Abstract
The poly (phenylene oxide)-block-poly (hexyl isocyanate) copolymers (PPO-b-PHIC)s were synthesized in various ratios of blocks using the organotitanium coordination method at room temperature. The copolymer particles were prepared by precipitation of concentrated solution of the copolymers in non-solvent under stirring. The synthesized copolymers and their particles were characterized by 1H and 13C nuclear magnetic resonance (1H and 13C NMR), Fourier transform infrared (FTIR) spectra, differential scanning calorimetry (DSC), polarized optical microscope (POM), thermogravimetric analysis (TGA) and scanning electron microscope (SEM). The effect of block ratio on the anisotropic-isotropic transition and liquid crystal texture of copolymers as a function of temperature and concentration of the solution was investigated. The polymeric particles tend to be aligned along the fibrillar direction by increasing the block ratio of amide in the copolymer.
Keywords
Introduction
Various copolymerization methods of PHIC reported in literature.

The optimal time and temperature of anionic copolymerization of hexyl isocyanate must be determined because at times greater or less than the optimum value, trimer formation or de-polymerization is possible. Due to the sensitivity of anionic polymerization and copolymerization of monoisocyanates to environmental factors and temperature, the coordination method was considered as a method with less sensitivity to these factors. Studies on the coordination of monoisocyanates by organometallic compounds back to 1967. Wher 33 concluded that the reaction of metallic catalysts with monoisocyanates did not produce a polymer. Metallic catalysts cannot form a stable complex with monoisocyanates and have to react with ether or phenolic compounds to form a ligand. William 33 proposed the formation of an ether ligand with a metal core to coordinate with monoisocyanates. Controlled coordination polymerization and copolymerization of monoisocyanates in the presence of titanium compounds at ambient temperature was first reported by Patten and Novak.34,35 In this reaction, the structure of the titanium compounds and the type of ligand attached to Ti are crucial to forming a stable complex. One of the advantages of this method is that it does not form circular trimers and reacts at ambient temperature. Wu et al. 31 reported the copolymerization of poly (ethylene glycol methyl ether) with hexyl isocyanate (polyether-block-polyamide) in the presence of titanium tetrachloride (TiCl4) as a catalyst and dry toluene as solvent at room temperature. We used this method for the synthesis of PPO-b-PHIC. Poly (phenylene ether) or PPO is one of the commonly used engineering polyethers with excellent thermal stability, low flammability, high dimensional stability, low moisture uptake, high electrical resistance, and good mechanical properties.36–38 Therefore, in this paper, a new family of polyether-block-polyamide LC copolymers was synthesized. This work aimed to study the copolymerization of PPO as a high-performance polyether with PHIC as a LC polyamide, the effect of block ratio on the thermal and optical properties at different temperatures and concentrations, and the geometry of copolymer particles according to the block ratio of copolymers. We hope that the synthesized copolymers will present in broad applications of liquid crystals and reinforcement materials.
Experimental
Materials
Hexyl isocyanate (97%), titanium (IV) chloride solution (1M, ≥99%), 4-Bromo-2,6-dimethylphenol (BDMP, 97%) purified by recrystallization with n-heptane (Merck, 99%), and anhydrous tetrahydrofuran (THF, ≥99.9%) were purchased from Sigma-Aldrich. Pure poly (phenylene oxide) (PPO) was synthesized in our laboratory (
Characterization
Proton and decoupled carbon nuclear magnetic resonances (1H NMR and 13C NMR) 500 MHz Bruker spectrometer were used to study the microstructure of polymer in CDCl3 solvent. Fourier transform infrared spectroscopy (FTIR) spectra were recorded in KBr disks using a Perkin-Elmer RXI FTIR spectrometer from 400 cm−1 to 4000 cm−1. The molecular weights of the polymers were measured by gel permeation chromatography GPC-Agilent 1100 in THF (flow rate = 1 mL/min, 23°C). DSC measurements were performed on a Mettler Toledo-DSC1 differential scanning calorimeter. 3 mg samples were weighed and placed into aluminum pans. Heating and cooling scans were carried out at 10˚C/min. Thermogravimetric analysis (TGA) was performed with a Mettler Toledo-TGA thermogravimetric analyzer under an N2 with a rate of 10˚C/min. X-Ray diffraction (XRD) data were collected on an X’Pert MPD-Philips diffractometer using a cobalt tube (40 kV and 40 mA). The resulting patterns were acquired in the range of diffraction angles from 10 to 70° in a step size of 0.02°/s. The surface morphology, size, and shape of samples were observed using scanning electron microscopy (SEM) with a KYKY-EM3200 (from 20 to 26 kV) microscope. A syringe pump (Allied syrn 200) was used to prepare polymeric particles. The LC phase of samples was identified using polarized optical microscopy (POM) (Leitz Wetzlar 307–148.002514687) equipped with a Zeiss camera.
Synthesis of poly (phenylene oxide)-block-poly (hexyl isocyanate)
The poly (phenylene oxide)-block-poly (hexyl isocyanate) (PPO-b-PHIC) was synthesized using organotitanium coordination polymerization reported by Novak et al.34,35 Scheme 1 shows different steps of PPO-b-PHIC synthesis. In a 15 mL test tube, PPO ( Different steps of PPO-b-PHIC synthesis.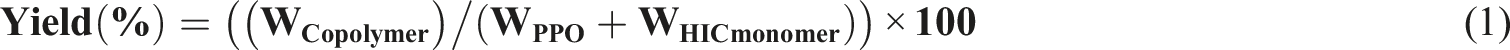

Preparation of polymeric particles
The polymeric particles were prepared from 1 mL concentrated solution of the synthesized copolymer in pure toluene (60% wt) at room temperature. The solution was injected in methanol as a non-solvent (20 mL, rotating at 500 r/min) using a syringe pump with a rate of 1 mL/h. The polymeric particles simultaneously appeared in methanol with the injection process. After 1 h, settled particles were separated from methanol and dried at room temperature under a vacuum.
Result and discussion
FTIR, 1H NMR, and 13C NMR analysis of copolymer
FTIR spectrum of PPO ( FTIR spectra of HIC monomer, PPO (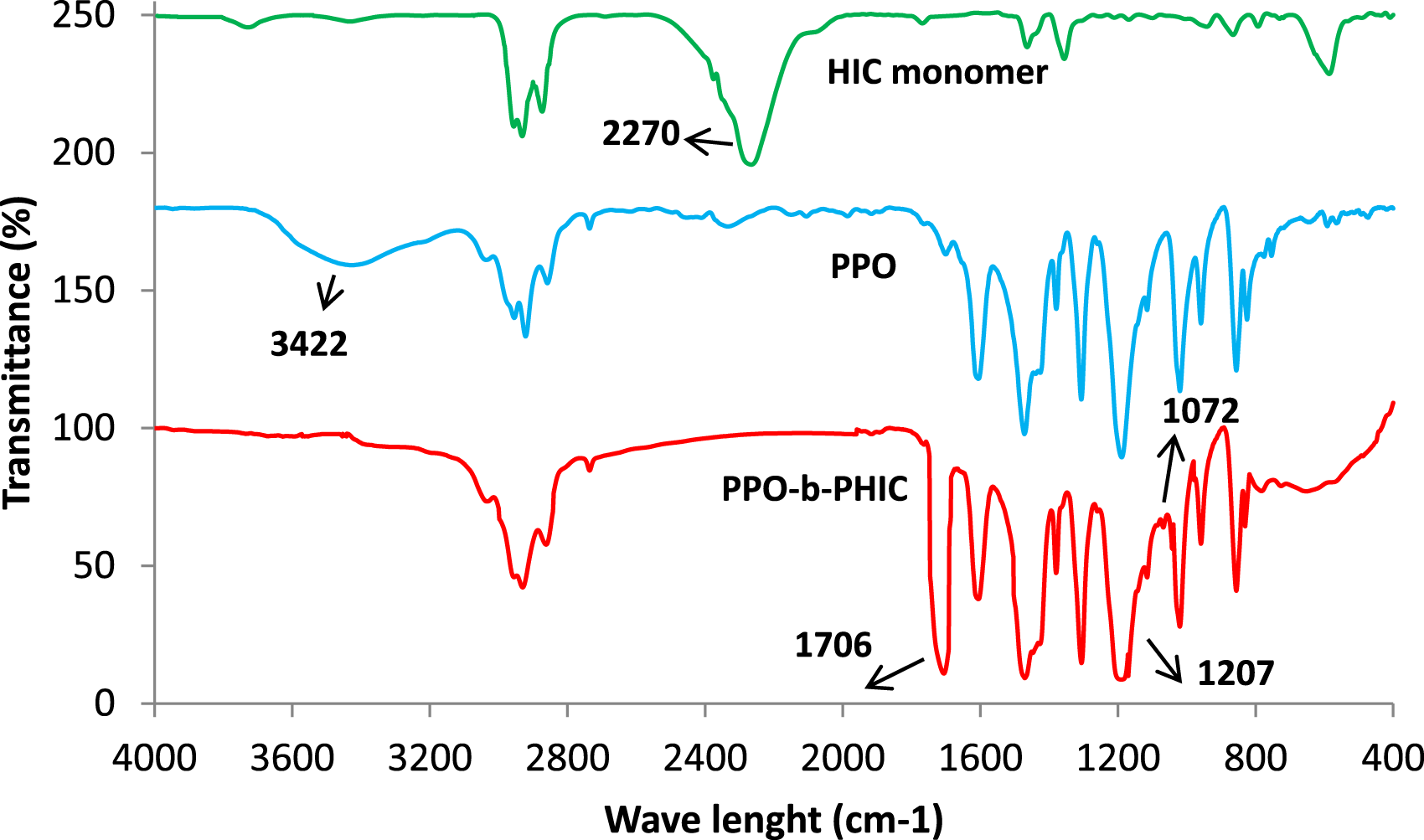
Figure 2(a) and (b) shows the 1H NMR spectra obtained for P1. All signals were assigned to the corresponding protons. It is clear that block copolymer has successfully been synthesized. The signals are seen at about 6.456, and 2.046–2.109 ppm corresponds to the aromatic protons (a) and methyl protons (b) of PPO, respectively.
43
The peak of the methyl group of PHIC (c) appears at 0.859–0.886 ppm. Hydrogen atoms of the methylene groups of PHIC were as follow: (d, e, f, 1.263–1.326 ppm), (g, 1.445–1.521ppm), (h, 3.615–3.682 ppm).19,20,23,24,30 The end of the chain (i) gives a weak peak at 7.074 ppm.30,35,44 Number-average molecular weight 1H NMR (a,b) and 13C NMR (c) spectrum of PPO-b-PHIC copolymer (sample P1).

The effect of the ether-amide block ratio on the physical properties of liquid crystals was considered. Therefore, the copolymer was also synthesized from PPO with 1H NMR spectrum of PPO-b-PHIC copolymer (P2 sample) (up) and BDMP-b-PHIC (P3 sample) (down). Results from 1H NMR of samples.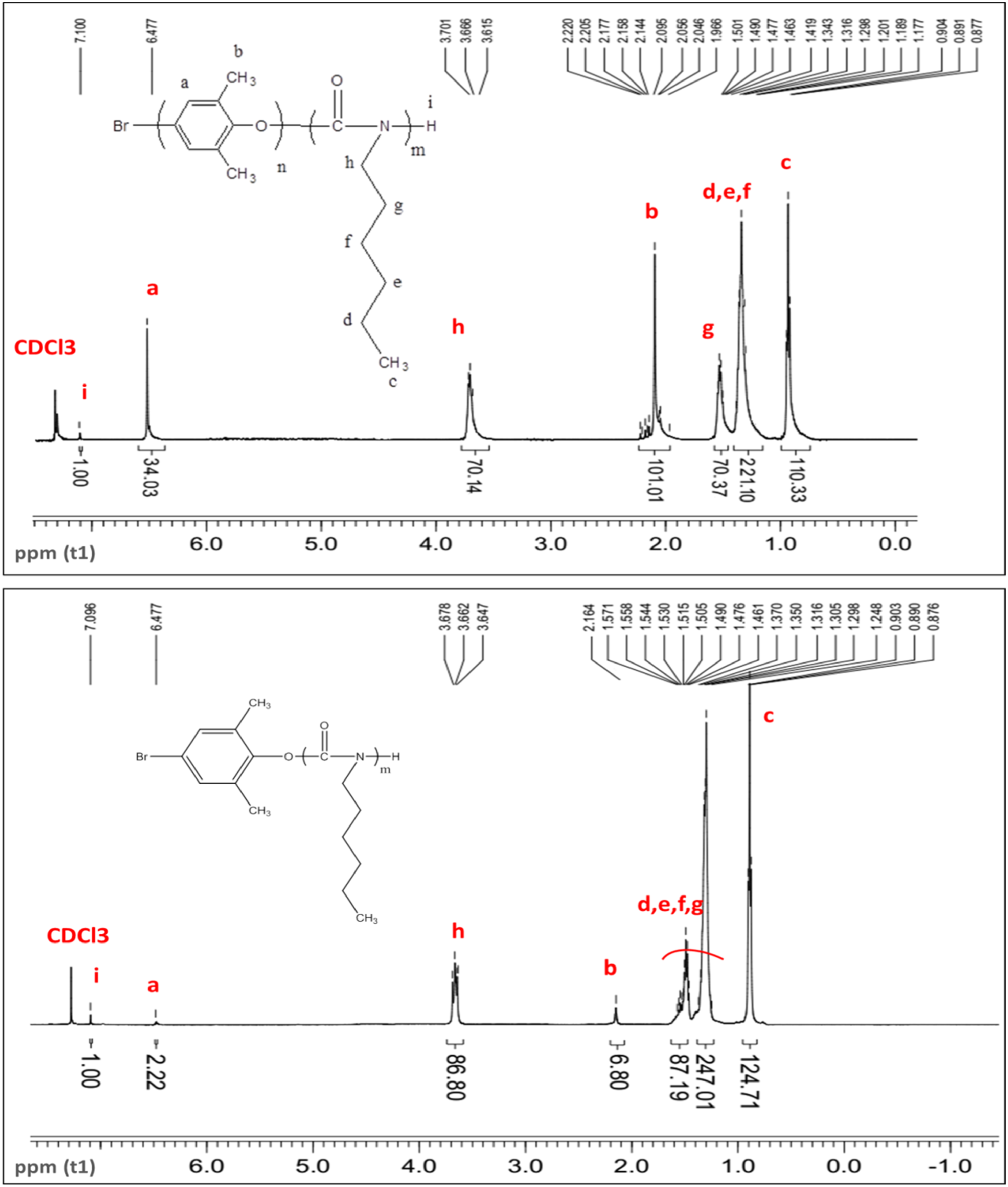

DSC analysis of P1, P2, and P3 samples:
The DSC results of samples are shown in Figure 4. The glass transitions of blocks (TgPPO and TgPHIC) for different ratios of n: m are listed in Table 3. TgPHIC was in good agreement with that reported by other researchers.23,24,31,50 The heating and cooling curves exhibit a broad endothermal peak from 20 to 90°C and a broad exothermal peak at 80 to 3°C, respectively due to conformation change of the PHIC chain (Tconf,tr).17,18,51 DSC curves of P1, P2 and P3 samples. Data obtained from DSC thermograph.

Peak intensity decreases with a decreasing number of ether units, indicating decreasing conformational energy of copolymers from rod to coil and vice versa. The steric hindrance due to the ether block restricts the chain conformation of copolymers. Size and optimal geometry of polymeric chains of samples synthesized in various ratios of n: m were estimated by material studio software (Figure 5). As can be seen, the molecular dimensions increased with the length of the ether block, which confirms the increase of rod to coil conformational energy. Rod to coil conformations and dynamics of the nematic LCPs was investigated by Warner et al.
52
using mathematical relations. The conformational behavior of poly-alkyl isocyanates (such as poly-hexyl isocyanate) was also investigated as a function of the relaxation time of chains using dipole relaxation analysis (DR).
53
Material studio simulation for samples (a) P1, (b) P2 and (c) P3.
POM analysis of samples
Figures 6 and 7 show microphotographs of the LC texture of the samples taken under a polarized optical microscope (POM) equipped with a heater (POM photos taken during heating), and the results are summarized in Table 4. Results show that the solution’s tendency to form anisotropic phases has increased with decreasing the ether-amide block ratio. This is because the PHIC is a LC block in the chemical structure of the copolymer. The higher percentage of LC block in the structure, the higher tendency of the copolymer to form an anisotropic phase.22,25–30,32,39,50,54 The phase transition of the solution is a function of two parameters of temperature and concentration. The orientation change of LC molecules as a function of free energy and temperature was discussed by Guo et al.
55
The effect of solution concentration (0.2, 0.4, and 0.6 mg/mL in toluene) and temperature (25–100°C) on the LC texture of samples was investigated. As can be seen, for 0.2 and 0.4 mg/mL of P1 in toluene, only the isotropic phase (I in Table 4) was observed (black under the polarized microscope). When the copolymer concentration in toluene reached 0.6 mg/mL (Figure 6(a) and (b)), the copolymer formed an anisotropic phase (AI in Table 4). The images show a mixture of colors without any shear or mechanical strain, indicating that the copolymer solution is birefringent at room temperature. The results obtained from POM micrographs are similar to those reported for other copolymers of PHIC.17,31 In Figure 6 (c),(d),(e),(f), with the decrease in the ratio of ether block in copolymer P2, the LC phase was observed for 0.4 mg/mL solution concentration at 25°C (Table 4). As the ether block ratio of the copolymer was increased, the rainbow circular nematic disclinations were observed under a polarized microscope. The emergence of the circular texture of the nematic phase is related to the strength-number of LC56,57 (In this case, the strength-number is equal to +1) (see supplementary data, Figure S1). The colors are due to the variation in the molecular orientation of LC copolymers in toluene. As the copolymer concentration in the solution is 0.4 mg/mL, the banded and circular textures coexist. Polarized optical micrographs of (a,b) 0.6 mg/mL solution of copolymer P1 in toluene at 25°C and 45°C and 0.4 and 0.6 mg/mL solution of copolymer P2 in toluene at 25°C and 45°C (c,d,e,f). Polarized optical micrographs of 0.2, 0.4 and 0.6 mg/mL solution of BDMP-b-PHIC (P3) in toluene at 25°C, 45°C and 60°C. Liquid crystal phase transition of copolymers (PPOn-b-PHICm) as a function of concentration and temperature (I: Isotropic, AI: Anisotropic).


Investigations by POM for BDMP-b-PHIC (P3) showed that all samples (0.2, 0.4, and 0.6 mg/mL) exhibit a LC phase at 25°C and 45°C. LC mesophase was also seen for samples with the solution concentrations of 0.4 and 0.6 mg/mL at 60°C (Figure 7 and Table 4). The thread texture of the nematic phase can be seen for 0.2 and 0.4 mg/mL concentrations. The disclinations with a strength-number of +1 were observed for a solution concentration of 0.6 mg/mL at 25–60°C (see Figure S2). Complex tensors of orientations are calculated to find the relationship between the texture and strength-number of liquid crystals.58–60
SEM analysis
Figure 8 shows SEM images of the particles of copolymers in different ratios of ether-amide blocks (n:m). As can be seen, the prepared particles change to a fiber-like shape by increasing the amide’s block length (P3 sample). The helical structure of PHIC19,23 tends to form fibers. Electrospinning of PHIC with a high molecular weight of 76,000 g/mol was also reported by Lin et al.
61
The particle size range in SEM images is 322.6 nm-2.847 μm (Figure 8 (b)), 445.1 nm-2.889 μm (Figure 8 (c)), and 279.1 nm–46.3 nm (Figure 8 (f)). SEM images of prepared particles as a function of ether-amide ratio (n:m) (a,b) P1, (c,d) P2, (e,f) P3.
XRD analysis
X-Ray diffraction of particles of PPO-b-PHIC copolymers (P1 and P2) and BDMP-b-PHIC (P3) shows three broad peaks from 13° to 22° (Figure 9). The appearance peaks were attributed to the physical structure of PHIC.17,22,62 The intensity of peaks increased with increasing the amide-ether block ratio which is in good agreement with particle shape in SEM images. X-ray diffraction pattern (solid state) of copolymers at room temperature a) P1, b) P2, c) P3.
TGA analysis
The thermal stability of the prepared particles and pure PPO was examined by TGA (Figure 10). It can be seen that P3 started to have a weight loss at about 158°C. In the TGA curve of P1, the weight loss occurs at 180°C. The weight loss of P2 appears in the temperature range similar to that observed with P1. The thermal stability of the three products is clearly lower than that of the PPO homopolymer. This is because of the low thermal stability of the PHIC block (160–200°C)18,22,31,51,63,64 in copolymer so that residual of P3 approaches zero at the end of the TGA thermogram (at 700°C). TGA thermograms of PPO and PPO-b-PHIC copolymers (P1 and P2) and BDMP-b-PHIC (P3).
Conclusion
Three different block ratios of the PPOn-b-PHICm and BDMP-b-PHIC were synthesized using coordination polymerization. DSC results clearly showed that the conformational energy of copolymers decreases with increasing the amide block ratio. The various nematic textures were observed for the solution of synthesized samples in toluene. The sample tendency to form the anisotropic orientation increases by increasing the amide block ratio. The formation of the anisotropic phase and the type of nematic texture are both temperature and concentration-dependent. A fibrous-like shape was observed under SEM for the particles obtained from the sample with an amide block ratio of 41.57. The particles of copolymers with ratios of 25.50:37.13 and 17.02:36.78 were irregular. The XRD was in good agreement with that obtained from SEM images. TGA showed that the thermal stability of copolymers increases with increasing the ether block ratio in molecular structure.
Supplemental Material
Supplemental Material - High-performance family of polymeric particles prepared from poly(phenylene oxide)-poly(hexyl isocyanate) liquid crystal block copolymer: synthesis and properties study
Supplemental Material for Improving Trendelenburg position effectiveness by varying cardiopulmonary bypass flow by Mohammad Ali Semsarzadeh, Arezoo Sh Dadkhah and Alireza sabzevari in Polymers and Polymer Composites
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
