) based on quintiles of the distribution in the national population.
Results
Characteristics of study participants, including those sent and not sent the research study questionnaire pack.
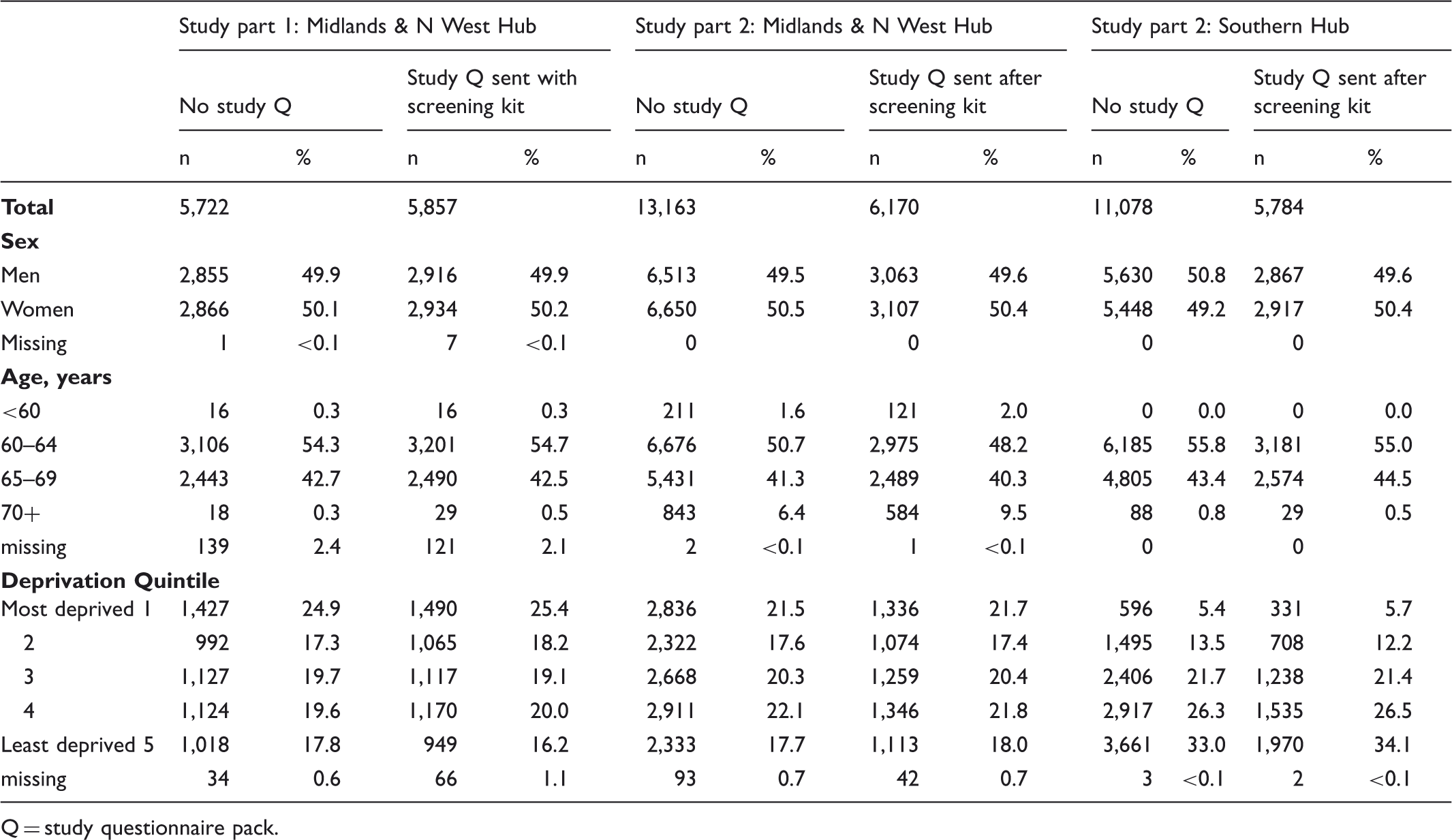
Q = study questionnaire pack.
Study part 1: screening test uptake* for Midlands & N West hub, in those sent and not sent research study questionnaire pack with screening kit.
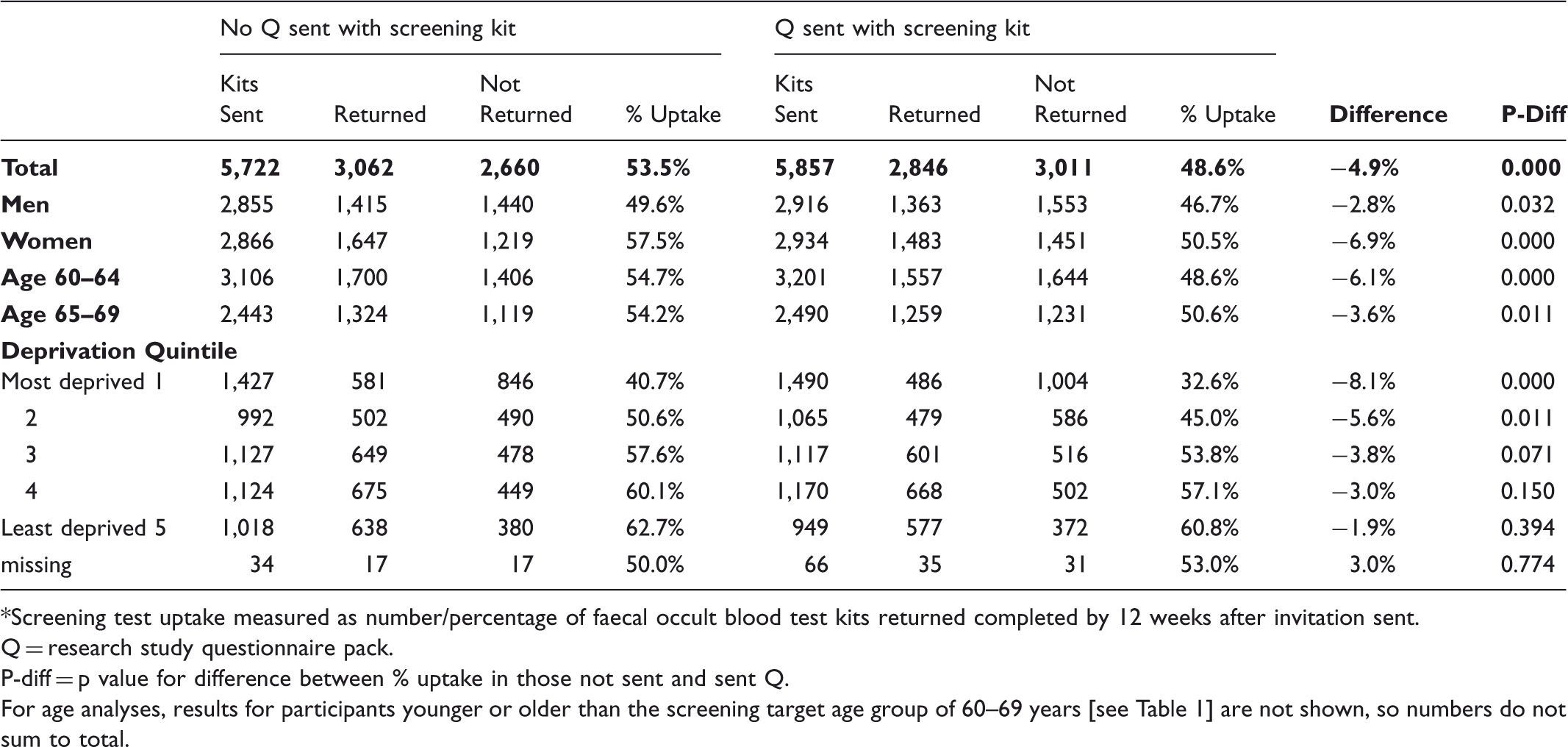
*Screening test uptake measured as number/percentage of faecal occult blood test kits returned completed by 12 weeks after invitation sent.
Q = research study questionnaire pack.
P-diff = p value for difference between % uptake in those not sent and sent Q.
For age analyses, results for participants younger or older than the screening target age group of 60–69 years [see Table 1] are not shown, so numbers do not sum to total.
Study part 2-screening test uptake* for Midlands and N West and for Southern hubs, in those sent and not sent a research study questionnaire pack shortly after their screening kit.
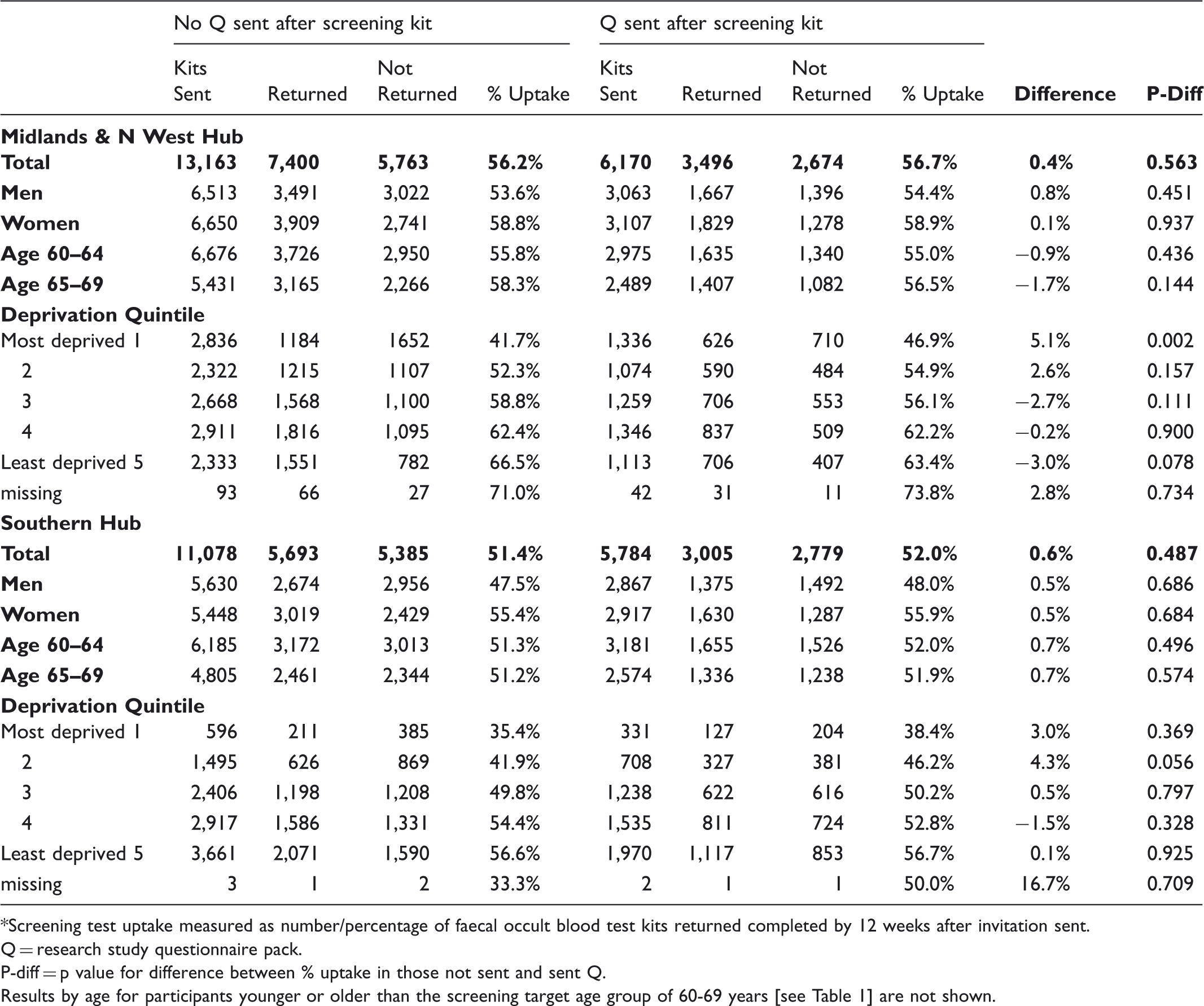
*Screening test uptake measured as number/percentage of faecal occult blood test kits returned completed by 12 weeks after invitation sent.
Q = research study questionnaire pack.
P-diff = p value for difference between % uptake in those not sent and sent Q.
Results by age for participants younger or older than the screening target age group of 60-69 years [see Table 1] are not shown.
Discussion
We found that including a research questionnaire and study information with a mailed FOBt kit was associated with significantly reduced uptake of screening for bowel cancer in an organized screening programme in England. Sending the same study questionnaire and information by separate mailing a few days after sending the kit did not affect uptake. These results underline the importance of conducting pilot studies for planned research associated with cancer screening programmes, and highlight apparent differences in the impact of research on programmes screening for different cancers. Our findings may also be more broadly applicable to programmes screening for other diseases.
Studies similar to ours, in the UK and in Europe, have found no evidence for an effect of adjunct research studies on uptake of mammography screening for breast cancer. In a study conducted in the NHS Breast Screening Programme, 6,400 women invited for breast screening by the Oxfordshire and West of London breast screening units were randomized to receive or not receive a self-administered research questionnaire with their standard invitation to screening. There was no difference in screening attendance between the two groups. 2 Two further studies investigating the effect on breast screening attendance of sending mailed invitations to take part in adjunct research projects around the time of breast screening invitation were conducted in Utrecht (the Netherlands) 3 and Tromsø (Norway). 4 Neither study found any effect of receiving adjunct research invitations on breast screening attendance. It was also found that conducting research interviews before inviting women to the NHS Breast Screening Programme in London (England) had no effect on subsequent screening uptake. 5
To our knowledge there has been one previous study of the effect on screening uptake of adjunct research in a bowel screening setting, conducted by O’Sullivan and colleagues within the setting of the pilot for the NHS Bowel Cancer Screening Programme in Coventry and North Warwickshire. 6 People sent an FOBt kit were selected to receive or not receive a self-administered questionnaire addressing their attitudes to participating in FOBt screening. The questionnaires were mailed two working days after the FOBt kits, and no significant difference in overall screening uptake between the two groups was seen. The results of the second part of our study are consistent with this finding.
We do not know why including a questionnaire within FOBt kit mailings reduced screening uptake, but sending questionnaires out separately 2–3 days later did not. The NHS Bowel Cancer Screening Programme, unlike those in the UK for other cancers, does not involve direct contact with a healthcare practitioner for the initial screening test. The FOBt kit mailings necessarily include information about the kit and how to perform it in written format. The questionnaire pack also contained large amounts of written information, and receiving both at the same time may have been overwhelming for some potential screening participants. This might be particularly likely for those with limited literacy skills and/or those already less likely to return a completed kit, and may explain the greater effect of questionnaire inclusion with the screening kit on uptake in more deprived socioeconomic areas. 7 There was no evidence for such an effect when the study documents were sent 2–3 days after the screening kit; indeed our results (Table 3) show a tendency for this kind of study mailing to be associated, if anything, with increased screening uptake in the more deprived socioeconomic groups.
Our study benefited from the availability of a well organized screening programme which routinely sends out large numbers of screening kits. The NHS Bowel Cancer Screening programme BCSS database allowed follow-up of patient behaviour from kit mailing to return, allowing accurate calculation of screening uptake with a very small percentage of missing information on age, sex and deprivation index. Individual randomization of potential participants to receive or not receive the study documents was not feasible. However screening invitation lists are organized both by birth date and by randomly-assigned NHS number (and, for those who have been screened previously, the date of completion of their previous screen), and there is no obvious reason why the pseudo-randomization used, based on these lists, should not result in an acceptably equal spread of patient characteristics across the questionnaire and control groups. Comparison of known patient characteristics (age, sex, quintile of deprivation index) between the questionnaire group and controls suggests that our randomization methods were adequate. The figures presented in this paper for screening uptake apply to the research setting, and should not be taken to represent accurately the operational characteristics of the NHS Bowel Cancer Screening Programme, either at the time of the study or subsequently. Programme definition of uptake is not identical to that used in this study.
Large-scale, well-organized screening (and other health) programmes with centralized electronic databases have huge potential as research resources. When research studies involve direct contact with programme participants it cannot be assumed that they will not have an impact on screening behaviour.
Declarations
The study was funded by Cancer Research UK. The funders did not influence study design, analysis, interpretation or the decision to publish. JW and JG had full access to study data and act as guarantors. The study was designed by JG, JP and VB, with analyses by JW. KS and MM contributed to database design and data entry, SS and SH to data collection; the manuscript was drafted by JW and JG with contributions from all authors.The authors have no competing interests to declare.
Footnotes
Acknowledgements
Midlands & North West Bowel Cancer Screening Programme Hub (Sara Lee, Hetal Kachiwala); Southern Bowel Cancer Screening Programme Hub (Julia Snowball, Katy Reed); NHS Cancer Screening Programmes: Claire Nickerson; the late Dr Joan Austoker (and her research team).
Funding
Cancer Research UK project grant C4023/A7506.
Bowel Screening Follow-up Study Investigators
Current: Professor Wendy Atkin, Professor Valerie Beral, Dr Jane Green, Professor Julietta Patnick, Dr Roland Valori. Professor David Forman was a study investigator between 2007 and 2010.
Bowel Screening Follow-up Study Collaborators
Mr Ronald Parker, Professor Philip Quirke
