Abstract
In this study, we firstly investigated the surface and protein adsorption properties of montmorillonite (MMT)/chitosan (CS) composite films with various MMT/CS weight ratios for metallic implants coating applications. Bicinchoninic acid (BCA) protein assay results show that the neat CS film exhibits a high concentration of bovine serum albumin (BSA) protein adhesion because the abundant carbonyl and amide functional groups on the surface of the CS film easily form hydrogen bonds with the copious carboxylic acid groups on the surface of the BSA protein. The MMT/CS composite films with MMT/CS = 3, 5, 8, and 10 possess a much lower BSA adhesion concentration than that of the neat CS film, as some of the carbonyl and amide functional groups on the surface of the composite films are replaced by the –Si–O–Si and –Al–O–Al groups. Among these MMT/CS composite films, the film with MMT/CS = 5 exhibits the lowest BSA adsorption concentration because it possesses a higher MMT content than those with MMT/CS = 1 and 3 and a smoother and non-porous surface than those with MMT/CS = 8 and 10. According to our results, MMT/CS composite films with appropriate MMT/CS weight ratios exhibit better surface and protein adsorption properties than neat CS for biomedical applications.
Introduction
Stainless steel, cobalt-chromium alloys, pure titanium, Ti-6Al-4V alloys, and shape memory alloys are widely used as metallic biomaterials. Nevertheless, when these metallic biomaterials are used as implants for surgery prosthetics and orthotics, they pose the risk of releasing metal ions. 1 Therefore, implant material surfaces are usually protected by bioactive coatings, such as hexamethyldisilazane, calcium phosphate, bioactive glass, various proteins, and chitosan.2-6 Chitosan, which is a bioactive polymer, is suitable for biomedical applications because of its non-toxicity, biodegradability, biocompatibility, and antifungal activity.6-8 Moreover, chitosan can also be used to derive or graft desirable bioactive groups because it contains many hydroxy and amino groups. 9 Inorganic additives are usually applied to prepare chitosan composites to improve the physical or chemical properties of chitosan. Montmorillonite (MMT), which is a functionalized layered silicate consisting of hydrous layered aluminum silicate with exchangeable cations and active hydroxyl groups, has been used to prepare MMT/chitosan (CS) composites to enhance the properties of chitosan for diverse applications.
Several studies reported that the mechanical properties of the MMT/CS composites could be significantly increased with an increase in the MMT content. Furthermore, the thermal stability of the MMT/CS composites is enhanced by increasing the MMT load.10-13 Shou et al. 14 developed a novel process to fabricate chitosan composites with the addition of functionalized MMT nanoparticles. They improved the low strength and significant shrinkage upon dehydration of chitosan when chitosan was used as a medical implant or heavy metal absorbent. Lewandowska 15 investigated the properties of composites containing chitosan with poly (N-vinylpyrrolidone) (PVP) and MMT. It was found that the mechanical properties and thermal stability of CS were improved by adding PVP and MMT because of the crosslinking reactions between the polymer chains and molecular interactions between CS, MMT, and PVP.
Wang et al. 16 proposed novel montmorillonite-nanosheet/chitosan (MMTNS/CS) hydrogels, which are suitable adsorbents for sewage treatment, drug carriers, microorganisms, and catalyzers because of their controllable porous structure and large specific surface area. They also reported that a self-assembled gel of Fe-chitosan/montmorillonite nanosheets (Fe-CS/MMTNS) is suitable for the elimination of methylene blue under visible light in the presence of H2O2. 17 Yadav and Ahmad 18 proposed novel MMT/graphene oxide (GO)/CS composite films prepared by a simple solution mixing evaporation method. According to their study, the MMT/GO/CS composite films exhibit better thermal stability and mechanical properties than chitosan films. Lertsutthiwong et al. 19 prepared a biopolymeric chitosan-montmorillonite using a cationic exchange reaction. They reported that chitosan could be used as an enhancer for the improvement of dye adsorption and antifungal properties of montmorillonite. Recently, Kimna et al. 20 developed vancomycin (VC) and gentamicin (GC)-loaded MMT/CS composite nanospheres for sustained antibiotic delivery at post-implantation bone infection. Several studies also reported that MMT/CS composite materials possess drug delivery applications.21,22 Kar et al. 23 and Vyas et al. 24 proposed a novel porous chitosan–organically modified montmorillonite–hydroxyapatite composite scaffold for non-load bearing bone tissue engineering applications. It has been reported that chitosan films are suitable for biomedical coatings.25-27 Although MMT/CS composites are promising for biomaterials, their surface and protein adsorption properties should also be carefully considered when they serve as orthopedic implants or stents coatings. Therefore, this study aimed to investigate the surface and protein adsorption properties of MMT/CS composite films.
Materials and methods
Preparation of MMT/CS composite films
The CS powders used in this study were prepared from crab shells and were purchased from C&B Co. Ltd., Taiwan. The CS powders had a molecular weight in the range of 10–50 kDa and a 90% degree of deacetylation. The MMT powders (Cloisite®25A) used in this study were purchased from WWRC Co. Ltd., Taiwan. CS (1.0 g) and MMT powders were blended to prepare MMT/CS powder mixtures with various weight ratios (MMT/CS = 0, 0.1, 0.3, 0.5, 0.8, and 1 g/g). Then, a 100 mL acetic acid solution (2 wt.%.) was added into each MMT/CS powder mixture and stirred to form an MMT/CS gel. The MMT/CS gel was stirred at room temperature for 2 h. Then, each MMT/CS gel was poured into a stainless steel tray and dried in an oven at 45°C for 15 h to form an MMT/CS composite film. The MMT/CS composite films were immersed in a 0.5 N NaOH solution for 1 h to neutralize excess acetic acid, and they were washed repeatedly with deionized water until the pH value of the solution approached 7. Each MMT/CS composite film was finally dried in ambient air at room temperature to obtain a dense composite film for the following characterization tests. The prepared MMT/CS composite films with MMT/CS weight ratios of 0, 0.1, 0.3, 0.5, 0.8, and 1 g/g are designated as CS, M1, M3, M5, M8, and M10, respectively, in the following context.
Characterizations
The functional groups of the MMT/CS composite films were detected using an attenuated total reflectance Fourier-transform infrared (ATR-FTIR) spectrometer (Spectrum 100, PerkinElmer). Each specimen was measured in the range of 4,000–400 cm−1 and 16 scans at a resolution of 4 cm−1. The surface morphology observations and chemical composition analysis of the MMT/CS composite films were performed using a scanning electron microscope (SEM) (5136 MM, Tescan instruments) equipped with an energy-dispersive X-ray spectroscope (EDS) (X-Act, Oxford). Prior to conducting the morphology observation, each composite film was coated with a thin gold film using a vacuum sputter coater. The surface chemical compositions of the MMT/CS composite films were determined using X-ray photoelectron spectroscopy (XPS) (K-Alpha, Thermo Scientific) with a monochromatic Al Kα radiation source of 1468.6 eV. The survey spectrum of each specimen was determined in the range of 0 to 700 eV in 1 eV steps. The high-resolution Si
The anticoagulant properties of the MMT/CS composite films were assessed by determining the amount of protein adsorbed on the film surface using a bicinchoninic acid (BCA) protein assay. The protein used in the BCA assay was bovine serum albumin (BSA). Both BSA and BCA were purchased from Bio Basic Inc. The phosphate buffer solution (PBS) and sodium dodecyl sulfate (SDS) used in the protein adsorption analyses were purchased from UniRegion and Sigma-Aldrich, respectively. The optical density (OD) values derived from the protein adsorption analysis were measured at 562 nm in a spectrophotometer (GENESYS 20, Thermo Scientific, USA), using a PBS solution as a blank cuvette.
The toxicity of the MMT/CS composite films was initially evaluated by observing the growth curve of
Results and discussion
ATR-FTIR analysis
Figure 1 shows the ATR-FTIR spectra of the neat CS and MMT/CS composite films with various MMT/CS weight ratios. As shown in Figure 1, the neat CS and MMT/CS composite films exhibit a broad characteristic peak at approximately 3000–3500 cm−1, which is attributed to the stretching vibrations of –NH2 and –OH groups. The characteristic absorption bands at approximately 1650 cm−1, 1563 cm−1, and 1411 cm−1 are attributed to the –C=O carbonyl stretching of amide I, the –NH bending of amide II, and the –CN stretching of amide III, respectively. Figure 1 also shows that –CH absorption bands appear at approximately 2852 and 2924 cm−1. The characteristic absorption bands at approximately 916 and 1011 cm−1 are assigned to the –Si–O–Si and –Al–O–Al groups, respectively, because of the presence of MMT. The signals of the –Si–O–Si and –Al–O–Al groups become more significant with an increase in the MMT content in the MMT/CS composite films.
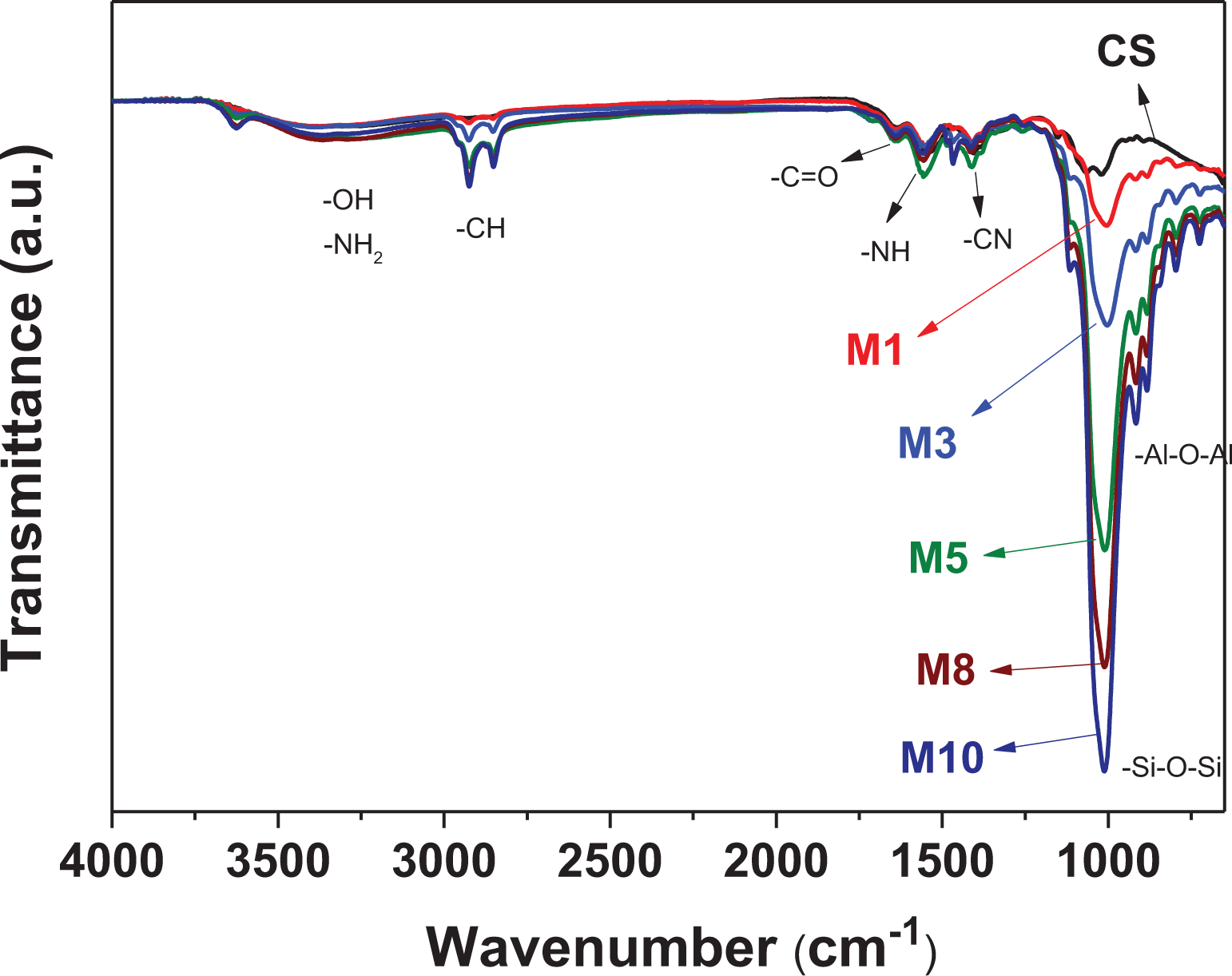
ATR-FTIR spectra of neat CS and MMT/CS composite films with various MMT/CS weight ratios.
SEM and EDS measurements
Figures 2(a) to 2(f) present the surface morphology images (1000×) of neat CS and MMT/CS composite films with various MMT/CS weight ratios. Figure 2(a) shows that the surface morphology of the neat CS film exhibits a smooth and homogeneous surface morphology. Figures 2(b)–2(d) show that the surface morphologies of M1, M3, and M5 MMT/CS composite films are slightly rougher than that of the neat CS film. Figures 2(e) and 2(f) show that the M8 and M10 MMT/CS composite films both exhibit a porous surface morphology because of their excessive MMT additive. Figures 3(a)–3(f) show the EDS mapping analysis of silicon for neat CS, M1, M3, M5, M8, and M10 MMT/CS composite films, respectively. In Figure 3(a), no silicon atoms can be detected on the surface of the neat CS film. Figures 3(b)–3(f) show that the amount of silicon atoms on the surface of the MMT/CS composite films gradually increases with an increase in the MMT content. Moreover, the silicon atoms were homogeneously distributed on the surface of the MMT/CS composite films, indicating that the blended MMT is well dispersed in the MMT/CS composite films.

SEM micrographs of the surface morphologies (1000×) for (a) neat CS, (b) M1, (c) M3, (d) M5, (e) M8, and (f) M10 MMT/CS composite films.

EDS silicon map results of (a) neat CS, (b) M1, (c) M3, (d) M5, (e) M8, and (f) M10 MMT/CS composite films.
XPS measurements
Figures 4(a) and 4(b) show the XPS survey spectra of the neat CS film and pure MMT, respectively. Figure 4(a) reveals that the XPS spectrum of the neat CS film exhibits significant characteristic peaks of C (C

XPS survey spectra for (a) neat CS, (b) pure MMT, (c) M1, (d) M3, (e) M5, (f) M8, and (g) M10 MMT/CS composite films. The inset shows the high-resolution Si
Figures 4(c)–4(g) show the XPS survey spectra of the M1, M3, M5, M8, and M10 MMT/CS composite films, respectively. Figure 4(c) shows that the XPS survey spectra of the M1 MMT/CS composite film are similar to those of the neat CS film, suggesting that only a small amount of blended MMT could be obtained on the surface of the M1 film. The inset in Figure 4(c) shows the high-resolution Si
Protein adsorption properties
The protein adsorption properties of neat CS and MMT/CS composite films were determined by performing a BCA protein assay. Each film specimen was rinsed three times with PBS, immersed in 5 mL of a BSA solution at 37°C for 24 h and then rinsed again with PBS. Then, the rinsed films were immersed in 2 mL of an SDS solution for 24 h. A 0.1 mL sample of each solution was mixed with 1 mL of the BCA solution in a cuvette to determine its OD value using a spectrophotometer. The concentrations of BSA that adhered to the films were calculated according to the determined OD values using a BSA concentration standard curve. Figure 5 presents the determined BSA adhesion concentration values of the MMT/CS composite films with various MMT/CS weight ratios. As shown in Figure 5, the concentration of adhered BSA on the surface of neat CS was determined to be 231.5 ± 2.4 μg/L. The concentrations of adhered BSA on the surfaces of the M1, M3, M5, M8, and M10 MMT/CS composite films were determined to be 226.2 ± 5.5, 107.6 ± 5.3, 76.4 ± 3.9, 82.5 ± 5.3, and 115.1 ± 5.5 μg/L, respectively. According to Figure 5, the amount of BSA protein that adhered to the surface of the M1 MMT/CS composite film was only slightly lower than that of the neat CS film. Nevertheless, the amounts of BSA protein that adhered to the surface of M3, M5, and M8 MMT/CS composite films were much lower than that of the neat CS film, suggesting that adding sufficient amounts of MMT into the CS film effectively improves the anticoagulant properties of the CS film.

BSA adsorption concentrations of neat CS and MMT/CS composite films with various MMT/CS weight ratios.
The neat CS film exhibits a high concentration of BSA protein adsorption because the abundant carbonyl and amide functional groups on the surface of the CS film easily form hydrogen bonds with the carboxylic acid of the BSA protein. The BSA protein that adhered to the surface of M3, M5, M8, and M10 MMT/CS composite films were much lower than that of the neat CS film. This is because numbers of the carbonyl and amide functional groups on the surface of the CS film decreased because of the presence of the abundant –Si–O–Si and –Al–O–Al groups. This indicates that MMT/CS composite films with sufficient MMT content should exhibit an enhanced anticoagulant property when used as an
Toxicity assessment
Figure 6 shows a plot of the lnOD600 values of
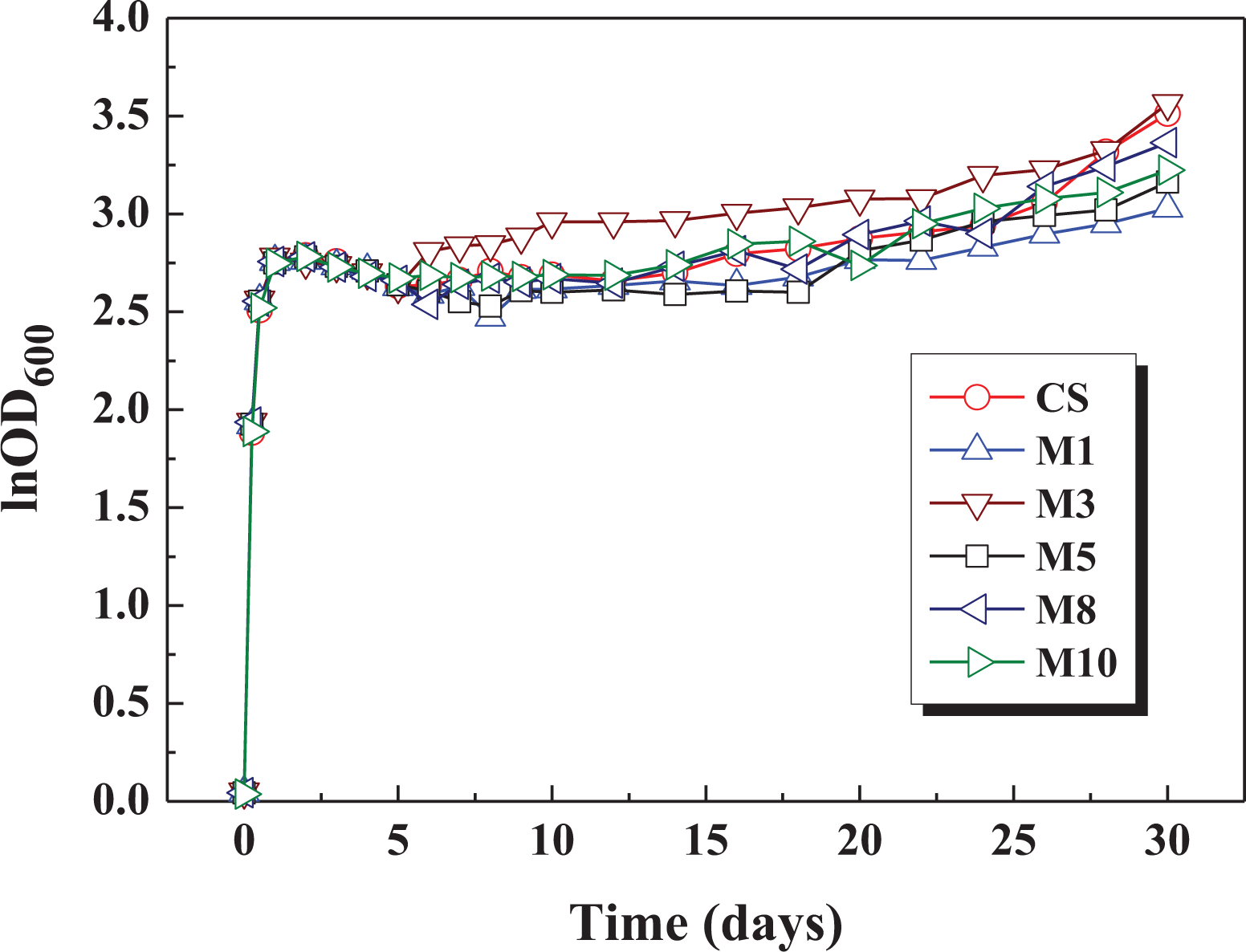
The lnOD600 values of
Conclusions
In this study, we demonstrated that MMT/CS composite films exhibit superior surface and protein adsorption properties when MMT and CS are mixed in the appropriate weight ratios. FTIR spectra reveal that the characteristic absorption bands of –Si–O–Si and –Al–O–Al groups on the surface of the MMT/CS composite films become more significant when the MMT/CS weight ratio of the composite films increases. SEM images show that the surface of the MMT/CS composite films is smooth when the MMT/CS weight ratio is below 5; however, it becomes rougher when the MMT/CS weight ratio is above 8. XPS spectra show that the characteristic peaks of MMT can be obtained on the surface of MMT/CS composite films when the MMT/CS weight ratio is above 3. BCA protein assay results show that MMT/CS composite films with sufficient MMT content exhibit better protein adhesion properties than neat chitosan films because the numbers of carbonyl and amide functional groups on the surface of CS reduce after blending with MMT. Preliminary toxicity assessments show that MMT/CS composite films do not exhibit significant toxicity to
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The authors gratefully acknowledge the financial support provided by the Ministry of Science and Technology (MOST), Taiwan, under Grant No. MOST 108-2221-E-197-017.
