Abstract
Over 400 million tons of plastic are produced annually. Plastic is nonbiodegradable and represents an abundant environmental challenge. This review focuses on novel polymer modification techniques to reduce waste in the recycling, reuse, and degradation context of a circular economy. The study focuses on the implementation of design-for-deconstruction thinking and an investigation into vitrimers – the dynamic, self-healing materials that can be reprocessed efficiently. Enzymatic, catalytic, and electrochemical methods are diagnosed as advanced approaches for the perfect recycling of waste plastics to high-performance materials using AI-assisted tools. These methods chemically reverse molecular damage to restore mechanical properties lost during recycling. Validation of the methodology is performed using FTIR, DSC, TGA, and SEM. While promising lab results, upscaling to industrial generation is difficult due to high catalyst cost, incompatibility of infrastructure, and insufficient standardized LCA and TEA data. Achieving a sustainable polymer circular economy requires a multifaceted cooperation between researchers, industry, and policymakers to achieve successful integration.
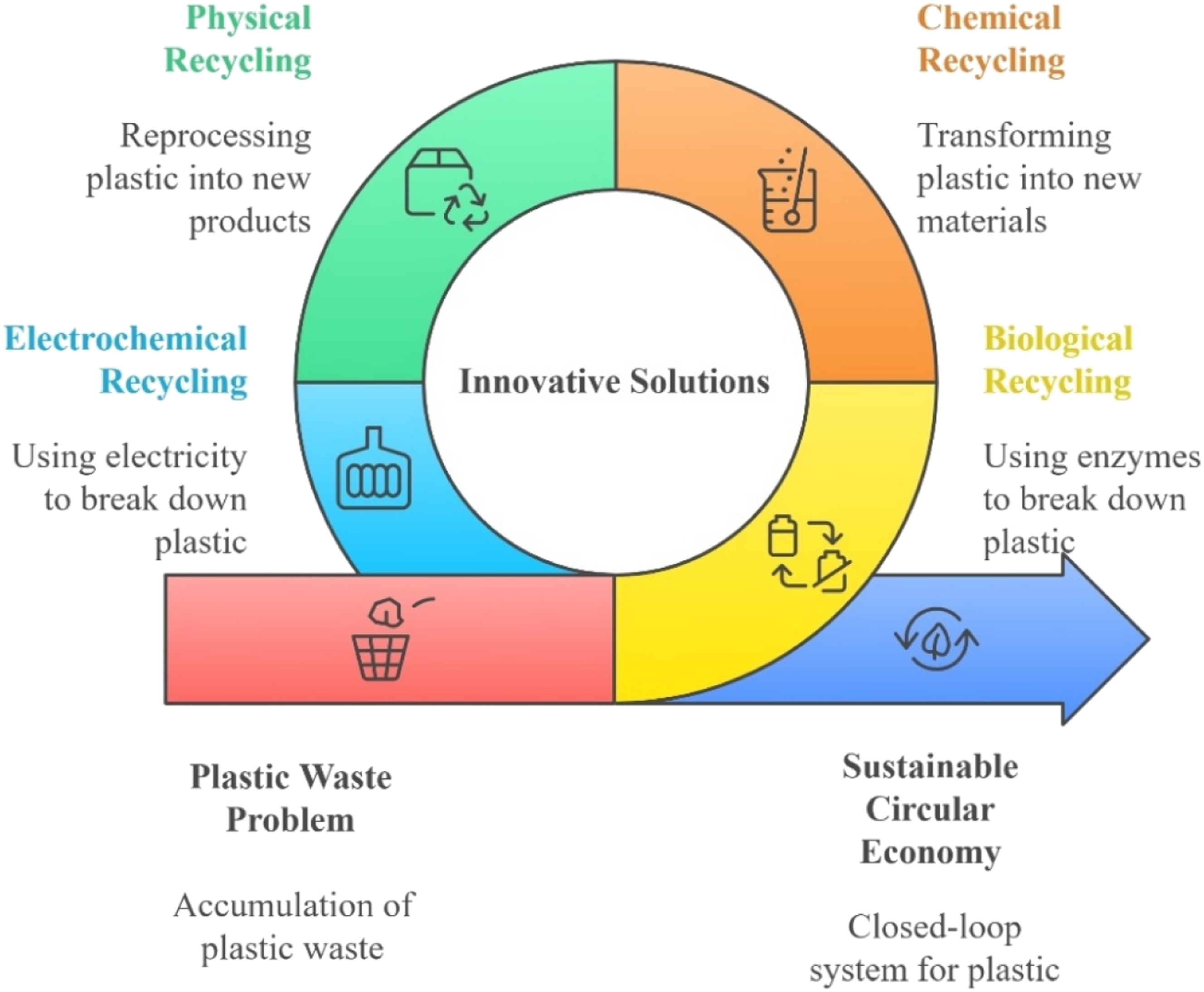
1. Introduction
Polymers are large molecules present throughout nature that play crucial roles in everyday life. The term “plastic” derives from “pliable,” meaning easily shaped or molded. Plastics, or polymers, consist of long chains of repeating units called monomers and can be engineered and formed for diverse applications.1,2 Plastic materials are widely used because of their convenience and broad availability. In almost every field today, people depend on plastics. Science, industry, and technology are all quickly increasing their demand for polymeric materials because they are flexible, long-lasting, and cheap. Applications include packaging, furniture, cosmetics, energy storage, electrical parts, medical devices, tissue engineering, and materials for implants that go into the human body.3–5 In nature, plastics can take hundreds to over a thousand years to break down completely.
By 2022, the world made an amazing 400 million tons of plastic, which meant that 267.68 million tons of plastic were thrown away that year. Even though people are still trying to build recycling systems all over the world, the world’s recycling systems were only able to collect and process 37.96 Mt of plastic, which is still only 9% of total primary production. Most of the rest of the trash was sent to its final destination: 40% went to landfills, and 30.66 Mt (34% of all collected trash) was burned. The rest of the waste, however, was not properly managed, and it ended up in uncontrolled dumpsites or leaked directly into the environment. 6 Using more plastic makes a lot of trash and harms the environment. Plastics are great for packaging because they last a long time and are easy to use, but most of them end up in landfills, where they can’t be recycled very well. Plastics break down slowly into microplastics that get into the body through food, water, and things we use every day. Microplastic pollution from this growing plastic waste threat is a serious threat to both human health and the environment. 7
The climate is getting worse and worse because of the environmental cost of making primary plastics. Making new plastics let out about 2.24 gigatonnes of carbon dioxide equivalent in 2019. This was 5.3% of all greenhouse gas emissions in the world. About 75% of these emissions come from fossil fuel extraction and refining before polymerization, which is very important. If growth is slow (2.5% per year), the greenhouse gas emissions from making primary plastic are expected to more than double, reaching 4.75 GtCO2e by 2050. This output would consume 21-26% of the remaining global carbon budget allocated to keeping average temperature increases below 1.5°C. If the annual growth rate were to accelerate to 4%, emissions would increase even more dramatically, by over three times, reaching 6.78 GtCO2e. In this scenario, primary plastic production would alone account for 25-31% of the remaining carbon budget for limiting global warming to 1.5°C. 8
Microplastics pose a direct threat to biological systems, inflicting fundamental harm that includes oxidative stress, DNA damage, and cellular dysfunction.9,10 Studies show that polystyrene microplastics lead to higher levels of inflammatory proteins like TGF-β and TNF-α in rat lungs, and raise inflammation signals such as TNF-α, IL-1β, and IFN-γ in mice. 9 People are exposed to microplastics by eating, breathing, and even through their skin, with these tiny particles being found in human blood, placenta, and organs.10,11 Every day use of oral care products, like toothpaste and toothbrushes, releases more than a million particles annually per person, 1.183 × 106 from toothpaste and at least ≥2.33 × 106 from toothbrushes. Microplastics trigger a cascade within the body that gives rise to issues such as mitochondrial disruption, chronic inflammation, genetic destruction, and tissues inability to defend themselves. 11 Despite more awareness of the issue, global recycling rates for plastic remain at 9%, and the amount of new plastic produced annually has increased by 8.4%. 6 And because most plastics can’t be naturally recycled, even traditional recycling methods are powerless to prevent them from becoming environmental waste.12,13 With each cycle of mechanical recycling, plastic grows weaker as its polymer chains degrade. This means that the best recycling rate is only about 31%, even when conditions are perfect. 14
Plastics need to be redesigned so that they break down naturally or can be sent to recycling systems to solve the problem of plastic waste. There are many ways to do this, such as chemical recycling, reactive extrusion, adding biodegradable parts, making copolymers, allowing enzyme-based breakdown, mixing incompatible plastics, using reversible bonds in thermosets, and mixing in bio-based materials.15–19
This review examined recent advances in modification techniques to reduce plastic waste and enhance recyclability through three objectives: evaluating emerging techniques like vitrimers and catalytic breakdown for high-quality recycling; assessing technical feasibility and economic viability using life cycle assessment and techno-economic analysis; and identifying barriers to scale these innovations from lab to commercial systems. Unlike previous reviews do not study single strategy recycling methods, this review offers new insights on alternative options together with actual recycling scenarios. This study also connected molecular design with experimental practice at the interface between chemistry, biology, electrochemistry, and materials science. It highlights “design-for-recycling,” linking performance, environmental impact, and economic factors that have historically been considered separately. Then, it brought in real-world complications through contamination and infrastructure compatibility to apply artificial intelligence to recycling optimization and catalyst discovery. Bridging polymer science with the needs of industry, it offers a guide to designing for the circular plastics economy (CPE).
2. Current status of plastic waste & recycling landscape
2.1. Sources and types of plastic waste
Plastic waste comes from all over, but the biggest source is packaging. In Europe, packaging accounts for about 40% of total plastic use, with an average person generating ∼35.9 kg of plastic waste annually. 20 The packaging sector consumes a lot of short-lived plastics lasting less than five years. It has been a major contributing factor to the massive increase in plastic waste. 21 In addition to packaging, much of the plastic waste is derived from synthetic textiles (such as polyester clothing), building products, consumer-ables, automotive parts, and electronic equipment. 3 The bulk of plastic waste from households is composed of PET (polyethylene terephthalate) bottles, HDPE (high‐density polyethylene) containers, LDPE (low‐density polyethylene) films, PP (polypropylene) packaging, and PS (polystyrene) trays. Approximately 40% of plastic waste is produced from various industries, such as manufacturing, processing, packaging, automobile, electric, and construction. This was referred to as commercial plastic waste.21–23 The rapid development of online shopping has caused a great amount of plastic waste, in particular the packaging waste, which is composed of HDPE and LDPE materials (64.8%), mainly protective film, pouches, and containers. 21
2.2. Processing routes: The waste hierarchy
The hierarchy of waste management establishes a framework that prioritizes at the top, waste prevention and, in descending order, reuse, recycling, energy recovery, and finally disposal (the least preferable option).
24
In 2022, the world created 436.66 million tonnes of plastic, from which it generated 267.68 million tonnes of waste. 178 Mt of that, however, were actually sorted and collected for mechanical recycling, while the other 6.66 Mt were traded. Figure 1 shows that managing plastic waste around the world is very inefficient. Landfilling made up 40% of the total, incineration made up 34%, mismanaged waste made up 17%, and recycling made up only 9% of 267.68 million tonnes. This distribution shows that there isn’t enough recycling infrastructure in place and that we need to improve waste management policies and move to a circular economy as soon as possible.
6
Global plastic waste management 2022.
6

Compared to landfilling and incineration, recycling has the least potential to cause global warming and uses the least amount of energy overall. This makes it the best choice for the environment. Incinerating waste to make energy has become a more common way to get rid of trash, especially in places where landfills are full. This method, on the other hand, does produce greenhouse gases and requires careful handling of the ash that is left over.24,25
2.3. Policies: Brief EU/US/Asia comparison
The EU has placed the world’s most ambitious regulations on how to manage plastic waste. The Single-Use Plastics Directive (2019) banned certain items, such as plates, cutlery, and straws, and required Extended Producer Responsibility (EPR) schemes. 26 The Packaging and Packaging Waste Regulation (PPWR) says that by 2030, all packaging materials must be designed to be reused or recycled in a way that makes sense from a business point of view. By 2035, there will be strict requirements for recyclable design and high targets for recyclability. There are now rules about how much recycled material must be in products. By 2025, 25% of PET beverage bottles must be made of recycled materials, and by 2030, 30% of all plastic bottles must be made of recycled materials.20,26 The European Union wants to collect 77% of plastic bottles by 2025 and 90% by 2029. At the same time, they want to cut the amount of packaging waste in half by 2040. 6
The United States lacks comprehensive federal legislation on plastic waste; instead, regulations are primarily enforced at the state and local government levels. The recycling rate in the United States has dropped a lot, from 9% in 2015 to only 5% in 2022. 76% of plastic waste (30.47 million tonnes) is still thrown away in landfills. 6 There are no federal laws in the United States that cover plastic waste, so states and local governments make the rules. In 2021, Maine was the first state to pass an Extended Producer Responsibility (EPR) bill for plastic packaging. Oregon, Colorado, and California followed. The California Plastic Pollution Prevention and Packaging Producer Responsibility Act of 2022 says that all plastics and single-use packaging must be able to be recycled or composted by 2032. New York, Connecticut, Illinois, Maryland, and Minnesota are some of the other states that have introduced similar EPR bills, but they are not all being put into effect. 27 The United States has the highest per capita plastic consumption in the world, at 216 kilograms per person per year. 6
Asian countries have different policy approaches that show how far along their economies are in terms of development. China used to be the world’s biggest importer of plastic waste. In 2018, the country banned plastic waste imports and then put in place strict Extended Producer Responsibility (EPR) rules and a five-year action plan to cut down on plastic pollution.6,28 The Tokyo-based Plastic Waste Management Institute says that Japan recycled 87% of its plastic in 2021. But 62% of this was through thermal recycling, and only 25% through material or chemical recycling. 29 India generates some 9.3 million tons of plastic waste annually, 20 percent of the global total. Recycling rates for individual polymer types differ significantly. 30 Every year, Bangladesh produces about 87,000 tons of single-use plastic waste — and 96 percent of that is thrown away immediately. The government enacted a ban on single-use plastics, which will take effect on 1st October 2024. 31
3. Literature review
Plastic pollution is a big problem for the environment. It can be found in all kinds of places in nature, from the deep sea and mountains to rainwater and human tissue. Researchers also found plastic particles in the placenta, the blood, drinking water, and marine life, among other things. We don’t fully understand the effects of plastics, especially microplastics, on the environment and our health, but they are very important. Many studies and groups, like UNEP, have shown that plastic pollution is a major threat to global sustainable development. UNEP says that every year, the world makes more than 400 million metric tons of plastic waste, which quickly builds up in the environment.32,33 While technology has made recycling plastic easier, the reality that plastics originate from finite sources in nature means we currently have a resource depletion problem that we must solve. So, beyond technical fixes, we need a broader approach that focuses on environmental, economic, and social strategies to ensure recycling is done in an environmentally sound way. For plastic solid waste management, it has to be environmentally friendly, acceptable to society, and good economics while keeping with the law. Moreover, this has led to an increased public concern in the efforts that are involved in recovering and recycling PSW as well as searching for environmentally preferable processes.34,35
Recycling reduces trash by using things a second time and cuts down on the costs to process materials. It’s going to require not just responsible consumer behavior, but production that is recycling-friendly. PET, HDPE, PP, and PS are among the most frequently used plastics in homes. When they are separated properly, they can be easily recycled. Machine learning portable gadgets with AI, cameras, and LEDs may help people separate their rubbish at home or at the recycling centre line. 23
There are two types of plastics -thermoplastics and thermosets. Knowing the difference between them is important for materials science, but also to make smart choices about how to manage and recycle plastic waste. In the plastic world, thermoplastics are like dough you can roll and use over and over. You can manipulate their shapes when you heat them. You can repeat this process again and again without altering the Material’s shape. Materials such as PET (bottles for water) and HDPE (shampoo bottles) can be made comparably. They’re much easier to recycle because thermoplastics melt when they get hot. This is particularly so when such ores are shredded and rerun. An increasing number of customers are using chemical recycling, which breaks plastic down into components of their original building blocks. This is particularly the case with materials, for example, plastics that are contaminated or degraded beyond mechanical recycling. 36 As thermosetting polymers cure, they cross-link, and once they do, it’s an irreversible process. This makes them stiff and heat-resistant. That stability is great for things that require mechanical strength and thermal resistance, like epoxy resins and electrical insulators, but it also makes recycling a lot harder. Because there are no established pathways to recycle thermosets, they typically go to a landfill or are incinerated. This gives environmental and health-related concerns. 37
Researchers are already developing “smart” thermosets that contain dynamic covalent bonds, and that can be reprocessed or broken down; this is a way to circumvent their usual drawbacks. For example, epoxy resins that are disulfide-based can be programmed to reform and melt when triggered, providing strength but also recyclability. Another new idea that’s full of possibility: “vitrimers,” materials that retain the structural virtues of thermosets while gaining reversible chemical bonding. This allows the materials to be formed and set at high temperatures. 38
Dreiling et al. (2025) developed a simple one-pot synthesis of thermoset polymers from a bio-derived monomer (DHF) with adjustable high-performance properties, which is environmentally degradable and recyclable, addressing the long-standing issue of sustainable and recyclable thermoset. 39
There are a couple of big obstacles to recycling plastics that have made it less efficient and effective:
Achilias, D. S. (2025) looks at how plastics often have things like food, glue, dyes, and other polymers on them that make recycled materials less useful. It costs a lot to filter and wash things for mechanical recycling to get rid of these contaminants, but the end product is still not good enough because there are still pollutants left over (which can even include microplastics in wastewater). 40
Fiorillo et al. (2024) studied that a major drawback of the mechanical recycling of plastics is degradation over time. Melting the plastic, then working with it in this way subjects its long molecular chains to both heat and stress — breaking them down, essentially, as when a rubber band loses its stretch after enough use. This makes it more brittle and lower-quality, so that it can usually not be used for the same thing one more time. Instead, it is often downcycled. For instance, you can recycle a PET bottle into garment fibers or carpet, but not another bottle. 41
Zhou et al. (2023), Plastics do not simply lose strength as they degrade; they melt and become more brittle differently. What this does is make them difficult to recycle, and results in lower-quality end products. 42
Polymerupdate Academy (2024) said that Multilayer plastics, such as those that package food and small sachets, are usually a mix of materials that can include PE, PET, and even thin layers of aluminum. This mix helps packaging work better, but complicates recycling enormously. The layers are difficult to tease apart because they have different melting points and chemical properties. Adhesives and barrier coatings make it still harder. Most multilayer plastics are discarded rather than recycled because of these technical and economic challenges. 43
Lahl et al. (2024) explored the multi-ingredient makeup of plastics. They typically contain a variety of additives, including plasticizers, flame retardants, stabilizers, colorants, and fillers. These are chemicals that may make things look better, function better, but that also render the recycling process much more difficult. Some additives can make recycling more difficult, alter the way recycled plastic behaves, or leave behind harmful chemicals. This is particularly troubling when it comes to older plastics with “legacy” additives, some of which may no longer be safe, especially in products that come into contact with food or skin. 44
There are all kinds of ways that researchers can alter things to make them more biodegradable or recyclable:
Graft, copolymerization, and functionalization are all chemical modifications frequently employed for the modification of polymer properties. A widely used example is that maleic anhydride has been grafted to polypropylene (PP-g-MA) to co-polymerize with polar polymers such as polyethylene terephthalate (PET). This has the advantage of having a higher mechanical properties in recycled blends.45–47
Bulatović et al. (2021) for biodegradable, as opposed to normal non-degrading, plastics. That will help to reduce the ever-growing pile of trash that does not disappear. Starch is in fine example of thermoplastic starch (TPS) as a biodegradable polymer with durability. One promising way to create TPS into materials that are as cheap, renewable, and completely biodegradable is to mix them with other biodegradable polyesters, particularly polylactic acid (PLA) and polycaprolactone. Individually, these polymers (PLA, PCL, and TPS) all have their own drawbacks, but often you can mix them just right to overcome each resistive property. Multicomponent Polymer Materials You can produce multicomponent polymer materials that meet the requirements of a functional performance and are totally biodegradable, even by single and economic processing measures. 48
Palacios-Mateo et al. (2025) demonstrated a novel method to determine if the hydrolase enzymes could be deployed for environmental cleanup in this study. On PET microfibers, they tested cutinize LCC^ICCG in three different settings: Buffered water, sewage sludge, and green compost. We employed typical temperatures for these procedures (55–70°C). We watched biodegradation by monitoring monomer release. We investigated several conditions, including various amounts of the enzyme (0–300 µg), substrate volume (0–200 mg/cm3), and mixing method (shaking/rotating). The enzyme was able to disintegrate as much as 16.6 mg of PET per cm3 water and 1.8mg/g dry weight sludge after 24 hours. Only in compost could it be degraded to at most 13.9 mg/cm3, presumably because this material was drier. Rotational mixing also reduced compost disintegration by about 35% relative to shaking. The findings are discussed within the context of current challenges, and recommendations for where interventions could be most successful in reducing microplastic pollution are also addressed. 49
Other recent work has focused on the catalytic and solvent-assisted depolymerization of (mainly) polyester plastics, in particular PET, to obtain their re-usable monomers for use in a circular economy. Zhao et al. (2024) surveyed several catalytic systems, from thermal through photochemical to electrochemical processes, highlighting the effect of catalyst selection (metal oxides and ruthenium complexes, for example) and reaction conditions on performance and selectivity. They emphasized that dynamic catalysts and mild conditions should also be useful for high-yield depolymerization, lacking in toxic side products. 50
Similarly, Weng et al. (2024) described larger advances in catalytic depolymerization of various biotic and nonionic. They also consider the primary issues driving the limitations of different processes and take a big-picture view of some of the major obstacles to achieving closed-loop recycling and upcycling. Communications like this can potentially further the development of more sophisticated means to address the problems associated with mounting polyester plastic waste and support its efficient recycling into useful chemicals and goods. 51
Notable among these is the chemical depolymerization of PET. Zhao et al. (2024) discussed a nifty way to upcycle PET: through bimetallic catalysts that convert the PET back into useful monomers at low temperatures. This contributes to the goals of the circular economy, in that it is bringing high-purity terephthalic acid (TPA) and ethylene glycol (EG) back for use in polymer production. 50 Olazabal et al. (2024) also have demonstrated that metal-free catalysts (TBD and potassium tert-butoxide) are highly effective for the glycolysis of PET at low temperature 100°C, with more than 90% yields of BHET in less than 15 min as an environmentally friendly approach to recycle. 52
Smart polymers being developed by scientists today are designed to be more easily deconstructed and recycled thanks to dynamic covalent bonds, including transesterification, imine, and disulfide bonds. Such special bonds can break and reform in response to heat or other environmental changes, allowing the polymers to possess practical abilities like self-healing, reshaping, and reprocessing, yet without losing any of their strength or structure.
Researchers are now deploying powerful covalent bonds — dynamic imine, transesterification, and disulfide bonding, among others, to cook up polymers that can smartly recycle or degrade themselves. These special bonds can break, then come back together when the temperature or something else in the environment changes. This allows the polymers to heal themselves, reshape, and be recycled, while remaining strong and maintaining their initial shapes. Vitrimers act like a solid thermoset at room temperature, but can flow and change shape when they are heated. That’s a case that should be looked at.
Recently, Wu et al. (2024) synthesized a biobased vitrimer based on citric acid and epoxidized cardanol glycidyl ether (ECGE). The ester and hydroxyl bonds constantly broke and reformed, so the material was extremely good at healing itself and remembering its shape. Even better: It could be repaired and reformed without adding further chemicals or catalysts, yet would continue to perform well mechanically. Smart material design of this sort raises a lot of potential for making plastics easier to recycle and enabling more environmentally friendly materials.53,54
Shirazimoghaddam et al. (2023) attempted to resolve the environmental issues of increasing plastic production by developing a green PET recycling system. PET has been depolymerized to monomers, mainly bis(2-hydroxyethyl) terephthalate (BHET), by glycolysis with ethylene glycol and sulfated niobia catalysts. Under mild reaction conditions, this gives an 85% yield and 100 conversion. With the help of an affordable and non-toxic, water-based catalyst, this process efficiently converts plastic waste into usable monomers. This aids in keeping the plastics economy circular and more sustainable. 55
Kordjazi et al. (2022) investigated PP/PET blends compatible with PP-g-MA, PP-g-GMA, and EPDM rubber. The morphology and rheological properties of the blends were also improved by the addition: 5 phr PP-g-MA had the best PET dispersion in the LDPE matrix, while it showed a higher storage modulus at lower frequencies. These are both important evidence that compatibilization was successful. 56
Future research should concentrate on scale-up materials with relevance to existing systems and apply life-cycle analysis tools to enable the circular plastic economy. Increasingly, research is directed toward new ways of modifying polymers so that they generate less plastic waste and are easier to recycle. People are putting chemicals like grafts and couplings through their paces to make various polymers play nice with each other, and break down properties faster. Physical methods, like blending plastics with biodegradable polymers, are helping mechanical recycling work better. Enzymatic and catalytic approaches hold great potential for breaking down plastics into molecular components, but many of these techniques are difficult to use on a scale large enough to make a dent in the growing volume of plastic. Meanwhile, the advent of so-called smart polymers, such as vitrimers, which have bonds that can be broken and reformed, is enabling the development of materials that can be reused, or even dismantled when their useful life comes to an end, contributing towards a circular use. While these enhancements look very exciting, there is still much work to do. Most of the new materials have not been tested outside the laboratory, and we still don’t know how much they cost, whether they can be used in industry, or whether they work with existing recycling systems. It’s also difficult to gauge whether these new ideas are truly sustainable in the real world because there is not enough life cycle and economic analysis. This review demonstrates the necessity to ensure compatibility between green design and effective recycling. It gives the goal of this paper, a strong base: to develop functional polymer modification to allow recycling of plastics, and not just help recycle them, but also help nature.
4. Methodology
This study is about finding ways to make plastics less harmful to the environment while still being useful. We’ll look into methods like catalytic solvolysis and making polymers with bonds that are easy to break so that we can get monomers back more quickly.57,58 We’re also looking to nature for ideas. For example, we’re embedding recycled polymer bits into a softer matrix to make materials more stable and even. 59 We’ll put the samples through repeated melting and extrusion cycles to test mechanical recycling in a practical way. 60 This will be done by adding compatibilizers and biodegradable fillers. Then, tested by tensile, DSC/TGA, and FTIR/SEM to see how strong, how they react to heat, and how they are built. Finally, some careful statistics will show us exactly how much these changes make things easier to recycle. It’s all about making plastics smarter without making things too complicated.
4.1. Materials and equipment
4.1.1. Raw materials
(a) Base Polymers: polyethylene (PE), polypropylene (PP), polyethylene terephthalate (PET), polystyrene (PS), polyolefins, etc., commonly used polymers. (b). Modifying Agents: Compatibilizers, chain extenders, crosslinkers, natural fillers (cellulose, starch, lignin). (c) Solvents and catalysts are used where needed.
4.1.2. Equipment
(a) Extruder/Twin-screw compounding machine, Injection molding machine, (b)Mechanical testing: Tensile and impact testers, (c) Thermal and structural analysis: FTIR, DSC, TGA, SEM, (d) Recycling simulation setup
4.2. Polymer modification strategies
Many modification techniques are available, including chemical modification, physical modification, surface modification, enzymatic modification, and catalytic modification processes. Here, several newly developed modification strategies and waste-management techniques are described:
4.2.1. Reactive extrusion
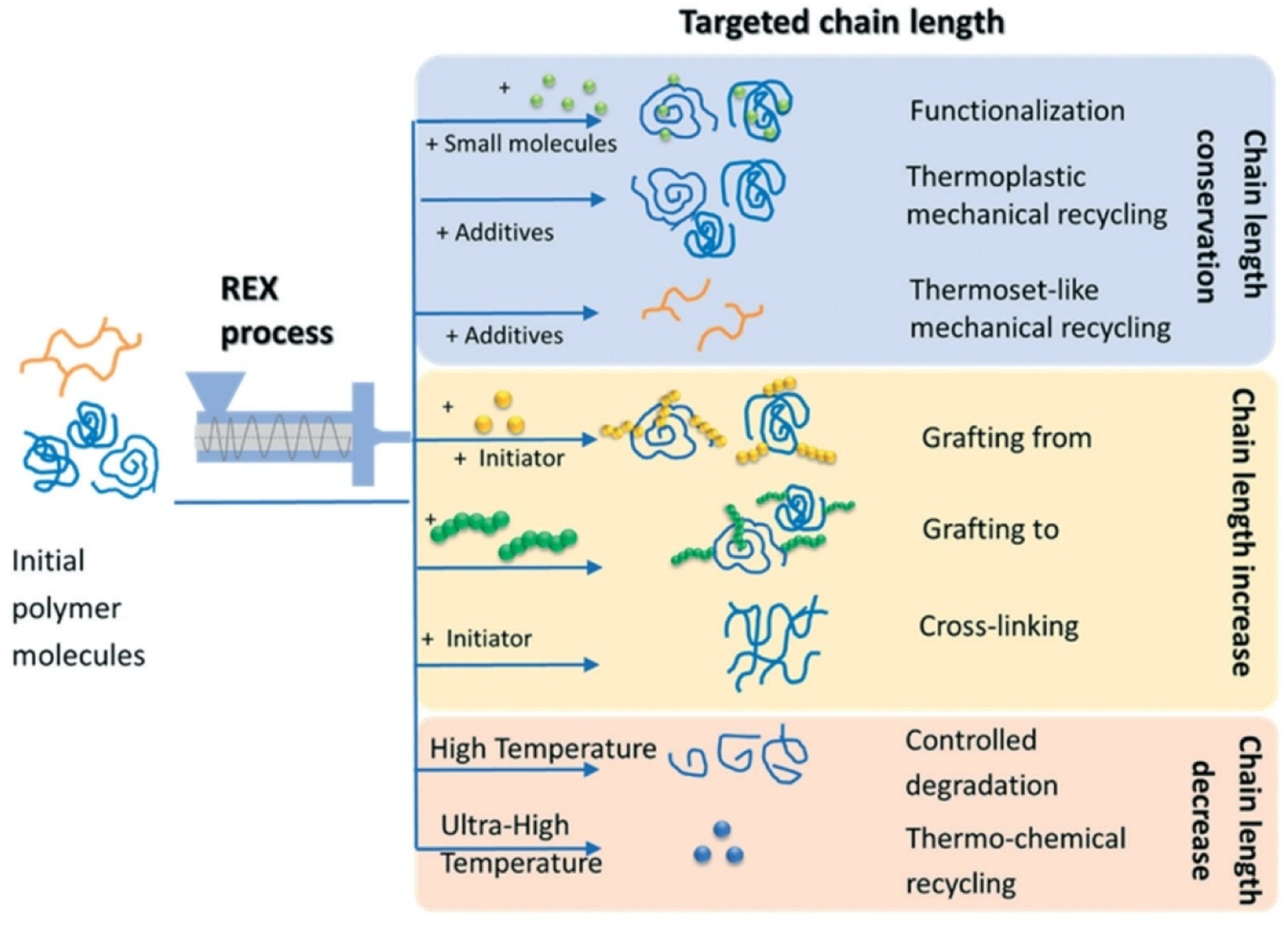
Reactive extrusion (REX) is a solvent-free, continuous process that combines extrusion and chemical reactions, making it highly effective for polymer waste modification and recycling. Using mainly co-rotating twin-screw extruders, REX provides strong mixing, heat transfer, and short residence times to enable rapid transformations such as depolymerization, grafting, crosslinking, and compatibilization. Free radical initiators and grafting agents (e.g., maleic anhydride) improve chain reactivity and blend compatibility, while chain extension restores degraded plastics. By integrating reaction and shaping in one step, REX lowers costs, avoids solvents, and produces higher-value recyclable materials.
18
Figure 2 shows that simplified reactive extrusion (REX) processes and their effects on average chain length, showing only cases starting from polymer molecules; additives are introduced along the extrusion.
61
Reactive extrusion (REX) processes and their effects.
61
Let’s describe the method with an example of Highspeed Reactive Extrusion of Low-Density Polyethylene. Here, Low-density polyethylene (LDPE, Agility™) with a melt flow index of 1.9 g/10 min was processed using an ultrahigh-speed co-rotating twin-screw extruder (KZW 15TW, 15 mm screw, L/D 60:1). The polymer was introduced at a feed rate of 9 g/min, with extrusion carried out at screw speeds of 100 and 2000 rpm. Before extrusion, LDPE was blended with Y-Zeolite and MCM-41 at loadings of 1, 2, and 5 wt%. The resulting composites were analyzed through thermogravimetric analysis (TGA) to assess thermal stability, differential scanning calorimetry (DSC) to examine melting and crystallinity, and oscillatory shear rheology to determine viscoelastic properties. The findings revealed that filler content and screw speed had a huge impact on how LDPE degraded, how much it crystallized, and how it flowed. 62
4.2.2. Solvent-based recycling
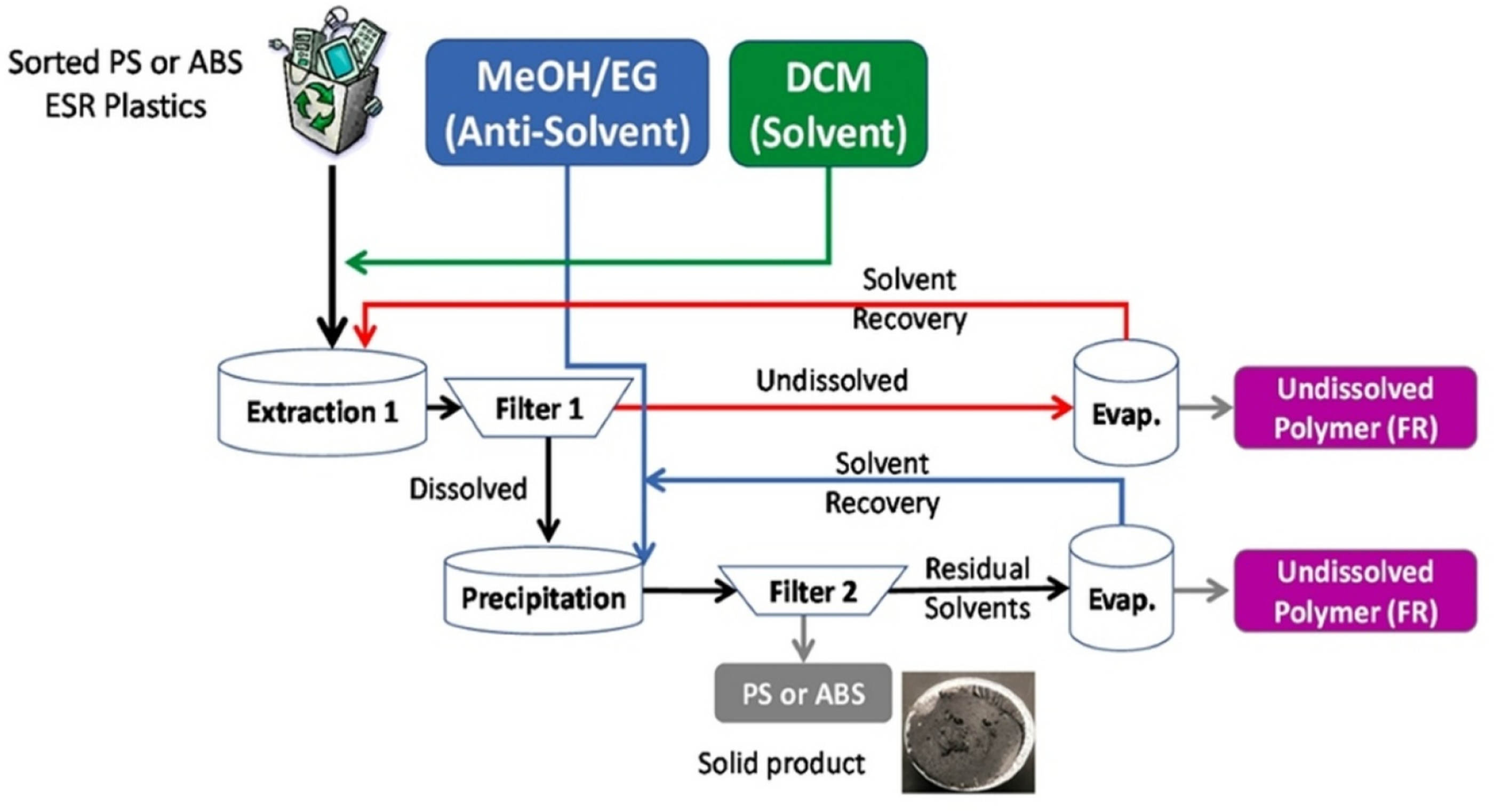
The solvent-based recycling approach physically dissolves and reprecipitates polymers. In other words, some solvents are employed for dissolving the polymers that should be recycled or reused, and not dissolving certain impurities and/or nonpolymeric fractions. In the present study, a manual selective hand cleaning and classification of plastics from electronic shredder residue (ESR) was performed to remove metals and other non-polymeric fractions. And then some of the solvents were used to apply to polymers, such as ABS and PS. These solvents were selected according to Hansen Solubility Parameters (HSP): they are based on the polymer dispersive forces, polarity, and hydrogen bonding with the solvent. Solvents, including dichloromethane (DCM), tetrahydrofuran (THF), toluene, and acetone, were used to see if they could dissolve the plastics. They then employed anti-solvents such as methanol and ethylene glycol to remove the dissolved polymers from the solution. Figure 3 explained the Dissolution, Precipitation process for polymer recovery. Dissolving and then precipitating polymers from mixed plastic waste has proven to be a very good process. It also eliminates additives such as flame retardants migrating into the antisolvent. Once they were recovered, the polymers were filtered, dried, and investigated in order to ensure that they appeared pure and free from any residual solvents. This would be a selective and efficient method to regain high-quality polymers from complex waste streams.
63
Dissolution precipitation process for the recovery of polymer.
63
4.2.3. Catalytic upcycling
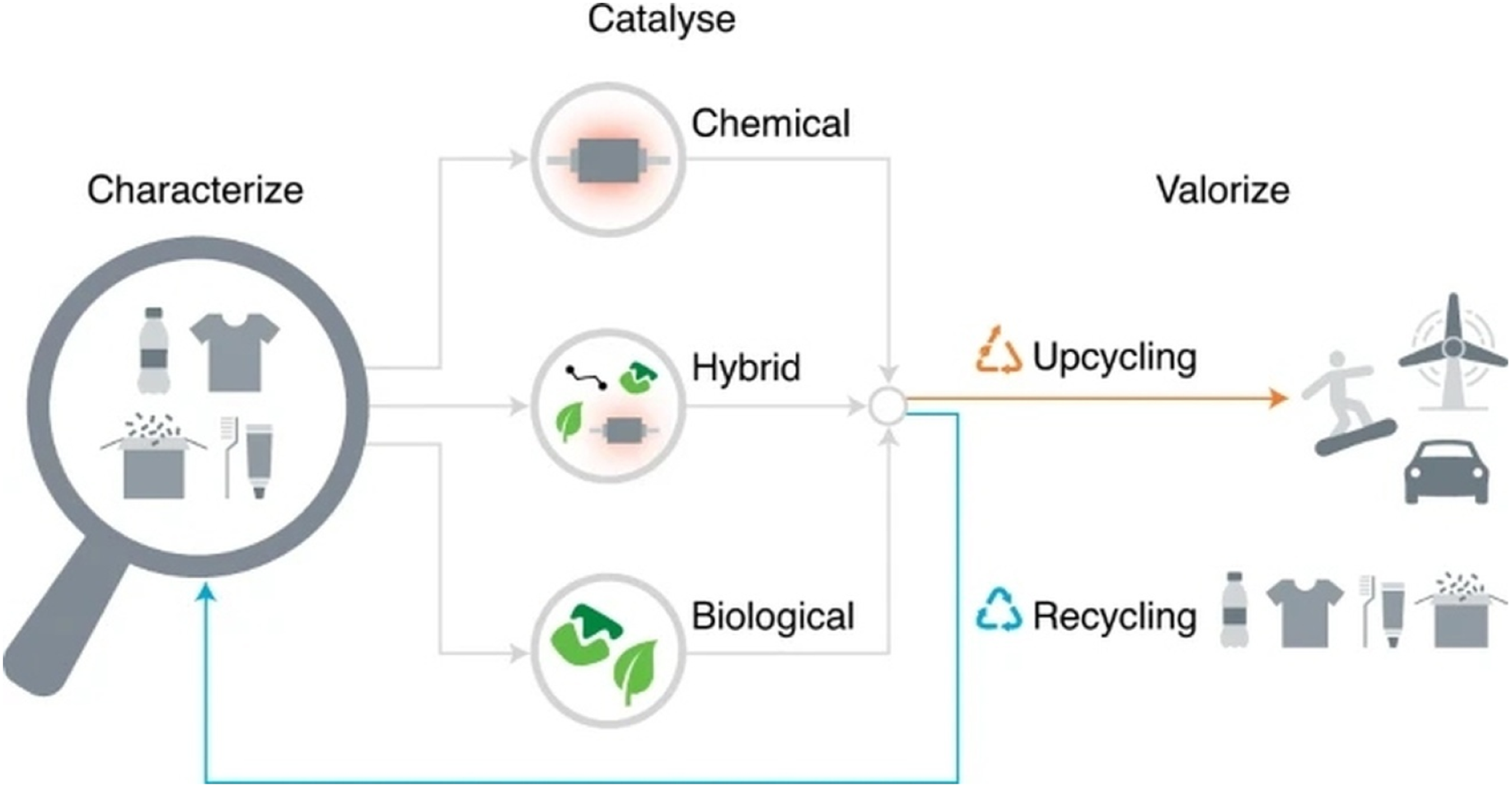
Catalytic upcycling is a promising way to cut down on plastic waste and make it easier to recycle by turning some polymers into products that are worth more or can be used again. Traditional thermal degradation often makes low-value mixtures, but catalytic methods use chemical or biological catalysts to control how polymers break down. Catalytic upcycling uses catalysts (metal, zeolite, enzyme, or hybrid systems) to break down big, inactive polymer chains (like polyethylene, polypropylene, PET, and polystyrene) into smaller molecules that can be used. Metal-catalyzed hydrogenolysis (Ru, Pt, Ni) breaks C–C bonds to make shorter alkanes, and zeolite-based hydrocracking makes aromatic compounds. Oxidative functionalization with transition metals introduces oxygen groups into polyolefins, making them more reactive, and olefin metathesis rearranges unsaturated fragments into useful monomers. Enzymatic approaches, such as PETase-driven hydrolysis, recover pure monomers like terephthalic acid from PET. These processes involve polymer chains sticking to catalytic sites, activating and breaking bonds through intermediates (carbocations, radicals, or carbenes), and making products that can be reused or that add value. Overall, catalytic upcycling provides a pathway to transform persistent plastic waste into useful materials, supporting a circular plastics economy. Here, Figure 4 shows upcycling and recycling of plastic material by catalyzing.
64
Upcycling and recycling of plastic material by catalyzing.
64
4.2.4. Chain extension & functionalization
Chain extension and functionalization are widely used strategies to make plastic recycling more effective and sustainable. In chain extension, broken or shortened polymer chains that result from use or reprocessing are linked back together with special chemical agents such as peroxides, carbodiimides, isocyanates, or epoxies. This rebuilding step restores the polymer’s molecular weight and improves its strength, toughness, and thermal stability, helping recycled plastics perform more like new materials. 65
Functionalization takes a different approach by adding reactive or polar groups-like hydroxyl, carboxyl, or amine-onto otherwise inert plastic chains. Figure 5 shows that C–H functionalization of polyolefins enables dynamic networks.
66
This makes different types of plastics, which usually do not mix well, more compatible (for example, PET with polyolefins) and also improves adhesion with fillers and fibers. Chain extension strengthens the structural integrity of recycled plastics, while functionalization makes them more useful and compatible with other materials. They work together to make mixed or broken plastic waste into stronger, more useful materials, which helps make the plastic economy more circular and sustainable.
65
C–H functionalization of polyolefins enables dynamic networks: (a) a route to replace non-recyclable thermosets; (b) amidyl radicals offer advantages over peroxide methods; (c) selective C–H functionalization forms diketoenamine-based dynamic, reprocessable polyolefin networks.
66

4.2.5. Enzymatic PET depolymerization
Enzymatic depolymerization of PET employs chemicals and enzymes such as Humicola insolens cutinase (HiC) to decompose crystalline PET under moist-solid conditions. This method skips the energy-intensive step of amorphization and makes terephthalic acid that is more than 95% pure and ready to be turned into new PET. 67 Enzymes have a hard time breaking down polyethylene (PE) and other polyolefins because they have strong carbon-carbon bonds. A chemo-enzymatic method is used to fix this. First, PE is chemically oxidized (for example, with m-chloroperoxybenzoic acid and ultrasonication) to add reactive groups. Then, enzymes like peroxidases make it even more oxidized. Finally, a mix of enzymes, alcohol dehydrogenases, Baeyer–Villiger monooxygenases, and lipases or cutinases convert the PE into hydrolyzable esters, which are broken down into ω-hydroxycarboxylic and α,ω-dicarboxylic acids. 19 Overall, these approaches demonstrate how enzymatic processes—often aided by mild chemical treatments—can transform a wide range of plastics, from easily degradable polyesters like PET to more inert polyolefins like PE, into reusable monomers and functional intermediates under environmentally friendly conditions.
A one-pot dual-enzyme system that can recycle mixed plastics without having to sort them out by combining an engineered polyester hydrolase (PES-H1 FY) and a polyurethanase (UMG-SP-2). This system breaks down PET, PBAT, and TPU into their monomers at the same time. Hexafluoro-2-propanol (HFIP) is used to dissolve and then reprecipitate plastics. This makes enzymes easier to get to and break down. This method is a useful and long-lasting way to break down mixed plastics into parts that can be used again in mild conditions. Figure 6 shows how the PES-H1 FY and UMG-SP-2 enzymes break down mixed polymers (PET, PBAT, TPU) into their monomers (TPA, AA, FAD, MDA). The process uses enzymatic depolymerization to break down mixed plastics after they have been dissolved and then precipitated. This makes them easier to recycle.
68
Different monomers come from enzymatic hydrolysis of PBAT, PET, and TPU: (a) PBAT breaks down into TPA and AA (not shown: butane-1,4-diol). (b) PES-H1 FY breaks down PET into TPA (without ethylene glycol). (c) TPU that has been broken down by PES-H1 FY and UMGSP-2 makes FAD and MDA. Other minor products are not shown for clarity.
68

4.2.6. Plasma surface modification
Plasma surface modification is an environment friendly way to improve plastics like PLA films without changing their bulk structure. In this process, ionized gases such as oxygen or argon bombard the surface, breaking bonds and adding new chemical groups. Oxygen plasma wets things more by adding polar groups, and Argon plasma roughens the surface such that it can bond better. In practice, this makes it simpler to print, coat, and recycle plastics without the need for solvents. 69 It also makes inks, coatings, and adhesives adhere better. Plasma surface modification uses cold plasma from gases like O2, N2, Ar, or NH3 to change the surface of plastics. Energetic ions, electrons, and radicals hit the surface and break C–H and C–C bonds within the top 100 nm, creating reactive sites. These sites react with plasma species to add groups like –OH, –COOH, C=O, or –NH2. Ion bombardment makes the surface rougher and bigger. With added monomer vapors, ultra-thin functional films can also be deposited. The process combines bond breaking, radical formation, functionalization, and surface restructuring for precise plastic modification.70–72
Fouling makes reverse osmosis membranes work less well. Plasma treatments that use gases like O2, Ar, N2, CO2, and air make the surface less likely to get dirty. When you graft hydrophilic polymers onto a surface, it is less likely to get dirty with organic matter. RO membranes are even better when advanced techniques are used, such as atmospheric plasma-assisted graft polymerization with water-soluble monomers. These techniques cut down on scaling and fouling and make the membranes more permeable to water. Figure 7 shows how plasma-induced surface activation changed polyamide RO membranes, followed by surface graft polymerization.
70
Polyamide RO membrane modification via plasma-induced surface activation.
70

4.2.7. Chemical pretreatment and chemo-enzymatic approaches
A very effective way to break down plastics that are hard to break down, like polyethylene, is to use chemicals. Chemical oxidation is the first step in the process. It usually uses ultrasonication and m-chloroperoxybenzoic acid. This oxidation adds reactive groups to the polymer backbone that would not be there otherwise, such as hydroxyls, carbonyls, and esters. These new functional groups make the polymer more chemically reactive and hydrophilic on the surface, which makes it easier for enzymes to attack it. Figure 8 illustrates how plastic polymers are converted to monomers or valuable molecules via chemical, enzymatic, or combined chemoenzymatic depolymerization pathways.68,73 Schematic illustrations of the chemoenzymatic cascade depolymerization of plastic.
73

An enzyme cocktail of catalase-peroxidases, alcohol dehydrogenases, Baeyer–Villiger monooxygenases, and lipases/cutinases hydrolyzes chemically oxidized polyethylene into esters and subsequently into monomers such as ω-hydroxycarboxylic and α,ω-dicarboxylic acids, achieving ∼27% conversion in preparative-scale trials (GC-MS, weight loss). AFM confirms nanoparticle size reduction, and FT-IR verifies ester bond formation and cleavage. This gentle, multi-step chemo-enzymatic process is a more environmentally friendly way to recycle polyolefin waste than traditional thermochemical recycling. 19
4.2.8. Vitrimers & dynamic covalent polymers
Dynamic covalent polymers, especially vitrimers, use associative bond exchange, which means that bonds can break and reform at the same time without losing the integrity of the network. The cross-link density stays the same during the exchange process. Dissociative systems are not the same as this. Transesterification, transcarbamoylation, and boronic ester exchange are all reactions that happen a lot.
74
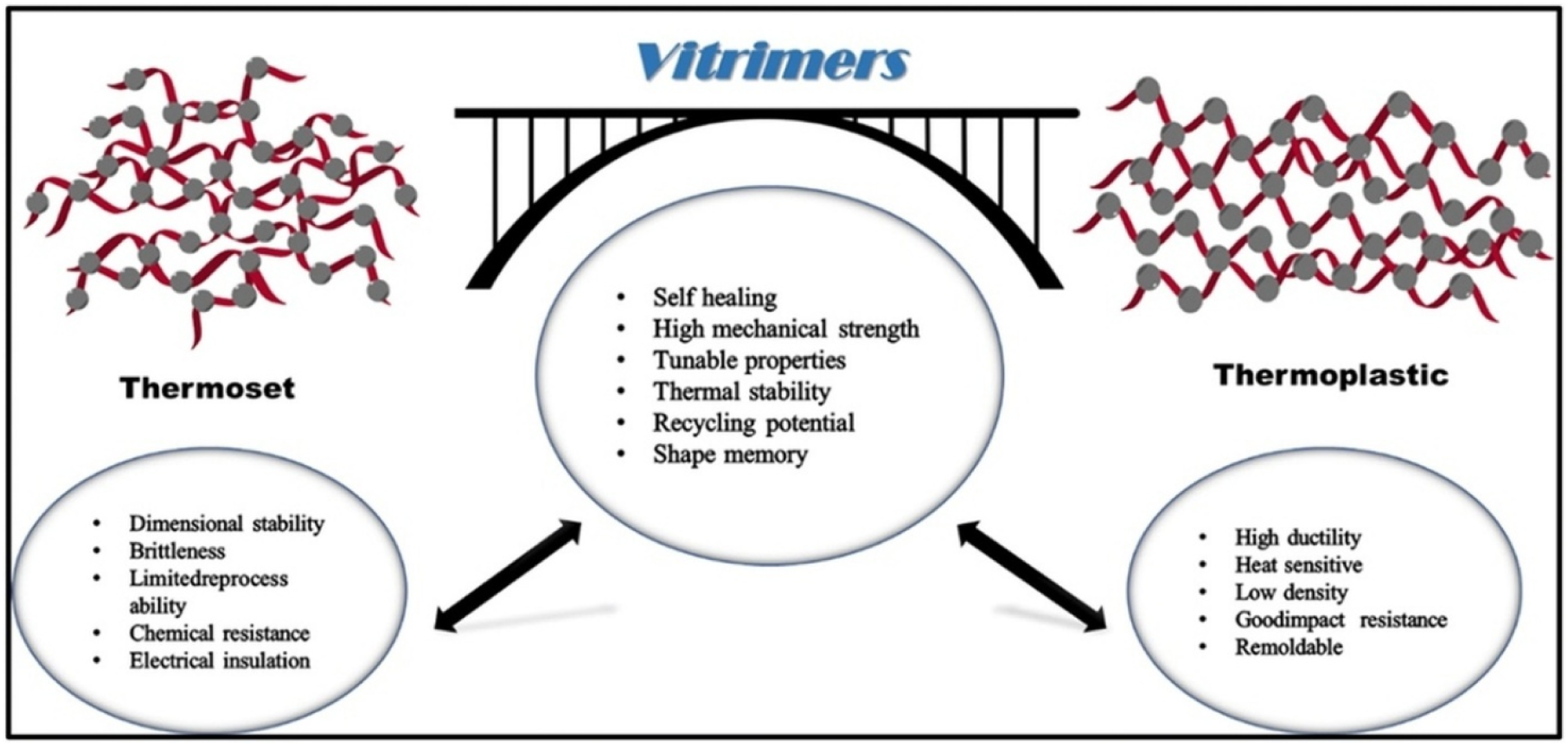
Vitrimers and dynamic covalent polymers (DCPs) can be recycled and reprocessed because they have reversible covalent bonds built into a network that is always cross-linked. In vitrimers, bond exchange reactions which are associated, such as transesterification or transcarbamoylation, allow the breaking and formatting of bonds at once. This maintains the cross-link density constant, so that only the topology of the network is allowed to distribute with heat or catalyst. The distinctions between thermoset and thermoplastic polymers are given in Figure 9. It demonstrates that vitrimers, in being both self-healing and recyclable while stable at high temperatures, are indeed having the best of both worlds.
75
Vitrimers as bridges between thermosets and thermoplasts.
75
Dynamic covalent polymer systems operate in either the 6 associative or dissociative mode. In systems where dissociative bonds valid for linkages, for example Diels–Alders, that reduce crosslink density before reformation on them are formed and ruptured the. In associative systems the network remains unbroken throughout. 76 At some high activation temperature these materials melt like thermoplastics through bond exchange. That allows the materials to be reshaped, self-healed and recycled many times. Upon cooling down, they regain their original mechanical performance. 74 To optimize the tradeoff among mechanical strength, stress relaxation rate, and healing efficiency. 77 Recent developments encompass catalyst-free networks, multi-dynamic bond networks for higher durability and use of bio-based monomers in line with the circular economy strategy. 78
4.2.9. Wax worm biodegradation of polyethylene
Polyethylene (PE) can be broken down by enzymes in wax worm larvae saliva (Galleria mellonella). These enzymes are predominantly phenol oxidases and hexamerins. These enzymes begin the process of breaking down and oxidizing the otherwise stable PE chains at low temperatures. 79
The gut microbiome of wax worms also helps break down PE, in addition to saliva enzymes. Bacteria like Bacillus, Pseudomonas, and Enterobacter stick to the plastic surface and release oxidative and hydrolytic enzymes (laccases, peroxidases, and esterases) that break down oxidized PE into smaller pieces. These are broken down even more into things like short-chain fatty acids and CO2, which are then used by microbes.
80
Tests have shown that wax worms can lower the mass of PE film in just a few days. FTIR and GPC analyses have confirmed that this happens by changing the chemicals and lowering the molecular weight.
81
Figure 10 shows a polyethylene sample with visible pits and cracks on the left, and broken fragments after after the decomposition by larvae on the right.
81
Picture of the naked eye before and after the decomposition by larvae of polyethylene.
81

This dual mechanism: saliva-driven oxidation followed by microbial enzymatic degradation offers a rapid, eco-friendly alternative to traditional plastic waste management, and highlights the potential for developing bio-inspired recycling technologies.
4.2.10. Cleavable linkers for sustainable deconstructable thermosets
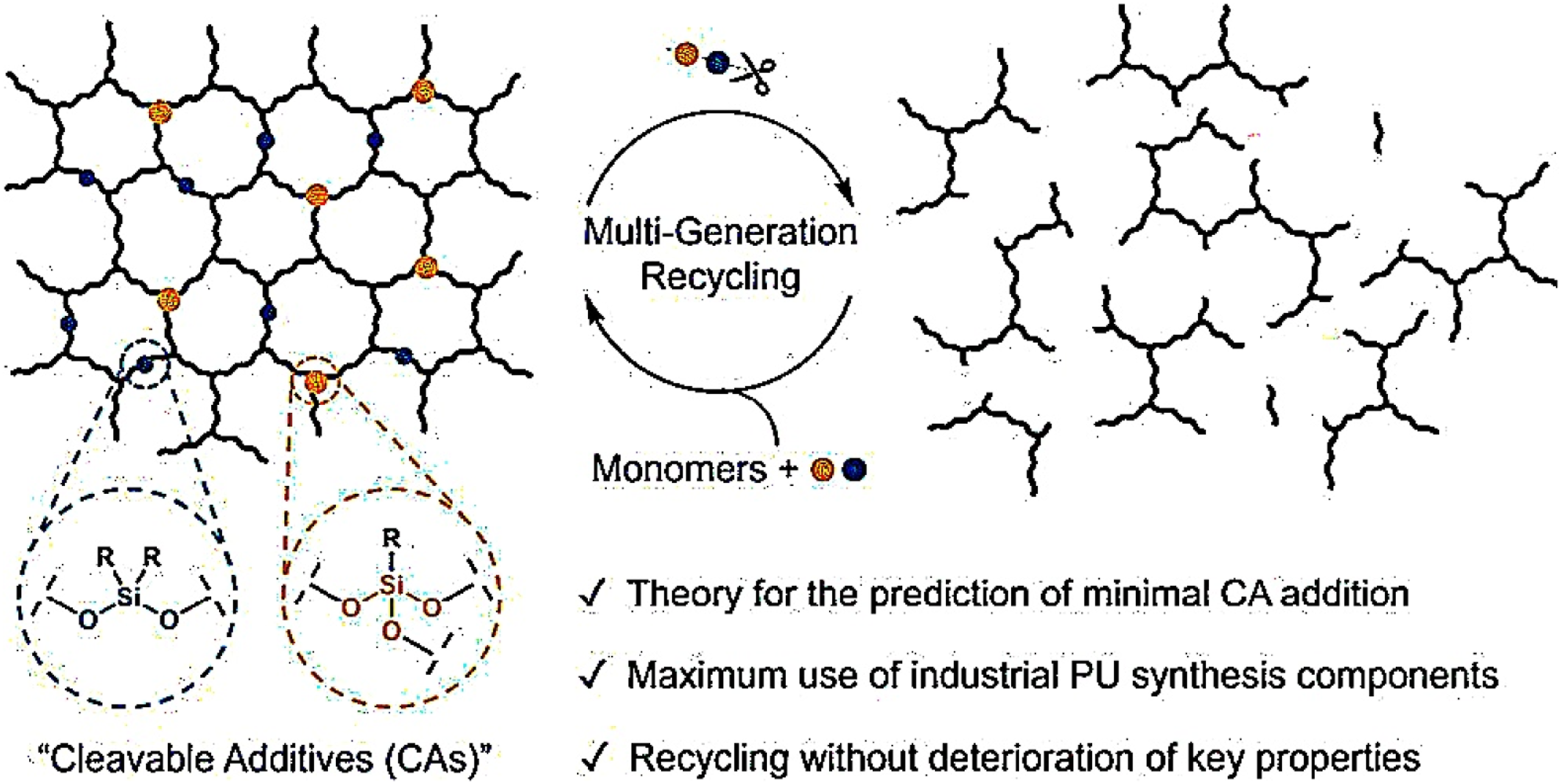
Deconstructable thermosets are built with carefully designed cleavable additives (CAs) that let the material be taken apart when desired, while staying strong and reliable during everyday use. Two main kinds of silyl ether-based CAs are used: bifunctional cleavable strands (BCSs), which can be made in 52% yield, and trifunctional cleavable junctions (TCJs), produced in higher yield (79%) from simple, widely available starting materials. These additives are integrated into polyurethane thermosets during synthesis. Under normal conditions, they remain stable, but when treated with a mild catalyst (FeCl3 in THF/methanol at 50°C), the Si–O bonds in the additives selectively break apart, leaving the rest of the polymer network, such as carbamate bonds, untouched. The amount of additive needed matches predictions from reverse gel-point theory. Figure 11 illustrates the use of cleavable additives (CAs) enabling multi-generation recycling of polyurethanes into monomers, maximizing industrial reuse while maintaining material properties. It takes about 0.3–0.4 equivalents (8.4–11.6 wt%) to completely break down BCSs, but only 0.10–0.15 equivalents (3.6–5.4 wt%) to break down TCJs. This makes TCJs 63% more efficient. After deconstruction, the pieces can be recovered in first-generation systems at a rate of about 81% and in second-generation systems at a rate of up to 85%. They can then be used to make new thermosets.
82
Recyclable industrial PU networks made possible by the small inclusion of cleavable additives (CAS).
82
This method is amazing because it lets recycling happen over many generations. Polydicyclopentadiene thermosets, for instance, have been recycled three times, with each round using 40–45 wt% of the recovered fragments. They still have strength, stability, and full recyclability. 83 Strand-fusing cross-linkers (SFCs), a more recent invention, push the idea even further. These join two chains of a network through four-way cleavable junctions. That change means that deconstruction can begin with just a fifth of the additive required in previous designs. They additionally make things work better by enhancing properties of matter that are thermomechanical in nature, for example, the glass transition temperature. 84
4.2.11. Electrochemical approaches to polymer recycling and upcycling
Electrochemical modification is an emerging, green route for recycling and upcycling of polymers. It uses electricity to change chemicals at low temperatures, so you can control exactly which reactions happen without having to use high temperatures or harsh chemicals like those found in traditional thermochemical processes. The main process is electrochemical depolymerization, which breaks down polymer chains by moving electrons at the surfaces of electrodes. This turns waste plastics into monomers, which are the building blocks for repolymerization. For instance, it was shown that heterogeneous electromediated depolymerization of highly crystalline polyoxymethylene (POM) could produce formaldehyde and 1,3,5-trioxane at room temperature in just 30 minutes.85–88 Figure 12 showed that electrochemical depolymerization of POM produces formaldehyde, oxydimethanol, and trioxane, with HFIP assisting by disrupting crystallinity and providing protons to enhance depolymerization.86,87
This is possible because anodic oxidation of 1,1,1,3,3,3-hexafluoro-2-propanol (HFIP) produces catalytic protons that help break up acetal chains and, at the same time, make polymers less crystalline. Electrochemical functionalization also changes the properties of polymers by adding functional groups to the backbones without breaking them down completely. This is accomplished through metal-free systems or metallaelectro-catalyzed pathways to direct electrolysis, resulting in high-valent metal-oxo species that transfer hydrogen atoms from polymer backbones to generate carbon-centered radicals for subsequent functionalization. Also, paired electrolysis strategies are the best because they use both anodic oxidation and cathodic reduction reactions at the same time. In this setup, one electrode changes the polymer, and the other makes useful byproducts like hydrogen gas or helps with other changes. This is a whole electrochemical device that kills the plastic life cycle.85–88
5. Results and discussion
This chapter provides a depth investigation of modified polymers produced by various approaches to reduce plastic waste or enhance recyclability. Characterization methods, including FTIR, DSC, TGA, and SEM, were adopted to verify the effectiveness of different modification methods in experimental studies.89–91 The results provide insight into how these changes impact the polymers’ heat conduction capability; their strength; their resistance to chemicals; and, in the context of breakdown in day-to-day use or during environmental waste disposal, what happens as they degrade. This tells us important things about how to make polymer systems that are better for the environment. 92 The findings help us gain insight into how some types of changes can be beneficial for addressing the global challenge of plastic waste management, while either maintaining or enhancing material performance targeted for various applications in industry. 93
5.1. Characterization of modified polymers
5.1.1. Fourier transform infrared (FTIR) analysis
It was found from FTIR spectroscopy that the structure of functional groups changed considerably due to polymer modification processes. The study of recycled polyethylene terephthalate (PET) showed that the chemical structure changed very little after three recycling cycles. FTIR spectra showed that the peak positions and intensities were almost the same as those of virgin material.
94
For polymer-modified systems, there were clear identification peaks at 997–1000 cm-1 for isotactic polypropylene bands and at 1014 cm-1 for siloxane (Si-O-Si) stretching vibrations. This made it possible to successfully find the amount of polypropylene in recycled materials. Figure 13 displayed FTIR spectra comparing neat PG 67 with 3% and 9% pure PP, showing a distinct absorbance peak at 997 cm-1 indicating PP content effects on the material.
95
The spectroscopic analysis of naturally weathered polypropylene showed systematic changes in carbonyl index values, with C=O stretching bands at 1760 and 1720 cm-1 assigned to esters and ketones respectively. The COO- antisymmetric stretching bands were observed in the 1620-1550 cm-1 region, with intensities stronger than C=O stretching bands, indicating substantial carboxylate group formation during degradation. The 3600-3200 cm-1 region showed medium to strong OH stretching vibrations, while vinyl groups were detected through C=C stretching modes in the 1660-1600 cm-1 region.
96
FTIR spectra of 0%, 3% & 9% pure PP modified asphalt binder.
95

Advanced machine learning approaches using convolutional neural networks successfully identified functional groups from FTIR spectra with high accuracy across 15 common organic functional groups, demonstrating 8728 gas-phase organic molecules analysis capability. 97 The μ-FTIR reflectance spectroscopy coupled with principal component analysis proved effective for classifying microplastics even when modified by photodegradation, showing higher sensitivity to degradation compared to traditional ATR-IR methods.96–99
When compatibilizers were added to polymer blends, the spectra shifted in a way that was typical for those blends. No new functional groups were found after mixing, which showed that the interactions were physical rather than chemical. 99 These FTIR-based identification systems have shown to be very useful for analyzing polymer reprocessing operations in factories.
5.1.2. Differential scanning calorimetry (DSC)
DSC study revealed significant alterations in the thermal characteristics of regenerated polymer systems. Modern DSC instruments are very good at finding thermal transitions, such as melting points, crystallization temperatures, and glass transition points that are specific to each type of polymer. 100 When recycled PET samples were analyzed with DSC it was observed that the molecules were breaking down further as a result of an increase in recycled content. The glass transition temperatures went down because the chains were breaking during reprocessing, and the glass transition temperature ranges got wider because the molecular weight distribution increased.101,102
Differential scanning calorimetry (DSC) research of recycled polypropylene showed that it behaved differently from virgin material when heated. Figure 14 illustrated the melting characteristics of PP: HDPE blends derived from DSC analysis. The melting point of virgin PP was 167.4°C and the crystallization point was 122.3°C, with a crystallinity of 31.1%. The melting point of recycled PP was lower at 164.9°C and the crystallization point was 122.7°C, with a crystallinity of 19.6%. The lower melting temperature and crystallinity in recycled PP were caused by chain scission during mechanical recycling, which made the material less strong.
103
Thermal degradation studies utilizing DSC indicated that biodegradable polyesters demonstrated peak combustion temperatures ranging from 292.9°C for PHA to 408.9°C for PCL, with total heat release values between 18-28 kJ/g, markedly lower than those of conventional polyolefins. The thermal degradation temperatures, from highest to lowest, were: PCL > PBS > PLA > PBAT > PHA.
104
The melting characteristics of PP:HDPE mixes derived from DSC: (a) vPP:vHDPE blends and (b) rPP:rHDPE blends.
103

5.1.3. Thermogravimetric analysis (TGA)
TGA analysis was a good way to check plastic waste pyrolysis because it showed us the temperatures and speeds at which the process breaks down. The thermal breakdown of recycled polymers started at 200–400°C and ended at 490–540°C, leaving behind less than 5% solid. Recycled polymer systems had higher temperatures at which they started to break down than new materials because reprocessing shortened the polymer chains. The amount of energy needed to start the pyrolysis of mixed plastic waste was about 196.45 kJ/mol.105,106
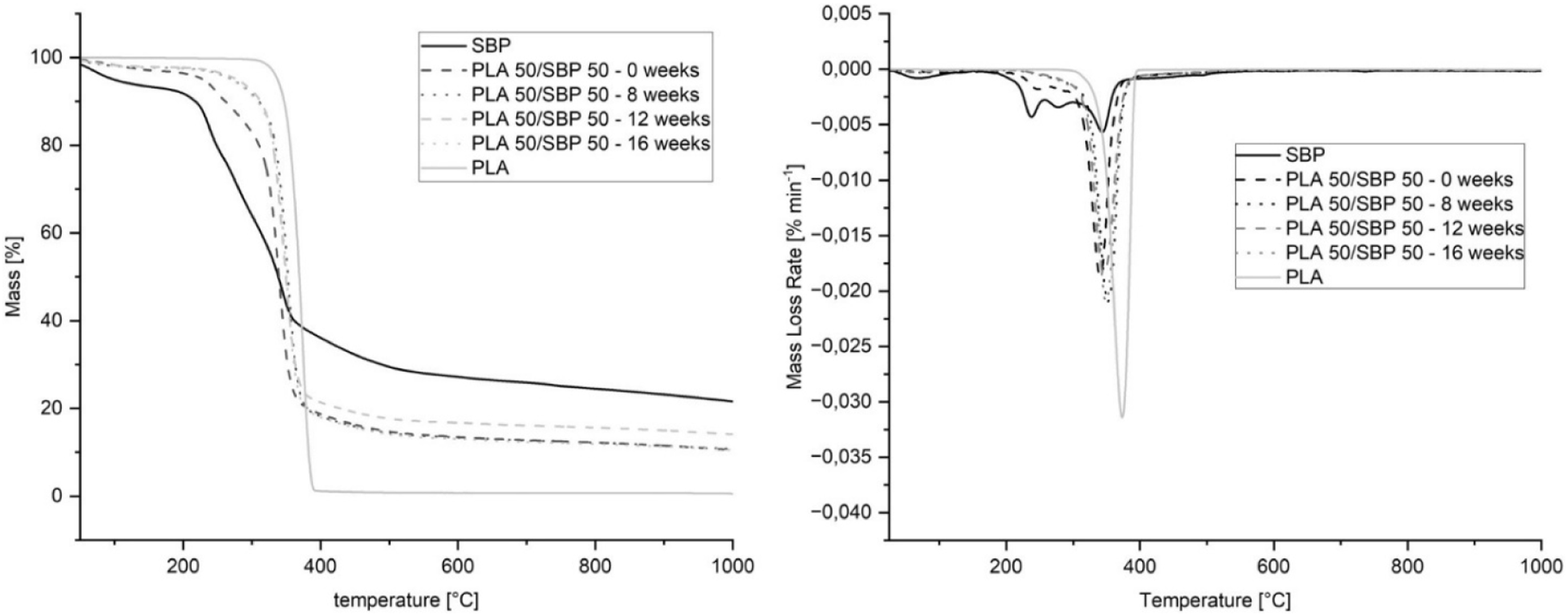
Advanced TGA analyses of polypropylene/biomass composites exhibited intricate degradation patterns. Studies have shown that adding more biomass to bio-composites makes them lose more weight at lower temperatures because hemicellulose and cellulose break down. On the other hand, adding more PP makes them lose more weight at higher temperatures.106–108 TGA thermograms of SBP, PLA and PLA/SBP blends at different aging times were shown in Figure 15, which presented the thermal degradation stability and weight loss process with temperature for each sample. These results provide important implications for sustainable composite materials from agricultural by-products. On the left, there is a TGA of HDPE-SBP composites, which are made of HDPE (high-density polyethylene) and SBP (sugar beet pulp). On the right, there is a DTG of HDPE-SBP composites, which are also made of HDPE and SBP.
107
TGA-FTIR analysis revealed that various biodegradable polymers disintegrate via unique mechanisms. PBS breaks down by cyclic intramolecular cleavage of O–CH2 bonds close to succinate groups, which makes anhydrides. PCL breaks down by unzipping from hydroxyl end groups, while PLA breaks down in a more complicated way that includes transesterification and cis-elimination reactions. Thermogravimetric studies showed that the final decomposition products depend on the atmosphere. For example, CO2 and water form in oxygen, but oligomers and unsaturated carboxylic acids form in nitrogen. The total heat release went down in the order PHA > PCL > PBAT > PBS > PLA, all of which were lower than polyolefins. This means that they are less likely to damage the furnace. 104
5.1.4. Scanning electron microscopy (SEM)
To study modified polymer scanning electron microscopy (SEM) is a highly effective tool. It gives high resolution image of surface morphology, particle dispersion, and also nanoscale structures to justify how structure and polymer nanocomposites are connected. The SEM study of chitosan derivatives shows that changing them a lot changes their shape. Chitosan that hasn’t been changed has surfaces that are smooth surfaces. Schiff base-modified and crosslinked derivatives, on the other hand, have rough, uneven surfaces because the amounts of crosslinking are not the same. Modified chitosan nanoparticles have rough, evenly spaced surfaces that are made up of sodium tripolyphosphate that forms in place from electrostatic interactions between tripolyphosphoric and amine groups. The sizes of the particles range from 27.1 to 38.6 nm (TEM) and less than 100 nm (DLS), which makes them good for biomedical use. 109 An upgrade to SEM, like FE-SEM, gives detailed information about polymer nanocomposites, such as how fillers are spread out and how they stick together. PMMA/TiO2 nanocomposites have spherical TiO2 particles that are 50 to 75 nm in size and rough surfaces because the nanoparticles stick together because they have a lot of surface energy. 110 SEM–EDS analysis confirmed elemental composition and effective incorporation of modifying agents into polymer matrices. 109
Using a 3 nm platinum coating and a 10 kV accelerating voltage is the best way to look at polymeric membranes with a SEM. This takes great pictures without hurting the polymer. Coatings on microfiltration membranes have difference in the average pore size is 3% and difference in the porosity is 13.3%. However, ultrafiltration membranes have pore diameters difference 15.9–20.0% and porosities difference 22.5–29.7%. This shows that the thickness of the coating effected the properties of membranes. The SEM surface images of the MF, UF and RO membranes at 5, 10 and 15 kV are presented in Figure 16. It demonstrates how the voltage that makes things move faster, affects contrast and ability to see features. In order to obtain clearer pictures, a thin film of platinum was then deposited on all samples. The Indirect-Freeze-Fracture approach does not alter the membrane structure and it is easier to investigate cross-sections in detail.
111
Comparison of surface images taken at different accelerating voltages. (a–c) MF, (d–f) UF, and (g–i) RO images at 5, 10, and 15 kV, respectively. All samples were coated with 3 nm of platinum.
111

SEM analysis showed that the cellulose films, which were smooth and uniform that started to show signs of surface degradation after only 7 days of being buried. The films got rougher and irregular over time because of sand particles from the soil stuck on to the surface. 35 days later, there found clear changes to the structure, and 56 days later the surface had broken down badly that deep cracks formed on the surface, getting worse with time. Other studies have also shown that PLA/acetyl tributyl citrate/chitosan composites break down in the same way on the surface 91 costs. From SEM image found that the soil-release polymer treatment made the surface of the polyester fibers become smoother by the soil-release polymer treatment and filled up the spaces between fibers that were overlapping, eliminating from where soil may settle. The density and distribution of polymer deposits varied depending on the type of treatment. Some treatments generated localized high-density areas that changed the overall surface properties. 112
5.2. Mechanical and physical properties
5.2.1. Tensile strength and elongation at break
Significant differences in tensile properties were found by mechanical testing after polymer modification and recycling. Relicensed recycled acrylonitrile-butadiene-styrene (ABS) compounds retained good tensile modulus stability, with the measured values close to those of reference virgin materials, regardless of both the levels of reprocessed content and the cycles that they underwent. Nevertheless, impurities of coating resulted in being more critical and decreasing up to 42% the impact strength for ABS and 28% for ABS/PC blends for a content in paint higher than the composite proportions. 113
In many types of polymers, virgin polymers have better tensile strength than recycled ones. For high-density polyethylene (HDPE), virgin samples got 21.81 MPa, while recycled samples only got 18.31 MPa. Blends of 50% recycled and 50% virgin showed values in between, at 21.05 MPa. Polypropylene virgin samples showed a tensile strength of 27.43 MPa, while recycled materials showed a tensile strength of 19.31 MPa. Polyvinyl chloride had the highest tensile strength, with 57.85 MPa for virgin samples and 32.83 MPa for recycled samples. 114
Technologically advanced recycling made massive leaps in the mechanics of how things worked. When the right processing methods were used, recent changes in the mechanical properties of recycled materials showed increases of 40%, 60%, 28%, and 64% in tensile strength, Young’s modulus, toughness, and yield strength, respectively. 115 Under the best processing conditions, recycled high-density polyethylene had tensile strength that went up by as much as 20.4% and elastic nominal stiffness that went up by as much as 93.5%. Nominal yield strain went down by as much as 50%. 116
5.2.2. Density and hardness
Density and hardness are the physical properties that give information about the structural strength and performance of modified polymeric materials. These parameters affected by polymer modification through blending, filler addition, or chemical alteration significantly and influencing both recyclability and mechanical durability. Variation of density usually occurs from differences in filler dispersion, void formation, and polymer chain packing. For example, adding high-density fillers such as glass fibers or carbon particles increases the composite density; on the other hand, introducing bio-fillers or creating micro-voids for lightweighting reduces composite density. 116
It is important for recyclable polymers to have a moderate density because a higher density can make transportation and processing more expensive, and a lower density might mean that the polymer doesn’t bond well at the interface and is structurally weak. Hardness, meanwhile, speaks to how well a polymer will withstand surface deformation and mechanical wear, something that’s key for cycles of reuse and recycling. Researchers have learned that materials can be made harder by adding rigid fillers or increasing the density of crosslinks. In contrast, adding flexible chains or cleavable bonds that may degrade during recycling could make it softer. 117
The surface hardness is closely related to the well distribution of the filled particles and their adhesion among them. These considerations influence how a material would perform through multiple recycling cycles without compromising its structural integrity. Therefore, density and hardness need to be well-balanced. The right hardness ensures the material can survive reuse, while the right density makes recycling a less laborious process and requires less energy. So, one answer to the riddle is that successful polymer modification should work to preserve structures that are both strong and lightweight, ensuring that materials remain tough and last over many lifetimes. 118
The influence of surface modification on physical attributes was found as per the contact angle and topography of the surface. The contact angles of water on plain polyester surfaces dropped from 65° to approximately 40° for surfaces that were modified with certain polymers. This indicates that the surfaces are relatively more hydrophilic. Optimal polymer formulations lowered the contact angles to as little as 23°, demonstrating that surface functionalization has been highly successful. 119
5.3. Chemical and environmental stability
5.3.1. Solvent resistance and chemical stability
The chemical resistance test indicated that modified polymers are more durable and recyclable. For recycling dirty or mixed-up plastic waste, solvent-based chemical recycling was more effective than traditional mechanical methods. With the PET method of dissolution precipitation, the polymer could be more effectively and with less energy recovered from one another because it retained the chemical bonding. Solvolysis was less profitable due to the high investment of cost capital as well as inefficient process; it turned out to be the best choice for recycling composites. Acid-based systems were more expensive, as the equipment was costly and fibers could be damaged. Other solvent systems, on the other hand, were less expensive. The most expensive way to recycle chemicals was with dynamic covalent reactions, mostly because catalysts cost a lot. But if prices go down in the future, it could be as simple as recycling by hand. Choosing solvents based on Hansen solubility parameters led to the creation of bio-based and eco-friendly solvent systems that dissolve polymers well at moderate temperatures. Ionic liquids also became promising green alternatives because they allow for faster processing at lower temperatures with less harm to the environment. 93
5.3.2. Biodegradability and environmental degradation study
An analysis of environmental degradation showed that modified polymer systems have different patterns of biodegradability. Biodegradable plastics in soil environments didn’t fully break down, which released microplastics and other breakdown products that were affected by the soil’s properties, the weather, and the type of polymer used. 120 Studies of soil burial degradation showed that biodegradable polymer films broke down faster than regular polyethylene-based material. als. Weight loss measurements indicated that decay accelerated markedly when the pre-striking of UV light was employed before the burial. 119
The temperature dependent biodegradation effects were considerable for end-of-life degradable plastic accumulation. Degradation of biodegradable polymer system, if degraded under higher temperatures, was promoted by the environmental conditions, such as humidity and microbial activity. 121 Cellulose films experienced total biodegradation during the 63-112 days of soil burial testing and three phases were observed: scum colonization, substrate decomposition by enzymatic action, with comsumption to CO2, water, and biomass. 91
Microbes degrade biodegradable plastics and consign them to soil systems, which is good for organic matter in the soil. Scientists found that PLA microplastics increased levels of total and dissolved organic carbon. That would mean that over the long run they could reshuffle the ranks of soil’s little organisms, providing some microorganisms more carbon to grow on than others. Certain biodegradable plastics, however, could leach heavy metals as they decompose. This could potentially harm the plants as well as the soil microbes. 120
5.4. Discussion on recyclability enhancement
The recyclability improvement obtained by different modifications has been proven to have a promising perspective for dealing with plastic waste problems. Mechanical recycling rate was greatly enhanced for the materials that were formulated with intentions of recycling, in particular by using compatibilizers and processing aids, which allowed the processing of mixed waste plastics. Thermal processing compatibility became an important requirement, as when tailored for this purpose, modified polymers continued to exhibit good melt flow characteristics and thermal stability even after several reprocessing cycles. Chemical recycling had the advantage by integrating mechanical treatments for soiled and composite waste flow, which could not be treated in a conventional processing line. Among them, the enzymatic depolymerization had great potential for closed-loop recycling (degradation route e) as a total monomer recovery can be obtained without quality loss. Pyrolysis followed by conversion to lubricant oils was the most profitable way to recycle chemicals. Gasification and other chemical recycling methods, on the other hand, needed special carbon capture systems because they released a lot of carbon dioxide. The integration of modified polymers into a circular economy demonstrated significant potential for resource conservation. When mechanical and chemical recycling methods were used together, they worked better together. Mechanical recycling dealt with clean waste streams, while chemical methods dealt with dirty materials. 93
When mechanical or chemical methods became too expensive, the addition of bio-based parts made it easier to recycle everything by giving controlled biodegradation pathways for end-of-life disposal. When looking at the emissions that could be avoided by not making new polymers, life cycle assessment showed that recycling technologies were better for the environment than incineration or landfilling. However, long-term studies that are very thorough and standardized testing methods are still needed to prove that degradation works well in conditions that are similar to those found in nature. 120
5.5. Machine learning and AI-assisted recycling optimization
Artificial intelligence and machine learning are changing the way polymers are recycled by making slow, manual steps into quick, data-driven ones that can work on a large scale. This will help solve the problem of plastic waste around the world more effectively. Using real data from an industrial facility, the Mask R-CNN model was able to classify things with 91.2% accuracy (mAP 91.1%), while the YOLO v8 model was able to do so with 86.7% accuracy (mAP 92.2%) and much faster inference speeds. This made it a great choice for real-time sorting systems. 122 Most impressively, graph neural networks and multi-objective optimization have found new ionic liquid catalysts for PET glycolysis that are 29% cheaper and 2.6% lower in CO2 emissions than what has been reported. Amazingly, about 47% of the AI-designed catalyst combinations work better than the best values found in the literature. 123
New developments in neural network-based property prediction are also changing the way we directly measure polymer performance from molecular structures. A hybrid CNN–MLP fusion model has been very accurate, with R2 values as high as 0.89 for predicting important mechanical properties like moduli, shear strength, and thermal transition temperatures. Poly-GNN architectures, on the other hand, speed up feature extraction by one to two orders of magnitude while keeping predictive accuracy the same across datasets of 13,388 polymers. 124 Deep reinforcement learning is also making polymer recycling better. For example, the Deep Deterministic Policy Gradient (DDPG) agent changes the conditions of a reaction in real time. Compared to traditional gradient-free methods, these methods cut down on the amount of work needed for experiments by 50–75%. They also make it possible for autonomous flow-chemistry systems to continuously depolymerize polymers. 125 Machine learning is speeding up the process of designing polymers that are good for the environment. Genetic algorithms employing virtual forward synthesis have yielded nearly one million candidates for ring-opening polymerization, while predictive models discern polystyrene alternatives characterized by minimal synthetic complexity and inherent recyclability. 126
AI is still changing the way we recycle polymers, even though there are still problems like small datasets, models that are hard to understand, and the fact that it’s hard to apply lab breakthroughs to real-world industrial systems. It gives researchers much better control over processes, speeds up the search for catalysts, and helps them design greener materials much faster and more effectively than traditional experimental methods ever could.
5.6. Environmental and economic impact
Strategies for modifying polymers to reduce plastic waste are good for the environment because they lower greenhouse gas emissions and help save resources. Using a mix of biomass and CO2 as renewable carbon feedstocks cuts the potential for global warming by 59%, which lowers the emissions from making polyurethane to 3.1 kg CO2-eq/kg. This is a 13% greater reduction than using either feedstock alone. Directly replacing CO2 in the production of polyols cuts the impact on global warming by 45% to 4.2 kg CO2-eq/kg without needing renewable electricity. 121 Chemical recycling and upcycling strategies make it possible to turn polypropylene waste that has been contaminated with chlorine into useful lubricants. This also gets rid of HCl emissions and chlorine product contamination. Catalyst regeneration keeps the liquid alkane yield at about 80%. 127 Depolymerization technologies use catalysts to turn polyesters back into monomers for repolymerization without damaging their properties. This makes a circular polymer economy possible. Lignin-graft-polyester biodegradable polymers show better biodegradability, thermal stability, and mechanical strength than regular petroleum-based polyesters, and they are much better for the environment. 128 These combined polymer modification methods all help to lower reliance on landfills, lower the extraction of fossil resources, stop pollution of the environment, and help achieve global climate change goals.
Research on recycling technologies indicates that they may reduce waste management expenses, generate employment opportunities, and facilitate the emergence of new markets. Mechanical recycling costs more than burning trash at first (677 tonne-1 vs. 66 tonne-1), but it is more profitable in the long run because the money made from selling recycled materials and the money saved by not having to throw them away add up to a benefit-cost ratio of 1.52. Life cycle analyses show that small recycling plants have trouble making money because they have to pay more for workers and shipping. On the other hand, big businesses save a lot of money by using less concrete and steel (over 7%) and lowering their overall lifecycle costs (5.9%). Without the cost of collection and transportation, chemical recycling techniques like pyrolysis could give you a 54% return on your money. The larger the plant, the more money you can earn. But such projects tend to be quite capital-intensive and require immediate access to a lot of waste, so governments, businesses, and communities have to work in conjunction with one another in order for them to function in low-income areas. 93
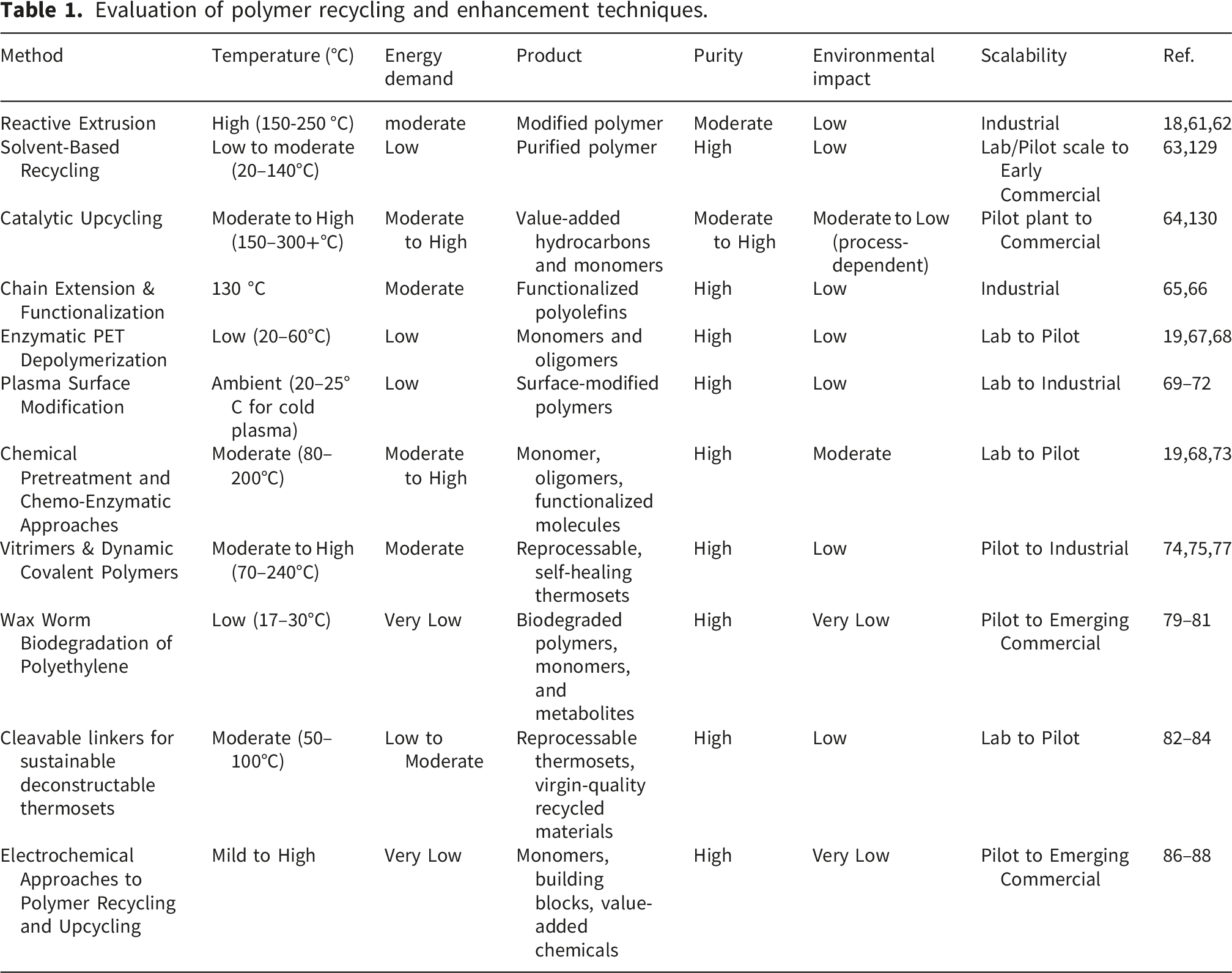
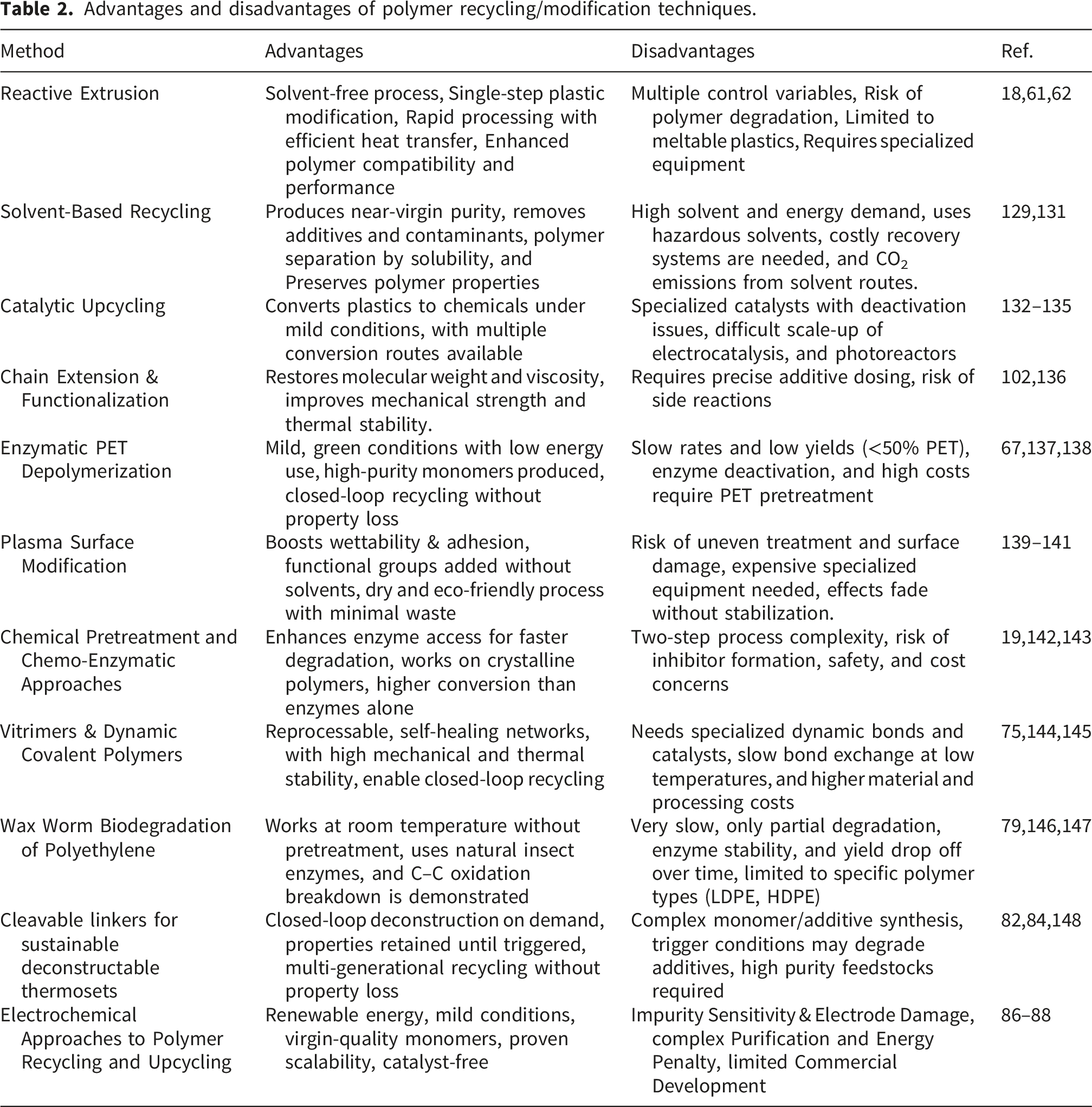
5.7. Comparison of polymer modification methods this study
Evaluation of polymer recycling and enhancement techniques.
Advantages and disadvantages of polymer recycling/modification techniques.
5.8. Life cycle assessment and techno-economic analysis for advanced polymer recycling
To determine whether advanced polymer recycling is sustainable and can be scaled in the real world, you have to use both life cycle assessment (LCA) and techno-economic analysis (TEA). Like an overview for the environmental effects of a material during its entire life cycle — everything from greenhouse gas emissions, energy use, and waste. TEA, by contrast, considers things like capital costs and operating expenses as well as how effectively the material goes head-to-head with new polymers. Mechanical recycling and PET glycolysis have the best performance in comparison. Their minimum selling prices are 9-73% lower, and their greenhouse gas emissions reductions are ∼7-88% compared with the typical chemical recycling process. Enzymatic Hydrolysis and Dissolution Produce Higher Quality Recyclates (2–27% higher melt flow rates), but they are expensive and more environmentally harmful. 149 They are significant factors impacting the environment in terms of electricity, steam, and solvent consumption. About producing terephthalic acid from new, enzymatically optimized PET recycling processes can save 69–83% of energy and reduce greenhouse gas by 17–43% across the whole value chain. 150
The best recycling technologies may never make it out of the laboratory if they cannot compete with new plastics that are very cheap or motion no real benefits for the environment. Without good LCA and TEA frameworks, it is difficult to ascertain what truly works. The plastics industry needs clear, standardized procedures for testing new processes that consider input energy sources, erratic waste streams, and the messy realities of how things function in real life. Only then can engineers, investors, and policymakers make decisions that truly advance us toward a circular economy.149,150 To move from a “take-make-dispose” plastics model to a circular system, you need creativity and solutions that work socially, economically, and environmentally. And you have to rigorously test, refine, and amplify those solutions.
5.9. Limitations of the study
There are a couple of important notes about these results. Laboratory testing conditions might not be representative of industrial processing environments, particularly in pollution levels and in the nature of used processing equipment found in actual recycling facilities. Sample preparation processes may introduce variability that isn’t representative of commercial recycling loads. That’s because the controlled environment of a lab tends to produce more uniform material than that produced at industrial plants that process many different streams of waste. 113
The economic analysis used current costs of materials and energy, but those numbers can vary quite a bit as recycling technologies mature and markets take different courses. The feasibility of various modification strategies also hinged on factors like feedstock availability, regulations in flux, and other disposal options that change. Market forces mattered a lot as well — namely, the prices of recycled materials and virgin plastics. Oil prices fall, and virgin plastics get cheaper. This devalues the return from recycled materials and makes the economy harder for businesses. 93
You can’t run a short study to understand the long-term effect on the environment. You have to stay vigilant for decades.” Stability tests and short-term biodegradation data can be of help, but they tell you only part of the picture. To really understand how they affect ecosystems, or if they could accumulate over time, we need to monitor these materials over much longer periods. There is also no way of knowing for sure how long biodegradable plastics will last, since claims on the subject are “all over the map” and there isn’t an industry standard for how the alleged biodegradability should be tested. 120
Researchers found it difficult to replicate their results because the labs were not all identical. Even small changes to how samples were prepared, such as making a minor tweak to the degree the microscope’s beam was focused before authoring an SEM scan, could alter what the material appeared like. Polymers decay as they interact with chemicals, are exposed to the environment, and age. You can’t be giving just one test or a little snapshot one time; try to capture what’s going on. It takes a combination of various analytical approaches, as well as long-term monitoring, to get the full picture.111,120
6. Prospect
The future of polymer modification is expected to focus on the production of polymers that can be simply recycled through controlled depolymerization and reprocessing. Recent progress has been made in the Design-for-Depolymerization, wherein the use of reversible covalent bonds, as well as materials with self-immolative polymer backbones, has led to the degradation of plastics to their monomers under mild conditions, indicating a truly circular material life cycle.
151
A path was drawn in Figure 17 where these emerging polymer recycling technologies could be scaled up by integrating the above developments in chemical and enzymatic, AI/ML optimization, robust analysis using life cycle assessment, and intensive stakeholder engagement. The following components jointly constitute the basis for achieving techno-economic feasibility and widespread acceptance of circular economics in the plastics industry.
33
Future directions for plastic recycling.
33

More recently, advances in microbial and enzymatic engineering have shown that enzymes such as PETase and MHETase work dramatically more efficiently after increasing their catalytic activity, thermostability, and pH tolerance with the help of directed evolution methodologies. Early examples for improving upon were making disulfide bonds to increase stability, and trying to improve divalent metal (like calcium) scattering sites in them has made them even more rugged than these already robust enzymes are against industrial processing conditions. 152 There is also increased interest in dynamic covalent networks, or vitrimers, as a promising approach to making thermosets and composites that are recyclable. These materials retain their structural strength, but can also be reprocessed with heat and even repair themselves if they are damaged. 75 And where disposing contaminated, or mixed plastics which cannot be recycled easily using conventional mechanical processes also becomes problematic, selective chemical recycling technologies like catalytic depolymerization, solvolysis, and pyrolysis start coming in vogue as eco-friendly initiatives. 153 Recent developments in catalytic and electrochemical recycling provide dramatic prospects for establishing economically sustainable circular polymer systems. These methods enable the conversion of plastic waste into monomers or new polymers by recovering building blocks, functionalizing polymer chains, and using paired electrolysis to precisely control catalyst activity and selectivity under mild conditions, even for complex multilayer materials. 86 Catalytic fast pyrolysis of 240 metric tons per day of mixed plastic waste demonstrates economic viability with a minimum selling price of $0.85/kg for ethylene, which is 1.4 times higher than fossil-derived ethylene at $0.61/kg. However, feasibility remains highly dependent on feedstock costs, which are three times higher than the actual pyrolysis process costs, while greenhouse gas analysis shows supply chain energy reductions mainly from eliminating fossil-derived chemical feedstocks. 154
If we don’t take action, global mismanaged plastic waste could nearly double from 62 million tons in 2020 to 121 million tons by 2050, and emissions from plastics are set to rise by about 37%, reaching 3.35 gigatons of CO2. But there’s good news: a strong four-part policy plan can change that trajectory. By requiring 40% recycled content, freezing virgin plastic production at 2020 levels, investing $50 billion in waste management, and adding packaging taxes, countries could cut mismanaged waste by 91% (down to just 11 million tonnes) and reduce emissions by one-third to 2.09 gigatons CO2. It serves as a reminder that coordinated action on a global scale can have a significant impact. 155
In the future, more and more effort should be dedicated to combining polymer-modification/recycling methods with full life cycle assessments (LCA) and technoeconomic evaluations. This will be a step towards making the environment and industry sustainable for real. To move from success in the lab to success at scale, we need new materials but also to engineer systems that work better as systems, that take into account how much energy they use, how much pollution they create, and how much total cost effectiveness we get from those materials.92,153 Biological (enzymatic) and depolymerization under mild conditions also have merits, especially with respect to polyesters and polyamides, where such a process is more selective and less energy-consuming. 92 To make genuine progress, researchers in chemistry and engineering, policymakers, and business innovators will need to collaborate more closely than ever before — moving from the welcome developments that researchers have proved in labs into innovative solutions that can transform how we make and use plastic. Just imagine a future where products are designed to be recycled endlessly, where sustainability is the inspiration behind innovation, and circular production is the standard. To reach it, scientists must collaborate; regulators must provide smart incentives; and businesses need to pledge that they will use closed-loop systems. When they all congeal, however, polymer modification becomes more than a scientific discovery; it turns into a means to make the future greener and circular as we turn trashy plastic waste into something that is useful to people and the planet.
7. Conclusion
Now, the problem of managing plastic waste must also evolve from traditional waste management to innovative molecular design of materials. In this review, we examined current polymer modification techniques that aim to overcome the fundamental drawbacks of traditional recycling; namely, material deterioration, contamination challenges, and the complexity associated with recyclable thermoset plastics. The results indicated that molecular modification is required to overcome the intrinsic degradation trend of plastics in each recycling treatment, according to this study. Methods such as dynamic covalent bonding in vitrimers or polymer chain repair using chain extension are efficient tools to tune material properties. Within here, new electrochemical methods allow for a mild depolymerization or functionalization of polymers with precise control now provided and an energy-efficient alternative to conventional recycling. By developing polymers that can be depolymerized at will, even strong materials such as thermosets can now be efficiently recycled in a pure-state process. Other methods such as catradic upcycling and enzyme-mediated depolymerization, demonstrated that mixed and complex plastic wastes, even multilayer packaging or polyolefins, could be converted into valuable homogenously reusable active monomers. The study highlights a central challenge, that is: the technical basis for a circular plastic economy is already in place (science), and could be implemented at scale, except for economic and industrial shortfalls. The absence of standardized life-cycle and techno-economic assessments complicates the evaluation of real feasibility and the attraction of major investments for a sustainable circular plastic economy.
Looking ahead, the polymer system will also need research collaboration linking industry and policymakers. Scientists have to concentrate on enabling their discoveries toward volume scalability and cost reduction, whereas industries and governments also need to participate in providing access to reliable feedstock supply, cheap catalysts, and new benchmarking evaluation methods. And when material design, on-the-ground testing, and conducive policies are all in place, polymer modification can move from the lab to reality and help shape global sustainability. It can result in the long-term beneficial treatment of the excessive problem of plastic waste.
Footnotes
Acknowledgments
The authors express gratitude to the following organizations for their cooperation in conducting this study: University of Rajshahi, the Bangladesh Council of Scientific and Industrial Research (BCSIR), and the Bangladesh Atomic Energy Commission.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
