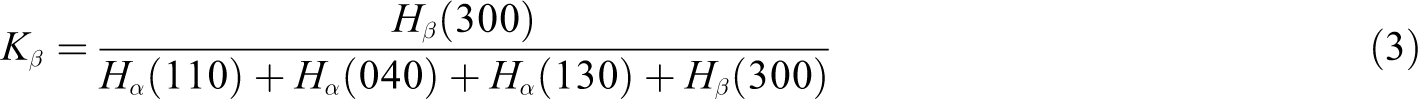Abstract
In this study, a new liquid crystal polymer, named as smectic liquid crystal polymer (LCPs-P), was synthesized by reacting diosgenyl 4-(10-undecen-1-yloyloxy) benzoate with polymethylhydrosiloxane using chloroplatinic acid as a catalyst. The mesomorphic properties of the LCPs-P were characterized with polarizing optical microscopy, differential scanning calorimetry (DSC), thermogravimetric analysis, and X-ray diffraction (XRD). Then, the influence of the LCPs-P as a new nucleator on the crystallization structures, thermal properties, and kinetic properties of isotactic polypropylene (iPP) was investigated by XRD and DSC. The experimental results indicated that the LCPs-P exhibited fan-shaped texture of a smectic A phase between 55°C and 318°C. The content of the β-crystal (K β) of the iPP blends mainly depended on the LCPs-P content and the crystallization temperature. By optimally combining the LCPs-P content with the crystallization temperature, a high K β could be obtained. When the content of the LCPs-P was 0.9 wt%, the maximum K β could reach 51% after isothermal crystallization for 1 h at 130°C. Moreover, the addition of the LCPs-P could increase the crystallization temperature and shorten the crystallization time of the iPP blends.
Introduction
As a kind of thermoplastic resin with good rigidity, great heat resistance, and easy molding, isotactic polypropylene (iPP) has always attracted attention due to wide application in various areas, such as industrial materials, household appliances, composite fibers, and portable containers. However, application exploration of the iPP has been hampered by several limitations owing to its poor impact strength, low temperature ductility, and dimensional stability of injection-molded products. The crystal structure of the iPP has a critical effect on its physical and mechanical properties. It is known that the iPP can exhibit in different crystal forms such as α
In general, the β-nucleator can be divided into organic compounds and inorganic compounds. The organic β-nucleators reported in the literature include some dyes, fused ring aromatic compounds, carboxylic acid derivatives, carboxylate complexes, and the aromatic amides. Some aromatic amides, such as 2,6-benzenedioic acid cyclohexamide and N,N-dicyclohexyl-2,6-naphthalene diamide (DND), exhibit excellent β-crystal nucleation effect. 32 Under appropriate conditions, the addition of 0.5% DND to the iPP can make the β-crystal content close to 100%. At present, some small molecule β-nucleators have been used in industrial application. Although small molecule β-nucleators have high nucleation efficiency, they also have some problems such as strong agglomeration, poor dispersibility, and easy precipitation from the iPP matrix.
As a new type of polymer additive, liquid crystal (LC) polymers with the ordered orientation have been used in the modification of the resins. Comparing small molecule modifier, the LC polymer nucleator with good compatibility can effectively prevent the reduction of material strength. Therefore, it is very important to study different kinds of the LC polymers as new nucleator with high efficiency and develop their applications in the iPP. In this work, a smectic LC polymer (LCPs-P), as a new β-nucleator, was synthesized. The LC properties of the LCPs-P were characterized by polarizing optical microscopy (POM), differential scanning calorimetry (DSC), thermogravimetric analysis (TGA), and X-ray diffraction (XRD). Then, the crystallization structure and thermal behavior of the iPP blends with the LCPs-P were investigated with XRD and DSC. The influence of the LCPs-P content and crystallization temperature on the content of the β-crystal of the iPP was discussed.
Experimental method
Materials
The iPP used in this study was T30S powder with a melt flow index of 2.3 g/10 min (230°C, 23N) and melting temperature of 166.8°C from the Jilin Petrochemical Co. (Jilin, China). Diosgenin was obtained from Wuhan Chemical Industry Co., Ltd (Wuhan, China). 4-Hydroxybenzoic acid was purchased from Shanghai Wulian Chemical Plant (Shanghai, China). Undecenoic acid was purchased from Liuan Chemical Plant (Liuan, China). Polymethylhydrosiloxane (PMHS, DP = 35) was purchased from Sigma-Aldrich Corporation (Saint Louis City, Missouri, USA). Chloroplatnic acid was purchased from Shenyang Jinke Reagent Factory (Shenyang, China).
Measurements
Fourier transform infrared spectroscopy
Fourier transform infrared (FTIR) spectra were obtained on a PerkinElmer spectrum One (B) spectrometer (PerkinElmer, Foster City, California, USA) using a KBr pellet.
Polarizing optical microscopy
The optical textures and phase transition temperature of the LCPs-P were observed with a Leica DMRX POM (Leica, Germany) equipped with a Linkam THMSE-600 (Linkam, UK) cool stage.
Differential scanning calorimetry
The thermal properties of the LCPs-P and blend samples were measured with a Netzsch 204 (Netzsch, Hanau, Germany) DSC equipped with a cooling system at a heating and cooling rate of 10°C min−1 at nitrogen (N2) atmosphere.
The blend samples were heated from room temperature to 210°C, kept at 210°C for 5 min to eliminate the thermal history, and then cooled quickly to the crystallization temperature selected in this study of 140, 135, 130, 125, 120, 115, and 110°C for 1 h, and then cooled to the room temperature and heated again to 210°C at a rate of 10°C min−1. The heating and cooling curves were recorded.
According to the following equation, the values of the β-crystal content (Φ β) can be obtained 35
where q is the ratio of the equilibrium fusion enthalpy for the α-crystal and β-crystal. The measured fusion enthalpy of the pure α-crystal and β-crystal is 170.0 and 168.5 J g−1, respectively, and q equals to 1.01. 36
Thermogravimetric analysis
The decomposition temperature of the LCPs-P was determined at N2 atmosphere using a Netzsch 209C TGA at a heating rate of 20°C min−1.
X-ray diffraction
The mesophase structure of the LCPs-P and crystallization structure of the iPP blend samples were performed by XRD, and the patterns were obtained using a nickel-filtered Cu Kα radiation with a DMAX-3A Rigaku powder diffractometer (Rigaku, Japan).
According to Turner-Jones equation, 37 the relative content of the β-crystal (K β) for the iPP can be calculated
where H β(300) is the typical diffraction peak intensity of the (300) plane of the β-crystal at 2θ = 16.0°, while H α(110), H α(040), and H α(130) are the peak intensities of other α-crystal assigned to the (110), (040), and (130) planes.
Synthesis of the monomer
The LC monomer (M LC) was prepared according to the method reported by Hu et al. 38 Yield: 43%, mp: 124.5°C. IR (KBr, cm−1): 3078 (=C−H); 2926, 2852 (CH3−, −CH2−); 1763, 1720 (C=O); 1641 (C=C); 1605, 1506 (Ar−).
Synthesis of the polymer
The synthesis route of the LC polymer (LCPs-P) is shown in Figure 1. The synthesis of the LCPs-P is described as follows. The monomer M LC (12.3 g, 17.5 mmol) and PMHS (1.6 g, 17.5 mmol) were placed in a three-necked flask, and then 50 mL of dry toluene was added. The reaction mixture was stirred under N2 and slowly heated to 60°C, and 2 mL of chloroplatinic acid catalyst (0.2 g of chloroplatinic acid dissolved in 50 mL of dry tetrahydrofuran) was added. Then, the reaction mixture was stirred for 48 h at 70°C. After the completion of the reaction, the toluene was distilled off, the concentrated solution was poured into methanol, and the precipitate was filtered off. The crude product was purified with ethanol and dried under a vacuum. Yield: 96%. IR (KBr, cm−1): 2927, 2854 (CH3−,−CH2−); 1765, 1717 (C=O); 1604, 1504 (Ar−); 1272–1016 (C−O, Si−O, Si−C).

The synthesis route of LCPs-P.
Preparation of the blend samples
Firstly, the iPP was put into oven and dried for 2 h. Then the iPP was mixed with different content of the LCPs-P (0.1, 0.3, 0.5, 0.7, 0.9, and 1.1 wt%). The dry mixture was fed into the internal mixer (SU-70C model), and blended for 10 min at 185°C with a screw speed of 70 r min−1. The pellets of the blends were placed between two glass slides at a hot stage, kept at 200°C for 10 min to avoid thermal history, then the samples were quickly transferred to the other hot stage with various temperature (110, 115, 120, 125, 130, 135, and 140°C), kept for 1 h, and quenched in ice water. The pure iPP was treated in the same way for a comparison.
Results and discussion
LC properties of LCPs-P
POM analysis
POM is used to observe the typical optical textures of the mesophase, and record the glass transition temperature (T g) or melting temperature, and the isotropic temperature. The POM results showed that the LCPs-P exhibited a color texture at 55°C and fan-shaped texture of a smectic A phase at 90°C. When the temperature rose to 318°C, the texture began to disappear. Therefore, the LCPs-P could exhibit good mesomorphism at the main crystallization temperature range of the iPP (i.e. between 90°C and 140°C), which meets the design of this study. The optical texture of the LCPs-P is shown in Figure 2.

Optical textures of LCPs-P.
DSC analysis
DSC is used to study the thermal properties of the LC polymer. According to the DSC curves, the corresponding phase transition temperatures and enthalpy changes can be obtained. In general, the T g and isotropic temperature (T i) can be observed for amorphous LC polymer. DSC curve of the LCPs-P, shown in Figure 3, displayed a glass transition at 64.0°C and a smectic to isotropic phase transition at 320.7°C, which agreed with the POM results.

DSC curve of LCPs-P.
TGA analysis
In general, the nucleator is required to have a certain heat resistance during the heat treatment of the modified iPP. The thermal stability of the LCPs-P was detected with TGA. The thermal decomposition temperature (T d), at which 5 wt% weight loss of the polymer occurred, was usually used to characterize its stability. As shown in Figure 4, the curve of the LCPs-P showed the T d of 333.4°C, which indicated that the LCPs-P had good thermal stability and could meet the requirement of processing temperature for the iPP.
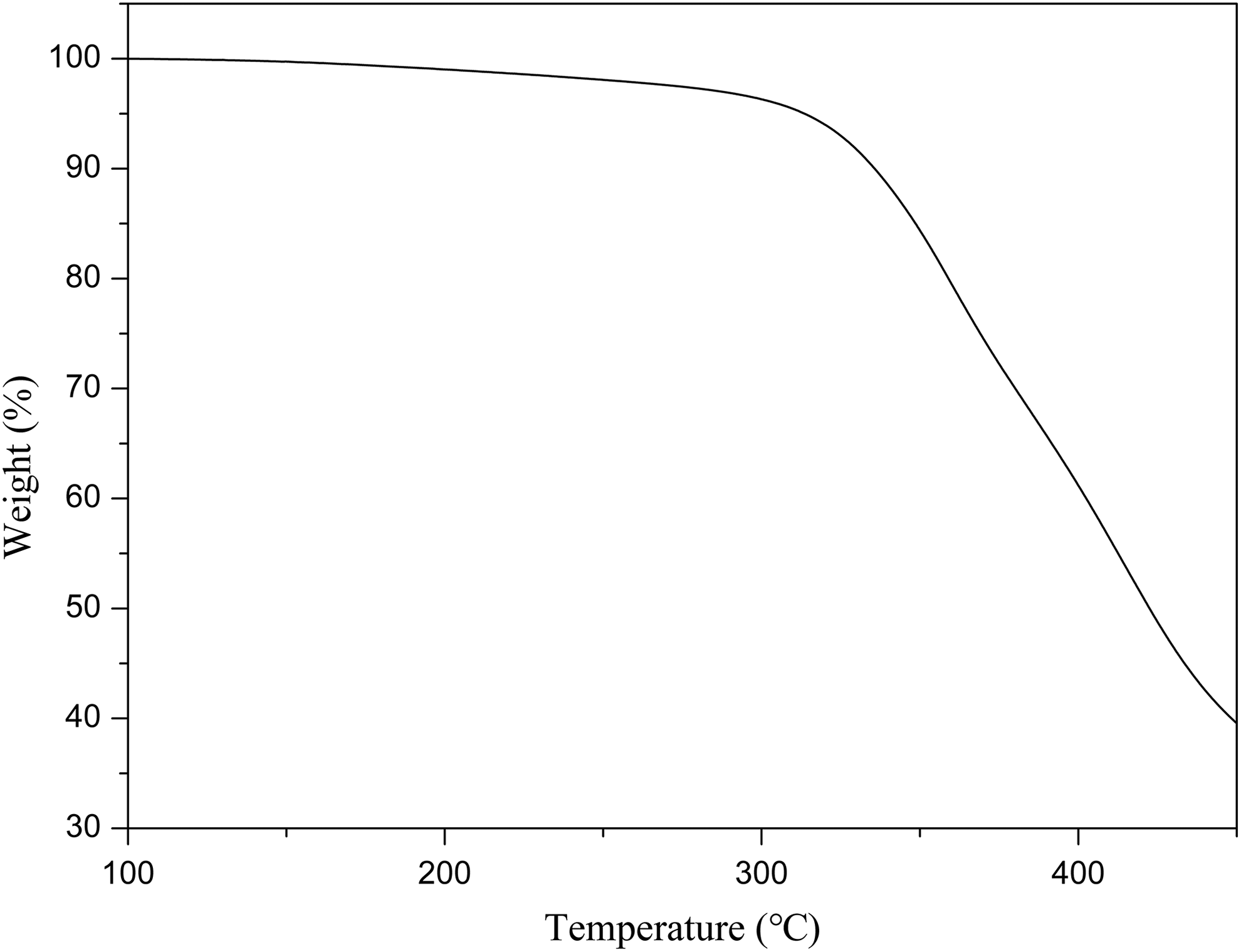
TGA curve of LCPs-P.
XRD analysis
XRD measurement is carried out to identify the mesophase structure, especially the determination of the smectic phase. Figure 5 shows the XRD pattern of the LCPs-P. The result showed that a sharp peak associated with the smectic layers appeared at 2 = 2.2°, indicating the LC molecules arranged in layers with short range, and a broad peak associated with the lateral packing appeared at 2θ = 16.7°, representing the average lateral distance between the adjacent LC groups. So the smectic phase structure of the LCPs-P was confirmed according to the POM and XRD results.

XRD pattern of LCPs-P.
Crystallization structure
Effect of LCPs-P content on crystallization structure
Figure 6 shows XRD patterns of the iPP with different LCPs-P content crystallized for 1 h at 130°C. As seen in Figure 6, the pure iPP exhibited five diffraction peaks at 2θ of 14.0°, 16.8°, 18.4°, 21.0°, and 21.8°, assigning to the (110), (040), (130), (131), and (111) reflections of the α-crystal, respectively. However, all the blend samples showed the characteristic diffraction peak of the β-crystal at 2θ = 16.0° attributing to the (300) reflection besides the diffraction peaks of the α-crystal. The results indicated that the addition of the LCPs-P could provide β-crystal nucleus to the blend system and induce the formation of the β-crystal in the iPP.

XRD patterns of the iPP blends with different LCPs-P content isothermally crystallized for 1 h at 130°C from melt.
The K β of the blend samples could be obtained according to equation (3). The effect of the LCPs-P content on the K β is shown in Figure 7. With increasing the LCPs-P content, the K β value tended to first increase and then decrease. The K β reached a maximum of 51% when the LCPs-P content was 0.9 wt%. The reason was explained as follows. The self-orientation ability of the LCPs-P could provide considerable heterogeneous nuclei number for the blend system throughout the process. Moreover, appropriate increase of the LCPs-P content was in favor of the formation of the β-crystal. When the blend system could not accommodate too much nucleator, the LC molecules would not be used as the β nucleus, which would lead to the decrease of the K β value.

K β and Φ β values as a function of LCPs-P content.
Effect of temperature on crystallization structure
The XRD patterns of the iPP blend with 0.9 wt% LCPs-P at various crystallization temperatures for 1 h are shown in Figure 8. As known, the formation of the β-crystal is very sensitive to the temperature. It was obvious that the typical diffraction peak of the β-crystal at 2θ = 16.0° could be observed at the temperature range from 115°C to 140°C. Moreover, with the change of the temperature, the growth rate of the β-crystal changed greatly. The K β showed a trend of first increase and then decrease with the increase of the crystallization temperature. The optimum crystallization temperature of the LCPs-P was 130°C, and the corresponding maximum value of the K β was 51%.

XRD patterns of the iPP blend with 0.9 wt% LCPs-P isothermally crystallized for 1 h at various crystallization temperatures.
Figure 9 shows the K β value as a function of the crystallization temperature. When the crystallization temperatures increased from 110°C to 130°C, the K β value rapidly increased from 4% to 51%. However, when the crystallization temperatures increased from 130°C to 140°C, the K β value decreased from 51% to 14%. This result indicated the upper limit temperature for β-crystal nucleation.

K β and Φ β values as a function of the crystallization temperature.
According to the competing kinetic and thermodynamic effect of the α- and β-crystals, 39 -41 the influence of the crystallization temperature on the K β value could be explained as follows. Compared with the α-crystal, the growth rate of the β-crystal was faster between 115°C and 130°C for the iPP blend with 0.9 wt% LCPs-P, so the formation of the β-crystal favored thermodynamically. This indicated that the upper limit of the β-crystal formation was 130°C. When the crystallization temperature was more than 130°C, the α-crystal became kinetically favored, so the K β value began to decrease.
Crystallization thermodynamics
Effect of LCPs-P content on thermal properties
According to the DSC curves in Figure 10, the pure iPP only showed a melting peak of the α-crystal at 166.8°C. However, with the addition of the LCPs-P into the iPP, the blend samples revealed two melting peaks, representing the melting transitions of the β-crystal at 151.0–155.6°C and the α-crystal at 165.0–167.9°C, respectively. When increasing the LCPs-P content, the corresponding peak area of the β-crystal first increased and then decreased, which were well in agreement with XRD result.

DSC melting curves of the iPP blends with LCPs-P isothermally crystallized for 1 h at 130°C.
The Φ β value was calculated according to equations (1) and (2). As could be clearly seen in Figure 7, the Φ β value increased with increasing the LCPs-P content from 0.1 wt% to 0.9 wt%, moreover, the change trend of the K β value from XRD and the Φ β value from DSC was consistent. In general, XRD was more accurate than DSC for evaluating the relative content of the β-crystal. It was very difficult to obtain the accurate enthalpy change because there was overlap between the melting peaks of the α- and β-crystals.
Effect of crystallization temperature on thermal properties
DSC curves of the iPP with 0.9 wt% LCPs-P content at different crystallization temperature are presented in Figure 11. No melting peak of the β-crystal was detected at 110°C for 1 h. However, two melting peaks appeared between 115°C and 140°C. The typical melting peaks of the α-crystal appeared between 164.1°C and 169.7°C, and that of the β-crystal could be seen from 148.6°C to 159.4°C. With the increase of the crystallization temperature, the peak area or enthalpy change of the β-crystal tended to first increase and then decrease. The peak area or enthalpy change reached a maximum value at 130°C. So higher or lower crystallization temperature was not in favor of the formation of the β-crystal. The dependence of the Φ β value, calculated from equations (1) and (2), on the crystallization temperature is shown in Figure 9. The values of the K β and Φ β for the bend samples with same LCPs-P content were different, but the general trend was same, which indicated that the results obtained from DSC and XRD were consistent.

DSC heating curves of the iPP blends with LCPs-P.
Crystallization kinetics
The crystallization behavior of the iPP blends with different LCPs-P content was studied. Their DSC curves are shown in Figure 12, and the corresponding thermal analysis data are summarized in Table 1. According to Table 1, compared with the crystallization process of pure iPP, the peak shape of the blend samples was sharper with the addition of the LCPs-P nucleator, and the corresponding crystallization peak shifted to the high temperature region. The crystallization peak temperature (T c) of pure iPP was 109.6°C, and the T c of the iPP blend samples was enhanced to above 110.6°C. Therefore, the addition of the LCPs-P could make the iPP blends meet the heterogeneous nucleation effect, and also increase the crystallization onset temperature (T on) and T c of the iPP. In addition, the ΔT (ΔT = T on − T c) of pure iPP was 13.6°C, while the ΔT of the modified iPP decreased. The ΔT value could show the crystallization rate of the iPP sample, so the addition of the LCPs-P nucleator could shorten the crystallization time.

DSC crystallization curves of pure iPP and iPP with LCPs-P.
Thermal analysis data of LCPs-P/iPP.

iPP: isotactic polypropylene; T on: crystallization onset temperature; T c: crystallization peak temperature; ΔT = T on − T c.
Conclusions
In summary, a smectic LC polymer (LCPs-P) was synthesized and characterized, and then used as a new nucleator to induce the formation of β-crystals in the iPP. The content of the LCPs-P and crystallization temperature had a very important influence on the crystallization structure and thermal behavior of the iPP blends. The general trends of the K β value from XRD and Φ β value from DSC were similar. With increasing the LCPs-P content and crystallization temperature, the corresponding K β and Φ β value first increased and then decreased. A higher content of the β-crystal could be obtained through controlling the LCPs-P content and crystallization temperature. The maximum K β value reached 51% when the LCPs-P content, crystallization temperature, and time were 0.9 wt%, 130°C, and 1 h, respectively.
Supplemental material
Supplemental Material, Supporting_information - Study on crystallization structure and thermal behavior of iPP induced by a new liquid crystal nucleator
Supplemental Material, Supporting_information for Study on crystallization structure and thermal behavior of iPP induced by a new liquid crystal nucleator by Xiaoxu Xu, Zhengzheng Hao, Chao Fan, Qifan Chen, Yanhua Lu and Yaoqing Wu in Polymers and Polymer Composites
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was financially supported by the Foundation of Eastern Liaoning University (2017ZD004), National Natural Science Foundation of China (51873084), Department of Education of Liaoning Province (ldxy2017002), and Science and Technology Committee of Liaoning Province (201602342).
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.