Abstract
This study presents manufacturing of Poly(ethylene glycol) diacrylate (PEGDA)- Hydroxyapatite (HA) composite by Digital Light Processing (DLP) technology. PEGDA along with HA extracted from bovine was mixed with a photoinitiator (PI) and water to achieve optimized rheology for DLP. The printing parameters for DLP were optimized. Chemical composition, microstructure and mechanical properties of the composite were characterized. The compressive strength of PEGDA-HA composite achieved a maximum of 20 MPa in Z direction. The flexural strength was 56 MPa and 34 MPa for horizontal and vertical orientations, correspondingly, indicating anisotropic behavior of the samples. The cytotoxicity assessments were performed. The 3-(4,5-Dimethylthiazol-2-yl)-2,5-Diphenyltetrazolium Bromide (MTT) assay and confocal imaging indicated that the scaffold powder exhibits minimal cytotoxicity at low concentrations (0.001 g/ml), with high cell viability and minimal cell death or membrane damage.
Introduction
Recently, interest in biocompatible polymer-ceramic composites has been growing rapidly.1,2 In the context of tissue engineering or biomedical applications, polymer-ceramic composites can offer enhanced biocompatibility compared to traditional ceramics.3–5 Individually, ceramic materials and polymers have certain disadvantages when considering their application in implantology. Ceramic materials are usually brittle, which can lead to fracture under mechanical stress. On the other hand, polymers often exhibit low mechanical strength and anisotropic properties, making them less suitable for load-bearing applications. 6
However, a composite of these two types of materials often demonstrates superior properties compared to each material used separately. 1 Ceramics provide excellent biocompatibility, and wear resistance, while polymers contribute elasticity, ductility, and ease of processing.7–9
Polymers like hydrolysable Poly(ethylene glycol) diacrylate (PEGDA),10,11 have attracted significant attention in biomedical and biotechnology fields due to their remarkable properties. The Poly(ethylene glycol) (PEG) component provides biocompatibility and hydrophilicity, while the acrylate groups allow to cross-link the polymer under suitable conditions. PEGDA is known for its excellent biocompatibility, causing minimal immune response when implanted in the body. 12
Hydroxyapatite (HA) is well known biomaterial. It is crucial for bone regeneration and integrates perfectly with natural bone tissue. 13 The combination of these two materials leverages the strengths of each: PEGDA’s flexibility, biocompatibility, and ability to form hydrogels, and HA’s osteoconductivity and structural similarity to bone mineral. This synergy results in composite materials that are highly suited for bone tissue engineering applications.14,15
Khalili et al., in their review paper, described recent advancements in the fabrication, characterization, and biomedical applications of PEGDA hydrogels. They highlighted PEGDA-HA biocomposites as permeable scaffolds for bone graft applications, emphasizing their mechanical properties and potential for in vivo bone regeneration. 11
With the introduction of the latest technologies, these polymer-ceramic composites can be easily produced. Additive manufacturing (AM) technologies allows to fabricate metals, ceramics, polymers, and composites with complex shapes and structures, offering a wide range of design possibilities.16–18
Digital Light Processing (DLP) is an AM process that utilizes light to selectively cure liquid resin, resulting in 3D objects with high resolution and precision. The process involves projecting an image of the 3D model in a liquid resin bath using a light source, such as a projector or a laser. As the light interacts with the resin, it cures and hardens the resin, forming a solid layer that corresponds to the shape of the object being printed. The liquid resin can also contain ceramic powder, allowing the printing of polymer-ceramic composites.19,20
Advancements in additive manufacturing (AM) technologies have made it possible to fabricate polymer-ceramic composites with complex geometries and tailored properties. 16 Among the various AM techniques, Digital Light Processing (DLP) stands out due to its ability to produce 3D objects with high resolution and precision. Unlike other methods such as Fused Deposition Modeling (FDM) or Selective Laser Sintering (SLS), DLP enables the fabrication of intricate designs by selectively curing a liquid resin using projected light. This process is particularly advantageous for polymer-ceramic composites because it allows precise control over the material’s microstructure and minimizes material waste. The addition of ceramic powders to the resin enables the production of composites with enhanced mechanical and biological properties.
Despite its promise, the application of DLP in fabricating polymer-ceramic composites faces several challenges. The inclusion of ceramic powder increases the viscosity of the resin, complicating the printing process. This can result in issues such as shape shrinkage, poor densification, cracking, and reduced mechanical performance. 19 Furthermore, achieving a uniform distribution of ceramic particles within the polymer matrix is critical to ensure consistent properties across the printed part.
Polymer-ceramic composites developments.

However, the overcoming of limitations associated with ceramic brittleness and mechanical integrity of printed parts remain critical in utilizing DLP technology in biomedical engineering. In this context, the present study aims to address these challenges by developing polymer-ceramic composites suitable for biomedical scaffolds.
This study introduces an approach to the fabrication of biocompatible polymer-ceramic composite scaffolds using DLP technology, with a focus on the development of PEGDA and HA composites.
The study highlights the successful customization of DLP printing parameters, which resulted in high-resolution scaffolds with robust mechanical integrity. The scaffolds demonstrated anisotropic mechanical behavior, with significant compressive and flexural strengths, making them suitable for bone tissue engineering. Furthermore, the biological evaluation revealed cell viability at lower scaffold concentrations, indicating the material’s biocompatibility and suitability for biomedical use.
Materials and methods
The polymer used was PEGDA (Mw = 575), supplied by Sigma Aldrich. Diphenyl(2,4,6-trimethyl-benzoyl) phosphine oxide (TPO, 97%, Sigma Aldrich) was used as the photoinitiator (PI). Deionized water was employed as a diluent to adjust the viscosity of the slurry.
Extraction of hydroxyapatite
Bovine tibia bones were cleaned with a sharp knife to remove residual muscle and visible impurities. The bones were then cut into small cylindric pieces of 2 cm in thickness and boiled for 6 h in a closed pot. The boiled bones were washed several times with distilled water and immersed in acetone for 3 h to remove invisible traces of fat. The samples after were dried in an oven (WGLL-85BE) at 250°C for 4 h. The thermally treated bones were calcined in a furnace (Atmosphere muffle furnace GCF 1400, Across International) at 900°C for 5 h, with a heating rate of 5°C/min.31,32 The calcined pieces were planetary ball milled (Vertical Planetary Ball Mill BKBM-V2) at 240 rpm for 6 h to obtain powdered HA. The HA powder was obtained after 325 mesh sieving to eliminate larger particles.
Preparation of PEGDA/HA slurry
Composition of the slurries.

3D printing of PEGDA/HA composite scufolds
Printing parameters for DLP.
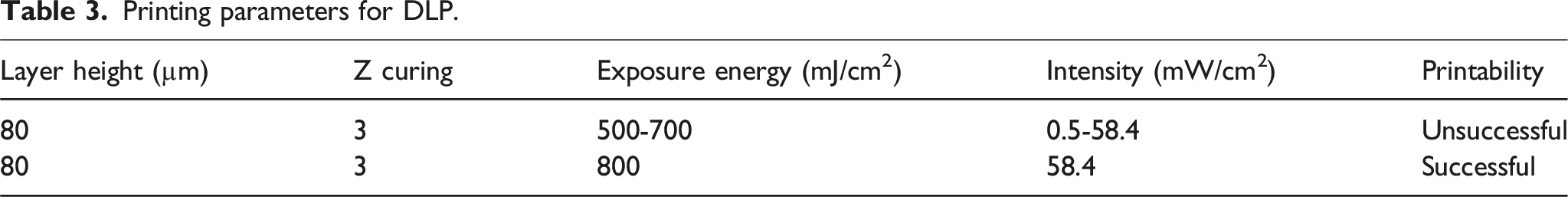
Type and dimensions of printed samples and mechanical properties.

Characterization
Particle size of HA was measured by laser scattering using a Mastersizer 3000 equipment (Malvern Panalytical B.V., Almelo, The Netherlands). Phase characterization was accomplished using X-ray diffraction (XRD) analysis. The data on the phase composition were collected by a MiniFlex 600 Rigaku Smart Lab SE diffractometer (Rigaku Corporation, Tokyo, Japan) with a D/teX Ultra 250 1D detector and processed via the JCPDS-ICDD database. Microstructural characterization was performed by scanning electron microscope (SEM, Prisma E, Thermo Fisher Scientific, Hillsboro, OR, USA). Samples were subjected to coating using a 30 nm layer of Ag to provide sufficient conductivity for SEM analyses. The viscosity of the slurries was determined by using a rotational viscometer (NDJ-1B, Shanghai Pingxuan Instrument Co., Ltd., China), applying rotor #2 and 60 rpm. Compressive strength of the samples was measured using universal mechanical testing machine (Instron Legend 5965 testing system, USA), equipped with a 5 kN load cell, with a crosshead speed of 20 mm/min at room temperature. Flexural strength was measured using 3-point bending method in accordance with ISO 23242:2020.
The metabolic activity (MTT) (3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide) assay was used to assess the cytotoxicity of tested composites by measuring metabolic activity of MDA-MB-231 (epithelial cancer) and HaCaT (human normal keratinocytes) cells exposed to samples SL6 and printed PEGDA in powder (0.5µm-1µm) form.
All cells were seeded at a density of 10,000 cells per well in a 96-well plate. After 24 h, they were exposed to powder suspensions at various concentrations and incubated for an additional 24 h.
Subsequently, the powder suspensions were removed, and the cells were washed twice with phosphate buffered saline (PBS). Each well was filled with 100 µL of fresh media and 20 µL of MTT reagent. The cells were incubated for 3 h to allow metabolically active live cells to convert MTT into formazan crystals. After incubation, the media and MTT reagent were discarded, and MTT solvent was added to dissolve the formazan crystals. The absorbance of the wells was measured at 570 nm using a spectrophotometer (BioTek Synergy H1 Multimode Reader), providing quantification of metabolic activity and indirectly of cell viability.
Human MDA-MB-231(ATCC: HTB-26 epithelial cancer) and HaCaT (ATCC: HTB-22 normal keratinocytes) cells were cultivated in Dulbecco’s modified Eagle medium with 5% v/v fetal bovine serum (DFBS) (Gibco/Termo Fisher Scientefic, Rochester, MN), 100 U/mL penicillin, and 100 μg/mL streptomycin at 37°C in a humidified atmosphere containing 5% CO2 (Memmert ICO105). Both cells lines were demonstrating similar results, therefore the results obtained from MDA-MB-231 are presented.
To visually illustrate cytotoxicity results, the DiO and propidium iodide fluorescent dyes were used to stain the cells and evaluate the effect of tested powders on their viability. The green fluorescence of DiO visualizes living cells, while the red fluorescence of propidium iodide facilitates the identification of non-viable cells. This comprehensive analysis provides valuable information about the efficacy and impact of the composites on the MDA-MB-231 cells.
MDA-MB-231 cells were seeded on coverslips enabling to reach a confluency of 70%. After 24 h, the cells were exposed to powder suspensions at the highest concentration of 0.1 g/ml and the lowest of 0.001 g/ml. After 24 h incubation, the cells were stained with 5 µM DiO and then propidium iodide was added to the cells at a concentration of 2 µl/ml. Following 30 min staining, the cells were washed 3-4 time with PBS and were fixed with Mowiol mounting medium for further microstructural study.
Results and discussions
For the calcination of bones, 900°C was chosen based on available literature, which indicates that higher temperatures lead to increased crystallinity and grain growth. In contrast, at lower temperatures, organic components and carbonates produced during processing remain present in the material, which can affect its purity and bioactivity.31,32 The X-ray diffraction (XRD) pattern of calcinated bovine bone is shown in Figure 1. The crystalline peaks in the XRD pattern closely match those of hydroxyapatite (HA), indicating that the thermal process successfully yielded pure HA. The crystal size of the powder, according to the Rietveld method and using Scherrer equation, is 42.4 µm. XRD of bovine bone powder versus synthetic HA.
The ATR-FTIR spectra (Figure 2) show the characteristic vibrational modes of bovine bone before and after calcination at 900°C. The raw bone spectrum contains peaks corresponding to both organic and inorganic components. Typically, amide I (∼1650 cm-1) and amide II (∼1550 cm-1) bands appear due to the presence of collagen. The major peaks around 1030-1100 cm-1, 600 cm-1, and 560 cm-1 correspond to phosphate (PO43-) vibrations. Additionally, carbonate (CO32-) substitution in hydroxyapatite is observed within the 1400-1500 cm-1 region. ATR-FTIR spectra of bovine bone before (a) and after (b) calcination at 900°C.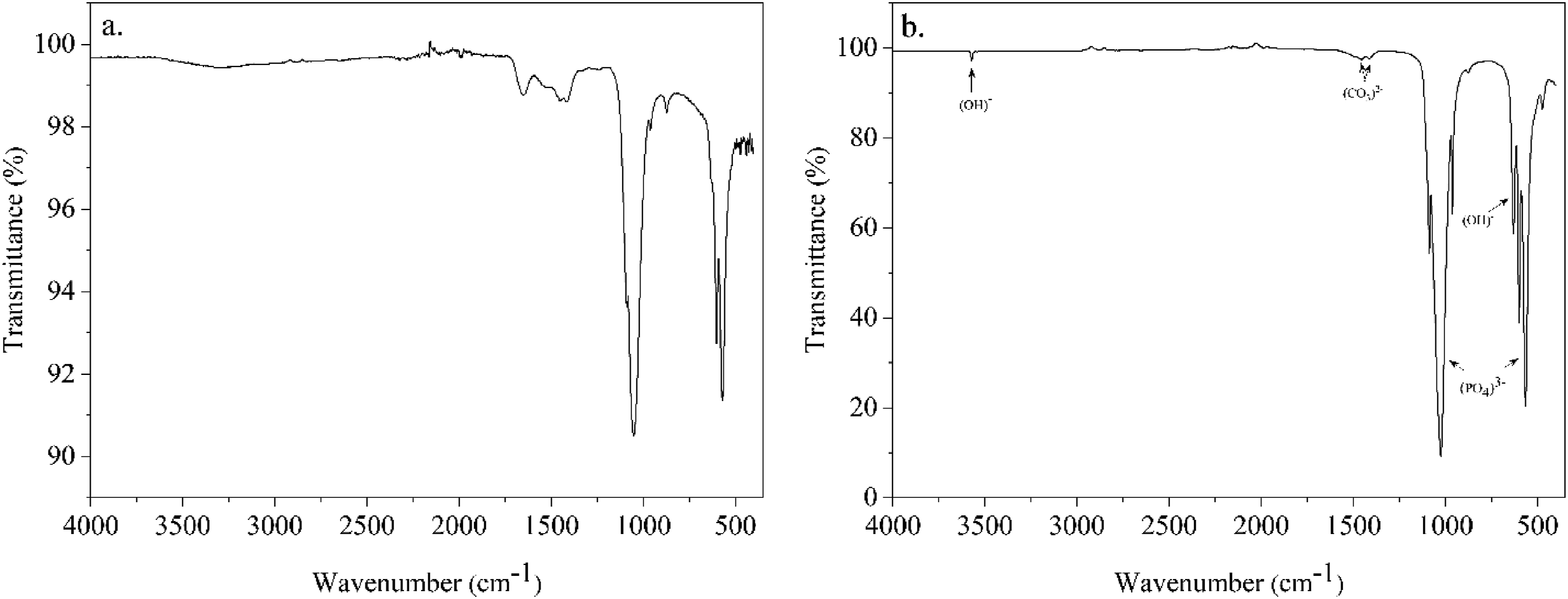
After calcination at 900°C, the organic components, including collagen, are completely degraded, as evidenced by the absence of amide bands. The sharp peaks around 1030-1100 cm-1 and 560-600 cm-1 remain, confirming the presence of hydroxyapatite. Furthermore, the peak near 3570 cm-1 corresponds to hydroxyl (OH-) vibrations, indicating the formation of a more crystalline hydroxyapatite structure.
Figure 3(a) illustrates the particle size distribution of the obtained HA powder. A bimodal distribution is observed, with the majority of particles within the 1–5 µm range (average particle size: 1.7 µm) and a smaller fraction in the 5–20 µm range (average particle size: 10 µm). Notably, the smaller particle fraction constitutes the majority of the powder (>90%). Bovine bone powder (a) Particle size distribution and (b) SEM micrograph.
The particle size distribution plays a crucial role in the printability of the ceramic slurry in DLP-based 3D printing. The predominance of fine HA particles (<5 µm) contributes to a higher surface area, which enhances the dispersion and stability of the slurry, improving homogeneity and reducing sedimentation during printing. 33 However, excessively small particles can significantly increase viscosity, leading to challenges in recoating and potentially limiting light penetration during photopolymerization. In contrast, the presence of a smaller fraction of larger particles (5–20 µm) can improve powder packing density, reduce shrinkage upon curing, and enhance mechanical integrity. Maintaining an optimal balance in particle size distribution is essential for ensuring smooth slurry flow, uniform layer formation, and high-resolution printing.
The SEM images in Figure 3(b) reveal that most particles are uniform in shape, with sizes ranging from 1–2 µm. Furthermore, the particles appear well-dispersed and show no evidence of agglomeration.
For the development of the photocurable ceramic slurry, PI with varying concentrations were used (Table 2). The concentration of the PI significantly affects the printability of the resin. 34 At lower concentrations (0.5 wt% on PEGDA), the initiator of polymerization was insufficient, leading to incomplete curing and failed prints. The minimum level of PI is essential for initiating the polymerization reaction necessary for 3D printing.
Increasing the PI concentration to 1.5 wt% enabled the curing process and made printing feasible. However, challenges emerged, as prints adhered to the resin vat instead of the building platform. This behavior suggests that while the higher PI concentration facilitated polymerization, other factors, such as slurry viscosity and interaction with the vat surface, required further optimization. Rapid curing at higher PI levels likely hindered proper adhesion to the platform.
To address these issues, 20 wt% water was added to the slurry (Table 2). Acting as a deluant, water effectively reduced slurry viscosity, improving its flow characteristics. This adjustment resolved adhesion issues and allowed for consistent layer formation, demonstrating that proper dispersion of the PI and resin components is critical for achieving reliable print quality. Mau at all. in their study demonstrate promising results of DLP 3D printing of PEGDA hydrogels for frontal sinus implants. It was shown that printing accuracy and Shore A hardness decrease with increasing content of water in the PEGDA hydrogel. 35
In addition to slurry composition, DLP process parameters significantly influenced printability (Table 3). In this study, various exposure energy doses (500, 600, 700 and 800 mJ/cm2) were systematically tested. However, these lower doses resulted in incomplete resin curing, leading to weak and under-cured prints. The optimal exposure energy was determined to be 800 mJ/cm2, which ensured successful polymerization and fully cured prints, achieving the desired mechanical integrity and structural accuracy. The layer thickness was set to 80 µm, with a light intensity of 58.4 mW/cm2. Table 4 details the sizes and densities of the printed samples.
The microstructure of the printed samples is shown in Figure 4. SEM images reveal globular forms of HA powder, approximately 25–35 µm in size. The surfaces of the samples are free of cracks, and the HA globules exhibit porosity, with pore sizes ranging from 0.5 to 1 µm. SEM images of printed sample with (a) 100 μm and (b) 20 μm magnification.
Mechanical properties
The mechanical properties of the printed samples were evaluated, focusing on compressive strength, flexural strength, and Young’s modulus. These properties are important indicators of the scaffold’s ability to support tissue regeneration and withstand physiological loads. The compressive strength for the cube-shaped samples was measured in three directions relative to the printing orientation: longitudinal (X), lateral (Y) and perpendicular (Z) to the layer orientation (Table 4).
The highest compressive strength was observed in the Z direction. This typically presents the strongest resistance to compressive forces due to the alignment of the layered structure. The higher strength in this direction suggests that the scaffold can effectively resist compression when forces are applied perpendicular to the layers. The lowest compressive strength was in X direction. This direction often shows lower strength compared to the Z direction due to possible weaknesses in interlayer adhesion or interlayer bonding. However, 13 MPa still shows a moderate resistance to compression. 36
These results suggest that the printed scaffolds are capable of withstanding typical physiological compressive forces, making them suitable for biomedical applications, particularly in tissue growth and regeneration.
Flexural strength was assessed to determine the resistance of the samples to bending forces, while Young’s modulus was measured to evaluate the stiffness and elastic behavior of the printed scaffolds. Rectangular prism samples were tested in both horizontal (H) and vertical (V) orientations relative to the printing layers. The flexural strengths of 56 MPa and 34 MPa indicate that the printed scaffolds have a significant capacity to resist deformation under bending loads. These values suggest that the scaffolds have adequate mechanical properties for bioscaffolds applications. 36 Flexural strength is critical to ensure that scaffolds can maintain structural integrity under physiological conditions where they may experience various mechanical stresses. 37
High values of Young’s modulus indicate that the printed samples have sufficient stiffness, which is useful for providing rigid support when the load is applied. 38
Cytotoxicity
Metabolic activity assay
The results from 5 individual experiments revealed that low concentrations (0.01, 0.003, and 0.001 g/mL) showed no significant difference compared to the control (P > 0.05), suggesting a non-toxic effect at these levels. Specifically, SL6 became non-toxic at 0.001 g/ml, demonstrating 91% metabolic activity compared to the control. Higher concentrations of SL6 powder (0.1, 0.05, and 0.03 g/mL) significantly reduced metabolic activity (P < .05), indicating potential cytotoxicity. Given that we initially tested metabolic effects using cancerous cells, our next step was to investigate if the composite might possess anti-cancer properties. However, our findings revealed that the composite demonstrated comparable toxicity in normal cells (HaCaT human keratinocytes), which mirrored its effect on cancer cells (data not shown) (Figure 4).
The results from 5 individual experiments revealed that lower concentrations of SL6, significantly reduced cytotoxicity on MDA-MB-231 cells in the powder suspension. Additionally, SL6 became non-toxic at 0.001 g/ml, demonstrating 91% metabolic activity compared to the control. Given that we initially tested metabolic effects using cancerous cells, our next step was to investigate if the composite might possess anti-cancer properties. However, our findings revealed that the composite demonstrated comparable toxicity in normal cells (HaCaT human keratinocytes), which mirrored its effect on cancer cells (data not shown) (Figure 5). (a) “Control” (non-exposed cells), (b) Cells exposed to “Sample SL6” after 24 h of co-culture.
Cell visualization
To visually assess the impact of SL6, MDA-MB-231 cells were exposed to 0.1 g/ml and 0.001 g/ml powder suspensions for 24 h, followed by staining with DiO live cell dye. The results (Figure 6) demonstrate that the majority of cells exposed to 0.001 g/ml powder suspension remained viable and alive throughout the experiment. Minimal propidium iodide staining suggests negligible cell death or damage attributable to the scaffold powder, despite some crystals being internalized by the exposed cells. SL6 is non- toxic at low concentrations. Metabolic activity of MDA-MB-231 cells exposed to different concentrations of SL6 powder. Data are presented as mean ± standard deviation (SD). Statistical significance was assessed using an independent two-tailed t-test, comparing each concentration with the control. ns: not significant (P > .05), *P < .05.
In contrast, cells exposed to 0.1 g/ml powder suspension were undetectable likely due to detachment prior to staining. These findings align with the metabolic activity results obtained under the same experimental conditions. The combination of live cell staining and the absence of propidium iodide staining indicates that the scaffold material was well-tolerated by the cells and did not induce significant cell death or membrane damage at the lower concentration of 0.001 g/ml. Several factors could explain the observed toxicity. One possibility is the finer particle size of the powder (approximately 0.5µm-1µm) used in cell viability study. These smaller particles could more easily penetrate cell membranes or be phagocytosed by cells, potentially disrupting cellular processes, or altering cytoplasmic pH, or increasing cytoplasmic density at higher concentrations. Additionally, PI TPO 1% can have an influence on heightening of cytotoxicity of the scaffolds powder in high concentration as shown in work of Popal at all. 39 Where authors investigated the cytotoxic and genotoxic effects of the photoinitiators BAPO and TPO on human oral keratinocytes and V79 fibroblasts. In line with our results, TPO exhibited concentration-dependent cytotoxic effects in both human oral keratinocytes (OKF6/Tert2) and V79 fibroblasts. However, in the micronucleus (MN) assay, TPO did not induce genotoxicity in either cell type, and no significant increase in DNA damage was observed.
Our results highlight the importance of optimizing scaffold material concentrations to balance efficacy and safety. While higher concentrations of SL6 demonstrate cytotoxicity, lower concentrations appear to be non-toxic, making SL6 a potential candidate for biomedical applications. However, the unexpected toxicity of our new composite underscores the need for further research to understand the underlying mechanisms. Understanding these mechanisms is essential for the further development and refinement of our composite materials, ultimately aiming to create safe and effective scaffolds for biomedical use.
Therefore, we plan to conduct additional studies to investigate the interactions between PEGDA/TPO cellular components and to explore potential modifications to reduce toxicity.
Conclusions
The study demonstrates that SL6 polymer-ceramic composites, offer enhanced mechanical properties. The development of a low-viscosity, was achieved by optimizing the concentration of the slurry adding water as a diluent. This adjustment significantly improved the printability and quality of the polymer-ceramic composites using DLP technology.
The compressive strength of PEGDA-HA composite achieved a maximum of 20 MPa in Z direction. The flexural strength was 56 MPa and 34 MPa for horizontal and vertical orientations.
The cytotoxicity of the composites were evaluated, showing promising results. The MTT and cell visualization techniques confirmed that the materials are biocompatible, with minimal immune response and no significant cytotoxic effects on both MDA-MB-231 and HaCaT cells at 0.001 mg/ml concentrations.
Footnotes
ORCID iDs
Author contributions
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Higher Education and Science Committee Ministry of Education, Science, Culture and Sport RA [grant number №22rl-050, 2022]; Confocal images were obtained using Biomedical Imaging Organization for South East Europe (BIO-SEE) grant from the Chan Zuckerberg Initiative.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
