Abstract
The effect of sodium dodecyl sulfate (SDS) on mechanical and thermal properties of chitosan-filled polypropylene (PP) composites was investigated. Mechanical and thermal properties of PP/chitosan composites were analyzed according to ASTM D 638-91, thermogravimetry analysis and differential scanning calorimetry, respectively. Results showed that the treated PP/chitosan composites had higher tensile strength and Young’s modulus when compared with untreated PP/chitosan composites, but lower in elongation at break in the same filler loading. Thermal analysis results showed that the thermal stability and degree of crystallinity of treated PP/chitosan composites were higher than untreated ones. Scanning electron microscopy and Fourier transform infrared studies revealed less detachment of filler from matrix on the tensile surface of treated PP/chitosan composites as an evidence of enhanced interfacial adhesion between filler and matrix due to the formation of covalent bonding between chitosan and SDS.
Introduction
Research on the use of materials from renewable sources as filler or reinforcing agent in plastic composites is growing. Numerous recent studies have revived the idea of exploiting natural fibers as an alternative to inorganic counterparts (i.e., glass fibers) for solving numerous problems in environmental and biomaterial engineering. 1 Chitosan is known as the second most abundant natural fiber after cellulose. It is extracted from crustaceous shells such as crabs, shrimps and prawns. 2 Currently, active researches have been conducted on the application of chitosan as biomaterials due to its biodegradability, biocompatibility, nontoxicity, 3 and so on, in spite of low cost, resource abundances, lightness and nonabrasive nature. 4
Plastics such as polypropylene (PP), polystryrene, polyethylene, and so on, largely dominated many materials for industrial application. 5 The use of these materials generates environmental problems after their service life when land-filled or incinerated. 6 This problem could be alleviated by reducing their consumption through blending with natural fibers in order to produce composite materials. 7 Plastic composites prepared using natural reinforcing fillers are inexpensive and could play a vital role in solving the environmental problems that we would otherwise have to face in future. 8
In this study, PP was used as the matrix and chitosan as natural filler in manufacturing the plastic composites. PP is used due to its excellent mechanical and thermal properties. 9–11 A large number of studies have reported the use of PP in natural fiber-reinforced composites 12–14 ; however, study of composite materials filled with chitosan was less reported. From our knowledge, chitosan is a polysaccharide that possesses the hydroxyl (–OH) and amine (–NH2 ) groups along the chain. 15 The existence of these groups contributes the hydrophilic character of chitosan, which decreases the interfacial adhesion with hydrophobic matrix leading to reduced mechanical properties. Therefore, several chemical modification methods have been developed in order to hydrophobize the chitosan and enhance the interfacial adhesion. 16,17
The interest in the use of chemical modification method is mainly due to the appearance of polar groups of chitosan, which contribute a substitution reaction with modifying agent. 18 Sodium dodecyl sulfate (SDS) is known as anionic surfactant and widely used in the synthesis of biomaterials. 19,20 The presence of SDS on chitosan chain is expected to decrease the hydrophilic character as a result of the formation of chemical interaction between polar head groups of SDS and polar groups of chitosan. 21 The current study was devoted to investigate the effect of SDS on mechanical and thermal properties of PP/chitosan composites.
Experimental
Materials
PP homopolymer used in this study was of injection molding grade S11232 G112 from Polypropylenas Sdn. Bhd. (Malaysia) with a Melt flow index (MFI) value of 45 g/10 min at 230°C. Chitosan (Hunza Nutriceuticals Sdn. Bhd., Malaysia) with average size of 80 µm and degree of deacetylation of 90% was dried in an oven for 24 hours at 80°C to eliminate the moisture content. Table 1 shows the properties of chitosan. Ethanol (98% v/v) and SDS in powder form were obtained from Aldrich (Penang, Malaysia).
Physical and chemical properties of chitosan.

Chemical modification of chitosan
SDS solution was prepared by dissolving SDS powder in ethanol at a temperature of 40°C. The amount of SDS used was 3% by weight of filler. The solution was then cooled down to room temperature. Afterward, chitosan was gradually added to the solution while slowly stirring. The mixture was mechanically stirred for 2 hours in order to be homogenized and left overnight. The next day, the precipitate was filtered and then dried in the oven at 80°C for 24 hours to remove ethanol residue.
Mixing procedure
PP/chitosan composites were prepared in a Z-blade Mixer (MCN ELEC Co., Taiwan) at 190°C and at a rotor speed of 50 rpm. PP was first charged to start the melt mixing. After 12 min, the chitosan was added. Mixing was continued for another 3 min. At the end of 15 min, the PP/chitosan composites were taken out and sheeted through a laboratory mill at a 2.0-mm nip setting. The sample of PP/chitosan composites was compression molded in an electrically heated hydraulic press. Hot-press procedures involved preheating at 190°C for 9 min followed by compressing for 3 min at the same temperature and subsequent cooling under pressure for 3 min. Table 2 shows the formulation of untreated and treated PP/chitosan composites at different filler loadings.
Formulation of untreated and treated PP/chitosan composites.

PP: polypropylene; php: part per hundred of polymer.
Tensile testing
Tensile tests were carried out according to ASTM D 638-91 on an Instron 5582. Five dumbbell specimens of each composition with a thickness of 1 mm were cut from the molded sheets with a Wallace die cutter (Lab Tech Eng. Co.). A crosshead speed of 50 mm min−1 was used and the test was performed at 25 ± 3°C.
Thermogravimetric analysis
Thermogravimetric analysis (TGA) and derivative thermogravimetric (DTG) analysis were investigated by TGA Q500 Perkin Elmer Instrument (Japan). Temperature is scanned from 30 to 600°C at a heating rate of 20°C min−1 using nitrogen flow of 50 ml min−1.
Differential scanning calorimetry
Differential scanning calorimetric (DSC) analysis was carried out using a Perkin Elmer TA Instrument DSC Q 1000. Samples were scanned from 20 to 250°C at a heating rate of 20°C min−1 using nitrogen flow of 50 ml min−1. The melting and fusion enthalpy of PP/chitosan composites were automatically calculated by the instrument software. The crystallinity of composites (X c) was determined using the Equation (1)

where ΔH
f is the heat of fusion of PP and PP/chitosan composites,
Morphology study
Study on the morphology of tensile fractured surface of PP/chitosan composites was carried out using a scanning electron microscope (SEM), model JSM 6260 LE JEOL (Japan). The fracture ends of specimens were mounted on aluminum stubs and sputter coated with a thin layer of palladium to avoid electrostatic charging during examination.
FTIR spectroscopy
Fourier transform infrared (FTIR) spectroscopy analysis of untreated and treated chitosan was carried out in ATR mode (Perkin-Elmer 1600 Series) scanned from 650 to 4000 cm−1, with a resolution of 4 cm−1.
Results and discussion
Figure 1 shows the effect of filler loading on the tensile strength of untreated and treated PP/chitosan composites. The tensile strength of untreated and treated PP/chitosan composites decreases with the increase of filler loading due to the lack of interaction between chitosan and PP matrix on the interface resulted from different polarities between hydrophilic chitosan and hydrophobic PP matrix. It has been known that chitosan has hydrophilic character due to the existence of –OH and –NH2 groups along the chain. 22 As the filler loading increases, thereby increasing the interfacial area, the worsening interfacial bonding between chitosan filler and PP matrix decreases the tensile strength. However, the tensile strength of treated PP/chitosan is higher when compared to untreated PP/chitosan composites. This result indicates the anchoring of PP chains and the presence of SDS in the interlayer spaces of chitosan. According to this anchoring, a better stress transfer from the matrix to the filler improves, leading to an increase in the tensile strength. The presence of SDS on the surface of chitosan reduces significantly the hydrophilicity. This might be due to the formation of intermolecular bonding between polar group of chitosan and polar group of SDS through the covalent bond.
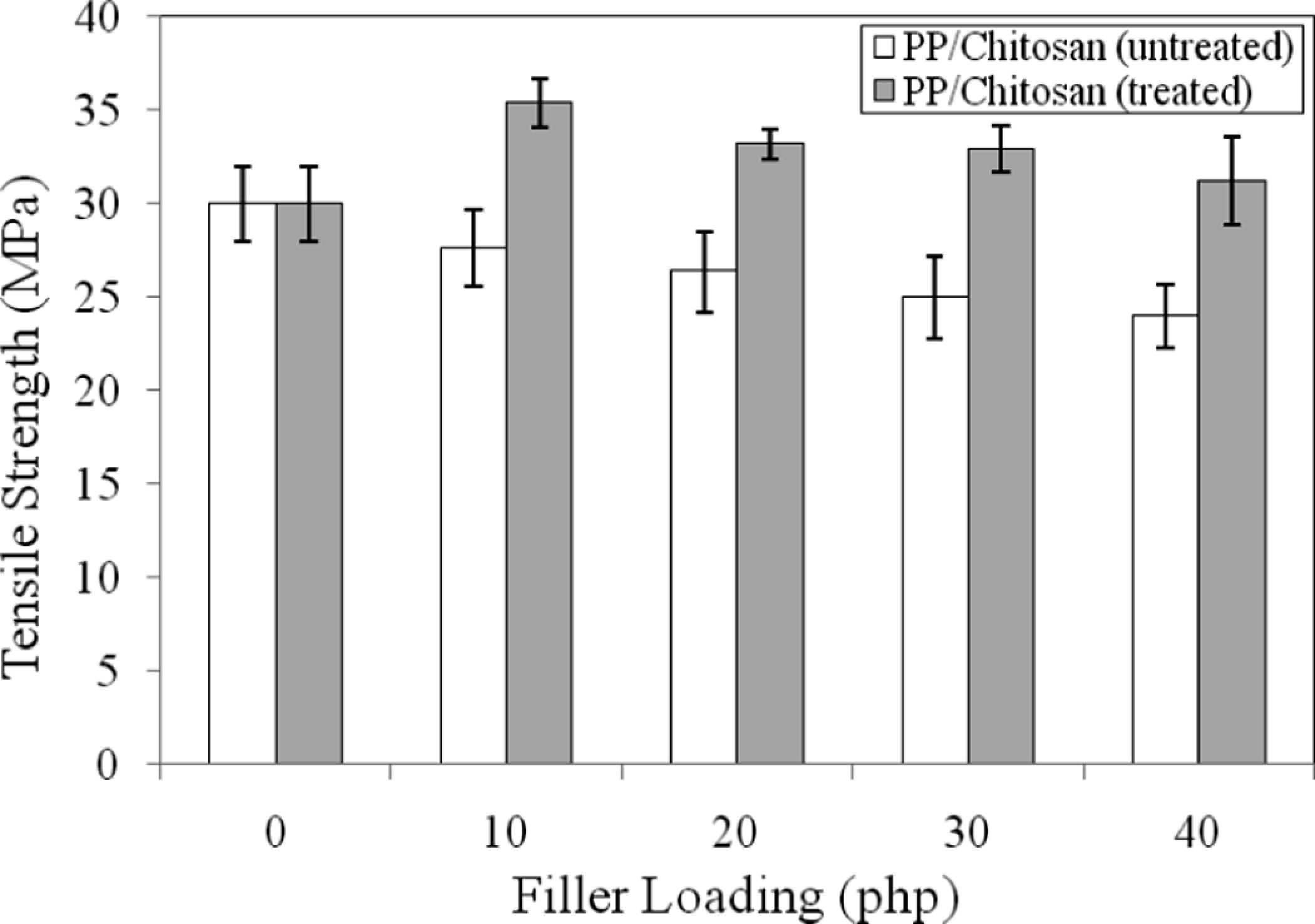
Effect of filler loading on tensile strength of untreated and treated PP/chitosan composites. PP: polypropylene.
Figure 2 shows the effect of filler loading on elongation at break of untreated and treated PP/chitosan composites. The increase in filler loading decreases significantly the elongation at break of untreated and treated PP/chitosan composites due to the formation of rigid interface between chitosan and PP matrix that hinder the mobility of PP molecular chain leading to the loss of flexibility of PP matrix. At a similar filler loading, the treated PP/chitosan composites show lower elongation at break compared with the untreated PP/chitosan composites. This is due to the presence of hydrophobic interaction between alkyl side chains of treated chitosan with SDS and alkyl chain of PP matrix at the interface, leading to decreased ductility of the composites.
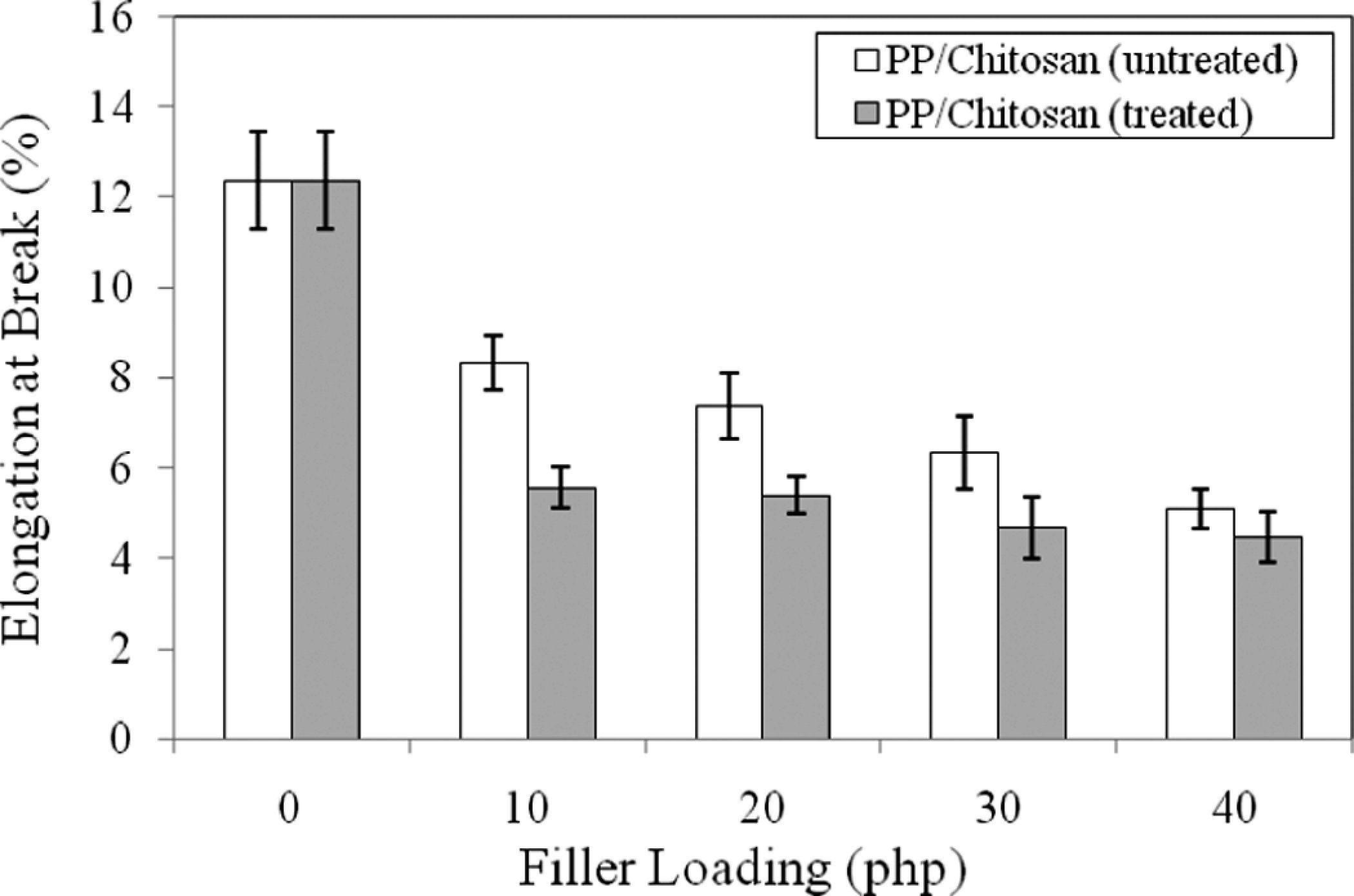
Effect of filler loading on elongation at break of untreated and treated PP/chitosan composites. PP: polypropylene.
On the contrary, the increase in filler loading increases the Young’s modulus of untreated and treated PP/chitosan composites as shown in Figure 3. Young’s modulus indicates the stiffness of composite materials and depends on the filler loading. 23 The stiffness of PP/chitosan composites is apparently stronger at a higher filler loading. Furthermore, the treated PP/chitosan composites exhibit higher Young’s modulus than untreated PP/chitosan composites. This observation reveals that the increasing rigidity of treated PP/chitosan composites with SDS is due to better interfacial adhesion in the composite systems.
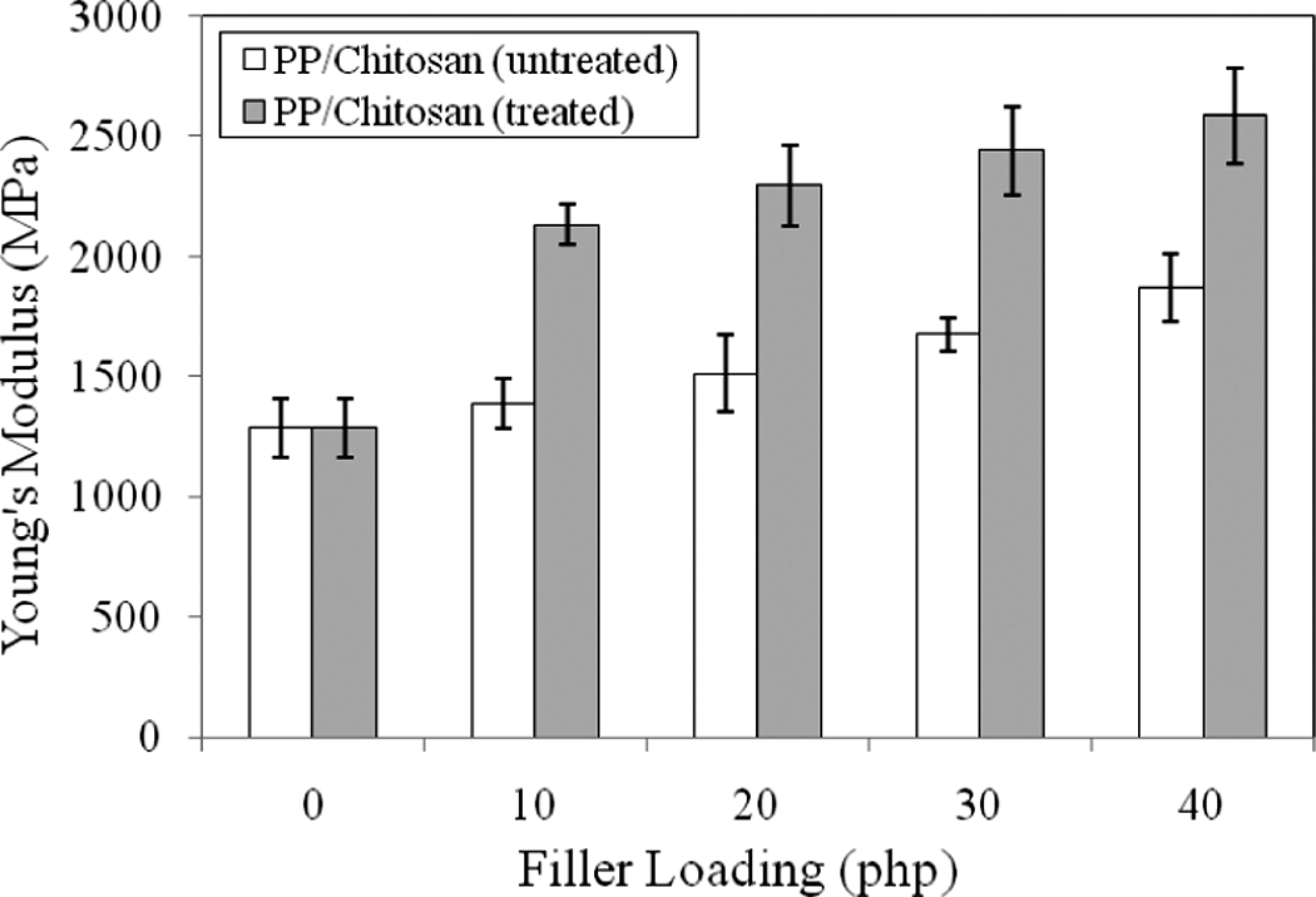
Effect of filler loading on Young’s modulus of untreated and treated PP/chitosan composites. PP: polypropylene.
Figures 4 –7 show SEM micrographs of tensile fractured surface of untreated and treated PP/chitosan composites. The micrographs of untreated PP/chitosan composites (Figures 4 and 5) show the appearance of voids that indicate the pulled-out filler from the matrix. This is mainly due to poor wetting of the filler by the matrix. The fracture occurred at the interface between chitosan and PP matrix. The different polarities between filler and matrix lead to lack of interaction on the interface, especially at a higher filler loading. 24 However, the less voids are observed on the tensile surface of treated PP/chitosan composites (Figures 6 and 7). A good wetting of the filler by the matrix is related to the presence of SDS, which hydrophobically modifies the chitosan, leading to enhanced interfacial adhesion with PP matrix.

SEM micrograph of tensile fractured surface of untreated PP/chitosan composite (20 php) at magnification ×200. PP: polypropylene; SEM: scanning electron microscopy; php: part per hundred of polymer.

SEM micrograph of tensile fractured surface of untreated PP/chitosan composite (40 php) at magnification ×200. PP: polypropylene; SEM: scanning electron microscopy; php: part per hundred of polymer.

SEM micrograph of tensile fractured surface of treated PP/chitosan composite (20 php) at magnification ×200. PP: polypropylene; SEM: scanning electron microscopy; php: part per hundred of polymer.

SEM micrograph of tensile fractured surface of treated PP/chitosan composite (40 php) at magnification ×200. PP: polypropylene; SEM: scanning electron microscopy; php: part per hundred of polymer.
Figure 8 exhibits TGA curve of untreated and treated PP/chitosan composites. The weight loss of untreated and treated PP/chitosan composites increases along the rising temperature. Table 3 summarizes the percentage of weight loss of untreated and treated PP/chitosan composites. It can be seen that PP degrades in one-stage, confirming that PP is composed of carbon–carbon bonds in the main chain, thereby allowing a temperature increase to promote random scission with associated thermal degradation and thermal depolymerization occurring at the weak sites of the PP main chain. 25 The incorporation of chitosan increases the thermal stability of PP composites indicated by the increasing of degradation temperature; however, at the range of 100–250°C, a slight increment in weight loss was observed in comparison to neat PP. This discrepancy is related to the presence of moisture content of chitosan obstructing the PP molecular chain, hence increasing the weight loss. The chitosan contains a significant level of water, which is released when the composites are produced. On the other hand, the treated PP/chitosan composites were observed to have lower total weight loss than untreated PP/chitosan composites, indicating better thermal stability. This result implies decreased interlayer spaces between the chitosan filler and the PP matrix due to the presence of SDS. The decreased interlayer spaces considerably enable in decreasing the rate of heat transfer, leading to suppress the weight loss and improve the thermal stability of composites. 26
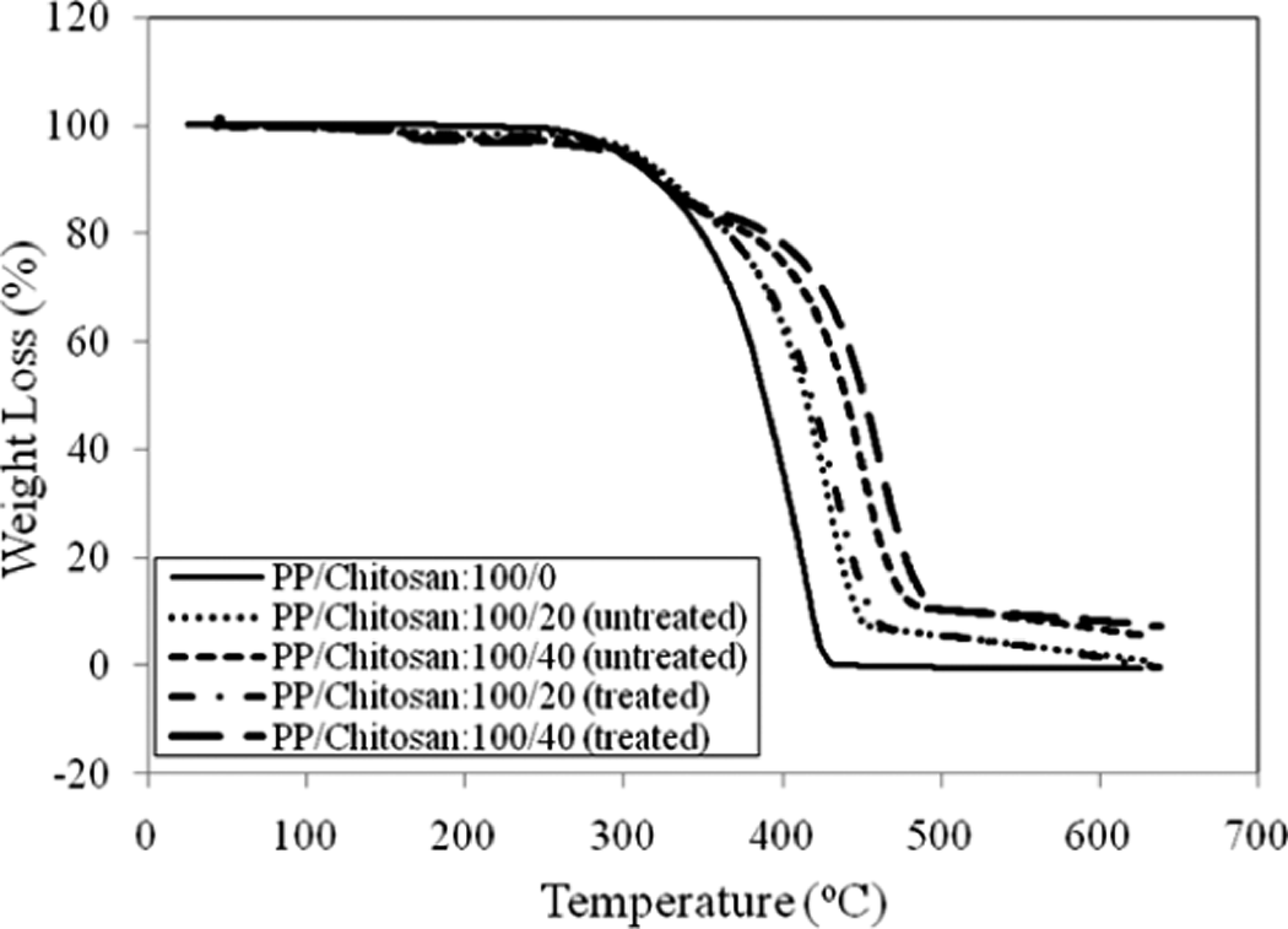
TGA curves of untreated and treated PP/chitosan composites at different temperatures. PP: polypropylene; TGA: thermogravimetic analysis.
Percentage of weight loss of untreated and treated PP/chitosan composites at different temperatures and filler loadings.
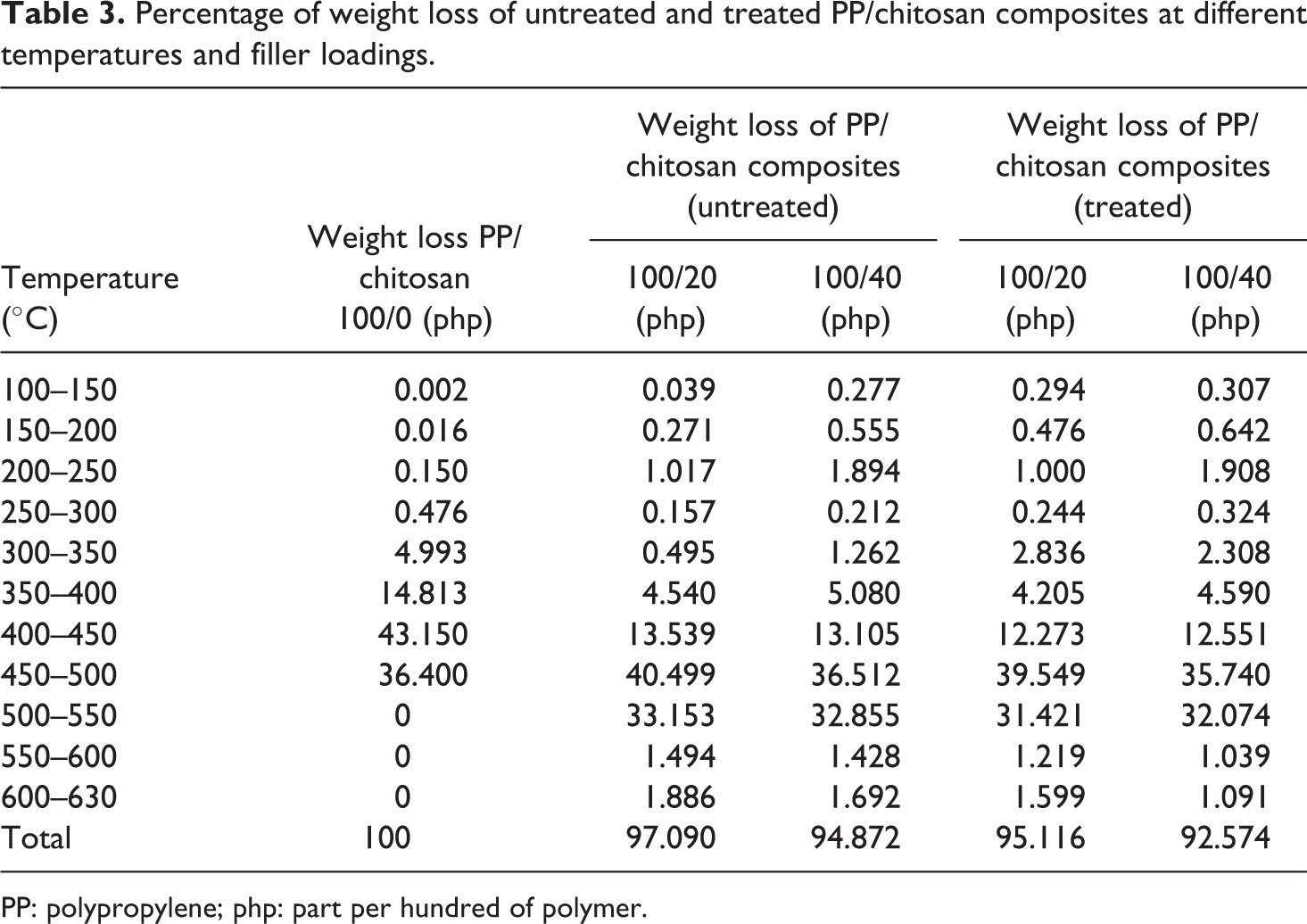
PP: polypropylene; php: part per hundred of polymer.
Figure 9 shows DTG curve of untreated and treated PP/chitosan composites. Table 4 summarizes the temperature peaks of untreated and treated PP/chitosan composites. It is clearly observed that PP decomposes in one-stage process by reaching the maxima at 415°C. On the other hand, untreated and treated PP/chitosan composites degrade in three-stage process. This points out that the chemical modification does not alter the degradation mechanism. The treated PP/chitosan composites show higher values at each peaks compared with the untreated ones. This is apparently due to the better interaction between the PP matrix and the hydrophobically modified chitosan in the interface, which maintains the structural orientation during heating process and decreases the weight loss.
Temperature peaks of untreated and treated PP/chitosan composites.
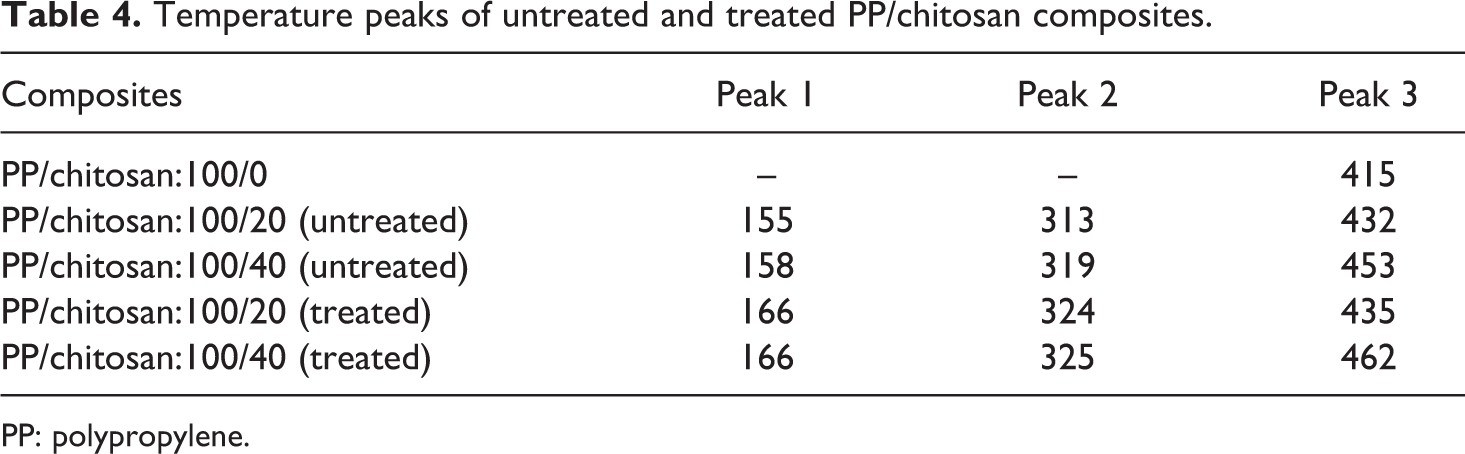
PP: polypropylene.
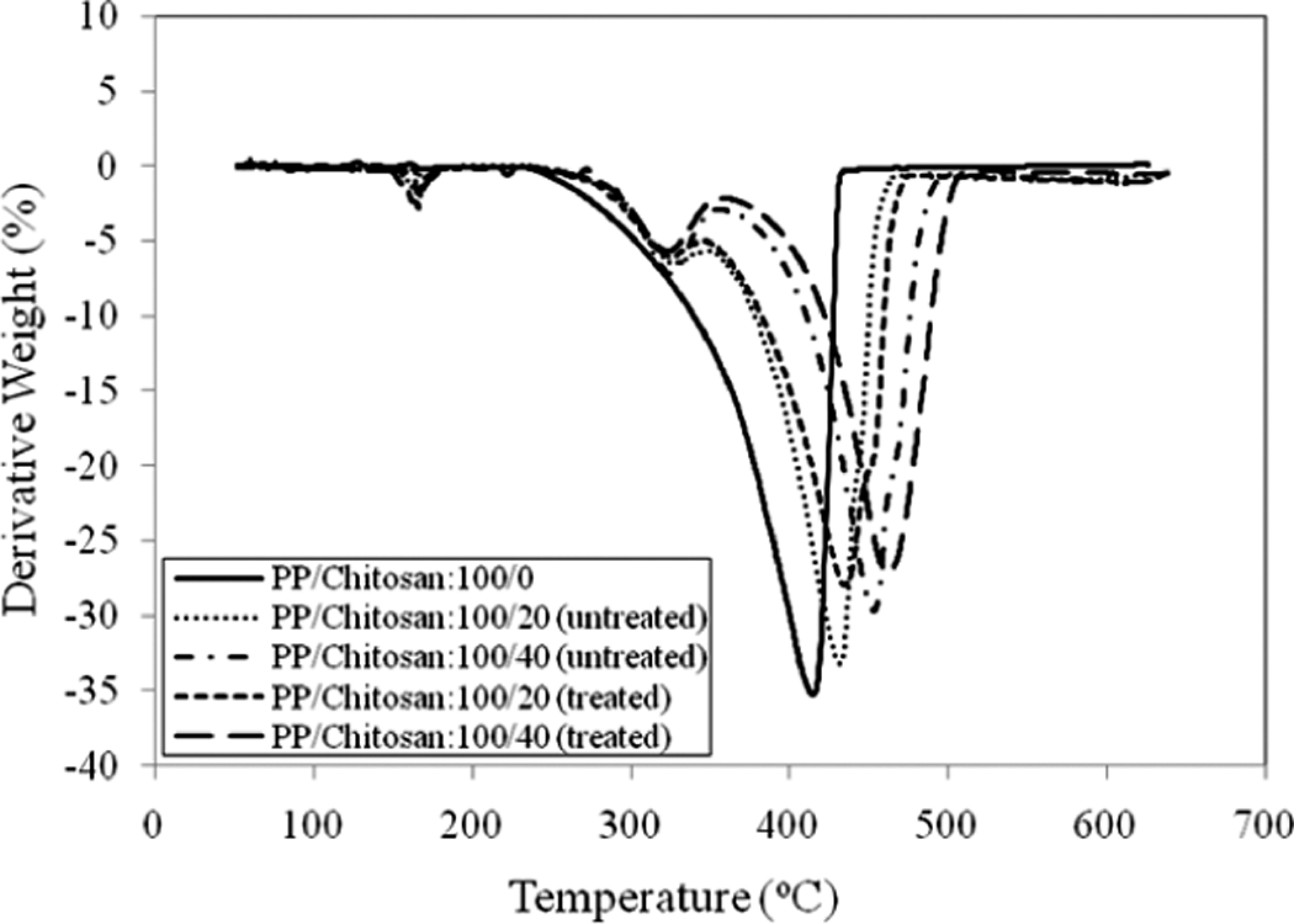
DTG curves of untreated and treated PP/chitosan composites. PP: polypropylene; DTG: derivative thermogravimetric analysis.
Figure 10 shows a representation of DSC thermograms of untreated and treated PP/chitosan composites. Table 5 lists the summary data of melting temperature (T m), fusion enthalpy (ΔH f ) and degree of crystallinity (X c ) of untreated and treated PP/chitosan composites. As can be seen, the incorporation of chitosan into PP matrix does not significantly change the T m. Moreover, it reduces the crystallinity of the PP composites. This phenomenon confirms the inability of chitosan to act as nucleating agent in PP matrix. The presence of chitosan blocks the nuclei—growth of the PP matrix during the crystallization process. However, the presence of SDS imparts a significant effect on the X c , where the treated PP/chitosan composites have higher crystallinity than untreated PP/chitosan composites. This result indicates that the treated chitosan with SDS could be the nucleating agent in PP matrix, which induces the nuclei growth and crystallization process. Otherwise, the T m does not significantly change when compared with untreated PP/chitosan composites.
Summary data of T m , ΔH f and X c of untreated and treated PP/chitosan composites.
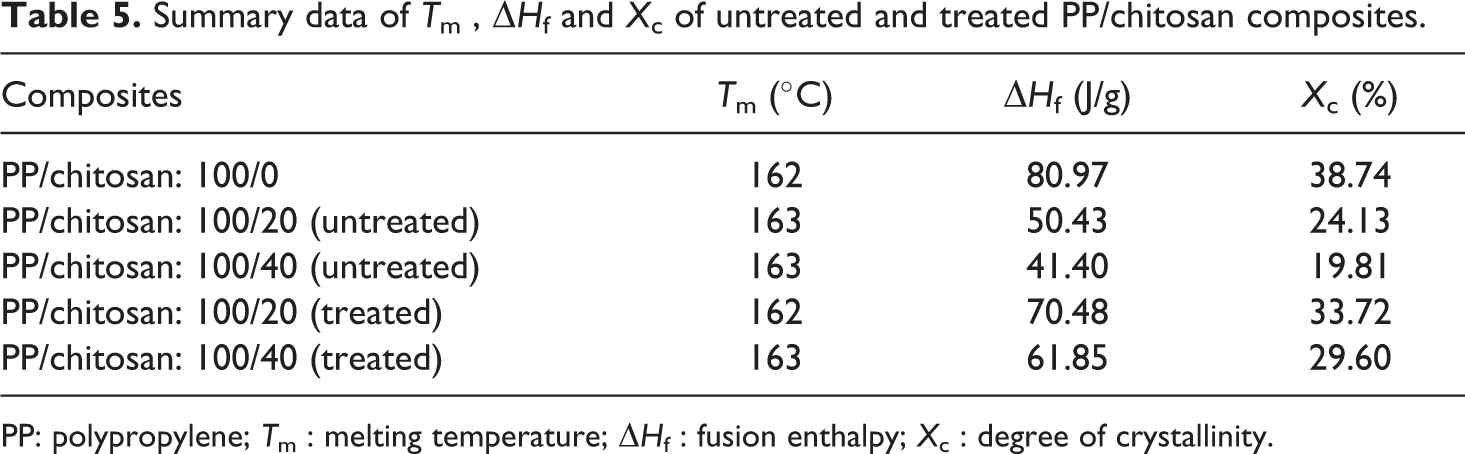
PP: polypropylene; T m : melting temperature; ΔH f : fusion enthalpy; X c : degree of crystallinity.
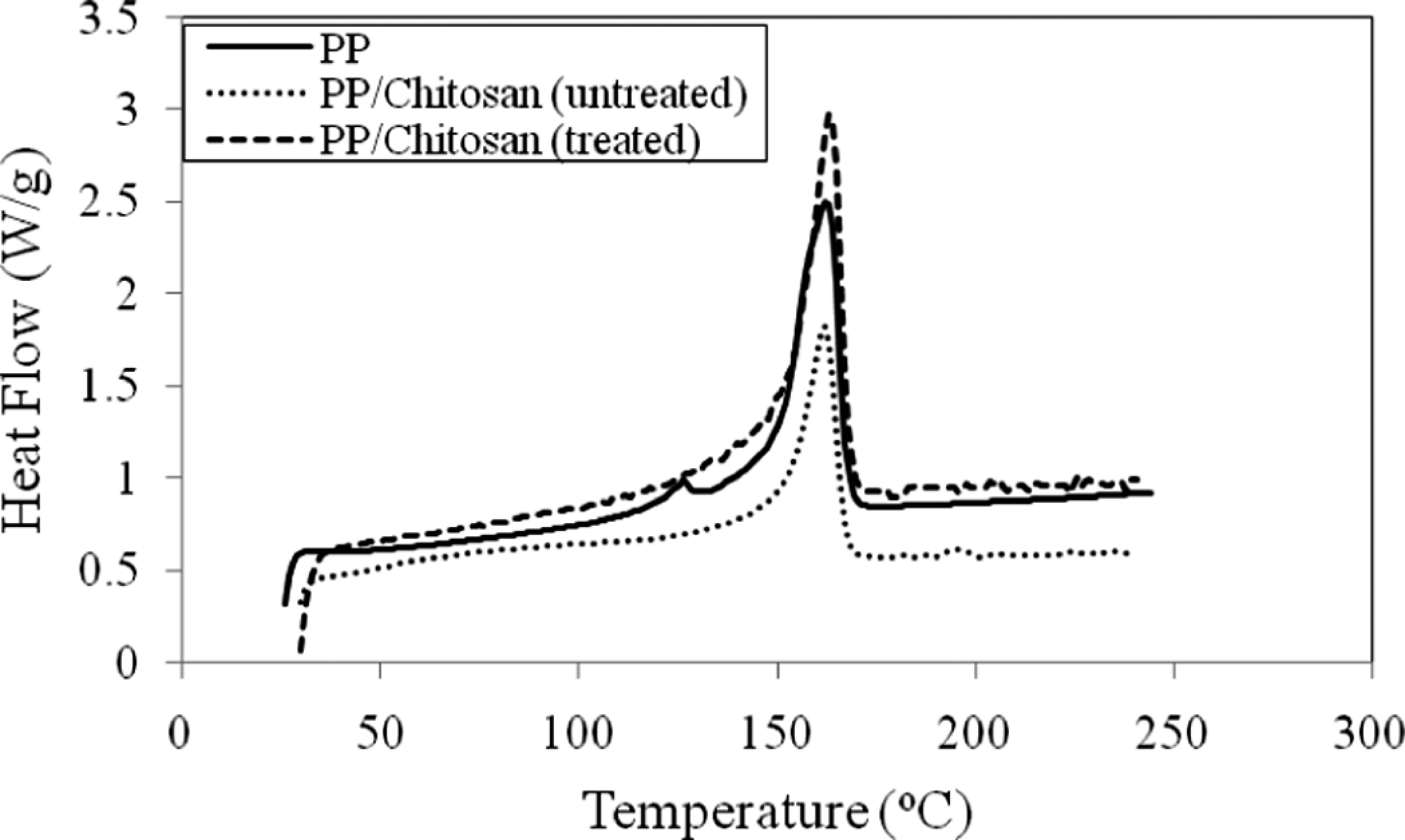
DSC curves of untreated and treated PP/chitosan composites (20 php). PP: polypropylene; DSC: differential scanning calorimetry; php: part per hundred of polymer.
Figure 11 illustrates the FTIR spectra of untreated and treated chitosan. As can be seen, the infrared (IR) spectra of untreated chitosan, the main characteristic peaks of chitosan, are at 3358 cm−1 (O–H stretch), 2872 cm−1 (C–H stretch), 1675 cm−1 (N–H bend) and 1590 cm−1 (C=O stretch). The IR spectrum of treated chitosan exhibits a significant absorption on the band of –OH group from 3358 to 3251 cm−1, indicating the reduction in hydrophilic character of chitosan. This is attributed to the presence of covalent bonding between polar head group of SDS and –OH group of chitosan as identified by the presence of the absorption band at 1722 cm−1. The formation of this bonding is also supported by the reduction of bands at 1675–1645 cm−1 and 1590–1565 cm−1, indicating N–H bending vibration and amide I (C=O) stretching vibration of chitosan chain. The schematic reaction between chitosan and SDS is shown in Figure 12.
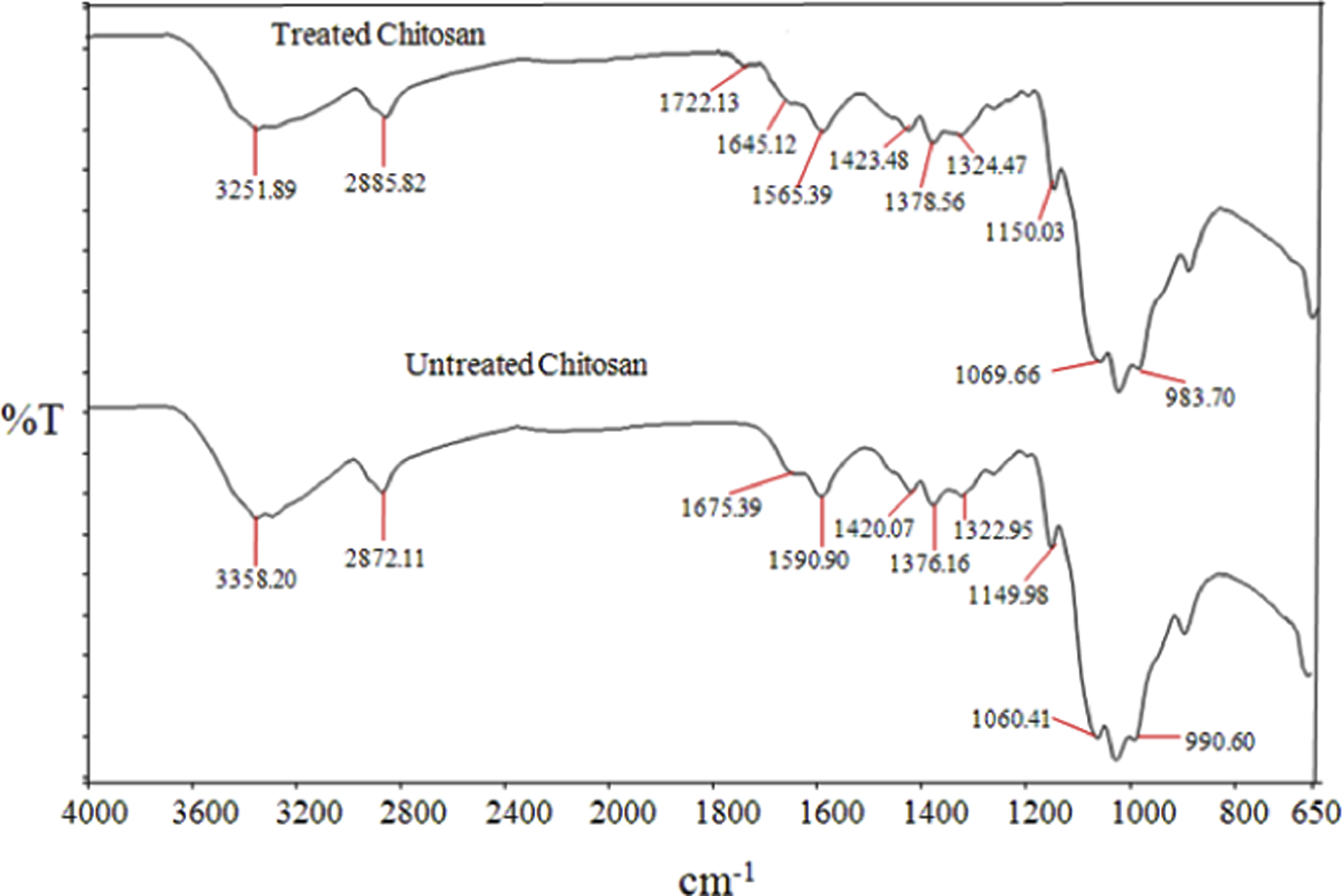
FTIR spectra of untreated and treated chitosan with SDS. FTIR: Fourier transform infrared; SDS: sodium dodecyl sulfate
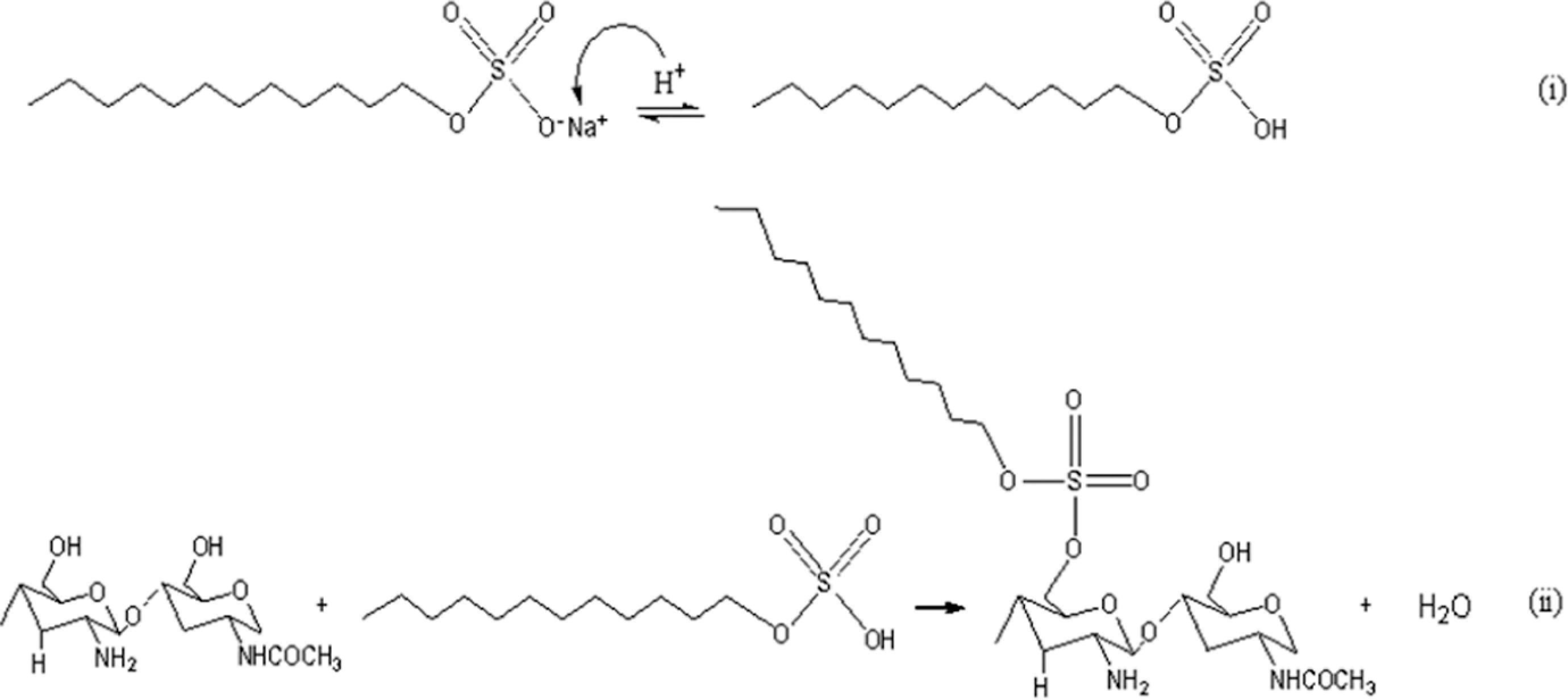
The proposed schematic reaction between chitosan and SDS. SDS: sodium dodecyl sulfate.
Conclusions
The effect of SDS on mechanical and thermal properties of PP/chitosan composites has been studied. The results of tensile properties showed that the incorporation of chitosan filler into PP matrix reduced the tensile strength and elongation at break but increased the Young’s modulus. The treated PP/chitosan composites were observed to have higher tensile strength and Young’s modulus but lower elongation at break than untreated composites. The thermal analysis results showed that the treated PP/chitosan composites had higher thermal stability and crystallinity. FTIR spectra and SEM micrographs revealed that SDS was successfully bonded onto the chitosan surface, leading to the enhanced interfacial adhesion with PP matrix.
Footnotes
Funding
Funding for this research was provided by Universiti Malaysia Perlis through university short-term grant.
