Abstract
The separation of one or a group of plastics from a plastics mix by flotation technique is an essential topic in plastic waste management. Basis of the technique refers to the selective adsorption of a depressant on the surface of the plastic. Consequently, the surface energy of plastic alters. Adsorption of the lignosulfonic acid sodium salt (SL) on the surface of the selected available plastics in the waste stream was studied at different SL concentrations and operative temperatures. Studied plastics were Polyvinylchloride (PVC), acrylonitrile-butadiene-styrene polymer (ABS), polystyrene (PS), polypropylene (PP), polyoxymethylene (POM), and polycarbonate PC. Based on the measured equilibrium adsorption capacities (

Introduction
To save natural resources, and avoid environmental impacts, waste plastics should be recycled. 1 In most waste streams, there is a mixture of different types and grades of plastics. As a preliminary step for the plastics recycling, the available plastics in a plastics mix should be separated from each other appropriately. Regardless of manual sorting, there are various developed separation techniques, including electric separation,2–4 selective dissolution,5–8 and density separation.9–12 The recently developed floatation technique13–16 is a safe, economical, and efficient separation technique for separating an individual and or a group of plastics from a plastics mix. The alteration of the surface energy of the plastic is the basis of the mentioned technique. Approaching and or distancing the surface energy of the plastic with the surface tension of the used liquid media, i.e., water, may cause sink and or float the plastic in the floatation tank, respectively.
Generally, the surface energy of the plastic changes by the adsorption of a chemical (depressant) on the surface of the plastics.
The adsorption process depends on several parameters,17–19 including the available active sites on the plastics surface, mono or multilayer of the adsorbed depressant on the polymer surface, the enthalpy of adsorption, the Gibb’s free energy of the adsorption, The indirect and direct interaction between the adjacent molecules of the adsorbed depressant, the solubility of the depressant (for a liquid-solid system), and the pressure (for a gas-solid system) and the temperature of the system.
In general, adsorption is an exothermic process. 20 Therefore, it is expected that with increasing temperature, the adsorption of the depressant on the surface of the plastic decreases. It is true for gas-solid systems. However, in adsorption from solutions, as observed in the floatation technique, temperature affects not only the adsorption process but also may change the solubility of the depressant in the liquid media. Since the solubility of a depressant is a crucial parameter in the adsorption process, the solubility factor should be considered in any investigation of adsorption from solutions.
So far, numerous depressants have been introduced in literature.13–16,21,22 However, among them, sodium lignosulphonate or lignosulfonic acid sodium salt (SL) as a biopolymer (Scheme 1) has distinguished characteristics, i.e., non-toxic, economy, water-soluble, renewable natural resource substance, and accessible in commercial grade in the market.
23
The chemical structure of used lignosulfonic acid sodium salt.
In many industries, namely food, building, dye manufacturing, and agriculture, it is applicable as a strong surfactant. Normally, SL extracts from broad and needle-leaved trees using sodium sulfite and pulp wastes. The guaiacyl, phenylpropane, syringyl, and p-hydroxyphenyl are the primary chemical parts of the highly branched three-dimensional structure of SL. 24 As shown in Scheme 1, SL has hydrophobic and hydrophilic groups in chemical structure. Because it is a salt, it readily dissolves in water. Simultaneously, the hydrophobic groups of the SL facilitate the adsorption of the substance on the surface of the plastic. Considering aspects mentioned above, Sl should be a suitable depressant for the sink-float of the plastics in aqueous liquid media.
To describe the adsorption of a chemical on the surface of a solid, two traditional mathematical models are available, Freundlich and Langmuir isotherms. 25 The Langmuir is a theoretical based model. It has several assumptions, including a reversible process for the adsorption and desorption, and equivalent and unit occupancy for active sites on the solid surface. However, the Freundlich isotherm is an empirical based model.
Based on the knowledge of the authors, there is no systematic study on the adsorption of SL on a group of traditional plastics (polymers) at different temperatures. In this study, the parameters of the models mentioned above were derived using the equilibrium concentration of the SL on the surface of the selected polymers at different temperatures. The studied polymers were acrylonitrile-butadiene-styrene polymer (ABS), Polyvinylchloride (PVC), polypropylene (PP), polystyrene (PS), polyoxymethylene (POM), and polycarbonate PC. Also, the separation of the studied plastics from each other in their mix was studied using the novel floatation technique with the SL and water as floatation chemical and liquid media, respectively.
Experimental
Materials
The specifications of the studied plastics.

Testing procedure, characteristic techniques, and equipment
All plastics were washed with de-ionized water and dried in a desiccator at ambient temperature before further use. The Brunauer-Emmett-Teller (N2-BET, Belsorp mini II, Microtrac Bel corp, Japan) adsorption method was used to determine the specific surface area of the used plastics (Table 1). Two grams of a specified plastic immersed in 10 mL of a known SL concentration (1–4 × 10−5 mol/L) glass baker at selected different temperatures 30, 50, and 70°C. After passing an hour to reach the equilibrium, the equilibrium SL concentration was measured by a UV-visible spectrophotometer (CECIL 8000 series, UK) at 204 nm using the Beer-Lambert law. 26 The measured equilibrium concentrations were used to determine the parameters of the Langmuir and Freundlich isotherms subsequently. The flotation experiments were performed as described earlier.13–16 For flotation experiments, 20 granules of plastic were introduced into the flotation tank. The flotation tank media (tap water) pH was 6.5 at the above-mentioned selected temperatures. The dimensions of the glass-made flotation tank were 20 cm (diameter) by 80 cm (height). The flotation tank equipped with an air-blowing system (13 cm circular air distributor diameter) with 4 L/minute as air bubble flow rate. The residence time of the granules in the flotation tank was kept at 1 min. All experiments were repeated three times, and the median values were reported.
Results and discussion

The initial and equilibrium SL concentrations (
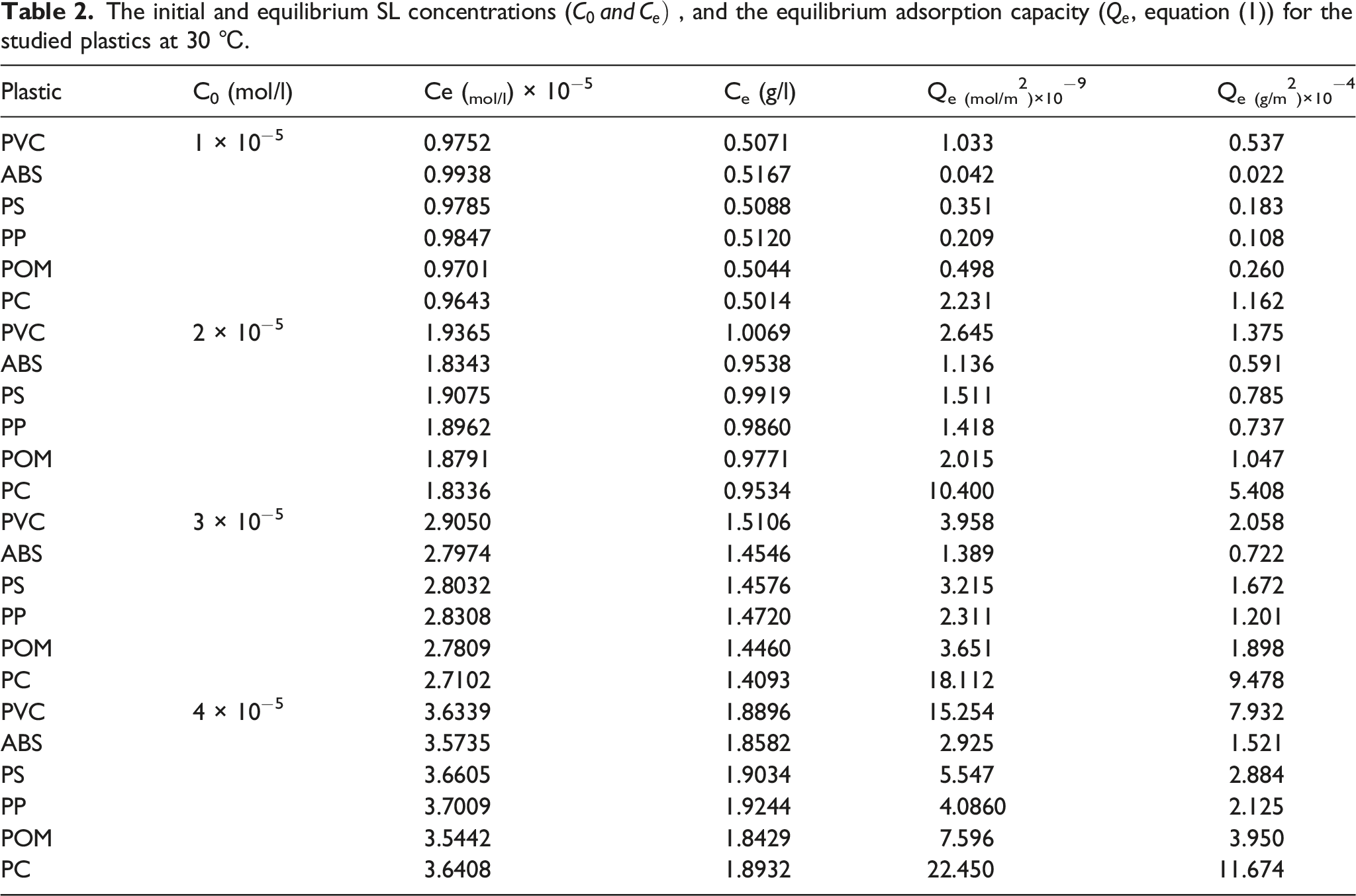
The initial and equilibrium SL concentrations (

The initial and equilibrium SL concentrations (

As an illustration for the effect of SL concentration on the equilibrium adsorption capacity, the
It concluded that, unlike the special surface area of the plastic, the initial concentration of the SL and operation temperature were effective parameters on the adsorption of SL on the surface of the plastic. It was in conformity with the earlier report. 19 The adsorption of a chemical on a polymer surface depends not several parameters, including the special surface area of the polymer and the chemical structure of the involved materials. It seemed, the latter parameter was more effective than the former in studied systems.


The Langmuir’s parameters,

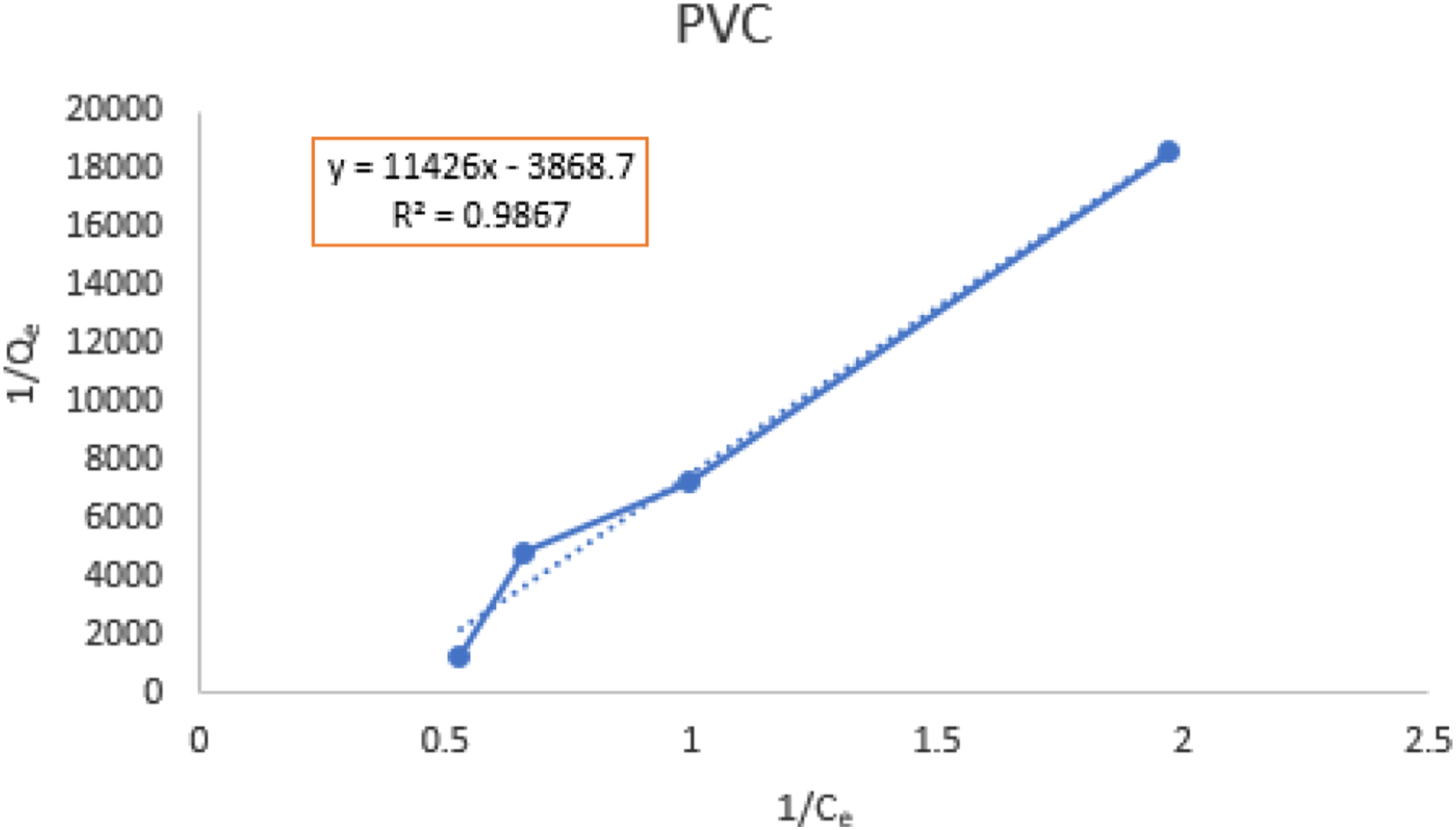
An illustration of the fitted line based on the measured experimental data for the PVC in the Langmuir’s model at 30

An illustration of the fitted line based on the measured experimental data for the PVC in the Langmuir’s model at 50

An illustration of the fitted line based on the measured experimental data for the PVC in the Langmuir’s model at 70
The Freundlich’s parameters,

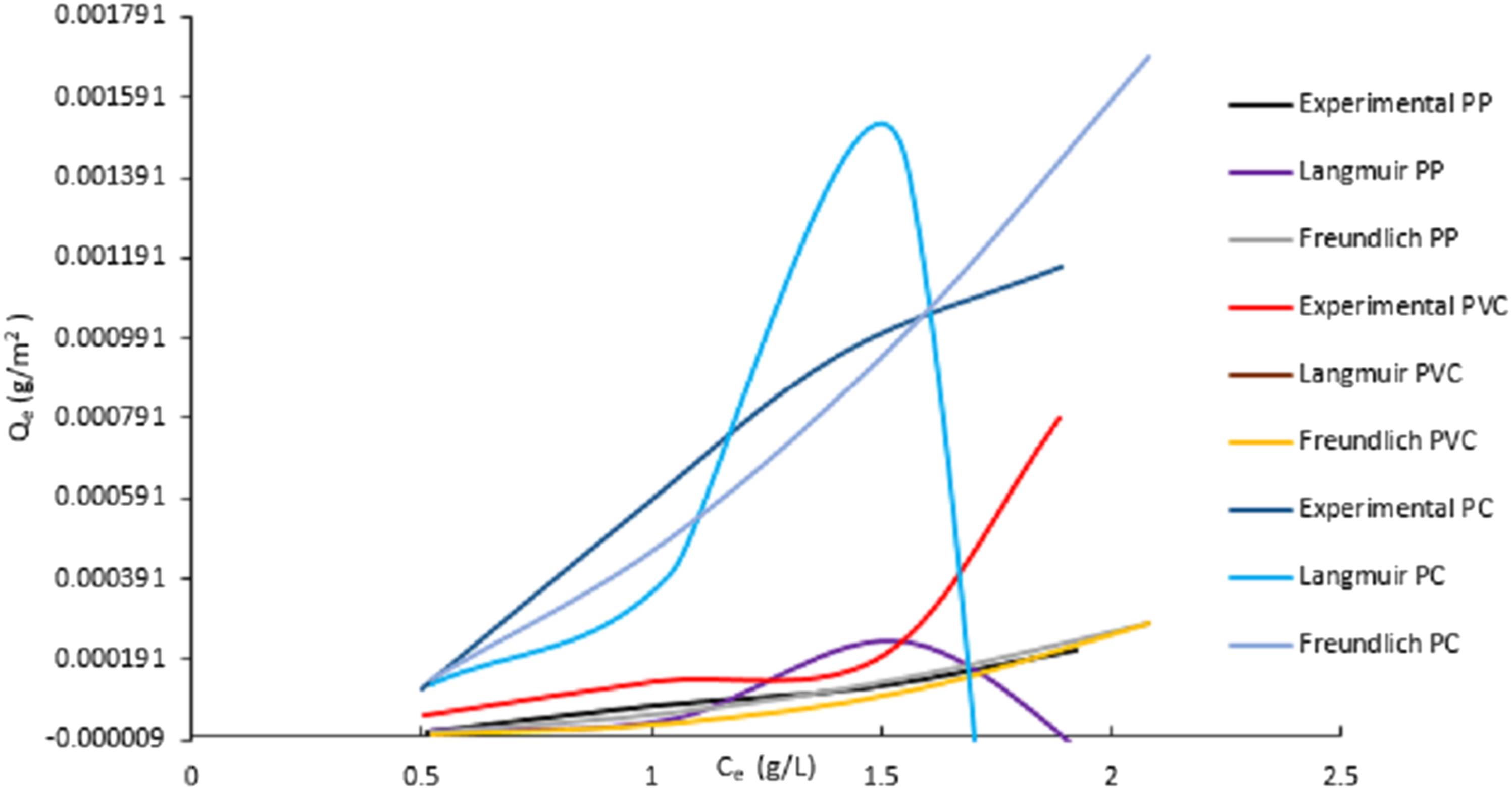
The comparison between the experimental and predicted

The comparison between the experimental and predicted

The comparison between the experimental and predicted
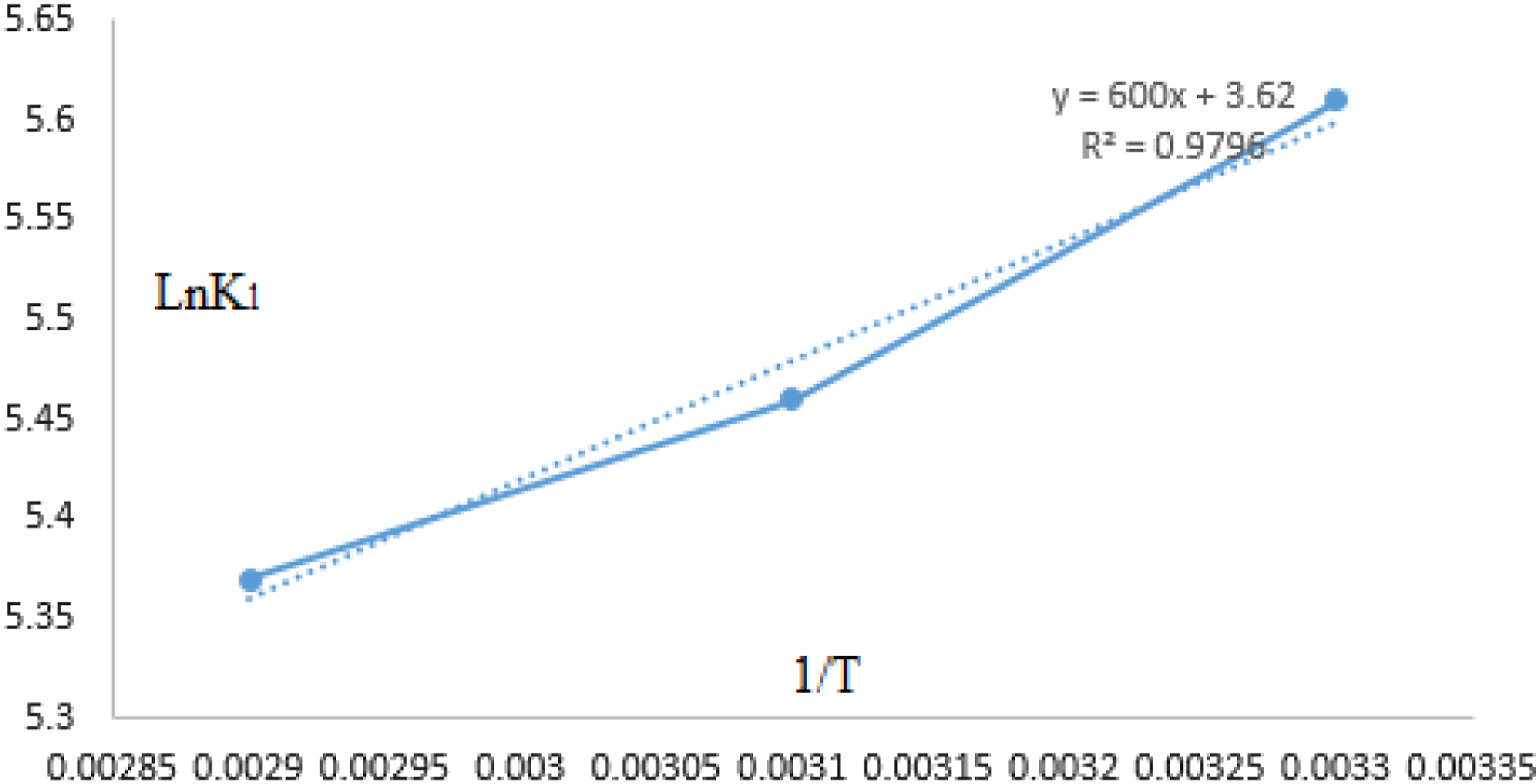
The relationship between the Langmuir isotherm constant ( The The thermodynamic parameters Gibbs free energy (



The flotation % of the studied plastics versus different SL concentrations at studied temperatures 30, 50, and 70

Concluding remarks
Based on the measured equilibrium adsorption capacities (
Suitable conformity was observed between the experimental results with predicted values by the traditional adsorption isotherms, Freundlich and Langmuir for studied temperatures. However, for the most studied plastics, the Freundlich model was more suitable than Langmuir. The measurement of thermodynamic parameters revealed spontaneous adsorption processes along with endothermic (except for PC) reactions. The relatively low thermodynamic values were proper evidence for the physical (not chemical) adsorption of SL on the surface of the selected plastics. The complete separation of PC from a plastics mix in all SL concentrations and temperatures was observed during the flotation process.
Footnotes
Acknowledgements
The authors sincerely thank the staffs of the polymer chemistry laboratory located at faculty of science, Ferdowsi University of Mashhad for their sincere cooperation. Approval no. 3/56351.
Author contributions
Atefeh Salari and Saeed Ostad Movahed designed the experiments. Dr. Saeed Ostad Movahed prepared the manuscript with contributions from all co-authors. The authors applied the SDC approach for the sequence of authors.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Data availability
The datasets generated during and/or analyzed during the current study are available from the corresponding author on reasonable request.

