Abstract
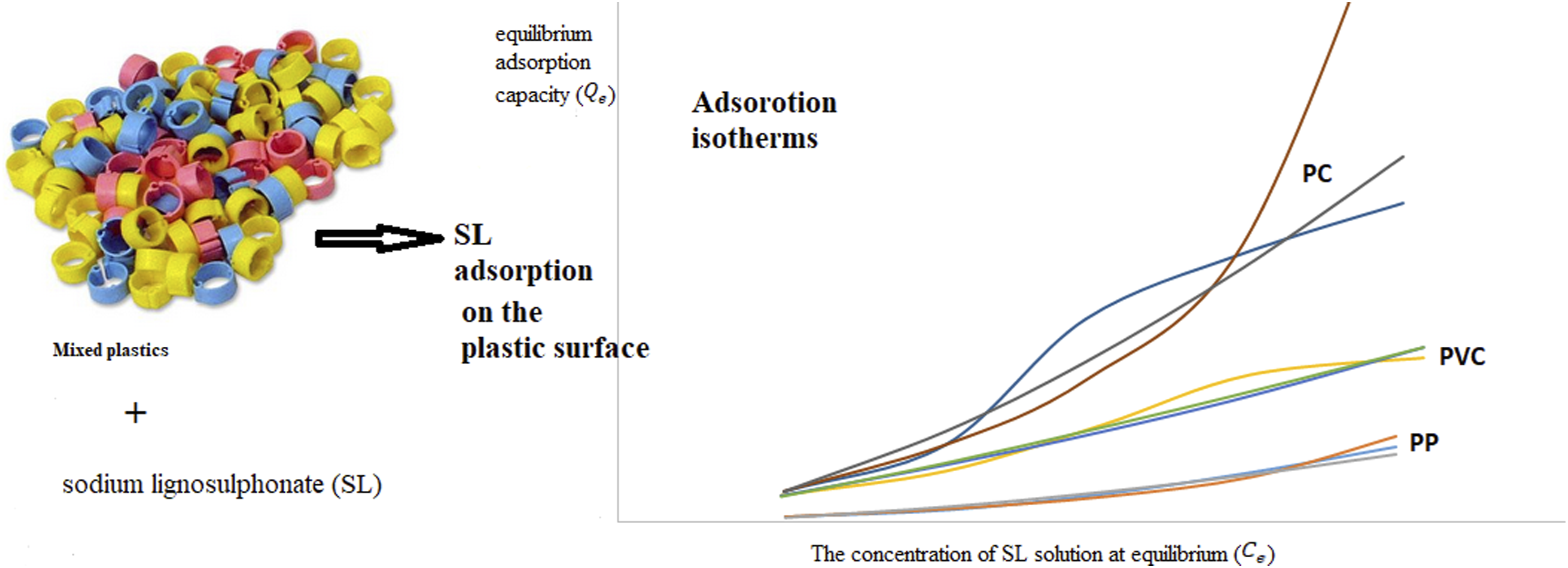
Separating different components of a plastic mixture is crucial in its recycling. Among the different separation techniques, flotation was selected as a cheap, non-toxic, and efficient process. Basis of the technique refers to the selective adsorption of a depressant on the plastics surface which cause the alteration in the surface energy of the plastic. Adsorption of the lignosulfonic acid sodium salt (SL) on the surface of the selected available plastics in the waste stream was studied. Plastics used in this study were Polyvinylchloride (PVC), acrylonitrile-butadiene-styrene polymer (ABS), polystyrene (PS), polypropylene (PP), polyoxymethylene (POM), and polycarbonate PC. It results showed that SL adsorbed on the surface of the selected plastics considerably. It was evidenced by the measured equilibrium adsorption capacities (
Introduction
The annual plastics production reached more than 400 million tones. 1 Consequently, plastic recycling is essential to protect the environment and resources. 2 In most cases, we face a mixture of plastics in the waste stream. For re-using them, they should separate from each other efficiently. Influential separation of an individual plastic from a mixture of waste plastics is a crucial factor for the quality assurance of recycled plastic. Several techniques have been developed for the separation of the plastic(s) from plastics mix, including gravity or density separation,3–5 selective dissolution,6–9 triboelectric separation,10–12 and flotation based separation.13–15 Among techniques mentioned above, the flotation technique is a simple, cheap, and efficient method. In this technique, a chemical adsorbs on the surface of a selected plastic and alters the surface energy of the plastic. The new formed surface energy makes the plastic surface more hydrophobic, and or hydrophilic depending on the chemical structure of the adsorbed agent. Subsequently, the treated plastic introduces into a flotation vessel filled with a liquid media (water). Depending on the new surface energy of the studied plastic, it may float on the top and or sink at the bottom of the flotation vessel.
Numerous chemicals used as a depressant to alter the sink-float behavior of the selected plastics.16,17
The sodium lignosulphonate or lignosulfonic acid sodium salt (SL) are prepared from needle-and broad-leaved trees through the treatment of sodium sulfite. It also can be prepared using pulp wastes. It appears as an odorless brown powder. It is cheap, nontoxic, accessible, and easily soluble in water and alkali materials. It is categorizes as an important renewable natural resource material.18–21 It is a highly branched three-dimensional biopolymer composed of phenylpropane, p-hydroxyphenyl, syringyl, and guaiacyl units. 22 It is used in numerous industries, including building, agriculture, dye, and food industries as an efficient dispersant.
The SL is a surfactant having hydrophilic and hydrophobic groups in chemical structure. The hydrophobic groups enable the SL to adsorb on a plastic surface efficiently. However, because it is a salt, it dissociates into several ions when immersed in water. Considering the points mentioned above, it should be a suitable volunteer as a depressant for plastic flotation in an aqueous liquid media.
The adsorption of a chemical on the polymer surface is a surface phenomenon dedicated to the accumulation of chemicals film on a solid (polymer) surface. The numerous aspects should consider during the adsorption-desorption process, including:23–25 • The availability and the number of suitable active sites on the polymer surface • The mono or multilayer of the adsorbed chemical on the polymer surface • The enthalpy of adsorption • The rate of adsorption and desorption of the chemical on and from the polymer surface • The Gibb’s free energy of the adsorption process • The direct and indirect interaction between the adjacent molecules of the adsorbed chemical
To understand the extent and degree of adsorption, two models, Langmuir and Freundlich isotherms are widely used. 26 The latter is an empirically based model. According to the Langmuir model, adsorption and desorption are reversible processes. Also, the active sites on the solid surface are equivalent and have unit occupancy.
Based on the knowledge of the authors, there is no systematic study on the adsorption of SL on a group of traditional plastics (polymers). In this study, the parameters of the models mentioned above were derived using the equilibrium concentration of the SL on the surface of the selected polymers. The studied polymers were Polyvinylchloride (PVC), acrylonitrile-butadiene-styrene polymer (ABS), polystyrene (PS), polypropylene (PP), polyoxymethylene (POM), and polycarbonate PC. The results were supported by conventional characteristic techniques, including contact angle (
Also, an efficient method based on flotation technique with the SL and water as flotation chemical and liquid media, respectively was introduced for separating PP from a plastics mix.
Experimental
Materials
The specifications of the studied plastics.

The chemical structure of lignosulfonic acid sodium salt.
Equipment, testing procedure, and characteristic techniques
All plastics were washed with de-ionized water and dried in a desiccator at ambient temperature for further use. The Brunauer-Emmett-Teller (N2-BET, Belsorp mini II, Microtrac Bel corp, Japan) adsorption method was used to determine the specific surface area of the used plastics (Table 1). 2 g of a specified plastic was immersed in 10 mL of a known SL concentration glass baker. After passing an hour to reach the equilibrium, the equilibrium SL concentration was measured by a UV-visible spectrophotometer (CECIL 8000 series, UK) at 204 nm using the Beer-Lambert law. 27 The measured equilibrium concentrations were used to determine the parameters of the Langmuir and Freundlich isotherms subsequently. The flotation experiments were performed as described earlier.13–15 For flotation experiments, 20 granules of plastic were introduced into the flotation tank. Subsequently, the number of floated granules was used for calculation of the flotation % (20 minus the number of floated granules divided by 20 and multiply by 100). The flotation tank media (tap water) had a pH of 6.5 at ambient temperature.
For flotation experiments, 20 granules of plastic were introduced into a glass-made flotation tank with dimensions, 20 cm (diameter) by 80 cm (height) and kept for 1 min. The flotation tank equipped with an air-blowing system (13 cm circular air distributor diameter) with 4 L/min as air bubble flow rate. All experiments were repeated three times, and the median values were reported. The contact angle (
Equation (1) introduces a useful adsorption parameter known as the equilibrium adsorption capacity ( 
Results and discussion
Table 1 shows the highest and the lowest special surface area values belonged to ABS and PC with the values of 7.29 and 0.80
The initial and equilibrium SL concentrations (

The Langmuir isotherm is a simple physical adsorption model. The Langmuir theory is bases on a kinetic principle.



The Langmuir’s parameters,

a

An illustration of the fitted line based on the measured experimental data for the POM in the Langmuir’s model.
The Freundlich’s parameters,
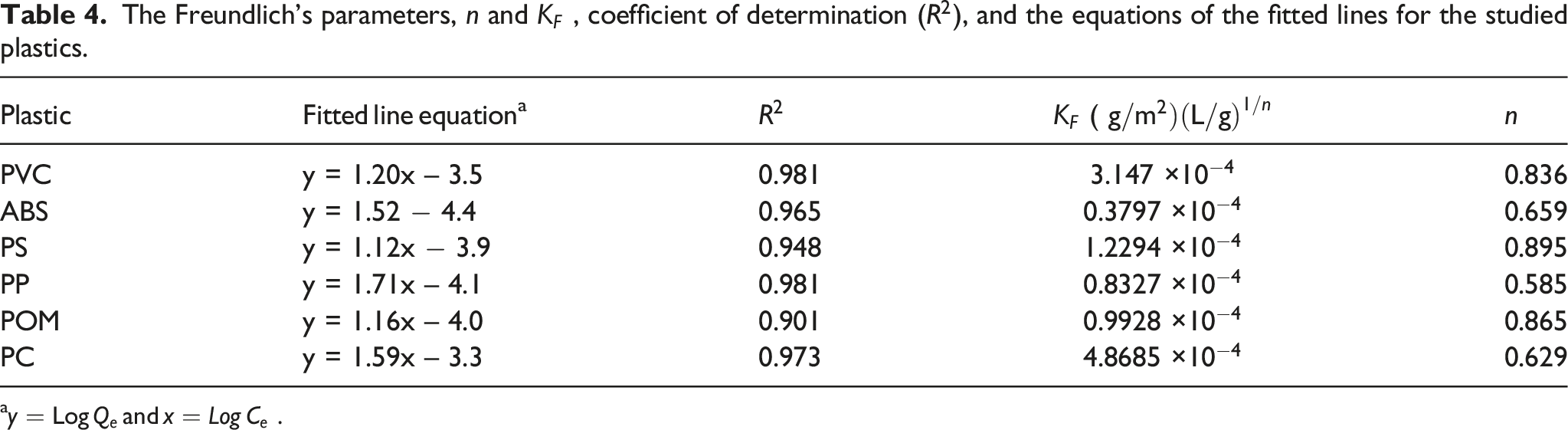
a

The comparison between the experimental and predicted

The SEM images of the PP and PC surfaces. The plastic’s surface un-treated and treated with the maximum SL concentration (

The AFM images of the PP and PC surfaces. The plastic’s surface un-treated and treated with the maximum SL concentration (
The PP, with a contact angle of 122.8°, was more hydrophobic than the PC with the contact angle of 98.1°. It was considering the high hydrophilic property of the SL results in higher SL adsorption on the PC surface. The SEM and AFM images for the treated plastics confirmed the above-mentioned facts.
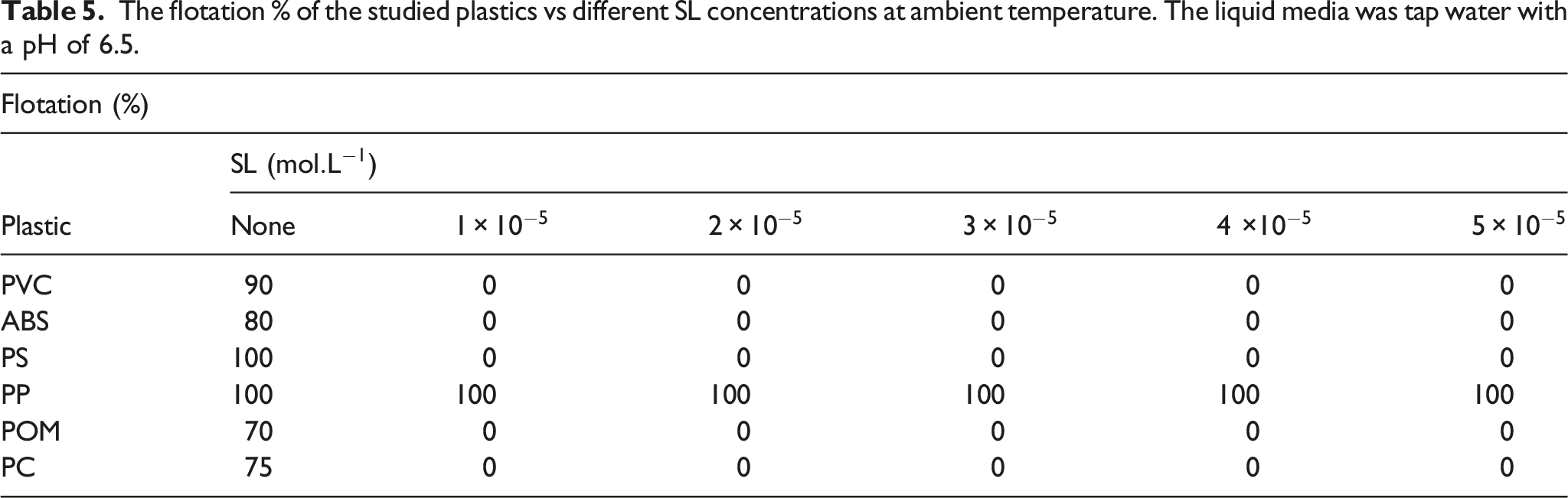
The flotation % of the studied plastics vs different SL concentrations at ambient temperature. The liquid media was tap water with a pH of 6.5.
Concluding remarks
From this study, we can conclude that the SL can be adsorbed on the selected plastics surface considerably. It was evidenced by the measured equilibrium adsorption capacities (
Footnotes
Author contributions
Fateme Shariatikia, Nadia Ostad Movahed and Saeed Ostad Movahed designed the experiments. Dr. Saeed Ostad Movahed prepared the manuscript with contributions from all co-authors. The authors applied the SDC approach for the sequence of authors.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The authors sincerely thank the staffs of the polymer chemistry laboratory located at faculty of science, Ferdowsi University of Mashhad for their sincere cooperation. Approval no. 3/53876.
