Abstract
Novel thermoplastic vulcanizates (TPVs) based on polyoxymethylene (POM) and methyl vinyl silicone rubber (MVQ) have been prepared by dynamic vulcanization process through a batch mixer. During the preparation of TPV blends, Di-(tert butyl peroxyisopropyl) benzene (BIBP) was used as the curing agent in order to make MVQ cross-linked and TPU was used to coat MVQ for improving the compatibility of MVQ and POM. In order to understand the influence of different compositions on TPV blends, five groups of experimental processes were described in detail. During these experiments, the amount of POM was reduced from 70phr to 30phr, that of MVQ was gradually increased from 18phr to 42 phr, and TPU was increased from 12phr to 28phr. In addition, the morphology and properties of TPVs were studied by DSC, FTIR, SEM, DMA and mechanical tests. The mechanical testing results showed that with the amount of POM decreasing and the total amount of MVQ and TPU increasing, the tensile strength of the TPV blends gradually was decreased, and the elongation at break was increased accordingly from 35.2 ± 6% of pure POM to 142.8 ± 11% of sample 5#.
Keywords
Introduction
Blending polymers are a class of materials in which two or more polymers have been mixed to form materials with desired properties. 1 Through blending modification, some defects of polymer materials can be improved, and the materials’ application will also be broadened. The first kind of blends has excellent tensile strength and rigidity, for example, polymers modified by glass fiber, glass bead, nanocalcium carbonate, potassium titanate whisker, or long glass fiber.2–6 Another class of composites are those which have good toughness and low-temperature resistance, such as those modified by thermoplastic elastomers, EPDM, POE, or SBR modified polymers.7–13 The third kind of polymer mixtures worth mentioning are modified by antistatic agents, for instance, using polymer antistatic agent, ionic liquid, carbon nanotubes, graphene, or conductive carbon black to modify materials for improving their antistatic properties.14–19 In addition to the above methods, dynamic vulcanization can also be used for polymer modification.20,21
As we all know, “dynamic vulcanization technology” refers to the combination of unvulcanized rubber elastomer and thermoplastic resin under the action of vulcanizing agent, high temperature and shearing. During the blending, the rubber phase is vulcanized under the action of cross-linking agent to obtain vulcanized rubber particles with micron sizes which are evenly dispersed in the resin. The composites prepared by dynamic vulcanization are called thermoplastic vulcanizates (TPVs) and the aggregated structure of which presents “sea-island” phase.22,23 Compared with traditional vulcanized rubbers, TPVs have the advantages of good processability, thermoplastic fluidity and recyclability.24,25 After decades of development, TPVs have been widely used in various fields, such as electronic equipment, automobile, cable, etc. In order to promote the research progresses in the field of TPVs, scientists have carried out in-depth researches on various types of TPVs by using various rubbers with different resins, and polypropylene is the most widely used resin in the preparation of TPVs. Such as, Nattapon Uthaipan 26 studied the surface properties and elastomeric behaviors of EPDM/EOC/PP TPVS with different octene contents, the results showed that EPDM and EOC form rubber particles with core-shell structure and disperse in PP matrix, the tensile deformation of TPVs decreased with the increase of 1-octene content. Xu et al. 27 prepared zinc dimethacrylate (ZDMA) by in situ method, and designed the shape-memory materials based on sea-island structured EPDM/PP TPVs via in-situ compatibilization of ZDMA. Prut et al. 28 prepared PP/EPDM TPVs by dynamic vulcanization method, and studied the morphology and dynamic viscoelasticity of vulcanized adhesive in depth, which included the rheological behavior was strongly affected by both the composition and the morphology of the blends. Tian et al. 29 used nitrile rubber and polypropylene to prepare dynamic vulcanizates, the results showed that the compatibility of nitrile rubber and polypropylene had a great influence on the particle size of nitrile rubber and the crystallinity of polypropylene, which could be improved by selecting compatibilizer. Nakason et al. 30 prepared thermoplastic vulcanizates from epoxidated natural rubber and polypropylene, in the process of dynamic vulcanization, the compatibility of phenolic modified polypropylene was studied. In addition to using polypropylene as the base resin, scholars have carried out other synthetic materials as base phase for TPVs research work.31–36 Chatterjee et al. 34 carried out researches on the preparation of TPVs maded by XNBR and PA12, and found that when the proportion XNBR to PA12 is 50:50, the blend shows the highest mechanical properties and superior thermal stability among all TPV samples. Kunanusont et al. 35 prepared TPVs of ethylene vinyl acetate copolymer and natural rubber by using peroxide vulcanization and Chatterjee et al. 36 studied the preparation and properties of silicone rubber/polyamide TPVs by dynamic vulcanization.
For POM resin, the research work on dynamic vulcanizates is very little. We can only learn some relevant knowledge from the research articles of Xiangfu Zhang and R. Nalini Uthaman.37–39 Xiangfu Zhang prepared NBR/POM thermoplastic elastomer by dynamic vulcanization, and found that using NBR with high acrylonitrile content was more helpful to prepare TPV with high tensile strength and oil resistance. In the Nalini Uthaman’ researches the EVA/POM thermoplastic elastomer and EPDM/POM thermoplastic elastomer were studied by dynamic vulcanization method, and the compatibilities of EVA-MAH to EVA/POM blends and EPDM-MAH to EPDM/POM blends were also investigated.
In this paper, our research goal is to modify POM resin with methyl vinyl silicone rubber (MVQ) by using dynamic vulcanization method. In the process of experiments, peroxide BIPB is used as initiator to crosslink silicone rubber. In addition, the second elastomer TPU is used in our study, which can cover and support the silicone rubber in the vulcanization process on the one hand, and improve the compatibility of silicone rubber and POM on the other hand. Finally, several analytical methods are used to evaluate the properties of POM/MVQ/TPU blends, such as mechanical properties test, differential thermal analysis, scanning electron microscope, FTIR and dynamic mechanical analysis. The results show that the POM/MVQ/TPU ternary TPVs prepared by dynamic vulcanization of BIPB have good mechanical properties, which provides a new idea for modifying POM.
Experimental
Materials
POM (MC90, MFR:9.1g/10 min, Mw:1.84 × 105, PDI:2.68) was manufactured by Shenhua Co., Ltd, China. MVQ (RBB2003, Shore A hardness:34, density:1.08g/cm3, tensile strength:7.0 MPa, elongation at break:640) was supplied by Dowcorning Corporation, America. TPU (WHT1570, Shore A hardness:73, density:1.19g/cm3, tensile strength:28.0 MPa, elongation at break:650) was produced by Wanhua Chemical Group, China. BIBP (FARIDA BIBP 90, melting point: 40–50°C, the content of BIBP is 90%, and CaCO3 is 10%) was produced by Fariada Chemical Co., Ltd, China. Antioxidant (Irganox1010) was purchased from Rianlon Corporation, China.
Preparation of POM/MVQ/TPU TPV blends
POM, MVQ and TPU were dried before the extrusion at 80°C for 2 h to eliminate the residual moisture in a convection oven. The TPV blends were prepared by a batch mixer (Haake Polylab QC) with a twin screw at 185°C. Figure 1 shows us the preparation process of POM/MVQ/TPU ternary TPVs. Fistly, when the rotor speed of the mixer was 50 rpm and the temperature rised to 185 °C, MVQ and TPU were added for mixing 2 minutes. Next, the speed of rotor was adjusted to 75 rpm, then POM was added and mixed for 2 minutes. Finally, when the rotor speed was set to 100 rpm, the curing agent BIPB was added to initiate the dynamic vulcanization reaction. After dynamic vulcanization and internal mixing for 4 minutes, the blends were taken out and processed into particles by pulverizer.
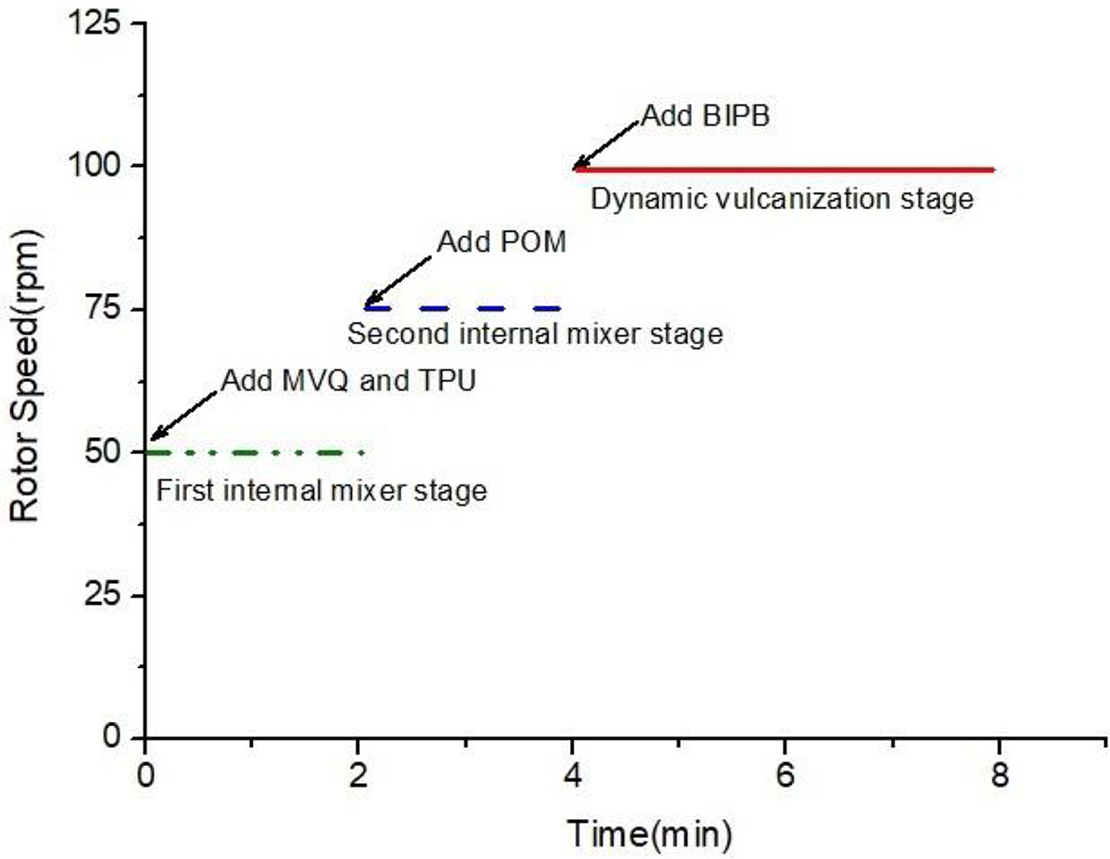
Preparation process of POM/MVQ /TPU ternary TPVs.
The specific formulation was listed in Table 1. To prevent MVQ, TPU and POM’s oxidative degradation during melting mixer, 0.25 phr Irganox 1010 was added to each experiment. The operating conditions of mixer process were as follows: the temperature of the mixter was 185°C; the rotor speed was 50 rpm, 75 rpm and 100 rpm respectively.
Experimental formula of POM/MVQ/TPU TPVs.

Characterizations
Scanning electron microscopy
The sample was fractured in liquid nitrogen, etched by the mixed solvent (the ratio of acetone to chloroform was two to three) at 40°C for 8 h, and then dried in a vacuum at 60°C. The surface of the sample was coated with a thin layer of gold. Phenom PRO PW-100-016 scanning electron microscope was used at 10 kV. 40
Differential scanning calorimetry
The differential scanning calorimetry (DSC) measurements were carried out by a differential scanning calorimetry (NETZSCH DSC 200F3). Some samples were first heated to 200°C and held for 5 min to eliminate previous thermal history, then cooled down to 30°C subsequently. Both the cooling and heating rates were 10°C/min. The experiments were carried out under a continuous high purity nitrogen atmosphere. 41
Fourier transfrom infrared (FTIR)
FTIR spectra of the specimens were carried out on a BRUKER Vertex 70 FTIR spectrometer (Bruker Daltonics Inc.). The test specimens were made into small pieces of films by using the melt-pressing method operated on the thermal platform. 41
Dynamic mechanical analysis (DMA)
Dynamic mechanical properties of the samples (60 × 10 × 3 mm) were measured under three-point bending mode by a NETZSCH 242E DMA system in a nitrogen atmosphere (50 ml/min). The measured frequency was 5 Hz, and the temperature scan was carried out from −100 to 100°C at a heating rate of 3°C/min. 42
Mechanical property measurements
All sample pellets were dried before the injection at 80°C for 1 h. The tensile and bending test splines were molded by injection using HAAKE MiniJet-Ⅱ(The mold model of tensile spline was 557-2298, the mold model of bending spline was 557-2296: 80mm × 10mm × 4 mm). The tensile properties of the specimens were tested by Universal Material Testing Machine (INSTRON 5966) according to ISO 527-2 method. The stretching rate was 50 mm/min. A Impact Testing Machine (INSTRON CEAST 9050) was used for measuring notched Charpy strength (the mold model of Impact testing spline is 557-2296: 80mm × 10mm × 4 mm). All test samples were placed in the Constant Climate Chambers (BINDER, Model KBF 240) for 40 h state adjustment at 23 ± 2°C and 50 ± 5% relative humidity. The mechanical results were calculated as a function of the original cross-section.
Results and discussion
Compatibility comparison between POM, MVQ and TPU
In previous studies, we found that the dispersion of toughened elastomer in POM matrix is closely related to the compatibility between the two materials. That is to say, if the compatibility of the two above materials is good, the toughened elastomer can form more uniform rubber particles with a small particle size in the POM matrix. The modified POM thus obtained has higher toughness and lower temperature resistance than that is made from materials with poor compatibility. This phenomenon can be explained by the principle of similarity compatibility. 43 The following formula can explain the compatibility law between polymer components:
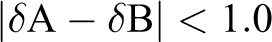
Here, δA is the solubility parameter of polymer A, and δB is polymer B’s. If the difference between the two parameters above is less than 1.0, the two polymers are compatible. If the difference is more than 1.0, an appropriate compatibilizer should be added.
By consulting the polymer solubility parameter manual, it can be found that the solubility parameter of POM is 20.9, and the solubility parameter of MVQ is 15.4. Therefore, it is difficult to obtain POM composites with good properties by dynamic curing modified POM with MVQ only. In order to solve the above problems, TPU with a solubility parameter of 20.4 can be selected as the third component in the experiment to form cladding layer on the surface of MVQ during the dynamic vulcanization process.
Morphologies of TPV blends
Figure 2 shows the scanning electron micrographs of POM/MVQ/TPU ternary TPVs with different compositions. Image (a) is the SEM of neat POM, from which we can clearly observe that the aggregate structure of neat POM is very uniform and smooth. Image (b), (c) and (d) are the SEM photographs of the ternary thermoplastic vulcanizates prepared dynamic vulcanization. The content of POM, MVQ and TPU in the vulcanizates ranges from 70 to 30 phr, 18 to 42 phr and 12 to 28 phr respectively, as showed in Table 1. It can be found from image (b) that the SEM of sample 1# shows us the large contiguous areas marked by the blue curves are very smooth in shape, which are the polyoxymethylene phases. The areas enclosed by the red circles are morphologically observed as cavities with different particle sizes. It can be inferred that these areas are elastomer particles formed by MVQ coated with TPU. This is because that the content of POM in sample 1# reaches 70 phr, which accounts for the majority of the blend composition, so it can form a continuous phase during the dynamic vulcanization process. During the dynamic vulcanization process, the silicone rubber is partially cross-linked under the action of the vulcanizing agent, and coated with TPU to form tiny particles dispersed in the POM matrix.

SEM images of POM/MVQ/TPU ternary TPVs with different compositions. (a) Neat POM, (b) POM/MVQ/TPU(70/18/12), (c) POM/MVQ/TPU (50/30/20), and (d) POM/MVQ/TPU (30/42/28).
When the composition of POM/MVQ/TPU TPVs changes, such as the formula of sample 2 to 5# in Table 1, the morphological structure of the blends will also change accordingly. As shown in image (c), when the content of POM in the TPV blends decreases to 50 phr and the total amount of MVQ and TPU increases to 50 phr, we can find that the smooth areas enclosed by the blue curves are reduced compared to image (b), and these blue areas are the POM phase. In addition, it can be found that the different blue areas are separated by the red folds wich are MVQ/TPU elastomer blends formed by the phase inversion. In addition, there is another part of the polyoxymethylene phase between these folds.
As the POM content of the TPV blends decreased to 30 phr and the total amount of MVQ and TPU is reached to 70 phr, it can be obversed clearly that SEM image of 5# sample showed that the red fold areas formed by the rubber phase of vulcanized MVQ coated with TPU ae more dense, and the areas enclosed by the blue curves are part of the polyoxymethylene phases, the other part of the polyoxymethylene is filled between the red folds.These phase structure changes during the dynamic vulcanization of POM/MVQ/TPU can be well explained by the theory of phase transition during polymer blending.
Differential scanning calorimetry of TPV blends
In this work, the influence of the content of MVQ and TPU on the crystallization behavior of the POM/MVQ/TPU ternary TPVs is evaluated by means of DSC analysis. From the crystallization curves of Figure 3, some regular information and useful data can be obtained for describing the crystallization behavior of TPVs blends. Firstly, the crystallization temperature of neat MC90 is 139.7°C, and that of TPU WHT1570 is 52.5°C, which means POM is easier to crystallize than TPU during the cooling process from high temperature to low temperature. Secondly, the area of crystallization peak of MC90 is significantly larger than that of WHT1570. That is to say, the crystallinity of POM is obviously higher than that of TPU. This is because there are a lot of hard segments and polyether soft segments in the molecules of WHT1570; the hard segment is crystalline, but the soft segment is amorphous. Thirdly, there is no crystallization peak on the DCS crystallization curve of MVQ RBB2003, which means that MVQ does not melt in the TPV blends system. This is because in the dynamic vulcanization system, the molecular chain of MVQ is undergoed a cross-linking reaction under the action of the peroxide initiator.
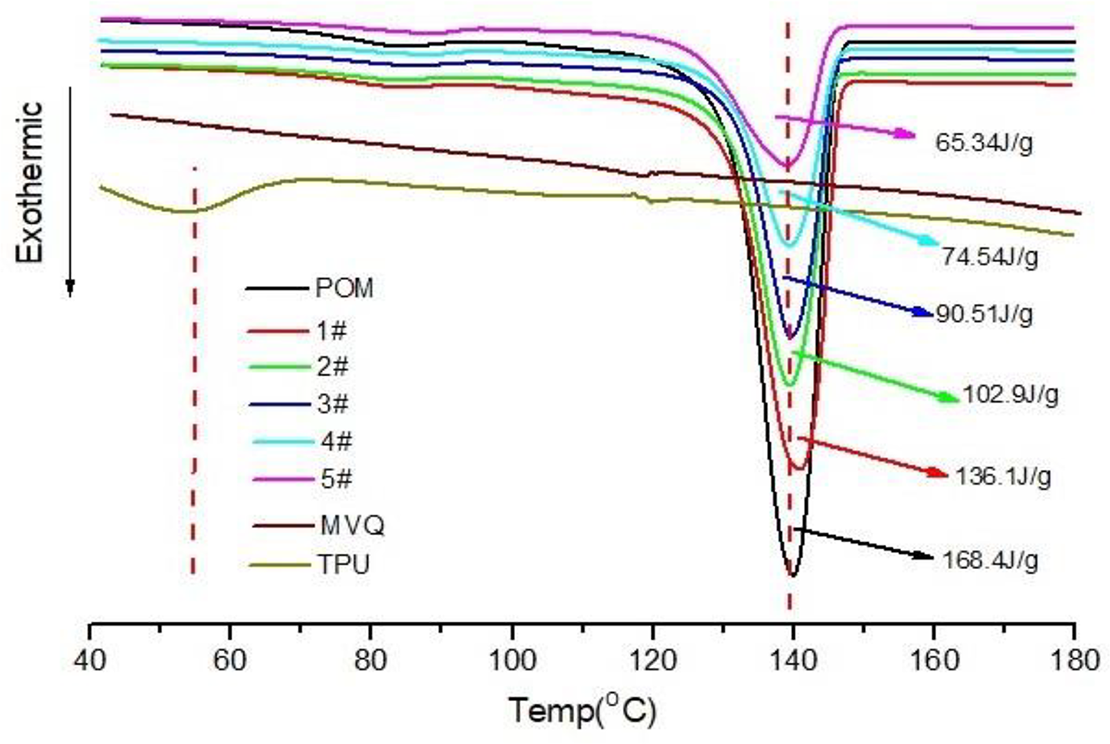
DCS crystallization exothermic curves of POM, MVQ, TPU and the POM/MVQ/TPU ternary TPV blends between 40°C and 180°C, the experiments were operated at the condition of reheating (20°C/min) from 30°C to 200°C.
For the TPV samples 1#, 2#, 3#, 4# and 5#, it can be seen from Figure 3 that their crystallization temperatures have little difference. However, the crystallization enthalpies of the above five TPV blends are very different. For examples, the crystallization enthalpy of neat POM is 168.4 J/g, and that of samples 1#, 2#, 3#, 4# and 5# are 136.1J/g, 102.9 J/g, 90.51J/g, 74.54 J/g and 65.34J/g, respectively. The crystallization enthalpy of all the five TPV blends is less than that of pure POM, which can be explained by the following viewpoint in my opinion.
The reason is that the crystallization of polymer melt is an exothermic process and only POM is a highly crystalline thermoplastic resin among the three kinds of polymer POM, MVQ and TPU in TPV blends, so when the content of POM in the blends decreases, the heat released by crystallization of the blends also decreases, and the corresponding crystallization peak reflected on the DCS curve gradually decreases.
Fourier transform infrared spectroscopy
Figure 4 shows the FTIR spectra of POM, 3# TPV material. The black curve is the infrared spectrum of POM MC90, in which 2972 and 2912 cm–1 are assigned to the CH stretching of POM molecular chain; 1081 and 881 cm−1 are assigned to the C–O and C–O–C absorption peaks of POM; the peak at 632 cm−1 is assigned to the OH bending of POM. The FTIR spectrum of 3# TPV is shown as the red curve, from which we can find the infrared characteristic peak of POM, such as the absorption peaks of CH, C–O and C–O–C. In addition, the peak at 2964 cm−1 is belonged to the Si–CH3 extended absorption of MVQ; 1620 cm−1 is assigned to the CH=CH2 vibration peak peak of MVQ; 1010 cm−1 represents the absorption peak of Si-O-Si in MVQ molecular chain. At last, the peak at 1756 cm−1 represents the C=O stretching vibration absorption of TPU; 3336 cm−1 is assigned to the NH vibration peak peak of TPU; 3695 cm−1 is belonged to the OH absorption of TPU.
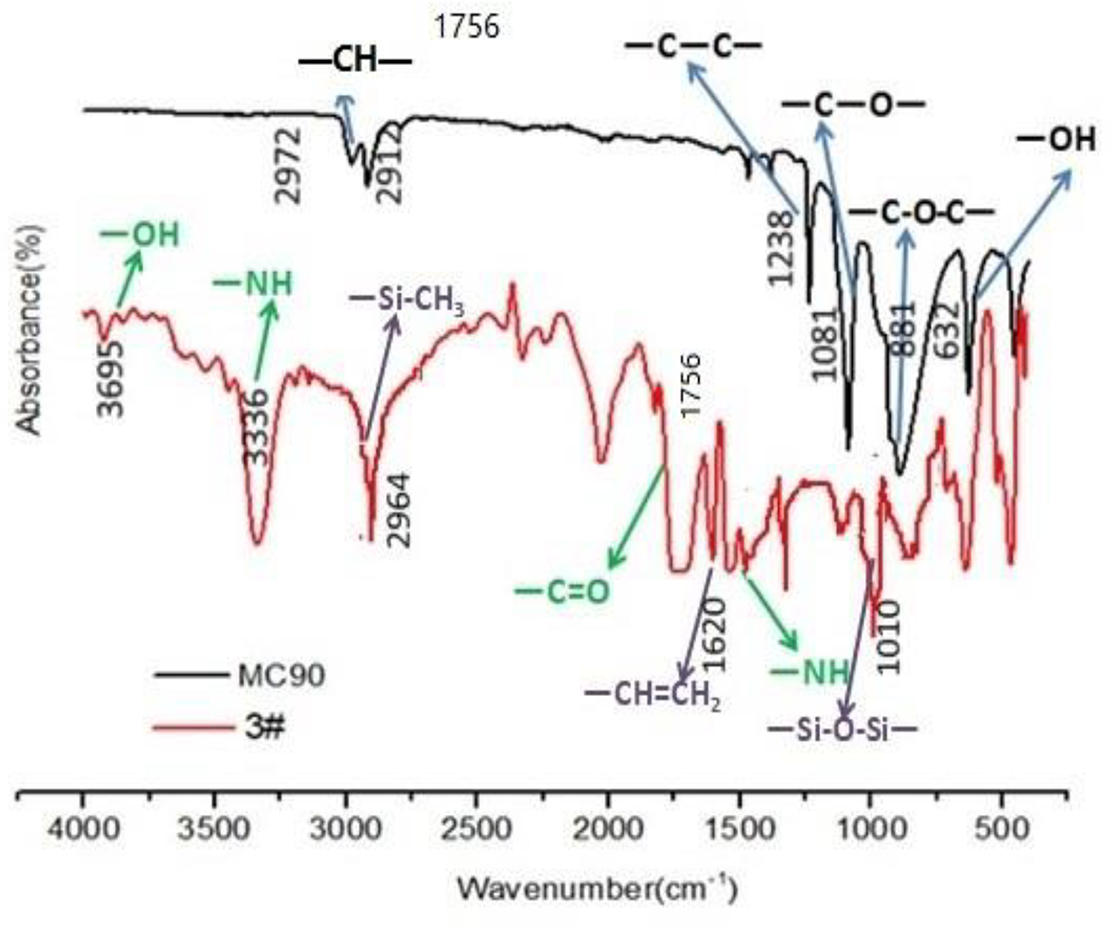
FTIR absorbance of POM and 3# TPV blend with 50 phr of POM, 30 phr of MVQ and 20 phr TPU.
Dynamic mechanical properties of TPV blends
It is well known that the energy storage modulus E′ can be used to characterize a material’s ability to store energy under the action of elastic strain. The larger the storage modulus is, the stronger the elastic recovery capacity and the greater the rigidity of the material are. The dynamic mechanical analysis curves of POM and TPV blends are shown in Figure 5. From image (a), it can be seen that the E′ value of the pure POM is the highest in the test range of –100°C to 100°C. This is because POM is a highly crystalline polymer, which has excellent mechanical properties, such as high strength and good rigidity. For the TPV blends, as shown in the storage modulus curves of 1# to 5# samples, the E′ value of which is decreased with POM content decreasing accordingly.
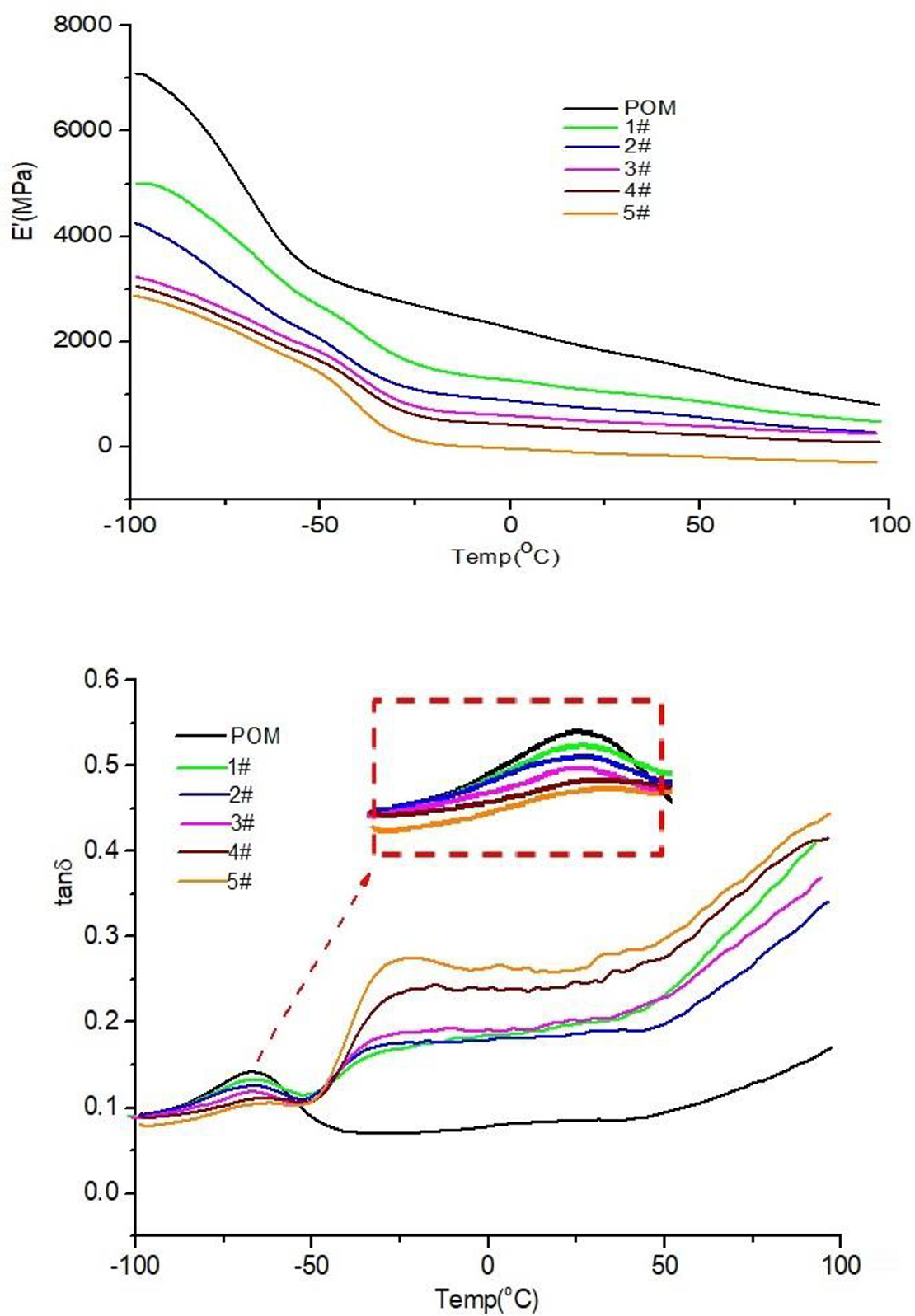
E′ and Tanδ curves of POM/MVQ/TPU ternary TPVs with different compositions.
From the curves of pure POM and TPV blends in image (b) it can be seen that pure POM has only one glass transition temperature Tg, which is about −67.5°C. But for samples 1# to 5#, each has two Tgs, one is equivalent to the Tg of pure polyoxymethylene, the other is about −24.5°C. We can infer that the first Tg belongs to POM, and the second one belongs to vulcanized silicone rubber coated with TPU. Another special feature is that the tanδ of pure POM is higher than that of all TPV samples when the temperature is below −50°C; when the temperature is more than −50°C, the tanδ of all TPV samples are higher than that of POM.
Mechanical properties of TPV blends
The tensile strength and elongation at break of POM/MVQ/TPU ternary TPVs with different compositions are shown in Table 2. One can observe that the tensile strength of pure POM is the largest and the elongation at break is the smallest. Just like the samples from 1# to 5#, as the amount of POM decreases and the total amount of MVQ and TPU increases, the tensile strength of the prepared TPV blends gradually decreases, and the elongation at break increases accordingly. Compared with pure POM, the five POM /MVQ/TPU blends prepared by dynamic vulcanization method in this study can be molded by injection on the one hand, on the other hand have good toughness and high elongation at break.
Mechanical properties of POM/MVQ/TPU ternary TPVs.

Conclusions
A detailed research has been made in this investigation to develop a novel TPV based on POM and MVQ. For the preparation of TPV blends, peroxide BIBP is used as the curing agent in order to make MVQ partially cross-linked; the function of TPU WHT1570 is to coat the vulcanized cross-linked MVQ and improve the compatibility of MVQ and POM. It can be clearly found from SEM images that when the total content of MVQ and TPU is more than 50phr, the vulcanized MVQ phase coated with TPU presents a microcosmic cocontinuous structure. DMA curves of five samples (from 1# to 5#) show that the POM/MVQ/TPU ternary composites prepared by dynamic vulcanization method have two glass transition temperatures. Finally, it can be found from the mechanical test results that with the amount of POM decreasing and the total amount of MVQ and TPU increasing, the tensile strength of the TPV blends gradually is decreased, and the elongation at break is increased accordingly from 35.2 ± 6% of pure POM to 142.8 ± 11% of sample 5#.
