Abstract
A comparative study was performed on the use of esterified alkaline lignin (AAL) and esterified organosolv lignin (AOSL) as the interfacial modifying agent to improve the properties of polypropylene-chitosan composites. Chitosan was chemically modified through a reaction with the esterified lignin in an alcohol medium. The composites were prepared using an internal mixer and hot-pressed method. Both modified chitosan showed a different chemical structure upon modification with the esterified lignins as confirmed by Fourier transform infrared spectroscopy spectra. With a lower molecular weight, the AOSL attachment on the chitosan surface resulted in more efficiency in decreasing hydrophilic characters. Tensile tests showed the increased tensile strength by 32.15% and 26.43% for AOSL-modified composites and AAL-modified composites as compared with the unmodified composites. Overall, the AOSL was superior in improving the mechanical strength and thermal stability of the composites, while the AAL exhibited the most apparent enhancement in ductility and crystallization.
Introduction
Nowadays, the use of natural fibers as reinforcing filler in the manufacturing of composite materials with desired properties has become a great of interest for a wide variety of applications, varying from automotive and military applications to use in sports equipment and civil infrastructures. 1 Natural fibers exhibit many attractive advantages over synthetic fibers, including low cost, low density, low processing requirements, less abrasion during processing, and biodegradability. 2 Moreover, with densities comparable to aramid fibers (1500 kg m−3), they can display specific strength and moduli higher than glass fibers. 3 Among the existing natural fibers, chitosan is known to possess excellent mechanical and thermal properties comparable to cellulose. Chitosan is extracted from crustaceous shells such as crabs, shrimp, and prawns.4–6 This biopolymer has extensively been used in many scientific and technological applications such as medicine, pharmacology, biotechnology, textile, and food industry, as well as fiber and plastic applications.7–9
Despite the advantages of chitosan, the main drawbacks of its using as reinforcing filler in polymer composites are the low degree of dispersion and poor interfacial adhesion which are attributed to the incompatibility between hydrophilic natural chitosan and hydrophobic matrix polymer. 10 This is demonstrated by the difficulty of the polar hydroxyl groups on the chitosan surface have in forming a well-bonded interface with a non-polar matrix polymer as the strength improvement relies on stress transfer on the composite’s interface, whereas the external force is applied. 11 If the interface is weak, failure will occur at fiber-matrix interface without any improvement on properties. 12 Consequently, this problem reduces the benefit of potential reinforcements in polymer composites.
To alleviate the issue of adhesion, interfacial interaction is modified in the composite system. To date, chemical modification is extensively applied with the priority to enhance the interfacial interaction in composite systems, because it is proven to be an effective method to reduce the hydrophilic character of natural fibers. 13 Nonetheless, chemical modification surely adds another step in the preparation of composites which consequently increase the production cost, declining one of the incentives to use natural fibers in composites. Therefore, the reagents used in the chemical modifications shall ideally be inexpensive and involved compounds obtained from renewable sources. 14
As one of the abundantly vegetal-derived materials, lignin is known because of its worldwide availability as a by-product of the wood pulping process. 15 Lignin is a highly stable polymer due to its complex aromatic structure which is connected by an ester-bridge. It contains large amounts of hydroxyl groups, both aliphatic and aromatic which provide high polarity. 16 In previous work, organosolv lignin had successfully been used as a “green” surface modifying agent in polypropylene-chitosan composites. It showed that tensile properties of treated composites with organosolv lignin were higher than the composites treated with synthetic reagents, implying the potential of organosolv lignin as a surface modifier in the composite materials. 17 In this study, two types of chemically modified lignins were used as a chemical modifying agent in polypropylene composites, and then their effectiveness was examined through the investigation of mechanical and thermal properties.
Lignin was extracted from lignocellulosic fiber using the organosolv process, and alkaline process (mercerization). The organosolv process consists of extracting lignin from lignocellulosic fiber by an organic solvent with water, while the alkaline process uses a strong base solution with water. Both processes are advantageous in resulting in lignin with low molecular weight and a large number of reaction sites, making it a more appropriate modifying agent. However, lignin is difficult to dissolve in common solvents due to the complexity inside its structure, constricting its chemical reactivity. 18 Therefore, a simple approach to enhance the lignin reactivity was carried out via an esterification reaction with acrylic acid. The lignin modification results in a pendant carboxylic moiety, providing a site upon which further reactive chemistry is possible. 19 This study is aimed to provide a comparative analysis of the mechanical and thermal properties of chitosan-filled polypropylene composites chemically modified with two kinds of different esterified lignin.
Materials and method
Raw materials
Physical and chemical properties of chitosan.

Extraction of organosolv lignin from lignocellulosic fiber
The extraction of organosolv lignin was carried out by using organosolv method developed by Hage et al. 20 Lignocellulosic fiber was modified with aqueous ethanol mixed with a catalyst, 1.2 w/w% sulfuric acid at 190°C for 1 h; the solid to liquid ratio was 1:8. The pre-modified fiber was then washed with warm ethanol. The washes were combined, and 3 vol% of water was added to precipitate the organosolv lignin. The organosolv lignin was centrifuged and directed to an oven for drying at 80°C for 24 h.
Extraction of alkaline lignin from lignocellulosic fiber
Alkaline lignin was extracted from lignocellulosic fiber according to the procedure conducted by Toledano and co-workers.
21
lignocellolusic fiber was cooked in 1
Preparation of esterified lignin
The chemical reaction of organosolv lignin and alkaline lignin with acrylic acid was carried out via an esterification reaction using the same procedure. Initially, 3.0 g of lignin sample was charged into the reactor together with 60 mL distilled water. The solution pH was adjusted to 8.5–9.0 with 2% NaOH solution. Acrylic acid was added slowly to the reaction mixture with constant stirring. Both pH and temperature were monitored during the reaction. Upon completion of the reaction, the product was separated by precipitation of the reaction mixture by 6
Preparation of modified chitosan
Chitosan fiber was chemically modified with acrylated lignin (acrylated organosolv lignin and acrylated alkaline lignin) in an ethanol medium under mild conditions. The acrylated lignin solution was prepared by dissolving the acrylated lignin in ethanol. The amount of acrylated lignin used was 3% by weight of chitosan fiber. Afterward, chitosan fiber was gradually added into the acrylated lignin solution and constantly stirred for the next 4 h. The chitosan solution was then filtered and dried in an oven at 80°C for 24 h.
Preparation of polypropylene-chitosan composites
Formulation of unmodified and modified polypropylene/chitosan composites at different filler content.

AAL: Acrylated alkaline lignin; AOSL: Acrylated organosolv lignin; php: part per hundred of polymer.
Characterization
Tensile tests were carried out according to ASTM D 638-91 on an Instron 5582. A minimum of five dumbbell specimens of each composition, 1 mm thick was cut from the molded sheets with a Wallace die cutter. A cross-head speed of 20 mm/min was used and the test was performed at 25 ± 3°C.
Impact tests were conducted on a Compact Charpy Impact Tester according to ASTM D-256, using notched samples. For each test, five specimens were measured and the average data were reported.
Thermogravimetric (TGA) and derivative thermogravimetric (DTG) analyses were performed with a TGA Q500 (Perkin Elmer Instrument). Samples were scanned from 30 to 600°C at a heating rate of 20°C/min using a nitrogen flow of 50 mL/min.
Differential scanning calorimetry (DSC) analysis was carried out with a DSC Q 1000 (Perkin Elmer TA Instrument). Samples were scanned from 25 to 250°C at a heating rate of 20°C/min using a nitrogen flow of 50 mL/min. The melting points and enthalpies of PP/chitosan composites were automatically calculated by the instrument software. The crystallinity of composites (X
c
) was manually calculated using equation (1)
The morphology of the tensile fracture surface of the PP/chitosan composites was investigated with a scanning electron microscope (SEM), JEOL model JSM 6260 LE. The fracture ends of specimens were mounted on aluminum stubs and sputter-coated with palladium to avoid electrostatic charging during the examination.
Fourier transform infrared spectroscopy (FTIR) was performed on unmodified and modified chitosan in ATR mode (Perkin Elmer 1600 Series). Samples were scanned from 650 to 4000 cm−1 with a resolution of 4 cm−1.
The number average molecular weight (Mn) and weight average molecular weight (Mw) of the lignin were determined by gel permeation chromatography (GPC) after acetylation of the lignin to allow dissolution in THF. 23 GPC analysis for lignin was performed using a Perkin Elmer instrument equipped with an interface (PE Series 900). Three Waters Styragel columns (HR 1, HR 2, and HR 3) ranging from 100 to 5 × 105 and a refractive index detector (Series 200) were employed, with a flow rate of 1 mL/min. The calibration was determined using polystyrene standards.
Results and discussion
Structural analysis of modified chitosan
Gel permeation chromatography was used to determine the number average molecular weight (Mn) and weight average molecular weight (Mw) of the lignin. The weighted average (Mw), number average (Mn) molecular weight, and polydispersity (Mw/Mn) of organosolv lignin and alkaline lignin were computed from the chromatogram. The organosolv lignin had an Mw of 3429 and an Mn of 2506, while the alkaline lignin had an Mw of 3988 and an Mn of 2621. These results indicated that both lignin extraction degraded the macromolecular structure of lignin to a noticeable extent. In addition, the low value of polydispersity for both kinds of lignin indicated a narrow molecular weight distribution.
FTIR analysis was used to examine a possible formation of chemical bonding of two types of esterified lignin with chitosan fiber. Thus, FTIR spectra for unmodified chitosan fiber, modified chitosan with acrylated organosolv lignin (AOSL), and modified chitosan with acrylated alkaline lignin (AAL), are illustrated in Figure 1. IR spectrum of unmodified chitosan exhibited the main characteristic peaks at 3358 cm−1 (O–H stretch), 2872 cm−1 (C–H stretch), 1675 cm−1 (N–H bend), and 1590 cm−1 (C=O stretch). Both modified chitosan clearly showed a difference in the absorption spectrum, indicating a difference in the chemical structure of chitosan upon the esterification. Distinct changes of the absorption spectrum taking place at the peak between 1760-1712 cm−1, and 3400-3200 cm−1 were due to the carbonyl groups (C=O) linked to the chitosan fiber as an ester bridge and the decreased content of hydroxyl groups (-OH) belonged to the chitosan, respectively. It was observed that the formation of the ester bond consequently resulted in the decreased hydrophilic character on the chitosan surface. Furthermore, there were slight shifts of the absorption peaks at 1675 cm−1 and 1590 cm−1 which corresponded to the amine bending vibration (N-H) and carbonyl stretch vibration (C=O) for both the modified chitosan. A relatively lower intensity of peak at 3292 cm−1 of modified chitosan with AOSL than that of the peak at 3301 cm−1 of the modified chitosan with AAL indicated a lower degree of hydrophilicity of the chitosan reacted with acrylated organosolv lignin. The hydrophilicity of chitosan decreased by 21.3% with acrylated organosolv lignin as compared to acrylated alkaline lignin which was 17.7%, concerning the unmodified chitosan. This outcome might be attributed to lower molecular weight of organosolv lignin than the alkaline lignin which provides to the higher organosolv lignin’s reactivity towards chitosan fiber. A possible schematic of the chemical reaction between chitosan fiber and esterified lignin is presented in Figure 2. Fourier transform infrared spectroscopy spectra of unmodified chitosan fiber, modified chitosan with esterified organosolv lignin, and modified chitosan with esterified alkaline lignin. A possible schematic of the chemical reaction between chitosan fiber and esterified lignin.

The surface modification of chitosan fiber through a chemical reaction with the esterified lignin was attributed by the formation of ester linkage (C–O–C) between the hydroxyl group (–OH) of esterified lignin and the carboxyl group (C–OH) belonged to chitosan fiber. The mechanism of chemical modification of the chitosan fiber followed a nucleophilic substitution reaction. In detail, lignin macromolecule (lignin–OH) used was chemically reacted with acrylic acid (CH2=CH–COOH) in a sodium hydroxide (NaOH) solution. The reaction was occurred through a nucleophilic substitution pathway that resulted a product that so-called acrylated lignin or esterified lignin (lignin–O–CO–CH=CH2). This product was brownish solid form. The chemical modification of chitosan was then conducted by mixing the esterified lignin (lignin–O–CO–CH=CH2) with the chitosan fiber in an ethyl alcohol solution. The reaction was observed to occur spontaneously between the both components via the formation of ester linkage, resulting a product of modified chitosan in the form of esterified lignin-modified chitosan (C3H3O–O –lignin–O– C56H102N9O38).
Mechanical properties of polypropylene-chitosan composites
Figure 3 exhibits stress versus strain traces recorded from tensile testing for neat PP, unmodified PP composite, modified PP composite with AOSL, and modified PP composite with AAL, respectively. All the PP composites contained 20 php of chitosan filler. It can be seen that all traces exhibited a similar trend whereas the incorporation of chitosan decreased the strength and strain of the composites, corresponding to poor interface interaction of PP and chitosan. Chitosan is a natural polysaccharide that possesses a hydrophilic character, while PP is a hydrophobic polymer. The difference of affinities diminished filler-matrix interfacial adhesion.
24
On the other hand, both modified PP/chitosan composites with esterified lignin showed a higher strength as compared to neat PP and unmodified composite. The higher strength of the modified composites with AOSL than that of the composite modified with AAL could be as a result of the lower molecular weight of AOSL that could facilitate the effectiveness of interfacial modification. Interestingly, the modified composite with AAL demonstrated a higher strain than the modified composite with AOSL and the unmodified composite. The elongation increased by 24.57% as compared with the unmodified composite, reflecting the enhanced chain mobility of the PP matrix within the composites containing AAL. Overall, it can be observed that the improvement of the composites' strength is dependent on interfacial compatibility. Although chemical modification did not affect the stress versus strain trace character of the modified composites; however, it influenced the measured strength at failure. Stress versus strain traces of tensile testing for neat PP, unmodified PP/chitosan composite, modified PP/chitosan composite with esterified organosolv lignin and modified PP/chitosan composite with esterified alkaline lignin. Chitosan content: 20 php. PP: polypropylene.
The tensile strength of unmodified and modified PP/chitosan composites at different filler content is shown in Figure 4. The increase of chitosan content dramatically decreased the tensile strength of PP composites. However, the modified composites showed higher tensile strength than the unmodified composites at the same chitosan content. Tensile strength of the modified composites with AAL improved by 26.43% as compared with the unmodified ones, indicating the enhanced filler-matrix interfacial adhesion. In meantime, the modified composites with AOSL showed improvement in tensile strength by 32.15% as compared with the unmodified composites, which was higher than the modified composites with AAL. This higher strength might be attributed to the lower molecular weight of AOSL than AAL that effectively facilitated the interfacial modification. Effect of chitosan content and acrylated lignin on tensile strength of polypropylene/chitosan composites.
Figure 5 shows Young’s modulus of unmodified and modified PP composites as a function of chitosan content. Young’s modulus of PP/chitosan composites increased with the increase of chitosan content. The incorporation of chitosan into the PP matrix improved the stiffness owing to the constricted segmental molecular motion of the PP chain. Young’s modulus of modified composites was remarkably higher than the unmodified composites. Young’s modulus of modified composites with AAL increased by 30.33% as compared with the unmodified ones. The rigid nature of filler, high crystallinity index, and the presence of modifying agent considerably play a vital role in the improvement of Young’s modulus of the composites. Moreover, Young’s modulus of modified composites with AOSL was higher about 55.25% than the unmodified composites. It is noticeable that the stiffness of the composites could be improved by enhancing the filler-matrix interfacial adhesion upon the presence of esterified lignin. Young’s modulus of unmodified and modified polypropylene/chitosan content at various chitosan content.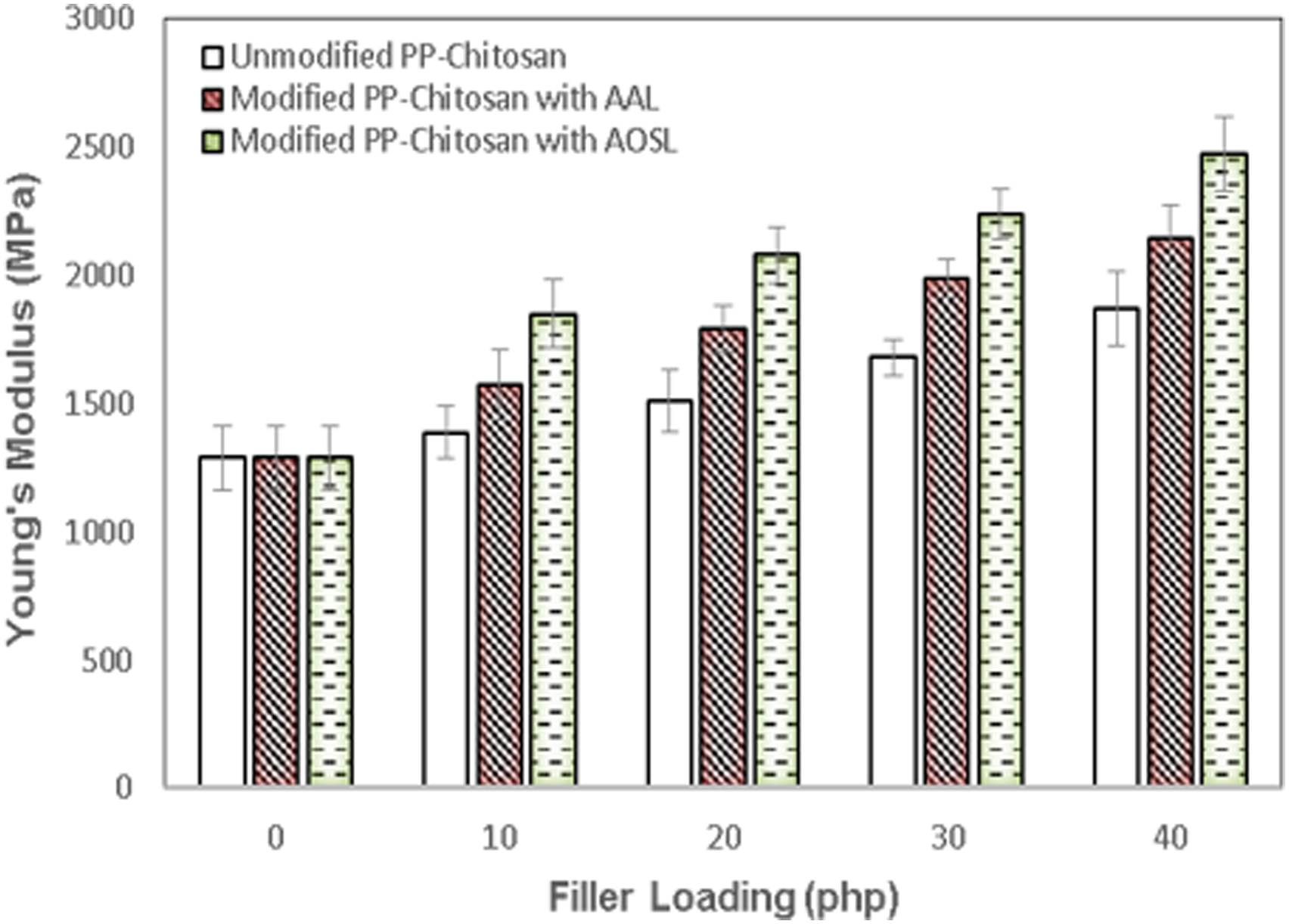
The impact strength of PP/chitosan composites at different filler content is depicted in Figure 6. All PP/chitosan composites exhibited the improvement of impact strength until 30 php of filler content, then it dramatically decreased at a higher filler content. The declined impact strength indicated that the filler–filler interaction was more favorable than the filler-matrix interaction at higher filler content exceeded 30 php, hence, creating stress concentration points, imparting the fracture process. The highest impact strength was found at 30 php filler content, considerably due to at this concentration the filler was likely well-wetted and homogeneously dispersed. Upon increasing the filler content, physical networks of filler agglomerated leading to the decreased impact strength. On the other hand, both modified composites were found to have higher impact strength than the unmodified composites, showing the higher capability of the modified composites to absorb a higher amount of energy to stop crack propagation. The presence of esterified lignin on the chitosan surface resulted in the improved impact strength owing to the enhanced interfacial adhesion. Lignin contains a large amount of carboxylic functional groups along its molecular backbone that can facilitate the toughening effect in polymer composites. The presence of appropriate amounts of lignin was obtained to induce the toughening which led to the increased toughness of the modified PP/chitosan composites. It was also found that the impact strength of the modified composites with AOSL was higher than the modified composites with AAL. This outcome conclusively deduces that AOSL modifying agent is more effective than AAL modifying agent in improving the fracture resistance of PP/chitosan composites. Impact strength of unmodified and modified polypropylene-chitosan composites as a function filler content.
Scanning electron microscope image analysis
SEM images of tensile fractured surfaces of PP matrix, unmodified PP composite, modified PP composite with AOSL, and modified composite with AAL are shown in Figure 7(a)–(d). The three composite categories contained 20 php of chitosan filler. As seen, SEM image of the PP matrix exhibited a quite smooth surface (Figure 7(a)). On the other hand, a few voids appeared on the fractured surface of the modified composite (Figure 7(b)), which demonstrates the filler detachment from the matrix. It indicates the poor wetting of the filler by the matrix. Consequently, the fracture occurred at the interface between chitosan and the PP matrix. The difference of affinities between the filler and the matrix led to a diminished interaction at the interface.
25
In contrast, a rather smooth surface associated with fewer voids was observed on both modified composites' surfaces (Figure 7(c) and (d)). This was related to a good wetting of the filler by the matrix since the modifying agents substantially hydrophobized the chitosan, leading to enhanced interfacial adhesion with the matrix. As a result, the filler pull-out traces in the matrix were considerably reduced in the modified composites. Scanning electron microscope micrographs of PP/chitosan composites with 20 php filler content, (a) neat PP, (b) untreated composite, (c) treated composite with esterified organosolv lignin, (d) treated composite with esterified alkaline lignin. PP: polypropylene.
Compared to the modified composite with AOSL (Figure 7(c)), the micrograph of the modified composite with AAL (Figure 7(d)) displayed the appearance of tear-traces on the surface which might correspond to the ductile behavior of the composites. It points out that the presence of AAL in the PP/chitosan composites contributed to the enhanced interfacial adhesion as well as the ductility. Additionally, the outcome of the better interfacial bonding between the chitosan and the PP matrix was reflected in the improvement of the tensile properties of the modified PP/chitosan composites.
Thermal properties of polypropylene-chitosan composites
Figure 8(a) and (b) presences the TGA and DTG curves of neat PP, unmodified PP/chitosan composites, modified PP/chitosan composites with AOSL, and modified PP/chitosan composites with AAL, respectively. Thermal decomposition of neat PP was occurred over a one-stage process between 300 and 415°C, resulting in gaseous products. This degradation confirmed that PP is composed of carbon-carbon bonds and that the degradation/depolymerisation takes place at the weak sites of the PP chain.
26
On the other hand, three distinct stages in the thermal decomposition could be observed for the PP/chitosan composites. They were approximately situated in the temperature ranges of 160–180°C, 260–340°C, and 400–490°C. These typical thermograms exemplified the similarity in the general patterns. However, the samples could be differentiated by their characteristic temperature and weights losses. The first stage was assigned to the release of typical strong hydrogen-bonded water and evaporation of volatile compounds from the samples, while the second stage corresponded to decomposition and depolymerization of both chitosan fiber and the modifying agent. The third stage might be attributable to the decomposition of char residue formed from the second stage. Effect of chitosan addition and chemical modification on thermogravimetric properties of polypropylene/chitosan composites. Chitosan content: 20 php (a) weight loss versus temperature curves, (b) derivative thermogravimetry curves.
Percentage of weight loss of unmodified and modified polypropylene composites containing of 20 php of chitosan filler at different temperature.

Figure 9 shows representative DSC curves for neat PP, unmodified PP composites, modified PP composite with AOSL, and modified PP composite with AAL, respectively, the latter three containing 20 php of chitosan filler. Table 4 summarizes the melting temperature (Tm), the fusion enthalpy (ΔHf) and the degree of crystallinity (X
c
) of all sample categories. The endothermic peak was observed at a maximum temperature range of 161–165°C for all composite categories. The addition of chitosan fiber demolished the metastable crystalline phase of the PP matrix that was observed at around 120–130°C. Moreover, the presence of chitosan apparently did not change the Tm of PP composites, even it did reduce the X
c
, indicating that the presence of chitosan in the PP composites became a barrier for nuclei-growth of the PP chain and therefore delayed the crystallization process.
28
On the other hand, a higher endothermic heat flow was observed for that of both modified composite categories as compared to that of the unmodified composites. It was seemed likely that the presence of acrylated lignin-based modifying agent resulted in the increased X
c
of the modified PP/chitosan composites. This finding indicated that the increased crystallinity of the modified composites was substantially attributed to the enhanced interfacial adhesion between the chitosan and the PP matrix. Then, it is worth noting that the hydrophobic segment of lignin might contribute to the nucleation process and the formation of spherulitic structure within the PP composites. The molecular structure and the chemical composition of lignin are expected as the factors affecting the nucleation process. When the fibers containing higher quantities of lignin were used some crystallization occurred.
29
Differential scanning calorimetry curves of polypropylene/chitosan composites with 20 php filler content. Melting temperature (Tm), fusion enthalpy ( Tm: melting temperature; Xc: crystallinity degree; ΔHf: fusion enthalpy; php: part per-hundred of polymer.

In addition, the crystallinity degree of the modified composites with AAL was found to be higher than that of the modified composites with AOSL which was probably due to the increased molecular chain mobility during the crystallization process as the acrylated alkaline lignin was also found to behave as a plasticizing agent in the composite system. Substantially, the different crystallinity degree obtained in the PP/chitosan composites was a consequence of the difference of nucleation process of polymer on substance since it was complex and affected by several factors; crystalline morphology the substrate, the surface topography of filler, the chemical compositions of surface, the wettability and surface energies of the substrates. 30 These factors might explain the lower Young’s modulus of the modified PP composites with AAL than that of the modified PP composites with AAL. Overall, the X c of the composites treated with acrylated lignin averagely decreased by 18.64% as compared with the composites treated with organosolv lignin. Nevertheless, the Tm did not significantly change, presumably due to the non-uniformity of crystal shape and lattice that resulted from different types of nucleation sites. 17
Conclusion
Polypropylene-chitosan composites modified with AOSL and AAL have been prepared. The properties of both modified composites were investigated and briefly discussed. The organosolv lignin obtained had a lower molecular weight than the alkaline lignin which in turn resulted in higher efficiency of chitosan modification up to 21.3% than the modified chitosan with AAL. Both esterified lignin substantially modified the chitosan surface which led to the enhanced interfacial adhesion within the composites. The mechanical strength of the composites modified with AOSL was higher than the modified composites with AAL. Interestingly, the modified composites with AAL demonstrated an improved ductility. SEM images clearly showed the appearance of fewer voids on the tensile surface of the modified composites. TGA and DSC results exhibited the improved thermal stability and crystallinity degree of the modified composites. The crystallinity degree of the modified composites with AAL was higher than the modified composites with AOSL. Overall, the obtained results suggested the advantages in the utilization of lignin-based modifying agents in the manufacturing of natural fiber-reinforced composite materials, thereby opening up the potential of renewable low-cost chemical reagent alternative to synthetic reagents.
Footnotes
Acknowledgments
FA Tanjung is grateful to the Ministry of Education, Culture, Research and Technology of Indonesia for research funding under the World Class Research (WCR) grant scheme (161/E4.1/AK.04.PT/2021; 297/LL1/PG/2021), and the Universitas Medan Area for the associated research facilities.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study is supported by Ministry of Education, Culture, Research and Technology of Indonesia for research (161/E4.1/AK.04.PT/2021, 297/LL1/PG/2021).
