Abstract
Herein, a novel polypropylene/polyurethane rubber (PP/PUR) was prepared based on thermoplastics vulcanizates (TPV)/layered double hydroxide (LDH) nanocomposite in the presence of 3-(triethoxysilyl)propylsuccinic anhydride (SSA) intercalated LDH (SSA-LDH). The effect of SSA-LDH was explored on thermo-mechanical, thermal, morphological, rheological, and mechanical properties as well as gel content, compression set, X-ray photoelectron spectroscopy analysis, and cross-linking density (CLD) of TPV based on PP/PUR with the weight ratio of 40/60. It was found that the crystallization, tensile strength, gel content, cross-linking density, young’s modulus, elongation at break, melting point, and damping factor significantly changed with increasing SSA-LDH content in PUR/PP TPVs. The reasons could be attributed to cross-linking induced by SSA-LDH and curing agent Bis-(triethoxysilylpropyl) tetrasulfide (also called (Si-69)) that grafted onto PUR chains, resulting in good dispersion of fine PUR vulcanized particles into PP matrix. This claim was confirmed by studying the rheological properties through Rheometeric mechanical spectroscopy and dynamic-mechanical analysis. Field emission scanning electron microscope images confirmed the improved compatibility between PUR and PP by adding SSA-LDH. The gel content and CLD of TPV samples without SSA-LDH were 20% and 2.75 mol/mL, respectively, which showed a significant increment to 25.5% and 4.5 mol/L upon adding 5 wt. % SSA-LDH. The highest elongation at break, tensile strength, and young’s modulus were 525%, 16.5 MPa, and 210 MPa, respectively which were observed for TPV5.
Keywords
Introduction
Thermoplastic elastomers (TPEs) refer to a blend of elastomer and thermoplastic polymer. 1 These materials are divided into two categories: TPO which can be prepared by simple mixing, and thermoplastics vulcanizates (TPVs) or dynamic vulcanizates which involves the dynamic crosslinking of the elastomeric phase.2-4 TPVs have a wide variety of applications in automobile parts, link protection, footwear, bundling, and clinical equipment due to their excellent properties such as magnificent wearability, low density, and moderately minimal effort. 5 During blending, the elastic phase is dynamically cross-linked in the presence of a suitable coupling agent to reduce the interfacial tension between two phases and also enhance the overall mechanical properties of TPV samples.6-9 Different investigations have indicated that these materials have properties similar to the ordinary vulcanized elastic. 10
Sulfur, phenol-formaldehyde resins, and peroxides are the most utilized curing agents for making TPVs. Peroxides have been long utilized to cross-link the rubber phase of the TPVs.11,12 Two simultaneous processes occur upon the incorporation of peroxides into TPE blend: vulcanization of rubber phase and degradation of thermoplastic phase. 13 However, in peroxide curing system the obtained TPV suffers from drawbacks such as unpleasant smell, a slight color change, and most importantly, deterioration in some mechanical properties such as reduced tensile strength and increased compression set.14,15 Lai and Wu employed a silane compound to prepare TPV from PP and new metallocene polyethylene. 16 They found that thermal properties and tensile strength remained constant at high concentrations of silane, while showing a decrease in energy dispersal during the tearing test. Wang et al. examined the effect of silane coupling agent and flame retardants such as dipentaerythritol and melamine phosphate on the intrinsic property of TPV based on PP/EPDM. 17 Their study confirmed enhanced mechanical, rheological, and dynamic mechanical properties of TPV upon the use of a silane coupling agent. Silane coupling agent have been widely used to prepare TPVs based on PP/ethylene-octene copolymer. 18 TPV based on PP/PUR compound can be produced by blending molten polyurethane rubber (PUR) and PP. It should be noted that there is still no report on the preparation of TPVs based on these polymeric materials.19-23 This research is thus aimed to explore the effect of SSA-LDH concentration on cross-linking density, compression set, morphological, thermal, mechanical, rheological, and gel content properties of crosslinked PUR/PP-based TPV. Also LDH nanoparticles are non-toxic and eco-friendly materials with conspicuous features like facile and affordable synthesis and powerful ion-exchange ability which leads to a promoted compatibilization between the rubber and polymeric phase of a TPV component, However, no report has addressed the application of this nano material on the synthesis of a TPV component.
Experimental
Raw materials
Homo-Polypropylene (HP552 R; melt flow index of 25 g/10 min @230
Preparation of SSA-modified LDH (SSA-LDH)
The silane-coating process of LDH nanofiller (Si-LDH fabrication) involved the following steps: LDH synthesis through decomposing urea by the hydrothermal approach .
24
In a typical procedure, 250 mL of the aqueous solution of MgCl2·6H2O (18.2wt. %) and AlCl3·6H2O (7.3wt. %) was mixed with a 100 mL of 28.5wt. % urea aqueous solution in a condenser-equipped flask. The mixture underwent 24 h of mild stirring at 150°C. Upon reaction completion, the white cream mixture was filtered and repeatedly centrifuged at 2000 r/min followed by over-drying at 80°C for 24 h. Subsequently, 150 mg LDH and 0.5 wt. % APTES were ultrasonically dispersed in methanol for 1 h. They reacted for 8 h at 50°C and pH=10 to achieve Si-LDH. The procedure of nanoparticle modification is illustrated in Scheme 1. Chemical Structure of bis-(Tri-ethoxypropyl) Polysulfide (SSA-LDH).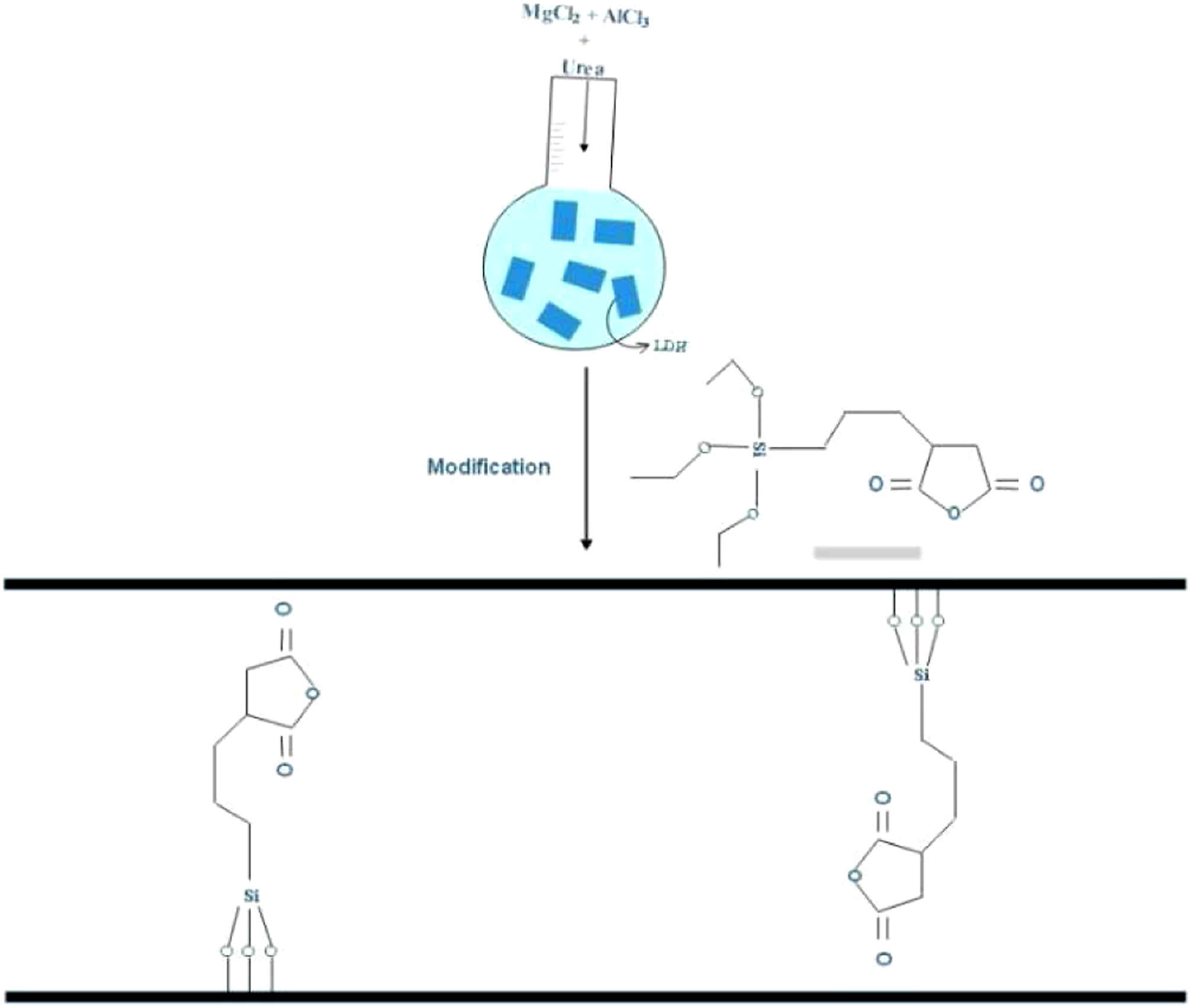
Preparation of the PUR-LDH mixture
Measured amounts of HTPB and calculated levels of LDH (0,1,3,5, and 7 wt. % relative to total resin) along with DBTDL (0.05 wt. %) were charged into a round-bottomed flask, heated to 85°C, and thoroughly mixed with a predetermined amount of IPDI corresponding to a fixed NCO/OH ratio of 1.5:1. The reaction system was vigorously stirred using a mechanical stirrer, under dry nitrogen atmosphere. The completion of the reaction was determined by the di-n-butylamine titration upon reaching the theoretical NCO percentage. Then, the mixture of PUR prepolymer and LDH was obtained.
Fabrication of TPVs
TPV formulation at a weight ratio of 60/40 and their nomenclature.

Characterization
Mechanical properties
Tensile properties
Tensile properties including elongation at break, young’s modulus, and tensile strength, were measured according to ASTM D412 using SANTAM Testing Machine (Iran) at a constant speed of 500 mm/min. All measurements were carried out under room condition at 25±2°C. Prior to testing, all samples were kept in this condition for 48 h.
Shore A hardness
Shore A hardness was determined based on ASTM D2240 (sample dimensions: 20×20×6 mm3).
Thermal properties
The crystallinity and melting temperature of TPVs were evaluated by exploring the thermal properties using DSC (Perkin Emler) analysis. DSC thermograms were recorded in the range of 25–200°C under N2 atmosphere at heating and cooling rates of 10°C.min−1. N2 purge rate was kept at 50 mL/min. The percentage of crystallization (Xc) was calculated according to equation (1)
Rheological properties
The rheological behavior of TPV samples was investigated using a parallel plate rheometer (MCR101: Anton paar, Austria). Based on the diameter of the parallel plates rheometer (PP35-SN20785), the samples were cut in a circular shape with an approximate diameter of 25 mm. The distance between the plates was set to 1 mm. The viscosity of samples was measured at 210°C while the shear rate ranged in 0.1–1000 s−1. Viscoelastic properties such as damping factor were determined at the angular frequency range of 500 to 0.5 s−1 and temperature of 200°C. Data analysis was performed using rheoplus/32 software (version 3.40, provided by the device producer).
Dynamic mechanical and thermal analysis
DMTA measurements were performed on the injection-molded samples with a dimension of 40 × 6 × 2.5 mm3. The temperature was first decreased to −150°C and then subsequently increased to 150°C at the rate of 0.7°C/min and a constant frequency of 15 Hz and 1wt. % strain. The maximum tan δ peak was specified as the Tg of PUR rubber.
Morphological properties
The morphology of TPV was characterized by a scanning electron microscopy (FE-SEM; Hitachi S-4800, Japan) operating at 15 kV for two virgin and etched samples. In etched samples, they were put in boiling xylene for 48 h to completely remove PP phase, oil, and uncross-linked PUR phase followed by 12 h of placement in an oven at 60°C to remove the solvent. The dried TPV was coated with gold prior to imaging by FE-SEM. The results were further assessed by ImageJ processing software to evaluate the rubber domain size of TPV samples. 120 rubber domains from three different broken specimens were examined to determine the rubber domain sample.
Cross-linking density
TPV samples (25 

The swelling ratio (Equation (4)) is defined as the ratio of the total volume of the swollen sample (V) and the volume (Vp) of the dry sample.
The reciprocal swelling ratio (1⁄Q) is considered as cross-linking density. By comparing equations (3) and (4), it is clear that 1⁄Q can be defined by φp.
Compression set
Compression set is an important elastic property of TPVs. The compression set shows the capacity of a material to recover within a particular time at a specific temperature. The compression is induced by a device consisting of two or more parallel plates between which the sample is placed. The sample is subjected to 25wt. % compression strain. To minimize the measurement error, the distance between the two plates was kept constant and determined by the ASTM standard D395-Method B. The distance between the two plates for specimens (type 1) was 9.5 ± 0.02 mm. The constant temperature of 70°C was also maintained over 22 h of the test period. The amount of compression set can be defined by equation (5)
In the above equation, Cb is the amount of compression set (test method B), which is communicated as the level of the first redirection. T0, Ti, and Tn denote the initial and final sample thickness and the distance between the two parallel plates, respectively.
Oil-Swelling
The oil resistance of the TPV specimens (10×10×10 cm3) was evaluated based on ASTM D-471 utilizing IRM 901 and IRM 903 oils. The specimens underwent 166 h of immersion in the oil bath at room temperature. Their weight was measured after removing the excess oil from the surface
In which, W0 and Ws are respective initial and post-immersion weight of the sample (g). The measurements were conducted in triplicates and the average value was recorded.
Results and discussion
Cross-linking density (gel content) and compression set
The gel content and cross-linking density (CLD) Vs SSA-LDH content were plotted in Figure 1. b. Both cross-linking density and gel content (wt. %) raising with increasing SSA-LDH content in TPV samples. The best results were obtained with adding 5 wt. % SSA-LDH to the TPV. The gel content and CLD of TPVs without SSA-LDH were 20wt. % and 2.75 mol/mL which showed a significant increment to 25.5 wt. % and 4.5 mol/L, respectively, with adding 5 wt. % SSA-LDH. As Si-69 activated by anhydride groups in SSA-LDH formulation, the cross-linking density was enhanced sharply by incrementing the amount of SSA-LDH in the mixture. Therefore, increasing the nanoparticle content beyond 5wt. % i. e 7wt. % or more percentage of it slightly declined the gel content and CLD due to the formation of larger SSA-LDH aggregation. An increase in the concentration of SSA-LDH resulted in a parabolic trend of gel content and CLD of TPV nanocomposites. It is important to note that one of the most characteristics of TPVs is their lower compression set defined as the elastic recovery of the rubber domain. This parameter is a measure of cross-linking density of elastomeric phase dispersed in TPV matrix. The higher level of cross-linking density, induced the lower the compression set.
25
As shown in Figure 1. a, the compression set of TPV samples reduced with increasing SSA-LDH content which can be due to the high solidity of nanoparticles and crosslink density of rubber domains. As mention above, an increment in the concentration of SSA-LDH could increase the number of active anhydride sites on the surface of SSA-LDH particles, which in turn, enhanced the cross-linking density and reduces the compression set of the system. Moreover, Si-69 grafting onto PUR polymer chains increased the probability of the chain anchor on nanoparticles, leading to excess cross-linking and immobile points on polymer chains, thereby this phenomenon can reduce the compression set of TPV samples. Scheme 2 shows the reaction between Si69 and PUR. The minimum value of the compression set was obtained for PUR/PP-based TPV containing 5wt. % SSA-LDH. Higher nanoparticle concentrations may result in the aggregation, hence increasing the plastic deformation and the compression set property. (a) Show the effect of SSA-LDH content on compression set and (b) show variation of the gel content and cross-linking density with different loading level of SSA-LDH content of PUR/PP-based TPVs, respectively. The mechanism of the reaction between Si-69 and PUR.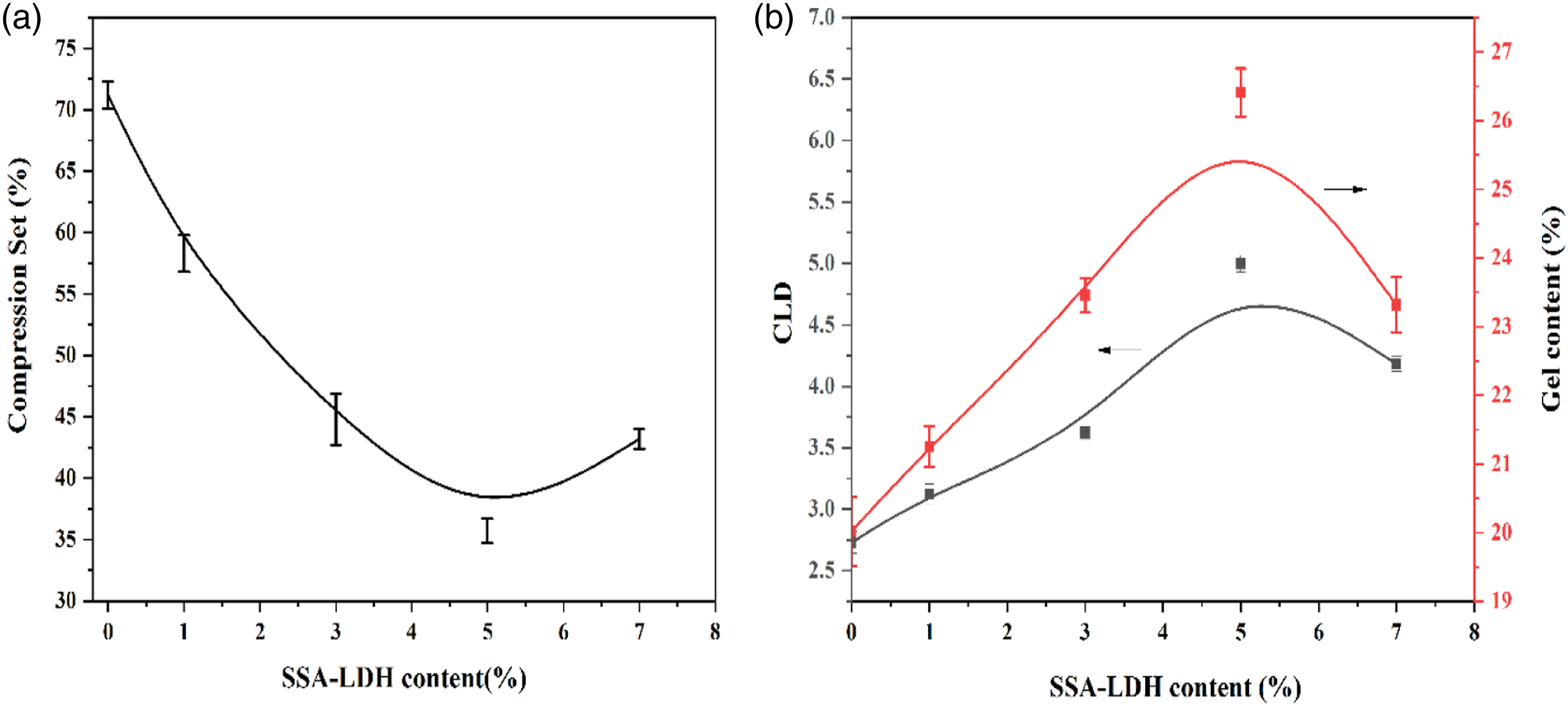

Tensile properties
Figures 2(a) and (b) show tensile properties of PUR/PP-based TPV blends (i.e., elongation at break (%), young’s modulus (Mpa), and tensile strength (Mpa)) as a function of SSA-LDH content, respectively. Elongation at break, tensile strength, and young’s modulus of pristine PUR/PP-based TPV blend were 248%, Mpa, 6.5 Mpa, and 12.5 Mpa, respectively. All of these values enhanced by increasing the SSA-LDH content of TPVs. The highest elongation at break, tensile strength, and young’s modulus were observed for TPV5 and they were 525%, 16.5 Mpa, and 210 MPa, respectively. Compared to pure TPV, the properties of PUR/PP-based TPV blends were improved significantly by the addition of SSA-LDH to the TPV samples. (a) Show the effect of SSA-LDH content on tensile strength and young’s modulus of PUR/PP-based TPVs and (b) show the effect of SSA-LDH content on hardness and elongation at break of PUR/PP-based TPVs.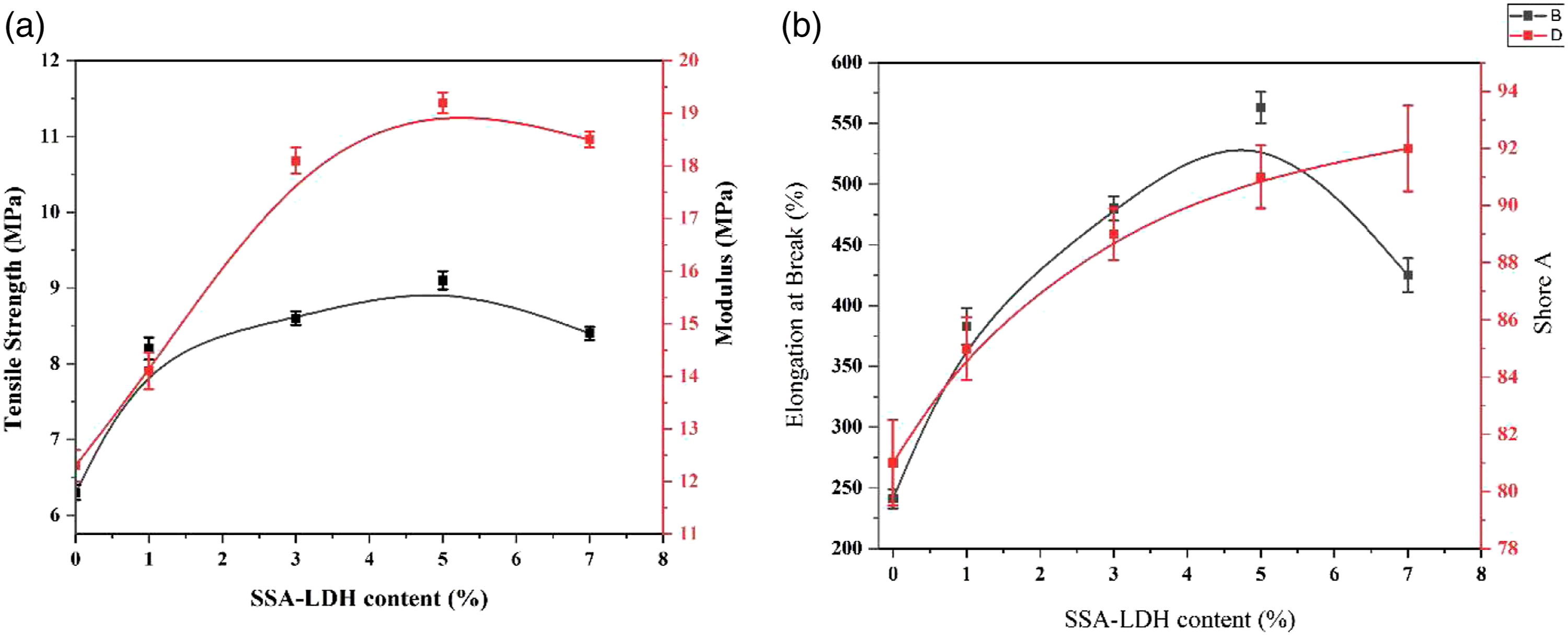
The size of the vulcanized rubber domain, as well as rubber-polyolefin interfacial adhesion, plays a crucial role in the mechanical properties of the TPV samples. The SSA-LDH particles are located at the phases interface of PP/PUR blend, forming stable interactions, which declines the interfacial tension while enhancing the interfacial adhesion. 26 The use of a compatibilizer can increment the interface thickness further promoting the stress transfer between dispersed PUR droplets and the PP matrix phase. Such a strong interfacial phase reduces the interlayer slippage, hence elevating the mechanical characteristics. Actually, the compatibilizer chains restricted the mobility of the matrix chains at the interphase, establishing a stable matrix-dispersed vulcanizate rubber boundary. Also linking the PUR chain to SSA-LDH will facilitate the dispersion of reduced nerve rubber domains within the thermoplastic matrix, resulting in better compatibility between two immiscible phases. This obviates the necessity of using an additional compatibilizer to improve the interactions between the two immiscible phases in TPV. FE-SEM analysis confirmed the better interactions between PUR and PP phases with adding the SSA-LDH, which improved the tensile properties of TPV samples. The most noteworthy improvement in the tensile strength was obtained in the sample containing 5wt. % SSA-LDH. Shore A hardness also increased by adding Si-69, which is attributed to the uniform distribution of the SSA-LDH particles and the crosslinked PUR domains in the PP matrix.
Differential scanning calorimetry (DSC)
The heating-cooling graph of PUR/PP-based TPV blends are presented in Figure 3. The pristine PUR/PP-based TPV had melting temperature (Tm, °C), melting enthalpy (Hm, J⁄g), and crystallinity degree (Xc) of 161.5 Heating-cooling curves of the prepared TPVs.
As previously mentioned, PUR can be grafted onto SSA-LDH and it can increase the cross-linking density of the PUR phase. This will cause higher interaction between rubber and thermoplastic phases and consequently intermolecular distance between the chains of PP phase, hence, it preventing chains from approaching each other, hence reducing the molecules compaction, and decreasing the crystallinity of the TPV samples. By increasing amorphous property of TPV blends, the melting point of polymer chains shifting to lower level as compared to the pure system, reducing the energy required to melt this structure.
Rheological properties
Regarding the importance of the melt processability in TPVs, the effect of SSA-LDH content was investigated on the rheological features of the samples. Figure 4 a, b demonstrates the variations in complex viscosity ( (a) Complex viscosity (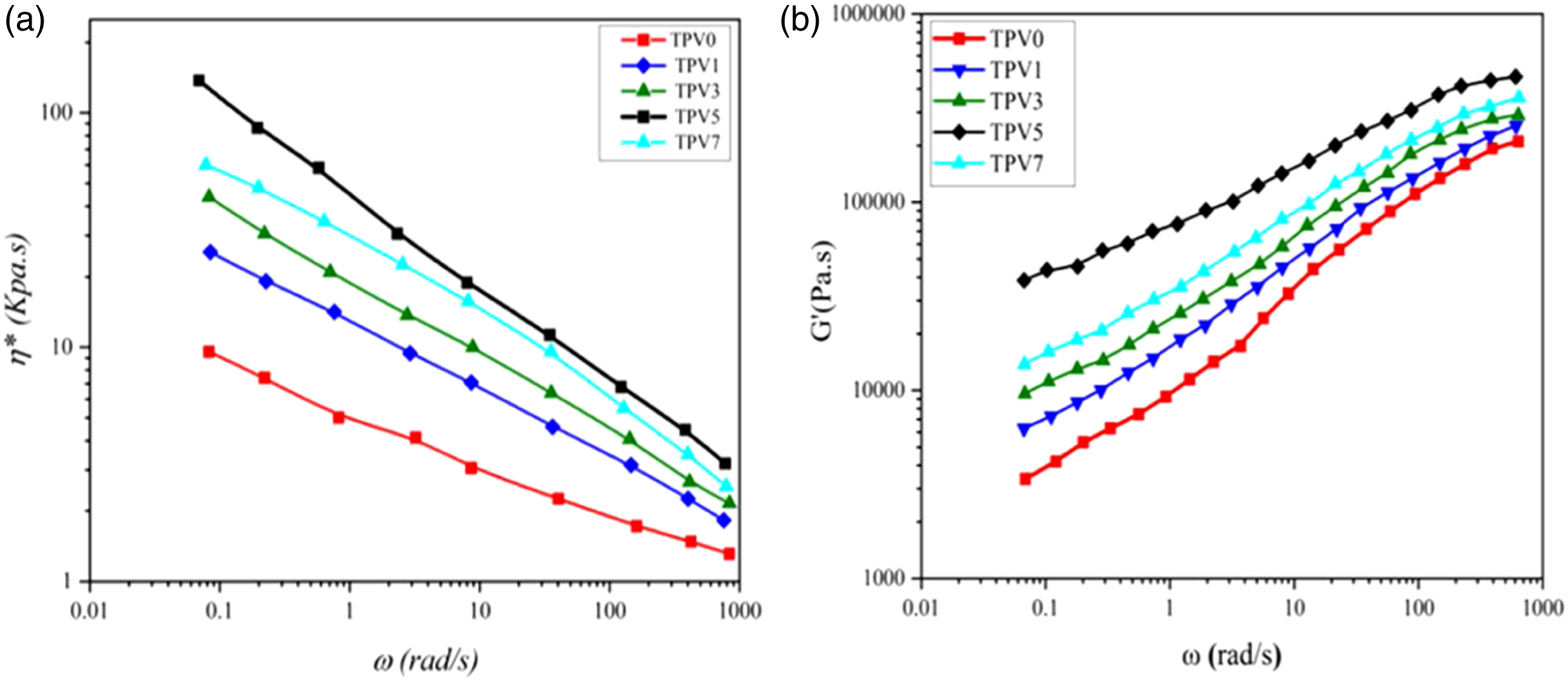
Figure 5 illustrates the temperature dependence of damping factor (tan Temperature dependence of damping factor (tan delta) of dynamically vulcanized TPV nanocomposites.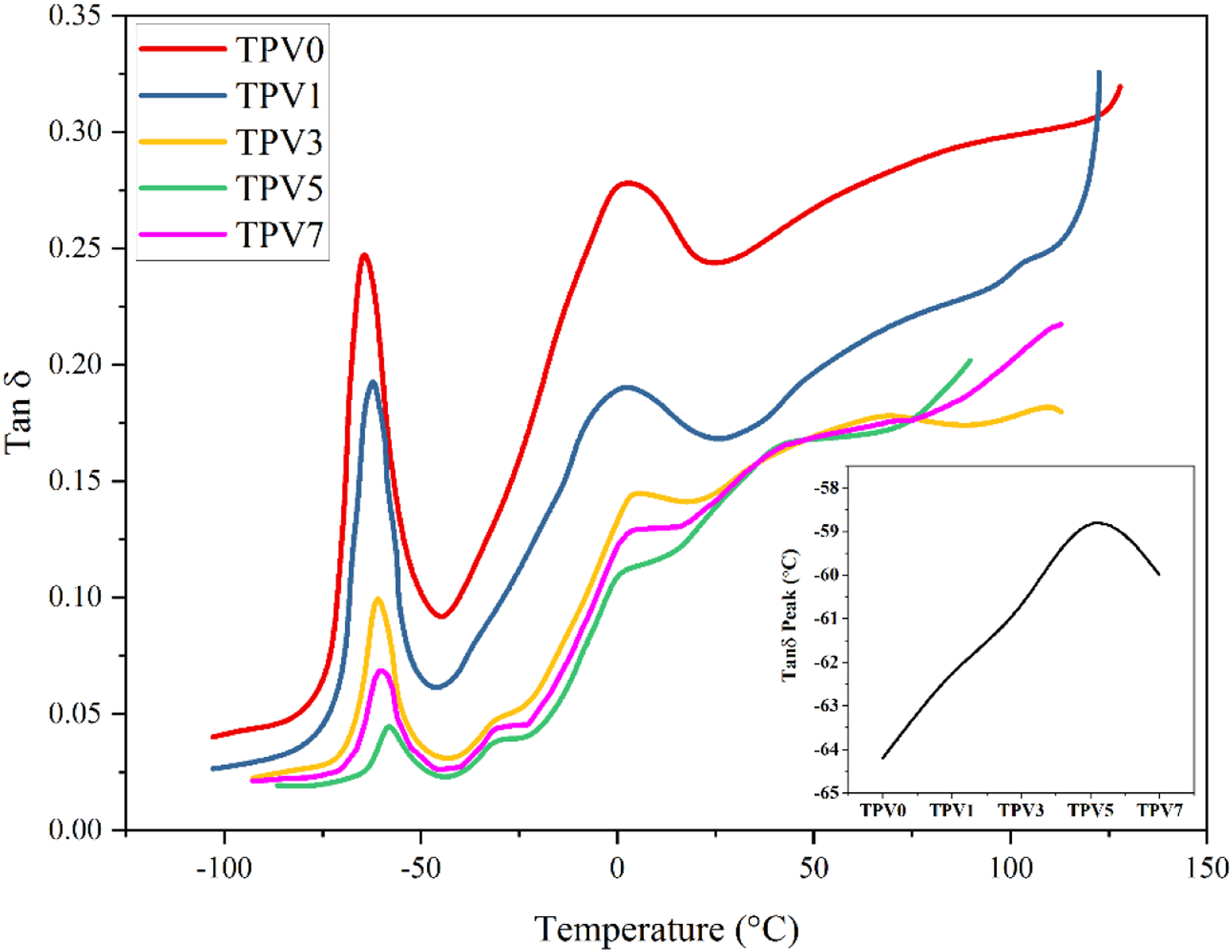
Morphological characteristics
Supplementary Figure S2 and Figure 6 (a, b, and c) illustrate FE-SEM photomicrographs of various PP/PUR/SSA-LDH TPV nanocomposites: untreated (virgin) and treated (etched by xylene) under various conditions for TPV0, TPV3, and TPV5, respectively. In the virgin cases, Figure S2 (a–c) shows the fractured surfaces of TPV samples with and without SSA-LDH. The white domains in the fractured segments were formed by stretch-induced crystallization when preparing fractured samples, which was often observed in the crystallizable polymer. Furthermore, SSA-LDH is reportedly a nucleating agent in the crystalline polymer systems it also confirmed that with DSC test. The wide particle size distribution can be due to the mixing under dynamic vulcanization. Based on Figures 6(a)–(c), the particle size of the etched sample was much bigger. The empty area in the etched images corresponds to the phase (i.e., PP) extracted by hot boiling xylene solvent during reflux condition. As can be seen, the size of the etched phase decreased with increasing the SSA-SSA-LDH concentration in TPV samples. This could be related to enhanced PP-PUR compatibility by addition of SSA-LDH in TPV samples. The major reasons for this observation are the crosslinking of PUR chains. Moreover, the smaller cured rubber particles were more dispersed in PUR/PP-based TPV sample containing 5 wt. % SSA-LDH. By increasing the nanoparticle content up to 5wt. % (for example, 7wt. %), the crosslink density and mechanical stress concertation of the rubber phase increased sharply, thereby lowering the rubber particle size and improving its dispersion within the PP matrix.
27
The excess of SSA-LDH particles showed a tendency to locate at PP/PUR interface, increasing the compatibility and interfacial adhesion between nonpolar matrix and relatively polar rubber phase. The variation trend of the effect of rubber particle size is shown in Supplementary Figure S3. Scanning electron micrographs determined for the etched PP/PUR TPV samples: (a) TPV0; (b) TPV3; (c) TPV5.
Figure 7 compares the oil swelling of the TPVs specimens with a reference one (TPV0) upon immersion in ASTM and IRM903 oil. All TPVs exhibited higher swelling levels in IRM 903 as compared with ASTM, implying their lower resistance against IRM 903 oil (since it has relatively higher aromatic contents). The oil swelling showed a decrement upon the incorporation of SSA-LDH,
28
reflecting that SSA-LDH enhanced the gel content of the rubber phase (reactivity effect) causing an improvement in the morphological features of the PP/PUR composites (as a result of the compatibilization effect) which promoted the oil resistance of PP/PUR TPVs. Noteworthy, SSA-LDH used in the TPV3 and TPV5 is an oil-resistant material. An increase in the SSA-LDH content up to 7wt. % significantly enhanced the swelling in ASTM and IRM 903. Moreover, TPV5 showed higher oil resistance which can be attributed to the SSA-LDH layers at the interface and its enhanced thickness. This issue was also confirmed by morphological examinations. The results indicated the oil resistance of PP/PUR nanocomposites, specifically those vulcanized by sulfur-silane, introducing them as a promising candidate for automobile, chemical, and aviation applications. Comparison of oil swelling for TPV0, TPV1, TPV3, TPV5, and TPV7 after immersion in (A) ASTM oil no. 1 and (B) IRM 903 oil.
X-Ray photoelectron spectroscopy analysis
Figure 8 shows the post-adsorption surface properties were examined by XPS analysis which shows the composition and chemical state of the adsorbents, offering valuable information on the fluoride adsorption. The binding energy (BE) of LDH 2p in SSA-LDH emerged at 610 eV, which shifted to lower values in the case of PUR-LDH hybrid and PP/(PUR-LDH) blend, implying a change in the chemical environment of LDH atoms in PUR-LDH hybrid materials and PP/(PUR-LDH) blend mostly due to the reactions between NCO of PUR polymer and OH of SSA-LDH. XPS spectra of LDH 2p in LDH, PUR-LDH hybrid materials, and PP/(PUR-LDH)- blend.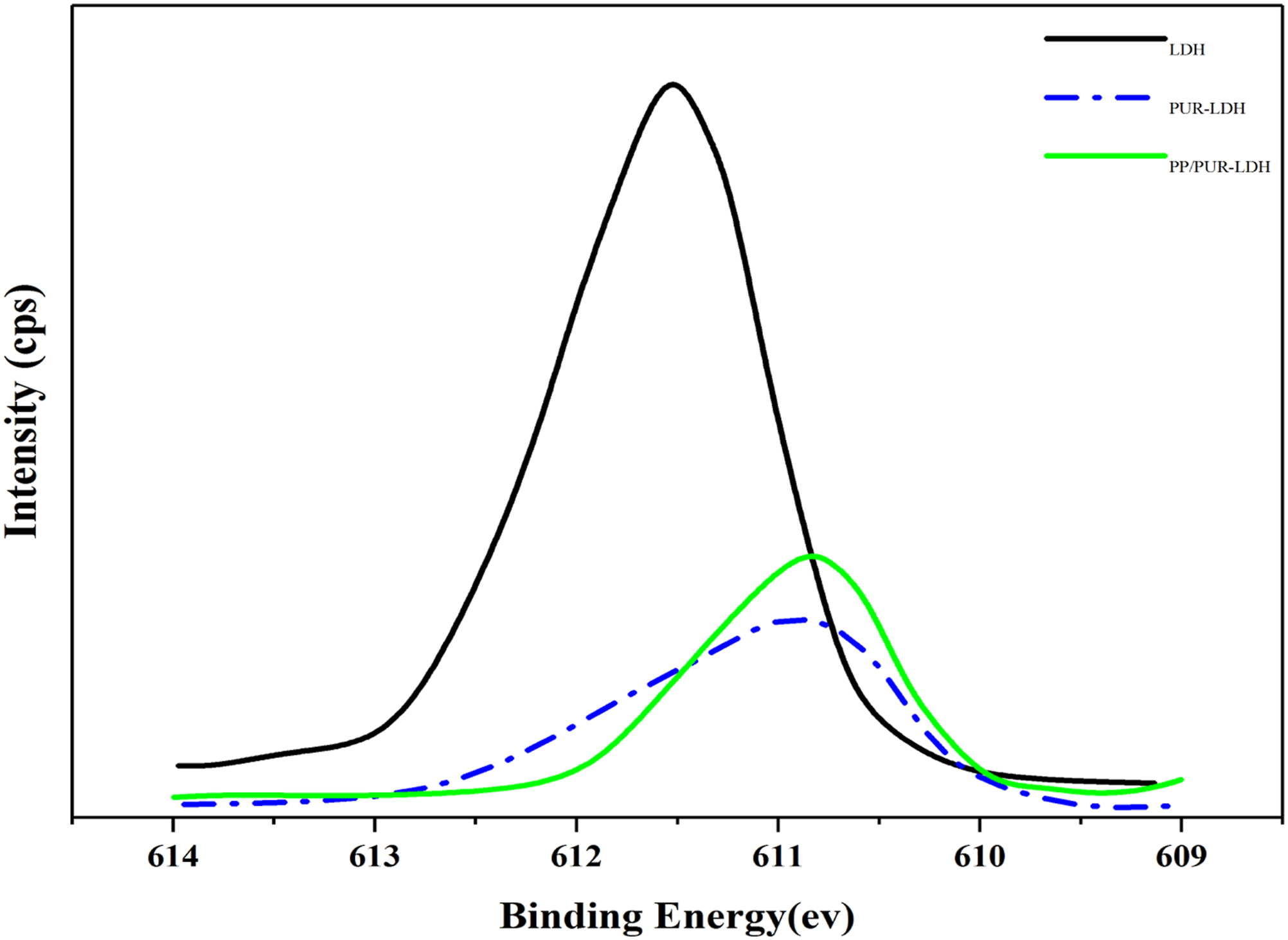
Conclusion
The PP/PUR-based TPV (weight ratio of 60/40) was successfully prepared which contained various levels of SSA-LDH nanoparticles (1, 3, 5, and 7wt. %). The fabricated TPVs were evaluated by morphological, mechanical, rheological, and thermal analyses. The gel content, compression set, and cross-linking density of TPVs were also investigated. The results showed an increment in elongation at break, cross-linking density, gel content, tensile strength, and viscosity by increasing SSA-LDH content of the blend, while compression set, melting temperature, melting enthalpy, crystallization, and damping factor showed a decrease. SSA-LDH induced cross-linking of PUR chains by reacting with Si-69 via its anhydride groups. Also, it was found that the best-desired properties can be obtained at the SSA-LDH content of 5wt. %. Furthermore, thermo-mechanical analysis and scanning electron microscopy showed an improvement in the compatibility between PP and PUR upon increasing SSA-LDH concentration. It should be mentioned that SSA-LDH served as a compatibilizer and also a curing agent in the cross-linking of the elastomeric phase. With increasing SSA-LDH in TPV samples, the amount of Si-69 increased in the blend followed by a dramatic enhancement in complex viscosity and elastic modulus of TPV samples.
Supplemental Material
sj-pdf-1-jid-10.1177_09673911221079126 – Supplemental Material for Preparation and characterization of polyurethane rubber/polypropylene-based thermoplastics vulcanizates nanocomposites with succinic anhydride intercalated layered double hydroxide
Supplemental Material for Preparation and characterization of polyurethane rubber/polypropylene-based thermoplastics vulcanizates nanocomposites with succinic anhydride intercalated layered double hydroxide by Ali Moshkriz and Reza Darvishi in Polymers and Polymer Composites.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
