Abstract
The absence of an effective technique for the individual separation of plastics within a plastic mixture remains a paramount concern in the domain of plastic waste management. Recently introduced, the floatation technique has emerged as a method for segregating specific plastics, such as polystyrene (PS), polyethylene terephthalate (PET), and polyvinyl chloride (PVC), from mixed waste streams. This separation process involves the utilization of traditional dual depressants (chemical agents). The study investigated the impact of pre-microwave irradiation of plastic surfaces at varying microwave output powers (20-100%) on the floatation behavior of each plastic. Additionally, the influence of depressant concentration (200-1600 mg/L) on plastic floatation was examined. The results revealed that pre-microwave irradiation at different output powers and varying depressant concentrations significantly affected the sink-float behavior of the studied plastics, with the exception of PET. It was observed that the microwave irradiation altered the number and type of active sites on the plastics' surfaces. Notably, there was no discernible regular trend in the flotation of plastic with an increase in microwave output power. The outcomes were substantiated by conventional identification techniques, including contact angle (θ) measurement, scanning electron microscopy (SEM) images, and attenuated total reflectance Fourier-transform infrared spectroscopy (ATR-FTIR) spectra analysis. Equations derived through the use of the design of experiment software (Design-Expert®) demonstrated a commendable alignment between the predicted and actual values of plastic flotation. This underscores the efficacy of the applied methodology in forecasting and validating the outcomes of the floatation process.

Introduction
The global consumption of plastics reached 440 million metric tons by the year 2022 and is projected to grow at a rate of 6%.1,2 This substantial increase in plastic usage can be attributed to their remarkable properties, which allow them to serve as substitutes for traditional materials such as wood, ceramics, and metals. Plastics are valued for their cost-effectiveness, lightweight nature, and inherent hygienic qualities. However, the surge in plastics consumption contributes to the generation of end-of-life articles that enter waste streams. The production of plastic articles entails significant investments of energy, financial resources, and labor. 1 In light of preserving natural resources and mitigating production costs, emphasis should be placed on plastic recycling. Moreover, recycling plastics serves to reduce adverse impacts on the human environment, considering that some plastics may take centuries to degrade and re-enter the environmental life cycle. Four primary strategies are employed for the recycling of plastics: reusing, mechanical recycling, chemical recycling, and energy recovery. 3 Reusing stands out as the most effective strategy for thermoplastics. Thermoplastics can undergo multiple cycles of re-melting in injection molding machines or extruders to produce new plastic articles. However, reusing is contingent upon the availability of individual plastic raw materials for the machine, excluding mixtures of different plastics. Sadly, in practical scenarios, the majority of plastic waste is of a mixed nature. This necessitates the separation of individual plastics before reusing. Various techniques exist for the separation of plastics, including methods based on density differences,4–7 selective dissolution,8–11 and triboelectric separation.12–14 These techniques are available for enabling recycling practices for the mixed plastic waste. However, the majority of the methodologies enumerated in the literature exhibit inherent limitations, such as elevated costs, deleterious emissions of solvents, and suboptimal separation efficiency. A recent innovation in separation techniques, specifically grounded in the sink-float principle for plastics, has been introduced.15–18 In the sink-float methodology, the alteration of the surface energy of plastic is facilitated through the adsorption of a chemical depressant agent onto the surface of the targeted plastic(s). 1 Upon descent of the treated plastic mixture into a flotation tank pre-filled with a flotation medium (typically water), plastics with similar or closely aligned surface energy to the surface tension of the medium precipitate to the bottom of the flotation tank. Conversely, plastics with disparate and lower surface energies remain buoyant on the medium’s surface. The efficacy of the sink-float behavior of a plastic particle is contingent upon various factors, irrespective of the depressant employed, encompassing parameters such as size, shape, surface characteristics, and density of the plastic particle. 19 The separation of acrylonitrile butadiene styrene (ABS), polycarbonates (PC), and polyoxymethylene (POM),1,15–17,20 polyethylene terephthalate (PET), polystyrene (PS), and polyvinyl chloride (PVC),2,16–17,21 POM, PS, and PC, 22 PVC and PET, 23 PET, POM, and PVC, 24 high-impact polystyrene (HIPS), PET, ABS, PVC, and PC,25–29 high-density polyethylene (HDPE), PVC, and PET, 30 ABS and PS,31,32 PS,PET,PC,PS, and PVC, 33 PS,PMMA,PET, and PVC, 34 PET, PVC, and PE, 35 and ABS and HIPS 36 via the sink-float technique have been extensively investigated using various depressants.
Within the scope of surface engineering, microwave irradiation emerges as a valuable modality for modifying the surface characteristics of solid materials. Operating within the frequency range of 300 MHz to 300 GHz and possessing wavelengths of 0.001 to 1 m, 37 microwave irradiation induces alterations in surface roughness, concentration, and type of functional groups affixed to the solid (plastic) surface.1,2,15,38 Moreover, microwave irradiation finds applicability in rubber recycling by disrupting crosslinks between rubber macromolecules in cured rubber.39–41 To the best of our knowledge, the extant literature on sink-float plastic separation techniques with the aid of microwave irradiation is limited.1,2,15,42–45 Existing studies predominantly focus on a restricted subset of plastics without exploring diverse depressants. Notably, polyvinyl chloride (PVC), polystyrene (PS), and polyethylene terephthalate (PET) are ubiquitous plastics in both industrial and household waste streams. As thermoplastics, their amenability to efficient separation from mixed waste streams renders them ideal candidates for reusing and recycling strategies.
In this investigation, the surfaces of the aforementioned plastic substrates were subjected to microwave pre-irradiation across varying microwave intensity levels ranging from 20% to 100%. Subsequent to pre-irradiation, these surfaces underwent treatment with select ubiquitous dual depressants, namely polyvinyl alcohol (PVA), methylcellulose (MC), polyethylene glycol (PEG), and tannic acid (TA), at concentrations spanning from 200 to 1600 mg/L, prior to their introduction into the flotation tank. The outcomes of the sink-float analysis were meticulously documented and thoroughly discussed, with a primary focus on addressing the implications for the effective management of plastic waste on a commercial scale. It is noteworthy that all employed depressants are characterized by their non-toxic nature and safety for human use. The majority of these compounds are recognized as food additives, exemplified by polyvinyl alcohol (PVA). Comprehensive assessments of sub-chronic toxicity and genotoxicity corroborate the safety of PVA for human exposure through various pathways in routine daily encounters.
Experimental
Materials
The key characterization of the used plastics, polystyrene (PS), polyvinyl chloride (PVC), and polyethylene terephthalate (PET).

Equipment and testing procedure
The pre-microwave irradiation of the plastic surfaces was conducted using a 30 L laboratory microwave oven (GOSONIC, GMO-530, Turkey) with a frequency of 2000 MHz and a maximum output power of 900 W (100%). The flotation tank and experimental procedures have been previously elucidated.1,2,15 In summary, 20 granules from each plastic type (a total of 60 granules) were pre-irradiated in the microwave oven for 20 seconds at specified output powers. Excessive irradiation was deliberately avoided to prevent an elevation in granule temperature, mitigating the risk of plastic deformation. Following microwave irradiation, the granules were treated with a selected dual depressants system (50-50 wt%) at various concentrations for 30 min before being introduced into the flotation tank. The flotation medium consisted of tap water with a pH of 6.5. After the 3-minute interval, the granules that sank and floated were collected from the flotation tank, and the results were recorded. The flotation tank, constructed from glass, featured a square base measuring 20 × 20 cm and a height of 80 cm filled up to 50 cm with tap water. Additionally, it was equipped with an air-blowing system at the tank bottom, providing an air bubble flow rate of 4 L/min. To assess the adsorption status of the depressant on the plastic surfaces, attenuated total reflectance Fourier transform infrared spectroscopy (ATR-FTIR) analysis was employed, utilizing an AVATAR 370FT-IR instrument equipped with a high-throughput ZnSe ATR crystal. The instrument's resolution was 4
Results and discussion
Plastic surface hydrophobic-hydrophilic properties
As elucidated earlier, the sink-float behavior of plastic particles in a liquid medium encompasses various influential parameters, including the chemical structure of the depressant, plastic particle dimensions and morphology, air bubble characteristics such as flow rate and size distribution, and the surface morphology of the plastic. 46 The selection of an appropriate depressant necessitates consideration of factors such as chemical structural compatibility with the plastic, commercial availability, cost-effectiveness, and safety considerations. In this study, attention was given to all aforementioned parameters in the choice of depressants. The surface properties of plastic materials such as roughness, as well as the type and concentration of functional groups affixed to the surface, hold significant importance in determining the sink-float behavior of plastic particles. Microwave irradiation emerged as a cost-effective methodology for modifying plastic surfaces.47–50 Additionally, it merits attention as an environmentally friendly technique.
Microwave irradiation has been documented to induce morphological changes, specifically in the surface properties of plastic materials.1,2 The magnitude and nature of these alterations play a key role in determining the ease or difficulty with which a depressant can adsorb onto the plastic surface. The hydrophobic-hydrophilic properties of a solid surface are intricately linked to its surface morphology. To evaluate the influence of pre-microwave irradiation on the aforementioned properties, contact angles (θ) between water droplets and the surfaces of four polystyrene (PS) samples were systematically quantified. The PS samples encompassed those lacking prior microwave pre-irradiation, those subjected to pre-irradiation at 100% output power (intensity level), and irradiated samples treated with a dual system of TA-PVA at two distinct concentrations, 800 mg/L and 1600 mg/L (Figure 1). Microwave irradiation was performed for a duration of 20 seconds. Concurrently, the plastic surfaces underwent immersion in a depressant solution at ambient temperature for 30 minutes. The untreated polystyrene (PS) sample exhibited a contact angle (θ) value of 91 degrees (Figure 1(a)), denoting the hydrophobic nature of the PS surface, as the angle surpassed 90 degrees. Following microwave irradiation, this angle decreased to 88.4 degrees (Figure 1(b)), indicating an augmentation in the hydrophilic property of the PS surface. Subsequent treatment of the irradiated PS surface with the dual system of TA-PVA resulted in further reduction of contact angles to 72.8 and 70.8 degrees for TA-PVA concentrations of 800 and 1600 mg/L, respectively. Evidently, microwave irradiation proved advantageous for enhancing the hydrophilic characteristics of the plastic surface. Unexpectedly, Figure 5 illustrates the flotation percentages of PS increased from 55% to 100% for unconditioned PVA-TA samples without and with microwave irradiation, respectively. It is apparent that, despite the marginal improvement in the hydrophilic property of the plastic surface after microwave irradiation, it adversely affected plastic flotation. This observation serves as evidence that factors beyond hydrophilicity, such as surface morphology, particularly roughness, may exert an influence on plastic flotation in aqueous medium.
34
Furthermore, an increase in TA-PVA concentration exhibited a positive impact on the hydrophilic traits of the PS surface. This phenomenon can be ascribed to the presence of O-H groups from the adsorbed polyvinyl alcohol (PVA) and tannic acid (TA) on the PS surface, contributing to its heightened hydrophilicity. Additionally, Figure 5 for the conditioned PS samples with PVA-TA solution demonstrates that the contact angle findings align with the sink-float results. The flotation percentages for the mentioned samples at two different PVA-TA concentrations of 800 and 1600 ml/L were 35% and 0%, respectively. The decrease in flotation percentage signifies an increase in sink percentage and consequently an elevation in the hydrophilic property of the plastic. Figures 2(a) to 2(d) present scanning electron microscope (SEM) images depicting the surface morphology of the aforementioned PS samples. A comparative analysis between Figures 2(a) and 2(b), representing unconditioned PS samples with PVA-TA solution, reveals the effectiveness of microwave irradiation on the surface morphology of the studied polystyrene. The surface roughness underwent inherent changes following irradiation, as evidenced by Figure 2(b) displaying a relatively roughened plastic surface post-microwave irradiation. However, upon conditioning with two distinct PVA-TA concentrations, the surface exhibited increased roughness, with varying degrees depending on the adsorbed PVA-TA values on the PS surface (Figures 2(c) and 2(d)). The additional roughness observed can be attributed to the presence of adsorbed depressants, namely PVA and TA. The contact angles ( The SEM images for four samples of polystyrene (PS) a) without microwave irradiation b)with 100% output power microwave irradiation c) conditioned with PVA-TA ( 800 mg/L) at 100% output power microwave irradiation d) conditioned with PVA-TA ( 1600 mg/L) at 100% output power microwave irradiation. The microwave irradiation was done for 20 s, Also, the plastic surface kept for 30 min in depressant solution at ambient temperature for images c and d.

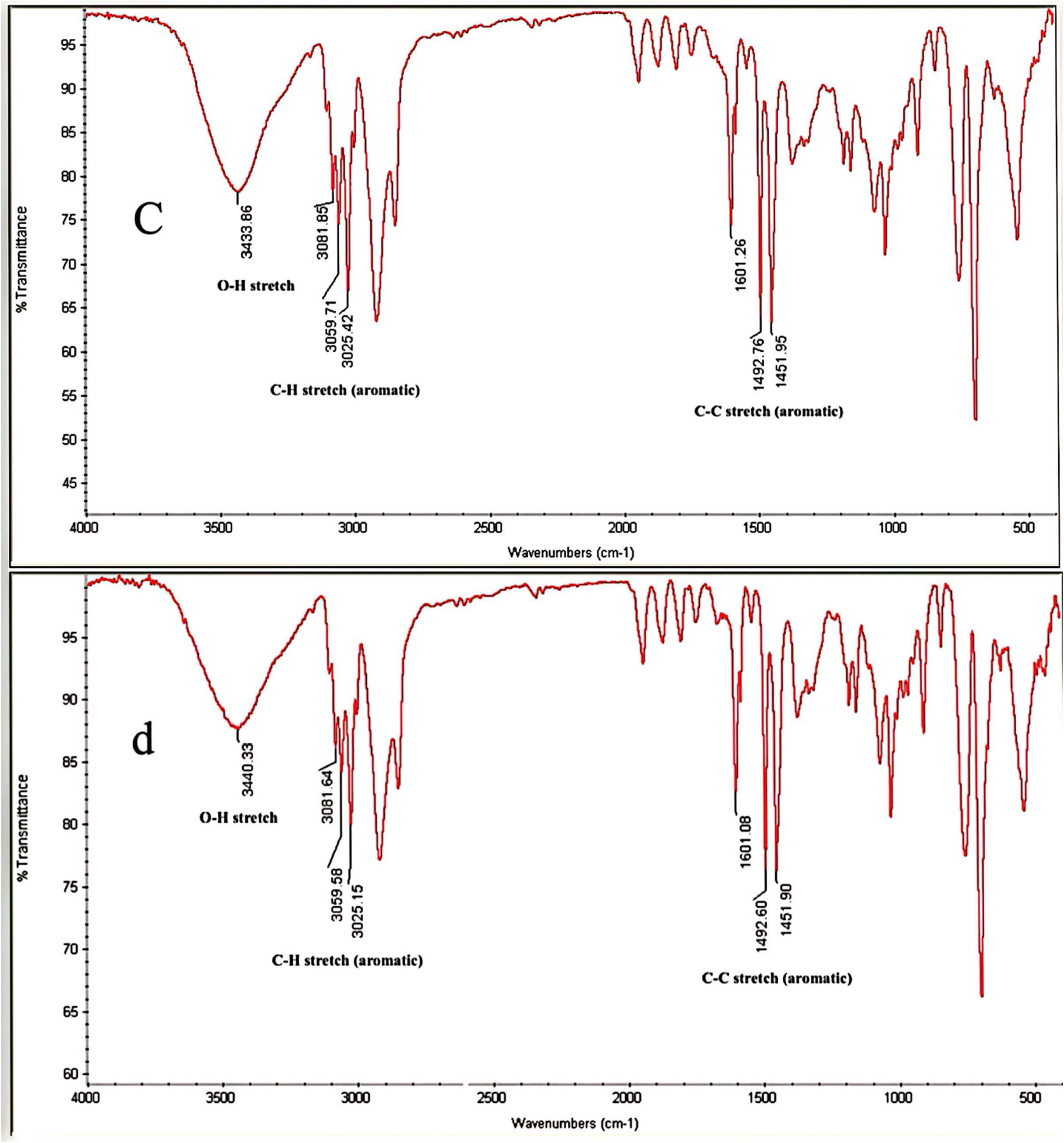
Consistent with the findings of contact angle measurements, the heightened hydrophilicity of the aforementioned plastic surfaces can be elucidated through the results obtained from attenuated total reflectance-Fourier transform infrared spectroscopy (ATR-FTIR) analyses of the plastic samples. Figure 3 illustrates the ATR-FTIR spectra for PS without microwave irradiation (a) and with 100% output power irradiation (b). A comparative examination of the spectra indicates that microwave irradiation induced alterations in the concentration and types of functional groups. The intensity (absorbance) of several groups underwent changes post-irradiation. Notably, the intensities of three specific peaks corresponding to C-C stretch in the aromatic ring between 1452 to 1602 The ATR-FTIR spectra comparison of polystyrene (PS) samples without (a) and with 100% output power microwave irradiation (b) The microwave irradiation was done for 20 s. The ATR-FTIR spectra comparison of polystyrene (PS) samples, with 100% output power microwave irradiation conditioned with PVA-TA at 800 (c) and 1600 mg/L (d) at 100% output power microwave irradiation. The microwave irradiation was done for 20 s, Also, the plastic surface kept for 30 min in depressant solution at ambient temperature.
The sink-float of the studied plastics
The influence of microwave irradiation on plastic surfaces is elucidated through varying microwave output power levels.1,2 Figures 5, 6, 7, 8, 9, 10 depict the flotation percentages of PVC, PS, and PET at different concentrations of dual depressant systems, namely PVA-TA, MC-TA, PEG-TA, PEG-PVA, MC-PEG, and MC-PVA, across various microwave output power levels (%). The pH of the flotation tank medium was maintained at 6.5, and when applicable, plastic surfaces underwent pre-irradiation with microwaves for 20 seconds before being introduced into the flotation tank. The microwave output powers were set at 20, 40, 60, 80, and 100%, with depressant concentrations adjusted to 200, 400, 800, 1200, and 1600 mg/L. Table 1 provides the densities of PET, PVC, and PS as 1.40, 1.35, and 0.98 g/cm³, respectively, and the surface energies as 44.6, 41.5, and 40.7 mN/m, respectively. Surprisingly, the sink-float observations (Figures 5, 6, 7, 8, 9, 10) revealed that, based on density alone, it was expected that PET and PVC would sink while PS would float. However, 30% of PVC floated, and 70% sank to the bottom of the flotation tank. For PS, the flotation percentage was 55%, indicating that 45% sank to the bottom. The polyvinylchloride (PVC), polystyrene (PS), and polyethylene terephthalate (PET).flotation (%) at different concentrations of PVA-TA for various microwave output power levels (%). The flotation tank medium pH was kept as 6.5 at ambient temperature. The microwave irradiation was done for 20 s, Also, the plastic surface kept for 30 min in depressant solution. The polyvinylchloride (PVC), polystyrene (PS), and polyethylene terephthalate (PET).flotation (%) at different concentrations of MC-TA for various microwave output power levels (%). The flotation tank medium pH was kept as 6.5 at ambient temperature. The microwave irradiation was done for 20 s, Also, the plastic surface kept for 30 min in depressant solution. The polyvinylchloride (PVC), polystyrene (PS), and polyethylene terephthalate (PET).flotation (%) at different concentrations of PEG-TA for various microwave output power levels (%). The flotation tank medium pH was kept as 6.5 at ambient temperature. The microwave irradiation was done for 20 s, Also, the plastic surface kept for 30 min in depressant solution. The polyvinylchloride (PVC), polystyrene (PS), and polyethylene terephthalate (PET).flotation (%) at different concentrations of PEG-PVA for various microwave output power levels (%). The flotation tank medium pH was kept as 6.5 at ambient temperature. The microwave irradiation was done for 20 s, Also, the plastic surface kept for 30 min in depressant solution. The polyvinylchloride (PVC), polystyrene (PS), and polyethylene terephthalate (PET).flotation (%) at different concentrations of MC-PEG for various microwave output power levels (%). The flotation tank medium pH was kept as 6.5 at ambient temperature. The microwave irradiation was done for 20 s, Also, the plastic surface kept for 30 min in depressant solution. The polyvinylchloride (PVC), polystyrene (PS), and polyethylene terephthalate (PET).flotation (%) at different concentrations of ME-PVA for various microwave output power levels (%). The flotation tank medium pH was kept as 6.5 at ambient temperature. The microwave irradiation was done for 20 s, Also, the plastic surface kept for 30 min in depressant solution.





In contrast, if surface energy were the predominant factor governing sink-float behavior, considering the substantial difference between the surface tension of water (72 mN/m) and that of the studied plastics, it would be anticipated that all plastics should float. Unexpectedly, the sink-float observations (Figures 5, 6, 7, 8, 9, 10) demonstrated that 100% of PET sank to the bottom of the flotation tank. This suggests that, while plastic density and surface energy play significant roles in predicting sink-float behavior, other factors are evidently involved in this phenomenon.
Figures 5, 6, 7, 8, 9, 10 distinctly illustrate that pre-microwave irradiation, without the use of a depressant, at various microwave output powers exerts no discernible influence on the flotation behavior of PET. Conversely, it is effective in the case of other plastics, namely PVC and PS. The flotation percentages for PVC and PS range from 30-80% at microwave irradiation output powers of 0 and 60%, and 55-100% at microwave irradiation output powers of 0, 80, and 100%, respectively. Figure 5 specifically demonstrates that conditioning PVC and PS with a dual system of polyvinyl alcohol-tannic acid (PVA-TA), in conjunction with pre-microwave irradiation, exerts notable effects on their flotation. Notably, PVC achieves the lowest flotation values of 5% and 10%, and even at a PVA-TA concentration of 400 mg/L, it reaches 0%, causing all PVC granules to settle at the bottom of the flotation tank. For PS, the corresponding values range from 100% flotation at a PVA-TA concentration of 200 mg/L for a microwave irradiation power of 40%, to lower values reaching 5% for various PVA-TA concentrations and microwave irradiation powers. Optimal separation results for PVC and PS are achieved at a PVA-TA concentration of 800 mg/L without pre-microwave irradiation, yielding 0% and 90% flotation for PVC and PS, respectively. In contrast to PS, PVC possesses inherent polarity, facilitating the adsorption of PVA and TA onto the plastic surface with greater ease. The hydroxyl (O-H) groups present in these depressants induce a heightened affinity for PVC, leading to its facile sinking in the aqueous medium. Once more, the combined system of methylcellulose (MC) and tannic acid (TA) demonstrated superior efficacy on PVC as opposed to PS, while manifesting negligible impact on PET (Figure 6). Flotation percentages for PVC ranged from 0 to 30% when pre-microwave irradiation was applied to the plastic surface. In the case of PS, flotation values varied between 30% and 85%, contingent upon the depressant concentration and the power of microwave irradiation (Figure 6). More or less, the scenario for the polyethylene glycol-tannic acid (PEG-TA) dual system mirrored that of previously investigated dual systems. However, a depressant concentration of 200 mg/L across all microwave irradiation powers exhibited heightened effectiveness for PVC, resulting in flotation values ranging from 0 to 5% (Figure 7). For PS, the highest and lowest flotation values were 100% and 5%, respectively. Interestingly, a 15% flotation was observed for PET at a PEG-TA concentration of 400 mg/L for 80% and 100% microwave irradiation powers. As depicted in Figure 9, the dual system of microwave-assisted methyl cellulose-polyethylene glycol (MC-PEG) exhibits diminished efficacy in influencing the flotation behavior of investigated plastic materials, particularly in comparison to previously examined dual systems within this text. This is observed for PS more clearly. Flotation percentages for PVC and PS vary between 5% and 80%, and 15% to 100%, respectively, across diverse microwave pre-irradiation power levels. Notably, PET does not exhibit any flotation percentage under the applied MC-PEG concentrations and pre-microwave irradiation powers. This outcome aligns with expectations, as the hydroxyl (O-H) group in none of the utilized depressants can be equated with tannic acid (TA). The dual system involving microwave-assisted methyl cellulose-polyvinyl alcohol (MC-PVA) (Figure 10) demonstrates similar results for MC-PEG but proves more effective on PVC than MC-PEG. Flotation percentages for PVC and PS were 5% to 80% and 50% to 100%, respectively, across various employed microwave pre-irradiation powers. Once again, PET does not exhibit any flotation percentage for the applied MC-PVA concentration and pre-microwave irradiation powers.
Overall, the results reveal varying flotation percentages dependent on plastic-type, microwave output power, and depressant type and concentration. No discernible regular trend is observed. The study concludes that the sink-float behavior of plastic is influenced by several parameters, including the adsorption phenomenon of a depressant on the plastic's surface. To comprehend this phenomenon, the desorption-adsorption mechanism of a chemical on a solid surface must be meticulously investigated.51-52 Adsorption of a chemical on a solid (polymer) surface is a surface phenomenon resulting from the accumulation of a chemical film on the solid surface. The desorption-adsorption process involves considerations such as: (A) The rate of adsorption and desorption of the chemical on and from the solid surface (B) The availability and number of suitable active sites on the solid surface (C) Direct and indirect interactions between adjacent molecules of the adsorbed chemical and with molecules on the solid surface (D) The enthalpy of adsorption and the difference between the enthalpy of adsorption of different effective sites (E) The equilibrium constant of the desorption and adsorption processes (F) The Gibbs free energy of the adsorption process and its negative sign for spontaneous adsorption (G) The mono or multilayer of the adsorbed chemical on the solid surface
In light of the findings of this study, the authors believe that microwave pre-irradiation alters the mono or multilayer capacity of the adsorbed depressant, as well as the numbers and types of active sites on the plastic surface, in diverse ways contingent upon the operating conditions. This alteration in the aforementioned parameters modifies the sign of the entropy of the system. Consequently, the sign of the Gibbs free energy also changes, resulting in spontaneous adsorption and/or desorption processes. However, additional studies are warranted to validate these conclusions in the future.
design of experiment
The suggested equations by a design of experiment software (Design-Expert


(a) The three-dimensional (3D) response surfaces to evaluate the interactive effect of microwave power % (P) and depressant concentration (PEG- PVA mg/L) on the floatation % of the PS. (b) The predicted values by software versus actual value for the same system.
The predicted flotation % is obtained through arbitrary substitution of C and P in the aforementioned equation. Figure 11(a) graphically represents this process. Additionally, Figure 11(b) contrasts the actual flotation % of conditioned PS with PEG-PVA at various concentrations and microwave output powers with the predicted values (Table 2) generated by Design-Expert® software. Notably, the predicted values exhibit reasonable concordance with the actual values.
Conclusion
Microwave pre-irradiation exerts a notable influence on the sink-float behavior of the investigated plastics, except for PET. No discernible regular trend is observed for plastic floatability with increasing microwave output power. Instead, this parameter's value is contingent on the irradiation level and depressant concentration. The employed dual depressants effectively adsorb onto the surfaces of the examined plastics, altering their hydrophilic-hydrophobic properties. These findings are substantiated by routine characterization techniques such as SEM, ATR-FTIR, and contact angle measurement. Equations derived through Design-Expert® software demonstrate satisfactory agreement between predicted and actual plastic flotation values.
Footnotes
Acknowledgements
The authors sincerely thank the staffs of the polymer chemistry laboratory located at the faculty of science, Ferdowsi University of Mashhad for sincere cooperation. Approval no. 3/58904.
Author contributions
Salva Jabbari, Saeed Ostad Movahed, and Shahab Jourabchi designed the experiments. Dr. Saeed Ostad Movahed prepared the manuscript with contributions from all co-authors. The authors applied the SDC approach for the sequence of authors.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Data availability statement
The datasets generated during and/or analysed during the current study are available from the corresponding author on reasonable request.
