Abstract
Water pollution caused by emerging organic pollutants such as the halogenated organophosphate flame retardants, has exacerbated the issue of water scarcity. An eco-friendly technology, e.g., the adsorption technique, requires the use of affordable and safe adsorbents. Agricultural waste materials are promising adsorbents for wastewater remediation due to their relative abundance, biodegradability, non-toxic and cost-effective properties. However, most agricultural wastes are not ideal for adsorption in their raw forms and may require physical or chemical pre-treatments/modification, in order to increase the materials' compatibility and natural adsorption properties. Cellulose is an important constituent of plant residues that can be used as a precursor for the production of greener and sustainable industrial nanomaterials. Therefore, in this review, a discourse on the most recent approach/strategies for the modification of a range of agricultural wastes is presented. The result from their applications showed agricultural wastes has great prospects as adsorbents. The challenges encountered in the synthesis of plant-based adsorbents and the development of 3-D structured nanocomposites from cellulose, to circumvent these difficulties is extensively reviewed. Furthermore, the prospects in the application of cellulose beads/ZnO nanocomposites (CB-ZnO) derived from the maize tassel, for the adsorption of the halogenated organophosphate esters from aqueous matrices are presented in this review
Keywords
Introduction
Water is essential for the sustenance of all forms of life and it is a valuable resource for industrial, agricultural and economic development. 1 The rapid increase in industrial activities, population surge and other environmental variables have caused a huge impact on water supplies globally, particularly in the African continent, resulting in a water-stressed situation. 2 Between 22-34% of the population in eight countries in the sub-saharan Africa continent has access to clean drinking water and predictions show that by 2025, about half of the African continent would be affected by acute water shortage.2,3 The pollution of water bodies by hazardous organic pollutants, particularly by emerging chemicals of concern (ECC) tends to exacerbate the water scarcity issue. ECCs are a group of chemical compounds that have the potential to adversely affect humans and the quality of the environment. 4 Pharmaceutical products, food supplements, wood preservatives, washing detergents, surfactants, plasticizers, flame retardants, pesticides, natural and synthetic hormonal products and some disinfectants waste items are among the harmful chemicals released by households and industries. 5 On a worldwide basis, a vast number of ECCs are released into the environment via anthropogenic activities. Organophosphate flame retardants (OPFRs) are one of such synthetic chemicals of interest.
The OPFRs are synthetic chemical additives which, when incorporated into or deposited on the surface of a fabric, wood or plastics produce a protective barrier over them, thereby reducing fire incidences.6,7 They are chemicals with the same primary phosphate unit and heterogeneous substituents that act as flame retardants.8,9 The brominated flame retardants (BFRs) such as polybrominated diphenyl ethers (PBDEs) were one of the first flame retardants with effective fire-retardant characteristics produced by industries. Due to their bio-accumulative, toxic and long-range atmospheric transport properties, they were listed as chemical agents of concern at the Stockholm Convention. Thus, the OPFRs, also known as the organophosphate esters (OPEs), were produced as an alternative to the BFRs, with an increasing global output of 300,000 tonnes in 2004 to 680,000 tonnes in 2015.9–11 The OPFRs were initially thought to be of minor environmental concern and assumed to be degradable, with little environmental persistence and low potential for “long-range atmospheric transport” (LRAT). 12 However, recent scientific findings and evidence contradict these ideas in various instances. For example, OPEs and their metabolites have been found in both indoor and outdoor environments, in rural, urban and industrial areas, remote regions, in food samples and breast milk of women.13–15 Furthermore, several OPEs have been linked to tumour formation in some organs of the body.14,16 Therefore, wastewater remediation requires efficient treatment systems for the removal of such harmful contaminants.
Water treatment techniques, which include micro and ultra-filtration, distillation, solvent extraction, reverse osmosis, ion exchange, precipitation, coagulation, biological treatment (aerobic and anaerobic) and advanced oxidation processes, have been successfully applied in water purification.17,18 However, the recalcitrancy caused by some compounds like the OPFRs, as well as the high operational costs, fouling effect and the generation of toxic intermediate by-products among others, tend to limit their efficiency and frequency of usage.19–22 Adsorption technique is an established and widely used technology in water treatment systems. Its easy application, simplicity of design, availability of a large range of adsorbents and excellent water purification performance makes it one of the most important water and wastewater treatment option to date.18,19,23,24 Adsorption is a separation process that relies on the change in concentration of a specific component at the interface between two phases. 25 Substances in the liquid phase (adsorbate) interact with the surface of the solid phase (adsorbent) and adheres to the solid surface during this process. 26 The contaminant of concern is the adsorbate present in the fluid phase, while the solid phase is any material capable of interacting with the pollutant as an adsorbent. Adsorbents, in general, have particular spatial and electronic characteristics with various activation sites that favour adsorbate interaction and adsorption.27,28 There are three steps to the adsorption process: 1) adsorbate transport in the fluid phase to the adsorbent’s surface (film diffusion), 2) migration of adsorbate into the adsorbent’s pores (diffusion within particles) and 3) adhesion of contaminants onto the adsorbent’s charged surface (surface bonding). 29 The adsorption of molecules onto an adsorbent is influenced by some variables which include the initial contaminant concentration, solution pH, temperature, interfering chemicals, adsorbate characteristics, adsorbent dosage, adsorbent surface type, particle size and adsorbent’s surface area. 30 Several researchers have recently concentrated on the use of biosorbents from renewable and less expensive precursors such as agricultural plant residues, for water purification purposes. 30
Activated carbon has been developed from agricultural residues and used for the biosorption of pollutants.31–33 Biosorption allows contaminants to be temporarily bound to the cellular porous structure of a biomaterial. 34 The functional groups present in the polysaccharides of plant residues (e.g., carboxyl, sulfhydryl, hydroxyl groups), in Chitin (e.g., amino, amido, acetamido) and nucleic acid (e.g., phosphates), can bind and sequester metals in agriculture-based adsorbents.34–36 The absence of intermediate by-products throughout the treatment process is a significant benefit of biosorption over other techniques. Furthermore, the biosorption process is reversible and the adsorbent utilized may be desorbed-regenerated for future use.27,29 The majority of plant residues are often not useful as adsorbents in their raw forms, but rather require some form of pre-treatment/modification procedures. These are generally required to increase or improve the surface qualities of the final adsorbent materials and their compatibility for the adsorption of target contaminant. Modification could be physical (thermal treatment) or chemical (use of safe chemical reagents) and the process incorporates or increases functional groups onto the adsorbent surface. Generally, chemical modification technique is the most often used and includes the oxidation, esterification, etherification and polymerization reaction strategies. 37
Agricultural waste, chitosan, carbon nanotubes, and activated carbon have all been successfully converted into composites in order to improve their adsorption properties. Organic and inorganic hybrid composite materials provide an appealing new type of functional materials with increased optical, thermal and mechanical features as well as of low toxicity, due to the synergistic interactions between the inorganic and organic elements. Khan et al. 38 synthesized nanocomposites (S-PANI and S-PANI/MWCNTs) from Polyaniline and multi-walled carbon nanotubes (MWCNTs), in order to study their electrical conductivity retention under isothermal cyclic aging conditions. In the first stage, nanocomposite of PANI(EB)/MWCNTS was prepared by the in situ oxidative polymerization of aniline with MWCNTs, using 14.8 g of K2S2O8, 1 M HCl and aqueous ammonia (1 M) solution. The oxidant was added dropwise into the reaction mixture and continuously stirred for about 22 h. The second step was to create S-PANI and S-PANI/MWCNTs by treating the PANI(EB) and PANI(EB)/MWCNTs composites with H2SO4 (1 M) in an ice bath (0–5°C), while stirring continuously for 4 h. According to Khan et al., the As-prepared S-PANI/MWCNTs nanocomposite had stronger electrical conductivity and better isothermal stability in terms of electrical conductivity retention than PANI and S-PANI. In a related study, Khan et al. 39 examined the adsorption performance of silica gel-impregnated polyaniline nanocomposites as adsorbents for the removal of organic dye from their mixtures. The polyaniline-modified silica gel was created by employing potassium persulfate as an oxidizing agent and conducting a basic in situ oxidative polymerization of aniline with various amounts of silica gel. According to Khan et al., significant electrostatic interactions between the dye-dissociated sulfonate groups and the protonated amino groups of the PANI-based silica adsorbents made PANI@SG more efficient for the separation of a mixture of organic colours. They asserted that chemical modification through the incorporation of acidic or basic surface function moieties could improve the cationic and anionic chemical adsorption performance, and increase the adsorption of charged organic species such as dyes. Shariq et al. 40 reported the successful in-situ polymerization synthesis of polypyrrole (PPy) and polypyrrole/molybdenum oxide composite (PPy/MoO3) by utilizing anhydrous ferric chloride (FeCl3) as an oxidant in an aqueous system. The PPy and PPy/MoO3 were both transformed into pellets and assessed for their ammonia vapour-sensing capacity at ambient temperature toward 1 M, 0.5 M, and 0.1 M ammonia concentrations, The PPy/MoO3 sensor was shown to be more effective than the PPy sensor in terms of sensing response and reversibility. Its sensor’s sensing response at 1 M, 0.5 M and 0.1 M ammonia concentration was found to be 79.3%, 38.3% and 28.9% respectively and its reversibility was 98.2%, 95.1% and 90.6% respectively.
Most often, the various modification techniques deal with the total mass of the biomaterial with little consideration towards isolating the active component of the biomaterial responsible for the adsorption properties. The isolation and modification of the most active component are deemed crucial since this will eliminate substances that may suppress the adsorption properties of the biomaterial and hence increase its adsorption performance. Cellulose is one of the main constituents of plant residue and it can be extracted and chemically modified and utilized as a nanocomposite for adsorption purposes.41–43 Several plant biomass, unlike wood, have soft structures and low carbonization energy which could be an issue, especially during thermal treatment. Therefore, the subsequent modification and fabrication of cellulose fibre into cellulose bead composites is an efficient way to circumvent these technological barriers
The application of agricultural residues for the removal of OPFRs from water received little attention in the past decade because the focus was more on removing organophosphate pesticides. In addition, there is scarce and scattered information in the literature on the current modification strategies employed in the conversion of agricultural residues into composites and the development of 3-D cellulose bead nanocomposites suitable for the removal of OPFRs. Therefore, this review presents an in-depth discourse on the current strategies for the modification of a range of agricultural wastes as adsorbents suitable for the removal of OPFRs. The feasibility of adsorbents produced from agro-waste materials and modified with zinc oxide for the extraction of organophosphate chemicals from aqueous media is further explored. Highlights of the halogenated-OPEs and their environmental/health impacts are presented in this review. Furthermore, the synthesis and prospects of 3-D cellulose bead nanocomposites as an effective adsorbent for the removal of halogenated organophosphate esters from aqueous matrices are extensively discussed
The organophosphate flame retardants
OPEs are phosphoric acid derivatives that occur as tri-esters (trialkyl), diesters (alkyl diaryl) and polyphosphates. They are synthesized commercially by reacting phosphorus oxychloride (POCl3) with other reactants.8,9 The Organophosphate (OP) tri-esters, OP-diesters and polyphosphates are examples of the structural composition of OPEs. The OP- tri-esters are the most common type of organophosphates, while the diesters of OPs are the degradation products of the OPs tri-esters. Thus, the esters of phosphoric acid are mostly referred to as tri-esters and less often as diesters and monoesters. 44 The tri-esters have three ester groups bonded with an alkyl or aromatic substituents while the OP diesters differ from tri-esters in that a hydroxyl group replaces an alkyl group of the tri-esters.
OPEs may be further categorized based on their different substituents (ester functional groups) as halogenated alkyl-OPs e.g., tris(2-chloroethyl) phosphate (TCEP), non-halogenated alkyl-OPs e.g., tris(2-butoxylethyl) phosphates (TBEP) and the aryl phosphates e.g., triphenyl-phosphate (TPhP).8,45 OPEs can function as flame retardants or plasticizers in polymers and textiles (cellulosic fiber origin), polyurethane foams (PUF), thermoset resins, thermoplastic materials, textile finishes, cellulosic polyesters and vehicle and electronic applications. 46 The halogenated OPEs are usually employed as flame retardants because the halogen substituents increase the lifetime (stability) of the chemical compounds in the products due to reduced mobility in the polymer molecule. 47 The different functional groups and the other constituents present in OPFRs result in a wide variety of physicochemical characteristics such as volatility, polarity and solubility. 15 These differences in features have been widely employed in the evaluation of the consequences and fate of OPE in diverse environmental matrices, notably in aquatic creatures. 47 Their hydrophilicity, vapour pressure (VP), octanol/water partition coefficient (Kow), Henry’s law constant (H) and bio-concentration factor (BCF) all differ significantly among congeners.
Health and environmental impact of the OPFRs
Due to their physicochemical features which are similar to persistent organic pollutants, halogenated OPEs are a threat to man and the ecosystems. There are recent studies that link these substances to the possibility of carcinogenic occurrences.14,48 The Ortho-containing trimethylphenyl-phosphate (TMPP) isomers, and the tris (1,3-dichloro-2-propyl) phosphate (TDCIPP) have all been found to be neurotoxic.47,49 Tris (1-chloro-2-propyl) phosphate (TCPP), which is believed to be carcinogenic, can accumulate in the liver and kidneys. The halogenated OPEs could be catalyzed and metabolized by enzymes in humans, resulting in the formation of daughter chemical compounds with high potential in disintegrating red blood cells (haemolytic effects). 50 Although these adverse effects are most obvious in animals, there has been speculation that they may cause harmful haemolytic and reproductive effects in people. 10 In addition, OPFR metabolites found in the urine of pregnant women has been linked to lower cognition and reflex abilities, poor language expression and low IQ in children. 51 The usage of OPEs as flame retardants also has some environmental implications. The halogenated OPEs do not easily degrade in the natural environment and evidence of their environmental persistence, bioaccumulation and other negative effects in aquatic creatures exist. 14 At 300 μg/L, TCEP, TPhP and TDCIPP can cause spine distortion in killifish and zebrafish larvae.8,52 TDCPP, at a concentration of 65 ng/L has been identified as the cause of reduced fecundity in Daphnia Magna and toxicity in zebrafish embryos and larvae. It could also cause reduced body weight and heartbeat rates, and low hatching and survival rates, even at concentrations as low as 600 ng/L. 52 These chemical compounds are easily leached into surface water through landfill aquifers. 53 Furthermore, the OP tri-esters can be released into the ecosystem via urban wastewater, industrial effluents and wastewater treatment works (WWTWs) discharges and atmospheric condensation.53–55 From there, some can spread via the water cycle and contaminate other water systems. As a result, there are more reports of OP tri-esters in aquatic media such as river water, seawater, drinking water and influent/effluent from treatment plants. 54 The OPFRs have been detected in other environmental media as well.56–59
Adsorbents for water remediation
Agricultural wastes are significant precursors for the production of adsorbents because they are inexpensive, renewable, non-toxic and abundant in nature. These include agro-industrial by-products, such as date stone, wood shavings and barks, coconut shells and fibres, bagasse, rice husk, corn stillage, fruit peels, fruit seeds, plant stalks, oil palm front etc.60–63 Large volume of unprocessed wastes that are often discarded by the agricultural and industrial sectors can contaminate and harm the ecosystem. The improper treatment of these wastes before disposal may result in similar issues. Many solutions have been proposed in recent years, for the appropriate disposal of agro-wastes. This includes the adoption of pollution legislation to prevent or reduce the movement of waste materials to other sites/places. 64 Another solution requires their valorisation into value-added products like adsorbents suitable for water purification. 65 Agricultural wastes are superior to other adsorbents in that they may be used in their original form or with little treatment (drying, grinding, pulverization, etc.) which reduces the cost of production.25,65 These wastes are widely employed as a source of AC production and as components of composite materials for the adsorption of a wide variety of contaminants, due to their low ash and carbon content.5,60,61,66 The availability and abundance of a precursor, its cost, simple regeneration qualities or ease of disposal, purity, production technique and the intended application of the product, are factors that influence the choice of a precursor for the preparation of adsorbents.61,66
Activated carbon
AC is the most widely used adsorbent for the adsorption of a broad range of emerging contaminants due to its special properties which include its large specific surface area, highly porous structure and high surface contact. 67 AC is a general term for a group of amorphous carbonaceous adsorbents that is highly crystalline with a well-developed internal pore structure. 26 The earliest adsorbent known in wastewater treatment is charcoal, which is the predecessor of current activated carbon. Its ability to cleanse water has been established since 2000 B.C. Raphael von Ostrejko was credited with producing commercial activated carbon and his ideas were patented in 1900 and 1901. 68 Pristine activated carbon’s surface is basically ‘non-polar and their surface functional groups facilitate the cohesive adhesion of the adsorbate molecules, thereby increasing their rate of adsorption. The types of AC, according to their particle size classification, are pulverized activated carbon (PAC) and granular activated carbon (GAC). AC can be further grouped according to their pore size as macroporous-structured AC (≥50 nm), mesoporous-structured AC (2–50 nm) and microporous-structured AC (2 - > 0.8 nm). 26 Various works on the removal of contaminants from water have been achieved by using GAC because they are more adaptive to continuous contact with adsorbate and do not require the removal of the carbon from the bulk fluid. Conversely, the use of PAC poses several practical concerns because of the difficulty encountered in separating the PAC from the liquid matrices. However, it is still employed for wastewater treatment despite these concerns due to its low operational cost.26,68 In terms of pore size, the mesoporous AC has been proposed as the best option for ECs removal, due to the minimal interference from other organic chemicals at the active pore sites of the adsorbent during adsorption. 69 The adsorption capacity of AC is an important property that is influenced by its specific surface area, pore size network and pore volume. 70 Adsorption capacity refers to the quantity of adsorbate that accumulates on the surface of a particular adsorbent, while removal effectiveness refers to the proportion of adsorbate transferred from the solution onto the adsorbent. 30 Functional groups like the carboxyl and phenol functional groups also influence the adsorption capacity of AC. These functional groups are prevalent in most activated carbon, due to the oxidation-reduction reaction that occur during the activation process. 27 In addition, electrostatic attraction, π–π interactions and other hydrophobic interactions play a role in adsorption. Physical adsorption could be used to control the adsorption process while chemisorption may occur in some instances. Physical adsorption involves some key mechanisms such as surface charge, steric interaction, π–π interaction, Van der Waals forces, hydrogen bonding, and dipole-dipole interactions. In contrast to physio-sorption, chemisorption involves the formation of a chemical bond between the adsorbent and the pollutant through the sharing of electrons. Chemical adsorption occurs where metallic ions and adsorbents with multiple functional groups are involved. 71 AC has substantial adsorptive capabilities because of its high internal porous network, which provides a wide surface area for adsorption.25,33,70 However, the overall adsorption performance of AC depends on the composition of the contaminant (particle size, functional group, surface charge, Kow, Kd, pKa), the AC structure and composition (particle size, surface area, pore diameter, mineral content) and some important environmental factors (pH, temperature, wastewater type). 26 Tiny pores on AC, for example, will not capture large adsorbate molecules while large pores, cannot hold smaller contaminants, whether charged, uncharged, polar or non-polar compounds. 72 Precursors commonly used to produce commercial AC are either from a botanical source, such as coconut shells, wood and nutshells, or carbonized plant debris (e.g., lignite, petroleum coke, peat and all types of coal). 73 However, the use of these types of activated carbon has generated concerns due to their high cost, unavailable raw materials, complicated production procedures and non-biodegradability of spent adsorbent. As a result, their practical use is limited.5,73,74 In recent years, many researchers have sought to develop adsorbents from affordable, easily accessible, renewable and eco-friendly precursors like agricultural residues.
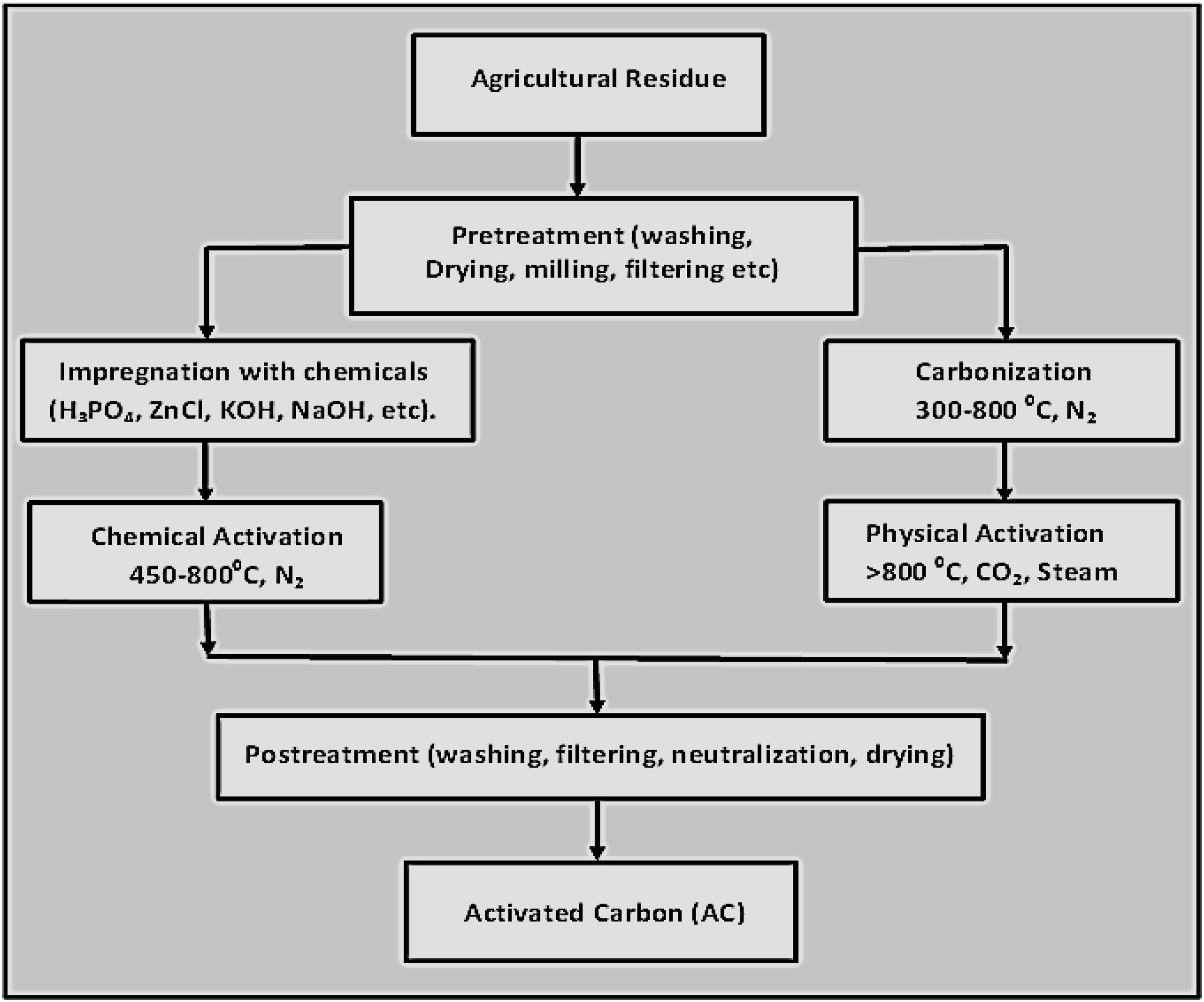
Conversion of agricultural wastes materials into an adsorbent
Two basic steps are involved in the preparation of AC from plant biomass. These are 1) thermal treatment (pyrolysis) of the carbonaceous biomass below 800°C in the absence of oxygen and 2) physical or chemical pre-treatment of the char produced.72,75 Carbonization is a non-toxic thermal process that converts raw materials into a non-porous solid char. During the carbonation process, the precursor (biomass) is pyrolyzed in an inert atmosphere and at elevated temperatures between 300-800°C.33,75 Pyrolysis is a type of energy recovery process that produces char, oil and gas formation as by-products.68,76 The amorphous by-products (i.e. tar) are burnt off, allowing the pore openings to expand and the number of pores to increase. The process parameters may be adjusted in order to increase the amount of biochar, oil, or gas produced, and all have potential industrial use. 68 The residual solid biochar (BC) produced after carbonization has characteristics that differ from the source biomass material due to the usage of heat which eliminates the moisture and volatile matter contents of the biomass. 72 The unique changes in the biochar are observed in its surface area distribution, porosity, pore shapes (micropores, mesopores and macropores), ash content and elemental content. The BC produced has a high amount of carbon but low oxygen and hydrogen content. 77 These changes in characteristics generally result in high reactivity, which makes the biochar suitable as an adsorbent material. Therefore, the char becomes a desirable by-product that may be used as a sorbent for air pollution and wastewater treatment. 78 However, the BC’s porosity after carbonization is low, which limits its application in adsorption systems. Furthermore, the tar (by-product) from the pyrolysis process may adhere to the surface of the BC, thereby blocking the pores and reducing the BC’s adsorption capacity. Thus, an activation phase is required in order to further convert the biochar to activated carbon.72,77,78 The particle’s dimension, temperature and heating rate are the process factors that have the most impact on the carbonization products. In terms of the treatment time and the necessary equipment, the carbonization of biomass offers a few benefits over the standard biological treatments. Pathogens and the possible organic pollutants present in biochar can be destroyed by high thermal treatment. 76 Conversely, the processing of activated carbon, which is frequently done at quite high temperatures, might lead to overheating and full combustion of the carbon material. 67
Physical/thermal Activation
Physical activation consists of two steps. In the first stage, the precursor is carbonized in an inert environment as previously discussed, thereafter the char is activated at high temperatures along with gaseous oxidants e.g. CO2, H2O(g) and on fewer occasions, the O2 molecule.25,27,30 Steam activation is a method that can be utilized for the removal of tar from BC by exposing it to gaseous oxidants at temperatures of above 800°C. It is a physical activation approach that can result in additional pore formation on the surface of the BC and the production of activated carbon suitable for adsorption. 77 Physical (or steam) activated carbons have substantial volume/area (0.15–0.50 cm3/g) of both micropores and macropores, but mesopore volumes are typically less than 0.10 cm3/g and contribute to less than 5% of their adsorption capacity. 79 Pyrolysis, oxidation, dehydration and decarbonisation activities impart functional/charge groups onto the biochar surface. At temperatures of between 300–400°C, alkane and alkene bonds in carbonaceous materials are normally converted into carboxylic acid, alcohol and aldehyde while hydroxyl, aldehyde groups and a few aromatic structures begin to form at temperatures of between 500–600°C. At temperatures beyond 600°C, ash content and aromatic carbon structure begin to develop. 80 The prevalent oxygenated functional groups and surface charges can be attributed to the low pyrolytic temperatures, which are useful for removing ionic pollutants. Conversely, higher temperatures (>600°C) may be required to remove oxygen groups due to their low thermal stability. In addition, nitrogen groups have better thermal stability, which permits them to be retained on carbon sites.81,82
Microwave pyrolysis
Pyrolysis of substances via microwave systems is a relatively recent thermal technology that uses microwaves as a heat source instead of the traditional heating sources like ovens and furnaces.70,77 Microwaves are non-ionizing electromagnetic radiation occurring between radio and infrared frequencies that range in wavelength from 1 mm to 1m and frequency from 300 GHz to 300 MHz, respectively.83,84 Microwave pyrolysis involves subjecting a dielectric substance to an alternating electromagnetic field where it emits energy via a process known as microwave heating or “dielectric heating”. During the process, ionic or molecular dipoles undergo translational motion caused by an internally produced electric field when microwave radiation pierces through a certain volume of material. As a result, polar molecules having dipole moments will rotate and align with an oscillating electromagnetic field. When the field changes, the molecules will reverse direction, vibrate, collide with nearby molecules, and emit heat energy to other molecules. In certain areas, the heat circulation is higher than the macroscopic temperature and is not homogenous. 85 The material structure is altered by the temperature’s nonhomogeneity and fast oscillation. Thus, the heat energy generated by the molecules' fast (million-per-second) oscillation causes volatilization and carbonization of the material.77,85,86 While dipole molecules are exposed to microwave radiation, thermal energy flows from the inside to the outside, enabling an effective heating process based on the material’s dielectric characteristics. 87
The microwave radiation approach is both internal and volumetric and thus, results in quick and shorter processing periods, low bulk temperature and reduced cost of production due to the heat energy released via ionic and dipole interactions.66,88 Other advantages of microwave radiation over the traditional heating technologies include selective heating, increased efficiency, fast start-up and shutdown during operation, lower activation energy required, enhanced safety, simplicity and reduced equipment size.70,89,90 However, the microwave as a heating source has only recently been used in the carbonization process, thus there are only a few studies that have reported on its usage in the creation of activated carbon.
Chemical activation
Chemical activation (ChA) can be applied for the production of AC via a one-step or two-step process. In the single-step procedure, prior to carbonization, the agricultural waste is impregnated with a chemical reagent such as acid, basic solutions, inorganic salts, dehydrating agents or oxidants and carbonized simultaneously at temperatures lower than those used in physical activation.27,30,33,68 In the two-step process, the biomass is first carbonized and the biochar formed is thereafter chemically activated. The inorganic constituents in the biochar are removed after the thermal treatment (under an inert atmosphere), in order to generate activated carbons with high surface area.
5
When an acidic or basic solution is mixed with carbonaceous material, the pyrolytic breakdown of the starting materials is accelerated, tar formation is repressed and the pyrolysis temperature is lowered.
70
HNO3, NaOH, KOH, H3PO4 and on rare occasions, K2CO3 are the most prevalent chemical reagents utilized for the activation of plant biomass.
72
It has been reported that the chemical introduced into the interior of the precursor particles reacts with the products formed from the thermal degradation of the precursor, thereby decreasing the emission of volatile matter and limiting the shrinkage of the precursor structure. Tar production is also prevented by chemical activation The precursor is converted to a highly carbonized product at a rapid rate in this manner and after the chemical is removed following heat treatment, a considerable amount of well-developed porous structure is generated.67,91 Chemical activation provides several benefits over physical activation and as a result, has been used in many types of research involving the fabrication of activated carbon from agricultural wastes. It is preferred because the material may be activated at a lower temperature and in less time. The activating chemicals aid in the destruction of biomass structural components and promote the creation of meso-micropores.
27
Furthermore, investigations have revealed that AC produced via the chemical activation procedures has a larger surface area and higher mesopore content in comparison to the AC produced via the physical activation techniques.30,66 The stages in the preparation of AC are shown in Figure 1. Activated carbon production steps.
27
Cellulose, an important precursor for the production of nanocomposites
Agricultural residues are largely composed of lignin (10–25%), hemicelluloses (20–30%) and cellulose (40–50%).67,92 Lignin is an amorphous phenolic polymer that is made up of phenyl propane units joined together to produce a complex structure. It contains hydroxyl, methoxyl and carbonyl functional groups in its chemical structure.93,94 Cellulose and hemicelluloses are produced from the polymerization of monosaccharides via dehydration and condensation reaction of glucose or other monosaccharides. The cellulose biopolymer is a linear natural polymer made up of d-anhydroglucose (C6H11O5) repeat-unit, linked together by 1,4-b-d-glycosidic bonds at the C-1 and C-4 carbon locations, to create a fundamental cellulose unit known as cellobiose. Cellulose is a low-cost, processable polymer that may be found in nature in plant cell walls, as well as in bacteria and tunicates. It has a significant amount of carbon and a proven ability for sorption. It has been identified from other biopolymers as an abundant, readily available and sustainable natural biopolymer. The cellulose fiber exhibits exceptional qualities such as low cytotoxicity, biocompatibility, low density, superior mechanical capabilities, high chemical stability and biodegradability. This makes it a preferable precursor for the synthesis of nanocomposites and a variety of industrial applications.95,96,97 The structure of cellulose is shown in Figure 2. The molecular structure of cellulose.
98

Cellulose may be regarded as a cellobiose isotactic polymer. 99 The reducing end of the cellulose molecule contains a loose hemi-acetal (or aldehyde) group at C-1, which has reducing qualities, while the non-reducing terminal has a free hydroxyl at C-4. In addition, the internal rings connected at C-1 and C-4 have non-reducing properties .99,100 The abundant hydroxyl groups on the cellulose structure serve as active sites for functionalization. These functional groups are vital in the nanocomposite preparation process. 33 In order to reduce the damage to the cellulose surface morphology during carbonization and increase its compatibility for the removal of a specific pollutant, chemical activation through surface modification is preferred Non-toxic chemical reagents are used to achieve chemical modification. 33 The main modification strategies for cellulose modification are oxidation, esterification, amination, etherification, and polymerization. Cellulose must first be isolated from plant biomass for chemical reagents to fully react with the hydrogen bonds in its internal structure during the modification process. 95
Extraction of cellulose from plant biomass
The pre-treatment of the plant biomass in order to remove hemicelluloses and lignin from the core cellulosic component is the first stage of the cellulose extraction process. Alkali and acid-chlorite or peroxide treatment are commonly used in this process.101,102 Alkaline treatments have long been regarded as one of the most cost-effective surface treatments. During alkalization, the amorphous areas that consist mostly of hemicellulose are removed from the cellulosic fibers, using by sodium hydroxide or potassium hydroxide. The second stage involves the simultaneous treatment of the alkalized biomass (pulp) with sodium chlorite acidified with glacial acetic acid or hydrogen peroxide/sodium hydroxide solution. This process removes the majority of lignin and non-cellulosic materials from the lingo-cellulosic biomass. The second stage is known as the bleaching or delignification process.101,103 The delignification process improves the crystallisation potential, enhances interfacial bonding and improves cellulose fiber compatibility for modification. As a result, the mechanical characteristic of the cellulose fiber is greatly enhanced. 78
Current strategies for the surface modification of agricultural residues
The thermochemical decomposition of lignin creates a char with a low amount of functional group, which limits its performance and the usage of raw agricultural residues. 77 Therefore increasing or adding specific functional groups onto the biochar’s surface require functionalization. Surface treatment via chemical reagents and metal impregnation of bio-char are two alternative approaches to biochar modification. The surface treatment adds functional groups to the biochar, while impregnation with metal or metal oxides results in nanocomposites. 104 Functional groups can be used to attach catalysts or catalyst precursors onto the surface of a material or to further modify the material. In addition, functional groups may serve as charged sites for catalytic reaction and pollutant bio-adsorption. 105 Therefore, surface-modified biosorbents and composites are of tremendous interest for specific catalytic applications. 106 Functional groups e.g., heteroatoms (O, S, P and N), carboxyl, carboxylic, phenolic, sulfonic and hydroxyl groups can be incorporated into bio-char via interaction with acids and alkaline reagents. 105 Higher adsorption of contaminants on the AC surface will thus be enhanced by the presence of functional groups. 27 The presence of carboxylic and hydroxyl functional groups on the bio-sorbent surface, for example, greatly enhances heavy metal adsorption via hydrogen bonding or other mechanisms. 107 Similarly, bio-char with amino surface groups creates active alkaline sites on the char surface, thereby considerably increasing its carbon sequestration properties.105,107 Oxidation, etherification, esterification, and polymer grafting are some modification strategies applied during the chemical modification of materials.27,33,108
Oxidative method
A variety of oxidants have been widely utilized for AC functionalization by chemical activation. The oxidation strategy introduces oxygenated functional groups onto the carbon surface. This occurs when the oxidizing agents are used to treat the carbon-based materials either in a liquid state (e.g., HNO3, H2SO4, H2O2, NH3SO4) or in a vapour state (e.g., oxygen, ozone, nitrogen oxides.79,109,110 The acid concentration, contact time, temperature and treatment period influence the intensity of the oxidation. The carbonaceous biomass could simply be boiled in concentrated acids e.g. HNO3 (which will cause significant changes in its textural qualities) or converted to AC before oxidation treatment. Acidic (oxygenated) functional groups such as phenolic, lactonic, carboxylic and anhydrides groups, as well as neutral or basic carbonyl and ether groups (e.g., quinone, chromen and pyrone groups), could be incorporated onto the surface of the material during the oxidation modification process. 79 Oxygenated groups promote the adsorptive removal of organic molecules except for aromatic compounds, due to the reduction in dispersive interaction between aromatics and the acidic property of carbon. 104 Jing and his colleagues chemically modified rice husk and investigated its effectiveness as an adsorbent for the recovery of tetracycline (TC) from water. 111 Raw rice husk bio-char was first pre-treated with a solution of 3 mol/L NaOH at a ratio of 1:10 (w/v). Under acidic circumstances, methanol was used to alter the carbonyl group via an esterification reaction to produce a chemically modified bio-char (MeOH-char). The FTIR analysis of MeOH-char revealed that the concentration of carbonyl groups reduced after modification, an indication that a reaction occurred between the carbonyl group on biochar and methanol. According to Jing and colleagues, the modified biochar demonstrated a 45.6% increase in tetracycline adsorptive removal in just 12 h. In a related study, the ability of modified cocoa shells to remove sodium diclofenac (DFC) and nimesulide (NM) (anti-inflammatories compounds) from simulated hospital effluents (effluent A and B respectively) was investigated by Sucier et al. 112 Activated carbon was produced from cocoa shell powder (CSC-1.0) via treatment with combined inorganic salts of ZnCl2 (400%), lime (20%) and FeCl3 (40%) at a ratio of 0:1, 1:1, 1:1.5, 1:2 and 1:10 (salt: bio-char). Thereafter, microwaved-cocoa shell carbon (MW-CSC-1.0.) was produced from CSC-1.0 by acidifying with 6 mol/L HCl and subsequent pyrolysis in a microwave oven for less than 10 min. According to Saucier and colleagues, the MWCS-1.0 successfully removed a maximum amount of 63.47 mg/g DCF and 74.81 mg/g NM from effluent A and B respectively. The FTIR result showed an increased concentration of oxygenated functional groups on MW-CSC-1.0. It can be deduced from this result that the interaction between the polar molecules of the pollutants and the major molecular groups of the developed adsorbent (OH, C = O, COOH) is largely responsible for the adsorption process according to data obtained from the MW-CS-1.0 characterization and from the kinetic, equilibrium and thermodynamic studies. In another study, Qu et al. 113 modified Rice husk bio-char with β-cyclodextrin (β-CD) via a microwave-assisted process and utilized it for the simultaneous removal of bisphenol A (BPA) and plumbum (Pb) from aqueous solution. During the preparation, H2SO4 was used to treat the pulverised rice husks which were then pyrolyzed at 300°C for 2 h, under N2 atm. Thereafter, the BC was treated with NaOH and glutaraldehyde (GA) and subsequently functionalized with -CD under MW irradiation (350 W for 15 min) to produce the grafted biochar microwave-CD (BCMW—CD). 113 Qu and his colleagues reported that the adsorption capacity of the synthesized BCMW-β-CD was higher at 209.20 mg/g for BPA and 240.13 mg/g for Pb (II) respectively, when compared to unmodified BC which had adsorption efficiency of 15.76 mg/g for Pb (II) and 56.61 mg/g for BPA. Further research into the adsorption process revealed that complexation and electrostatic interactions were responsible for the Pb (II) ion’s adhesion onto the BCMW-CD, while the removal of BPA was mostly due to attraction between the adsorbent, contaminant molecules, and π-π stacking interactions. 113
Esterification reaction
This involves the interaction of cellulose’s free hydroxyl groups with acids, resulting in the addition of carboxyl groups and the subsequent formation of cellulose esters as trivalent polymeric alcohol.
37
In order to add carboxyl groups to the cellulose substrate, cyclic anhydride reagents e.g. succinic anhydride, are more commonly utilized. Esterification reagents like acyl halide, EDTA dianhydride, maleic anhydride and citric acid anhydride could also be used.
114
After the esterification process, further processing, such as treatment with a saturated solution of sodium bicarbonate (Na2CO3), may be required to improve the surface characteristics necessary for binding contaminants from aqueous solutions. Na2CO3 contains carboxylate functions with stronger chelating capabilities than carboxylic groups, hence, its preference for post-treatment.
37
Han et al.
115
chemically modified Wheat straw (WS) with citric acid and investigated it for the adsorptive recovery of copper ions and methylene blue (MB) from solution. About 20–40 mesh fractions were obtained from milled dried straw and used for chemical modification. The chemically modified wheat straw (MWS) was produced by adding powdered wheat straw into 0.6 mol/L citric acid at a 1:12 ratio (straw/acid, w/v) and stirring at 20°C for 30°min. Next, the citric acid-impregnated wheat straw was dried and thereafter, thermo-chemically esterified via the reaction between the modified straw and the acid at an oven temperature of 120°C for 90°min. Han et al., reported that the quantity of Cu2+ and MB adsorbed by the MWS was 39.17 mg/g and 396.9 mg/g at 293 K, respectively. Similarly, Swede rape straw (SRS) residues (
Etherification
The reaction of alkaline cellulose with an etherifying agent produces cellulose ethers via the partial or complete replacement of the cellulose hydroxyl functional groups with ether groups under suitable conditions. Chemically modified walnut shells (MWNS) were produced by Cao et al. 117 using diethylenetriamine (DETA) and epichlorohydrin as the etherifying and crosslinking agents respectively. The MWNS was used to adsorb a model azo-Reactive Brilliant Red dye (K-2BP). Briefly, the walnut shell (WNS) was crushed and sieved to produce powders with particle sizes ranging between 65 to 75 microns. Thereafter, 1g of WNS was first treated with 1.25 M NaOH (15 mL) and 10 mL epichlorohydrin to obtain M1-WNS which was further treated with 2.5 mL DETA and 0.125 M NaOH (15 mL). Next, the mixture was stirred mechanically and etherified for 1 h at 65°C, to form a cationic amino-modified walnut shell (MWNS). The FTIR spectra of the unmodified WNS adsorbent revealed only carboxyl groups as its constituents, but on the modified -MWNS, the FTIR result showed the presence of numerous amine groups. The authors concluded that the highest K-2BP adsorptive capacity (Qm) determined by the best fitting model (Langmuir) at 313 K was 568.18 mg/g, which was approximately ten times higher than the raw material. In another study, Zheng et al. 118 produced adsorbents from maize stalks and studied their adsorption performance for the separation of cadmium from an aqueous solution. During the preparation, dried corn stalk was pulverized and sieved to obtain a 2–4 mm size. The chemical modification of the powdered stalk was performed by soaking 1g of the powdered stalk in 1 M/NaOH and the mixture stirred at 120 rev/minute for 18 h at 40°C. Afterwards, the mixture was treated with 98% acrylonitrile solution (10 mL) and stirred for 30 min at 120 rev/min to produce acrylonitrile-modified corn stalk (AMCS). The result from the FTIR analysis revealed the presence of cyano groups on the AMCS, which confirms the successful modification of the corn stalk. Zheng and his team reported an increase in adsorption performance from 3.39 (raw corn stalk) to 12.73 mg/g (modified corn stalk). Findings from this study showed that the complexation reaction generated in the cellulose between the nitrogen and oxygen of the cyano- and hydroxyl group respectively, may be responsible for their effective surface modification.
Polymer grafting
Graft polymerisation combines polymers into a single physical unit.
119
Grafting procedures are grouped into two categories: “grafting onto” and “grafting from.” The ‘grafting onto’ method involves attaching a target polymer to the OH of the pre-synthesized polymer’s/cellulose surface with a bonding agent or coupling agent
120
while the “grafting from” method refers to polymer chain growth from the surface of the functionalized polymer by the reaction between the surface functionalities of the polymeric material and the relevant monomers. The graft polymerisation process includes the following approaches: ring-opening polymerisation (ROP), atom transfer radical. Polym. (ATRP
Nanocomposite production
Surface modification with metallic nanomaterials
Biochar engineered with nanomaterials yields amazing biochar–based nanocomposites with advantageous properties of both materials. The following two approaches are generally employed in the preparation of biochar–based nanocomposites: 1) incorporating nano-metal oxides or hydroxides onto biochar and 2) pre-treating biomass or post-treating pyrolyzed biochar with a metal salt.130,131 Nanometal oxides (NMOs) and metal salts are nanoparticles utilized in the making of nano-adsorbents with excellent adsorptive properties. The term “nano” refers to various particles in which at least one of their dimensions is not more than (or equal to) 100 nm. 132 Metallic nanoparticles can sequester pollutants of various molecular sizes and hydrophobic characteristics, due to their high porosity, small particle size and reactive surface. These nanoparticles also allow the production process to utilize its constituents without any form of leaching out after nanocomposite formation. Furthermore, they can be chemically regenerated after being exhausted. 133 Nanoparticles have some unique features in addition to their large surface area, such as high reactivity and catalytic potential, that make them superior adsorbing materials than ordinary materials. 18 Due to these properties, scholarly interest in nanotechnology has risen globally. However, some challenges encountered in their application tend to limit their usage. As the size of these metallic species reduce from micro to nanoscale, the surface energy increases thereby lowering their stability. As a result, the nanoparticles become easily agglomerated, due to increased Van der Waals forces or other attractive forces. 134 Furthermore, their nano-size may cause an excessive loss of pressure and mechanical strength, rendering the nanoparticles ineffective in fixed beds and other flowing systems. In order to increase the usage of nanoparticles in wastewater remediation, a sustainable carrier or support material is required. Therefore, nanomaterials are used to develop hybrid adsorbents or nanocomposite by impregnating or coating metallic salts or nanometallic oxides (NMOs) particles into/onto porous supporting matrices of larger size.133–135 Activated carbon, carbon nanotubes, chitosan, agricultural residue and other polymers have been effectively coated with metal nanoparticles.136–139 The most commonly utilized metallic salts include Zn, Mg, Fe and Co among others.140–143 while others include noble metal nanoparticles such as silver (Ag) and gold (An)139,144,145 and nanoscale zerovalent metals (NZVI) [Zn (II), Cu(II), Cd(II) and Co(II)]. 134 The Ag and Au nanoparticles have the capacity to enhance the quantity of surface atoms on biochar which, in turn, increases the surface energy of the substrate to which they are attached. 139 Thus, the functionality and reactivity of the composites are improved. Furthermore, Ag and Au, when utilized for water filtration, have been shown to exhibit antimicrobial activity.146,147 Metal oxide nanomaterials such as SiO2, ZnO, MgO and TiO2 have been extensively explored due to their capacity to function as disinfecting photocatalytic agents and UV blockers, while serving as a second component in the nanocomposites.134,143,147 Metals may form functional groups such as Fe-O, Mn-O and metal-O-metal, which help with adsorption, particularly for ions. 148
Magnetic Nanocomposites have recently become a focal point in the water purification industry due to their ease of separation and collection via magnets. Materials that consist of an inorganic magnetic substance enclosed within an organic polymer are referred to as polymer-based magnetic nanocomposites. 149 When subjected to alternating or static magnetic fields, these magnetic nanomaterials can disperse in various materials and exhibit unique properties.147,150 The ferromagnetic features of magnetic adsorbents make them easy to be removed via magnetic attraction, after the completion of the adsorption process. This is an indication of their suitability for recycling and reuse. Iron chloride, iron oxide, ferric nitrate, Zero-valent iron and metal-doped iron oxides are the most often used compounds for magnetic modification.141,147,151
Gan et al. 142 produced zinc–biochar nanocomposites from sugarcane bagasse and determined their adsorption capacity for the removal of Cr(VI). Briefly, pulverized bagasse (20 g) was chemically activated with Zn (NO3)2 solution (20%), stirred at a speed of 130 r/min for 24 h at 30°C and pyrolyzed in a furnace. The XRD analysis revealed the presence of Zn on the surface of the Zn-biochar, thereby confirming the successful modification. The authors concluded that a maximum adsorption capability of 45.79 mg/g for Cr (VI) was achieved. In a similar report, Omo-okoro et al. 144 synthesized silver-nanocomposite from maize tassels via physical/chemical activation. The physical Ag-activation of the Maize tassel (PAMTAg) was produced via thermal treatment while the chemical treatment produced chemically activated Ag-maize tassels (CAMTAg). These nanocomposites were utilized for the removal of PFAS from aqueous solutions. The maize tassel-silver nanocomposites (MTAg) were prepared by dispersing 0.8 g; (particle size: ≤45 μm) in a mixture of 55 mL of distilled water and 25 mL of different percentages of silver nitrate (1, 2.5, 5 and 10%) which resulted in MtAg biochar. Some MtAg biochar were physically activated by using steam at 400°C and nitrogen at 600°C, which yielded PAMTAg while others were chemically activated by using H3PO4 to form the CAMTAg nanocomposites. From their results, Omo-okoro and colleagues observed that the maximum adsorption capacity of CAMTAg adsorbent for PFOS and PFOA were 454.1 mg/g and 321.2 mg/g respectively. In another study, MgO-biochar nanocomposites were produced by Zhang et al. 143 from five typical plant wastes: cottonwoods (CWs), sugar beet tailings (SBTs), sugarcane bagasse (SB), peanut shells (PSs) and pine woods (PWs). These nanocomposites were used for the separation of nitrates and phosphates from solutions. Each biomass was soaked with Magnesium chloride hexahydrate (MgCl2.6H2O) for 2 h and the mixture heated at 600°C for 1 h under N2 flow. Thereafter, the MgO-biochar formed from the pyrolysis process were crushed and sieved into two fractions of <0.5 and 0.5–1 mm. According to Zhang and co-workers, the peanut shells and residual sugar beet nanocomposites had the highest adsorption capacities with Langmuir adsorption prediction as high as 835 mg/g and 95 mg/g for phosphates and nitrate respectively. Mubarak et al. 151 in their study, produced unique magnetic bio-char from waste empty fruit bunch (EFB), impregnated with ferric chloride hexahydrate and pyrolyzed via microwave irradiation. Its adsorption effectiveness was determined through the recovery of methylene blue (MB) from solution. The dry biomass (EFB) was pulverized and filtered to particle sizes of less than 150 μm. Thereafter, varying quantities of the pulverized biomass were impregnation in ferric chloride hexahydrate at various ratios. The optimum conditions utilized for the production of highly porous magnetic biochar were microwave energy of 900 W, radiation time of 20 min and 0.5 g (FeCl3: biomass). The authors concluded that the synthesized magnetic nanocomposites had a maximum adsorption capacity of 265 mg/g for MB removal (99.9% removal efficiency) from an aqueous solution. In a similar study, Yang et al. 141 developed unique magnetic BCs from sawdust for the removal of HgO from simulated combustion flue gas. Apart from determining the bio-chars performance for the HgO removal, other parameters such as the impacts of the impregnation mass ratio of FeCl3/sawdust and reaction temperature were also examined. The sawdust was submerged in a solution of FeCl3 for 2 h with constant agitation and thereafter, the FeCl3-laden sawdust was pyrolyzed for 1 h at varying temperatures (800, 700, 600, 500°C) and a N2 flow rate of 0.5 L/min, in order to). Yang et al. observed that the ideal pyrolysis conditions required to form the magnetic biochar (MBC600) were a temperature of 600°C and a mass ratio of 1.5 g (FeCl3/sawdust). Due to the favourable high pyrolysis achieved, the pore volume and surface area of the MBCs were greatly enhanced when compared to the unmodified BC600. Findings from this study showed that MBC600 had a high HgO removal performance (ηT > 90%) between a wide reaction temperature range (120−250°C). 141
Cellulose bead nanocomposite
Although the functionalization of agricultural residues via thermal treatment is known to improve their compatibility and adsorption capacity for pollutants, the low carbonization energy of several plant biomass could be problematic, especially during thermal treatment. Unlike wood which undergoes thermal treatment at high temperatures, the surface morphology of plant biomass may be destroyed during pyrolysis because they easily burn off at low temperatures (160–200°C) and generate soft ash. Furthermore, there is a risk of organic matter seeping from the biochar, which might lead to re-contamination of the final treated water (secondary pollution).127,152 Cellulose is an important constituent of agricultural residues that can be used as a precursor for the production of greener and sustainable industrial nanomaterials. It has some unique characteristics, which include high flexibility, durability, renewability, non-toxicity and relatively high tensile strength. Therefore, the extraction of cellulose fiber from agro-wastes and its subsequent chemical modification and fabrication into microbeads or aerogels is a solution to these challenges. The mixing of cellulose solutions with organic/inorganic metal nanoparticles yields cellulosic nanocomposites which can be shaped into beads. Hydrogel and beads made from polysaccharide-based materials are the most sophisticated sort of adsorbent among all the other physical forms of adsorbents.
121
Cellulose beads (CB) can be either porous or nonporous and are often referred to as pellets, granules, microspheres, beaded cellulose or pearl cellulose. Porous CBs are characterized by high specific surface area and low density with other distinct attributes which include I) diameters greater than several micrometres; 2) preparation by dissolving, shaping and regeneration of cellulose or its derivatives and 3) exclusively composed of cellulose and shaped into their spherical forms by the re-establishment of the hydrogen bonding network and typical cellulose–cellulose interaction as shown in Figure 3.153,154 Scanning electron microscope images of the surfaces of cellulose beads (a and b) with different porosity and morphology.
153

Preparation of cellulose bead
A generally accepted strategy for cellulose bead production includes the following processes: (i) the use of suitable solvents for dissolving cellulose or its derivatives, (ii) gelation and coagulation of the polysaccharide solution into spherical particles, (iii) sol−gel transition and pore adjustment, (iv) Gel drying processes. 153 Cellulose beads are usually prepared via crosslinking cellulose with additional functional groups or coupling agents, in order to create a water-insoluble cross-linked network and improve their structure and suitability for a specific purpose. The transformation from dissolved polysaccharides to solid particles and, thereafter, the bead shape, may be controlled by changing the process variables. 155 For example, beads made with higher cellulose concentrations will be less porous than beads made with less concentration. The morphology, pore size and internal surface area of the bead can be controlled by altering the temperature and content of the coagulation medium. Nitric acid, ethanol, acetic acid, hydrochloric acid, sulphuric acid and water have been used as solvents for the coagulation of cellulose solution.156,157 Thus, the coagulation process (regeneration) determines the physical bead qualities such as density, specific area and pore size structure. 158 Although the polysaccharides, solvents and regeneration processes that are used to make cellulose beads may differ, all the procedures have one common factor: the shaping of the beads using either the dropping or dispersion techniques.
Dropping technique
The dropping technique employs various types of processes which include the traditional (conventional dropping), spraying/atomization, jet cutting and dropping under the influence of electrostatic force. The conventional dropping procedures include the use of syringes as well as other dropping devices that create droplets at the needle tip (orifice) which fall freely into a gelation or coagulation bath under gravitational attraction. 159 Various sizes and forms, ranging from extremely flat plates to spheres, may be created by adjusting solution viscosity, orifice diameter, ejection speed, bath temperature or the solution distance between the syringe tip and the coagulation bath.155,160 In order to create droplets of a specific size and shape, a variety of standard technological instruments can be used. The automated approach which utilizes spraying/atomization is a process in which atomizer nozzles completely fragmentize a stream or spray of non-compressible liquid, resulting in the creation of poly- or mono-dispersed droplets in the air or vacuum. Examples of the atomizer’s nozzle device are vibration atomization and pressure Jet atomization. The use of an automated device that operates at a higher speed is advantageous, especially for large-scale manufacturing. 159
The electrostatic technique is another dropping method that involves droplet formation with the aid of an electrically charged field when the polysaccharide solution is expelled via a charged nozzle. The liquid is forced out as droplets from the orifice’s tip by the charged force field. When the surface of the droplets formed from the polysaccharide solution is broken at the opening, it acquires an induced electrostatic charge. The subsequent electrostatic repulsion between liquid droplets inhibits coalescence while the fragmentation and dimensions of the droplets are affected by factors such as the viscous nature of the solution, nozzle/orifice diameter, proximity from the coagulating bath and voltage applied. The jet-cutting approach expels cellulose solutions (instead of droplets) through a narrow orifice at a high velocity, resulting in a continual liquid flow that is chopped into spherical particles via a spinning knife instrument (jet cutting). The chopped spherical particles are collected in a coagulation bath.
The ejection and cutting of the cellulose stream into the coagulation media can also be done by underwater pelletizing. The size and form of the beads produced by this technique may be controlled by adjusting the knife geometry and rotation speed, in addition to the ejection speed and jet nozzle diameter adjustments.153,161 Due to the size of droplets that may be generated, the diameter of cellulose beads produced via dropping procedures is generally limited to a range of roughly 0.5–3 mm.153,162 Only a few studies have been published on cellulose aerogel beads and the vast majority are created by syringe-dropping of cellulose dissolved in alkali liquids. Figure 4 shows some common types of dropping devices used in bead shaping.
159
Examples of dropping devices: (a) conventional dropping method influenced by gravity, b) vibrating nozzle dropping method, c) electrostatic forces and d) a mechanical cutting device.
159

Dispersion technique
When a solution of cellulose or a cellulose derivative is dispersed in an immiscible solvent of opposite polarity at a high rotating speed, emulsions, which can be stabilized with the aid of surfactants, are produced. These emulsions include dissolved polysaccharide droplet particles that may be consolidated into beads of the same size while stirring in a reactor or with the aid of moulds.163,164 Unlike methods that rely on dropping techniques, no specific equipment is required to produce beads with consistent features in this method. Thus, dispersion-made beads of various diameters, ranging from 10 to 250 μm, are obtainable using this method. 153
Cellulose beads can be chemically modified before or after the shaping procedures, and then, applied for the removal of contaminants from aqueous solutions. For instance, Moosavi et al. 140 extracted cellulose from raw kenaf core, shaped it into cellulose beads and functionalized them with three materials: activated carbon, iron oxide, and cobalt-iron oxide (AC, Fe3O4 and Co-Fe2O4). This yielded AC-cellulose beads, AC-Fe3O4 cellulose beads and AC-CoFe2O4 cellulose beads respectively. All the adsorbents were utilized for the recovery of cationic methylene blue (MB) dye from aqueous media. The Co-Fe2O4 nanoparticle used for the modification was synthesized from the reaction between CoCl2.6H2O and FeSO4.7H2O solutions. The beads were prepared by dissolving 3 g of the extracted cellulose in NaOH/urea/distilled water solution mixed at the ratio of 7:12:81 at −13°C, under rapid mechanical stirring. Thereafter, AC was added to a portion, while the magnetic cellulose beads were formed by functionalizing AC with 5 wt% Fe3O4 and CoFe2O4. Spherically modified cellulose beads were obtained by adding the modified cellulose mixture (dropwise) with the aid of a syringe pump, into a coagulation bath of sulphuric acid (250 mL, 10 wt%) at 20°C. Results from this study show that the adsorbent beads modified by AC-Fe3O4, AC-CoFe2O4 and AC, had improved thermal stability with maximum adsorption efficiency of 54, 53 and 50 mg/g, respectively. 140 In a similar study, Maaloul et al. 165 developed spherical-shaped cellulose from bleached almond shell (BAS), which were applied as bio-adsorbents for copper (II) elimination. The cellulose was made by gently dispersing 0.4 g of dried BAS into 10 g of ionic liquid [1-butyl-3-methylimidazolium chloride (BmimCl)] solution for 2 h at 120°C with constant stirring. Thereafter, the cellulose hydrogel beads were formed by dropping the previously cooled cellulose-IL solution into a coagulation bath of purified water (1 mL/min), to form cellulose-based beads derived from almond shells (CBBAS). The hydrogel beads were washed with purified water, frozen overnight at 20°C, steeped in 100% ethanol for 6 h, and dried at 30°C. The authors concluded that the CBBAS bio-sorbents showed a high adsorption capacity of 128.24 mg/g for copper (II) ions from aqueous solutions. Liu et al. 166 investigated the use of TEMPO-mediated oxidation method and Fe3+ crosslinking for the modification of CBs formed from cotton linter pulp, which yielded Fe(III)-carboxylated cellulose beads (Fe-CCBs) adsorbents. The functionalized CBs were used to recover low levels of bromide in pharmaceutical effluents. During the preparation, 3 mmol of sodium bromide and TEMPO reagent (0.0975 mmol) were added to 5 g of CBs immersed in 500 mL of deionized water. Then, in phases, 10% (w/w) NaClO (100 mL) was combined with the mixture and the pH stabilized at 10 using 0.5 M NaOH. Subsequently, the wet carboxylated cellulose bead (5 g) was immersed in 10 mM FeCl3 metal salt solutions to produce Fe(III)-carboxylated cellulose beads (Fe-CCBs). According to Liu et al., an adsorption efficiency of 82.33% was achieved for the elimination of the bromide ions by the modified CBs.
Adsorption of OPFRs from aqueous system
Application of agricultural waste materials for the adsorptive removal of halogenated OPFRs
Adsorbents derived from agricultural wastes as precursors for the adsorptive removal of contaminants.
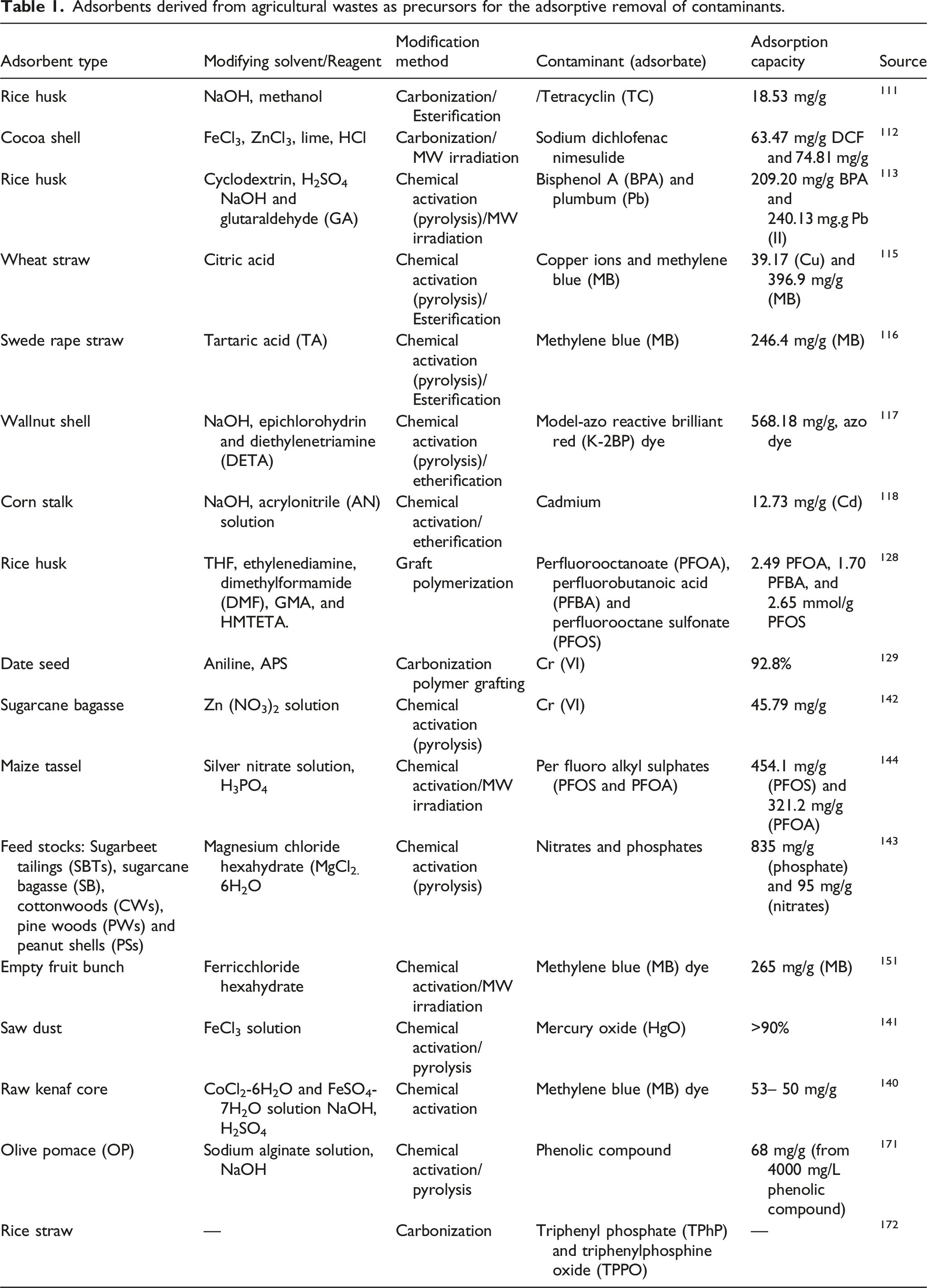
Du et al. 172 produced bio-chars (pyro-char and hydro-char) from rice straw and applied them for the removal of the aromatic OPFRs-triphenyl phosphate (TPhP) and triphenylphosphine oxide (TPPO). The study was performed in order to determine the effect of the properties of the biochars on their sorption performance. During the preparation, the rice straws were carbonized in a muffle furnace at 300, 450 and 600°C for 1 h under oxygen-limited conditions to produce pyrochar. Alternatively, hydrochar was made by hydrothermal carbonization of the straws. In this case, 10.0 g of the rice straws were mixed with 50 mL DI water at a solid/liquid ratio of 1:5 and heated at 200°C for 8 h to produce hydrothermal rice straw biochar (HRS200). 172 The FTIR spectra of the pyrochar revealed the presence of highly aromatic (C = C) and less aliphatic (C-H) functional groups, which is an indication that the pyro-chars' polarity has decreased and their aromatic properties have improved. On the other hand, oxygenated substituents provided by C–O and C = O groups dominated the surface of the hydrochars. Findings from this study show that the pyro-chars synthesized at elevated temperatures exhibited higher and faster sorption than the hydro-chars produced at lower temperatures. The sorption of the aromatic OPFR was shown to be more effective when chemically functionalized pyro-char was generated at high temperatures. In another report, Tetrakis (hydroxymethyl) Phosphonium Chloride (THPC), a constituent of OPFRs, was adsorbed from aqueous solution by using commercial biochar, as reported by Akech et al. 173 Three different chemicals, NaOH, CaCl2 and H3PO4, were used to chemically activate the bio-char under basic and acidic conditions. Samples of the biochar developed were immersed in 4M solutions of CaCl2, NaOH and H3PO4 at ambient temperature and for 24 h. The ratio of NaOH to bio-char was 3:1 (w: w); whereas the ratio for CaCl2 and H3PO4 to bio-char was 7: 1 (w: w). Akech et al. reported that the NaOH-treated biochar had the maximum adsorption capacity of 2.44 × 102 under optimized conditions. They further observed that the adsorption performance of the activated bio-char adsorbent (52.1%) was better than the non-activated bio-char (43.6%) under similar experimental conditions.
The potentials of maize tassel for the adsorptive removal of halogenated OPFRS from water
The availability and abundance of a precursor, its cost and purity, simple regeneration qualities or safe disposal, as well as the production technique and intended application of the product, all influence the choice of a precursor as an adsorbent.19,66 The Maize tassel (male inflorescence of the maize plant), is a waste product that is non-toxic and readily available in large amounts at little or no cost and hence, meets these requirements. After the maize cob has been harvested, local communities often dispose the Maize stalk containing the tassels in considerable amounts. 174 These wastes can, however, be used as raw materials in the production of value-added goods. Maize tassel is an agricultural waste product that is widely applicable as an adsorbent for removing organic and inorganic pollutants from aqueous media. Its efficiency as an adsorbent for the removal of contaminants from aqueous solutions has been widely reported.127,174,175 The Maize tassel contains functional groups such as carbonyl, carboxylic and hydroxyl groups that contribute to its adsorption capacity. 176 It is composed mainly of mesoporous pore structure (2–50 μm), making it an ideal material for the biosorption process. 176 The Maize tassel can be pulverized into powder and pyrolyzed to produce activated carbon. This conversion process increases its surface area. The chemical modification occurs when the Maize tassel powder (MTP) reacts with inexpensive, safe chemicals, via the modification strategies earlier discussed. This imparts a specific property or functionality on the MTP surface and increases its effectiveness as an adsorbent. The modification of MTP pores via pulverization and physical activation or the immobilization of metallic particles in the form of nanoparticles or single atoms/ions on the MTP to produce composites also results in surface functionalization of the tassels. 177 Cellulose can be extracted from the Maize tassel fibers, modified and shaped into beads that are suitable for the removal of the halogenated organophosphate esters. Thus, the fabrication of Maize tassel-derived cellulose bead nanocomposite for the removal of halogenated OPFRs and their metabolites will provide a potential route for the effective remediation of contaminated water.
Challenges in the use of plant-based nanocomposites for water treatment and perspectives for future research
The challenges encountered during the development of nanocomposites from plant-based biomass have been documented and include: 1) the low adsorption capability of plant biomass for negatively charged pollutants, 2) secondary pollution that may occur from the possible seepage of organic contaminants from the biochar, 3) the soft tissue of some plant biomass, which, if used alone, tend to clog column systems and severely limit the flow of eluent used to remove the adsorbate from the adsorbent surface in desorption studies, 4) the possible damage of the surface morphology of plant biomass during pyrolysis, due to their low carbonization energy and 5) the difficult separation and recovery of BC adsorbent from water. In addition, lignin, hemicelluloses and cellulose have been identified as important adsorptive mediums for the removal of organic pollutants. Therefore, agricultural waste materials containing small amounts of cellulose and hemicellulose may not be considered as good precursors for pollutant adsorption in aqueous media. Fabricating a cellulose bead adsorbent that possesses high adsorption capacity with easy application and regeneration capabilities, effectively addresses these technical barriers. Cellulose beads are particularly well suited for application in the area of adsorption in water treatment due to their ease of separation from aqueous media for re-use (unlike activated carbon) and their ability to minimize backpressure and agglomeration during column experiments. In addition, the possible damage to the surface morphology of the plant biomass during pyrolysis will be circumvented through the use of cellulose beads. Zinc oxide nanoparticles (ZnO-NP) can be used to modify the cellulose beads. The adsorption capacity of ZnO-NP for a variety of contaminants is very high due to their unique antibacterial, antifungal, UV filtering and strong catalytic and photochemical activities. Furthermore, ZnO-NP can impart changes to contaminants speciation in an aqueous medium and as a result, their bioavailability, while at the same time enhancing the stability and antibacterial characteristics of the beads. Thus, the incorporation of ZnO-NPs into a cellulose bead with subsequent heat activation at 100°C for 3 h will yield a cellulose-zinc oxide (Ce-ZnO) nanocomposite bead. The subsequent application of the modified cellulose bead in an aqueous medium at pH below the isoelectric point (IEP) of the ZnO will impart a cationic functionality to the bead and increase their adsorption capacity for anionic contaminants like the OPFRs. The halogenated OPFRs will be protonated and subsequently adsorbed by the Ce-ZnO nanocomposite via electrostatic attraction and surface complexation mechanisms between the protonated P = O groups and the charged cellulosic beads, under acidic conditions. Significant electrostatic interactions will only occur when the contaminant and adsorbent have opposing (negative/positive) charges.
The knowledge gap identified in this review is that the studies conducted on the use of agricultural residue as precursor materials for the adsorption of organophosphate compounds have so far concentrated mostly on the removal of organophosphate insecticides, with less focus on the removal of halogenated OPFRs. Only a few studies (among others) on OPFRs removal have been identified, either due to the few research conducted or the poor documentation of previous research. Therefore, more research in the usage of agricultural residues for the removal of OPFRs from aqueous medium is required in order to breach this gap. Furthermore, most of the research and development of agro-based bio-sorbent is currently done on a laboratory scale since the up-scaling process is quite expensive. Synthetic OPFRs solutions are often utilized in this case. Thus, global research into the development of sustainable agro-based adsorbents, suitable for the removal of OPFRs from real effluents and on a larger scale is recommended.
Conclusion
Water contamination and remediation have become a global issue. Emerging pollutants, e.g., the OPFRs are a cause for concern. Carbonaceous materials like the commercial activated carbon and graphene are some adsorbents that have been used for the adsorptive removal of the chlorinated-OPEs, although the precursors for these adsorbents are either scarce or expensive. The viability of agricultural wastes as a precursor for the production of adsorbent materials for adsorption systems has been verified in the literature. Furthermore, there is very little information that relates to agro-based materials as adsorbents for the removal of OPFRs. Current modification strategies which could be applied for the conversion of agricultural residues to adsorbents include the oxidative, etherification, esterification and polymer grafting. Cellulose is an important biopolymer that is contained in agricultural residues and can be used as a precursor for the production of adsorbents. This is due to its important characteristics such as its renewability, non-toxicity, inexpensive and modifiable surface structure. This paper confirms the increase in the research and development of agro-based composite materials in recent years, due to their huge benefits over conventional activated carbon-based materials. The role of cellulose biopolymers in the preparation of composites and their properties and prospects for environmental remediation has been identified. Over the past ten years, there has been a great deal of research on the use of cellulose beads modified with inorganic nanoparticles for the remediation of polluted water. Data on the adsorption capacity acquired from the application of modified cellulose-bead adsorbents reveals their remarkable adsorption efficiency for the removal of both organic and inorganic contaminants. Their unique performance has been attributed to 1) the functional moieties incorporated onto their surface during modification, and 2) the interaction of the electrostatic forces and complexation mechanisms that occur between the adsorbate and adsorbent during the adsorption process. Nanomaterials like the ZnO-NP can be engineered into cellulose to produce cellulose–ZnO bead nanocomposites with excellent adsorptive efficiencies derived from the advantageous properties of both materials. Thus, the introduction of 3-D structure cellulose bead composites derived from plant residues, has opened up several new opportunities for the removal of OPFRs and other emerging contaminants. This will aid in reducing the problems caused by water pollution.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Tshwane University of Technology.
