Abstract
The non-uniform structures of coal deposits make potential well distribution on the coal surface inhomogeneous. Deep potential wells adsorb methane molecules more easily than shallow potential wells. The methane adsorption heat release differs according to the depths of the potential wells. During isobaric adsorption, the adsorption heat in the high-temperature stage is significantly higher than that in the low-temperature stage. A lower adsorption pressure results in greater adsorption heat variation during a temperature increase. During the isothermal adsorption process, the adsorption heat is higher in the low-pressure stage, with the preferential adsorption characteristics of deep potential wells.
Keywords
Introduction
An adsorption potential well refers to a location with lower potential energy on the coal surface, which exhibits methane adsorption ability (Gürdal and Yalcin, 2001; Wang et al., 1996, 2013). The adsorption potential well depth is related to the mutual attraction between coal and methane (Ma et al., 2011a, 2011b). Owing to the variety of the oxygen-containing functional groups (Lu et al., 2014; Zhong, 2004) and side chains on the coal surface (Liu and Feng, 2012; Zhou et al., 2017a), as well as the fractal characteristics of coal surface morphology (Liu et al., 2003; Zhou et al., 2017b), the methane adsorption potential wells on natural coal surfaces exhibit obvious heterogeneity (Karacan, 2000; Wang, 2000). Similar to potential energy, adsorption sites with different adsorption capacities form rough equipotential adsorption potential surfaces (Fu et al., 2005). The sunken part is known as the potential well, and the depression potential energy is referred to as the potential well depth (Figure 1). Obviously, the depth of the potential wells (−ε) on the coal surface is inhomogeneous. By means of molecular simulation, Jiang et al. (2007) found that the methane adsorption capacities of the various oxygen functional groups and side chains differ, and their potential well depths range from 4 to 9 kJ/mol. Certain experimental measurements of the methane adsorption heat values were 0–30 kJ/mol (Guo and Wu, 2008; Lui et al., 2003; Zhang et al., 2004). According to the experiment conducted by Zhao et al. (2011), the methane adsorption amount in coal is highly sensitive to temperature variation at 303–543 K. These phenomena indicate that there are not only certain adsorption potential wells with depths far greater than the methane molecule energy, but also numerous adsorption potential wells with depths close to the methane molecule energy in coal. From calculations, Nie and Duan (1998) found that the potential well depth on the coal surface agrees with the experimentally measured adsorption heat values.

Equipotential line of solid surface.
Coalbed methane (CBM) is an unconventional natural gas that exists in coal reservoirs. Methane adsorption in coal is dependent on two factors: the adsorption potential well parameters (potential well depth and number of potential wells) and methane gas state parameters (such as adsorption pressure and temperature). CBM thermal recovery technology has been developed in recent years. In the CBM thermal recovery process, the variation rules of the methane adsorption heat of coal are not clear, which makes accurate evaluation of CBM thermal recovery efficiency challenging. Therefore, the variation law of the adsorption heat of methane and coal with inhomogeneous potential wells is studied in this paper. This will provide a new theoretical basis and application foundation for the accurate evaluation of CBM enrichment characteristics, CBM production and exploitation efficiency improvement, and popularisation of new methods and technology for CBM exploitation.
Kinetic process of methane adsorption by coal
In 1916, the state equation for monolayer adsorption was deduced by Langmuir, based on statistical thermodynamics (Yan and Zhang, 1979). The temperature-dependent adsorption parameters were obtained as follows
Compared to
Then, substituting b into the Langmuir equation
From equations (2) and (3), kT is a constant value under isothermal conditions. Furthermore, the adsorption parameter b of the deep adsorption potential wells (
From a microscopic viewpoint, methane adsorption in coal is the process of the potential energy of methane molecules being converted to coal surface energy and thermal energy by means of collisions between the methane molecules and adsorption site. In heterogeneous potential wells, when an adsorption site has a potential well depth that is smaller than the energy of the collided methane molecule, the adsorption site cannot trap the methane molecule, and adsorption cannot occur. However, when an adsorption site has a potential well depth that is larger than the energy of the collided methane molecule, the adsorption site can trap the methane molecule, and adsorption can occur, while the coal surface energy is reduced, and the remaining energy is converted into heat energy and released. Therefore, methane adsorption in coal is an exothermic process. Moreover, as the distributions of potential wells differ, the heat released from the equivalent methane adsorption varies.
Relationship between adsorption site coverage and adsorption heat
During methane adsorption in coal, the coverage of the heterogeneous potential wells on the coal surface mainly depends on the temperature and adsorption pressure. For all adsorption sites with a potential well depth of −ε on the coal surface, the coverage rate θ-ε is determined by the Langmuir equation
Substituting equation (1) into equation (4), we obtain
From equation (5), in the coal and methane adsorption equilibrium, the coverage of the potential wells with different depths obeys the law of the logistic (S) curve with the parameters of adsorption pressure p and temperature T. It should be noted that not all of the adsorption potential wells can adsorb methane molecules under conditions of any temperature and pressure (p, T), as illustrated in Figure 2. Under conditions of any temperature and adsorption pressure, deep potential wells (ε = 25–30 kJ/mol) exhibit nearly saturation adsorption, while shallow potential wells can scarcely undergo adsorption, and the coverage of medium-depth potential wells (ε = 5–25 kJ/mol) increases as the depth of the potential wells increases. When the adsorption pressure increases or temperature decreases, the adsorption of the adsorption sites occurs from the deep to shallow potential wells. Conversely, when the adsorption pressure or temperature decreases, methane desorption of the adsorption sites occurs from the shallow to deep potential wells.

Coverage rate of (a) isothermal and (b) isobaric adsorption at different potential well depths.
Experimental study on adsorption characteristics of heterogeneous potential wells
The test systems for measuring methane adsorption by coal at high temperatures include an inflation device or stable system, precise heating device, adsorption chamber, methane storage cylinder and system for gas measurement. The inflation device or stable system is used to load the methane adsorption container, and the axial pressure is controlled by the stable device. The precise heating device used for heating the adsorption container has an accuracy of 0.1 K and the maximum attainable heat is 873 K. The dimensions of the cylindrical coal mass sample taken from the 3# coal seam (Tunliu mine, Lu'an Mining Area) were 100 mm (diameter) × 150 mm (length). The internal dimensions of the stainless steel absorption container were the same as those of the coal sample, and it was connected to both the methane storage cylinder and gas measurement system by means of a steel pipe with an internal diameter of 4 mm. The experimental system principles are presented in Figure 3. The equipment is thermally stable and can be used in a controlled environment; therefore, very high accuracy is provided by the temperature supply and measurement. The system operates from room temperature (RT) to 573 K.

Schematic of test system.
In order to study the methane adsorption characteristics in coal with inhomogeneous potential wells, isothermal and isobaric adsorption experiments at different temperatures were carried out by the test systems. Seven different temperature points between 333 and 513 K were set in the experiment. In the isothermal adsorption experiment, methane was injected into the adsorption container at the required pressure after achieving a vacuum. The injection valve was closed and the container was heated at the adsorption equilibrium gas pressure. Each temperature stage lasted for 6 h after achieving the stabilised methane pressure before moving to the next stage. Five gas pressure points were recorded at each temperature point. In the isobaric adsorption experiment, the constant desorption pressure was set after adsorption equilibrium at 333 K, following which methane desorption was carried out under each temperature point. During this process, each temperature point was maintained above 8 h, and the methane desorption equilibrium was reached when the pressure was maintained constant. The methane adsorption amount was calculated by measuring the initial adsorption amount and methane desorption amount under a constant pressure at different temperature points.
Phenomenon of coal-adsorbing methane adsorption in coal with heterogeneous potential wells
The adsorption amount, temperature and adsorption pressure of the isothermal and isobaric adsorption experiments are displayed in Table 1. Firstly, the isothermal adsorption process at different temperatures was analysed. The curve fitting of the isothermal experiment data is displayed in Table 2, based on the Langmuir formula (equation (4)). It is indicated that, in the temperature range from 333 to 513 K, the lean coal adsorption capacity during isothermal methane adsorption increased with an increasing adsorption pressure, and the increasing rate slowed. The adsorption curve is similar to the Langmuir curve. The parameter a represents the maximum value of the methane adsorption amount (L) in coal under different temperature conditions by increasing the adsorption pressure. As opposed to the ideal Langmuir model, the fitting parameter a for the methane adsorption of the natural lean coal with inhomogeneous potential wells decreases with an increasing temperature. It is demonstrated that, at any temperature in this experiment, potential wells with depths less than the methane molecular energy existed on the coal surface, which could not absorb methane molecules by increasing the adsorption pressure. With the temperature increase, the average kinetic energy of the methane molecules increased, and the number of potential wells with depths less than the methane molecule energy increased, so that the maximum coal adsorption capacity (parameter a) decreased.
Adsorption capacity and pressure of methane in coal at different temperatures.

Curve fitting of adsorption capacity and pressure of methane at different temperatures.
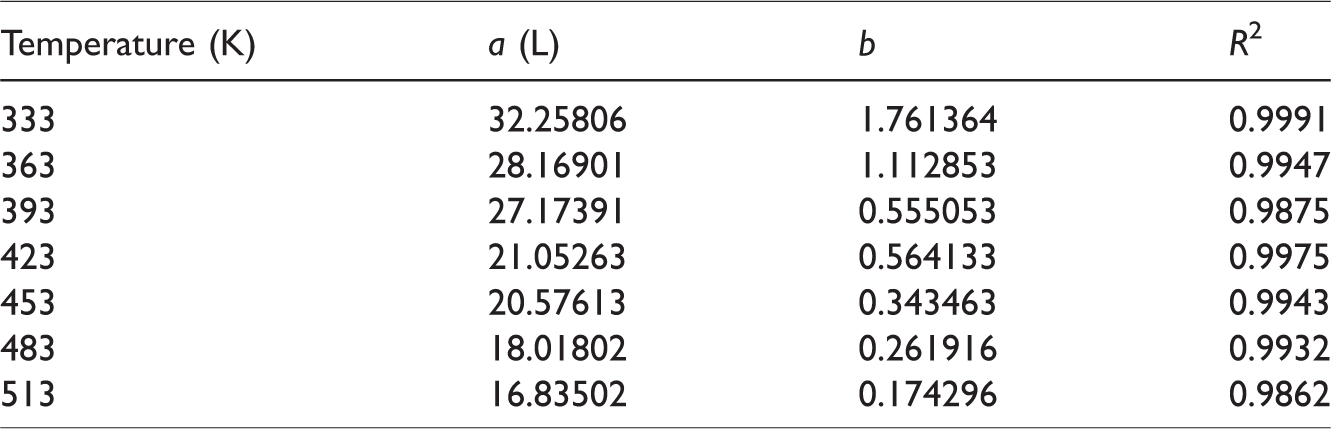
The methane adsorption amount variation with the temperature increase in the isobaric adsorption experiment is illustrated in Figure 4. It is indicated that the coal methane adsorption amount decreased with the temperature increase. When the adsorption pressure changed from 0.1 to 0.7 MPa, the adsorption amount variation decreased with the temperature increase; that is, the sensitivity of the methane adsorption amount to the adsorption pressure decreased with an increasing temperature.
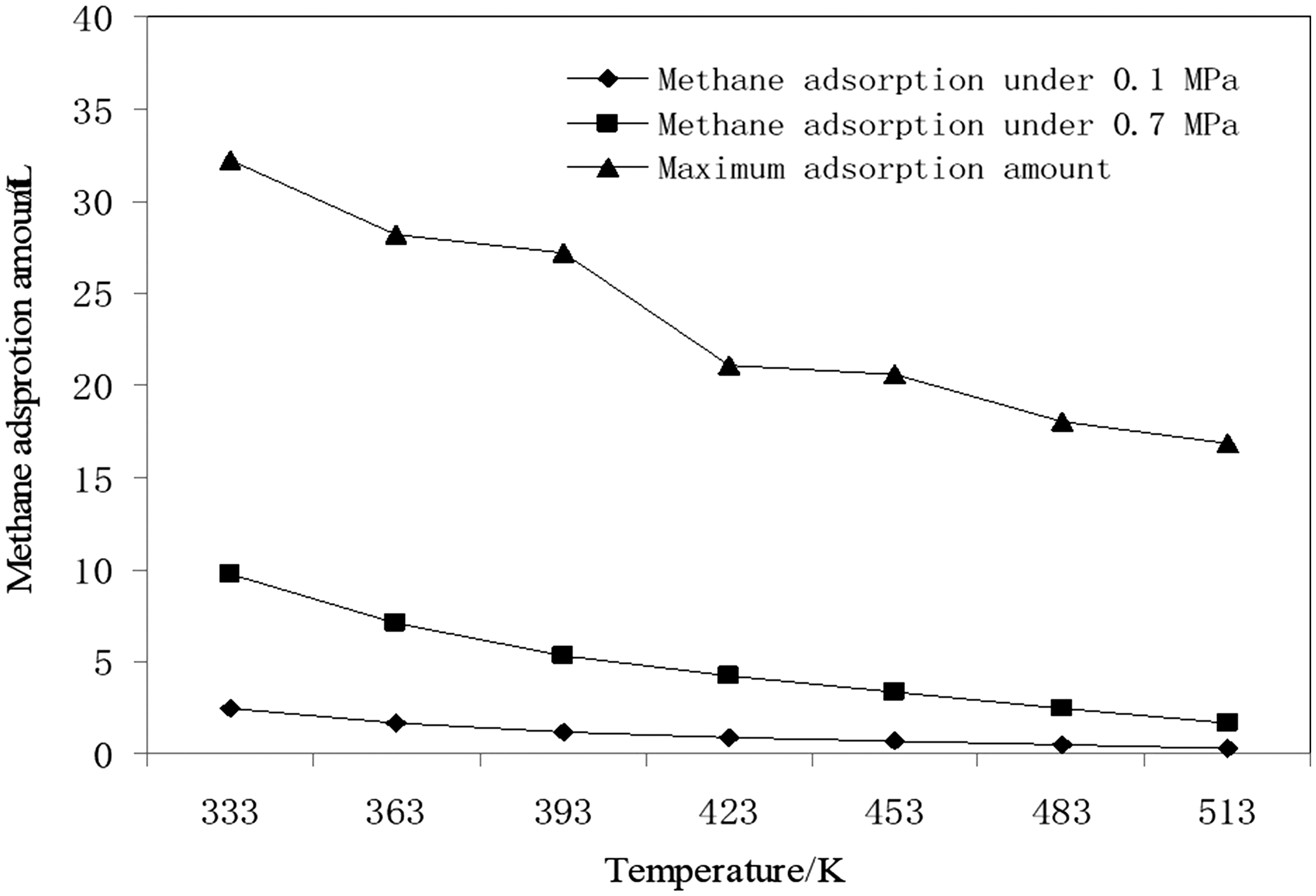
Variation of adsorption capacities with temperature at different adsorption pressures.
This is one of the main reasons for the low efficiency of CBM extraction by means of adsorption pressure reduction. For methane molecules that are tightly bounded in deep potential wells, only changing the methane molecule energy distribution through increasing the temperature by means of the CBM thermal recovery technology can effectively cause their adsorption state to change.
Influence of temperature and adsorption pressure on coal and methane adsorption heat
Owing to the differences in the adsorption sites of potential well depths and coverage priority, the methane adsorption heat in coal varies with changes in the system temperature T and adsorption pressure p. In experimental analysis, the influence of temperature and adsorption pressure on the adsorption heat can be analysed by isobaric and isothermal adsorption experiments, respectively. However, the total number of adsorption sites in a natural coal surface cannot be determined experimentally. Using the data in Table 1, the maximum adsorption amount at 333 K was regarded as the saturated methane adsorption amount. The isothermal and isobaric adsorption processes were curve fitted in stages, respectively, in order to calculate the adsorption heat.
Adsorption heat variation in isobaric adsorption process
The influence of temperature on adsorption heat can be analysed by curve fitting in stages during the isobaric adsorption experiment. As illustrated in Figure 5, the temperature section of the 333 to 423 K was regard as the low-temperature stage of the isobaric adsorption experiment. Conversely, the temperature section of 523 to 513 K was regarded as the high-temperature stage of the isobaric adsorption experiment. From equation (2)


Curve fitting of isobaric adsorption of methane at different temperatures.
Here,
Curve fitting of isosteric at different temperatures and adsorption heats.
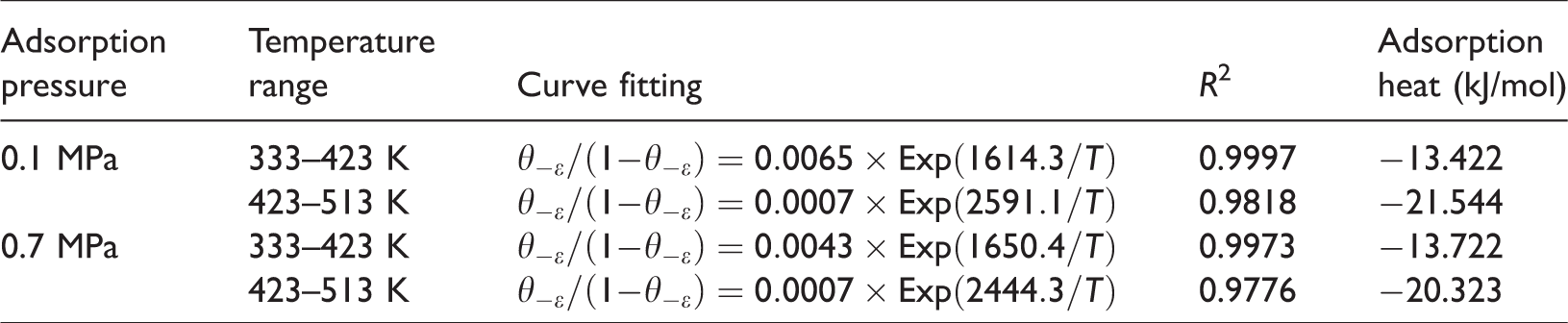
2. Adsorption heat variation in isothermal adsorption process
The influence of adsorption pressure on adsorption heat can be analysed by the curve fitting in stages of the isothermal adsorption experiment. From equation (4)

As illustrated in Figure 6, the adsorption pressure section from the first to third measuring points was regarded as the low adsorption pressure stage of the isothermal adsorption experiment. Conversely, the adsorption pressure section from the third to fifth measuring points was regarded as the high adsorption pressure stage of the isothermal adsorption experiment. The adsorption heat of the different adsorption pressure stages can be obtained by substituting the curve fittings and temperature T into equation (1).

Curve fitting of isothermal adsorption at different adsorption pressures.
The proportionality constant bm should be determined in the adsorption heat calculation. As a constant that is independent of the temperature and adsorption pressure, bm can be calculated by curve fitting of the b values (Table 2) under different isothermal conditions and corresponding temperature T. The calculation value of bm was 0.0032, which was applied to the calculations of the adsorption heat for all isothermal adsorption processes. The curve fitting and calculation results are displayed in Table 4.
Curve fitting of the adsorption capacity and pressure of methane in coal at different temperatures.

The curve fittings of the isothermal adsorption experiments exhibit high correlation coefficients. In the isothermal adsorption of 333 to 513 K, the calculated coal and methane adsorption heat ranged from 1.686 to 3.593 kJ/mol. Both the adsorption rate b of the coal and methane, and the adsorption heat in the low-pressure stage were significantly larger than that in the high-pressure stage. The differences in the adsorption heat in the different pressure stages ranged from 1.686 to 3.593 kJ/mol. It is indicated that, during isothermal adsorption, owing to the low coverage rate of the adsorption sites, the methane molecules were more likely to adsorb on the adsorption sites with deep potential wells during the low-pressure stage, so that the adsorption heat was higher. With an increase in the adsorption pressure, the coverage rate of the adsorption sites with shallow potential wells increased gradually, resulting in decreased adsorption heat.
Conclusions
For heterogeneous potential wells, methane adsorption in coal exhibits the following characteristics.
In the methane adsorption process, methane molecules prefer to be adsorbed by deep adsorption potential wells, rather than shallow adsorption potential wells. When the adsorption pressure increases or the temperature decreases, the adsorption occurs from the deep to shallow potential wells at the adsorption sites. Conversely, when the adsorption pressure or temperature decreases, methane desorption of the adsorption sites occurs from the shallow to the deep potential wells. Owing to the heterogeneity of the adsorption potential wells on the coal surface, a smaller adsorption amount results in a greater potential well depth being occupied by the adsorbed methane molecules, and the sensitivity of their adsorption states to adsorption pressure is weakened. During isobaric adsorption, the adsorption heat in the high-temperature stage is significantly higher than that in the low-temperature stage. A lower adsorption pressure results in greater adsorption heat variation during the temperature increase. During the isothermal adsorption process, the adsorption heat is higher in the low-pressure stage. As the adsorption pressure increases, the coverage rate of the adsorption sites with shallow potential wells increases gradually, leading to a decrease in the adsorption heat.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: the National Natural Science Foundation of China (General Program No. 21373146), Shanxi scholar support plan, Program for the Outstanding Innovative Teams of Higher Learning Institutions of Shanxi (2014), and excellent innovation project of graduate students in Shanxi (20150016).





