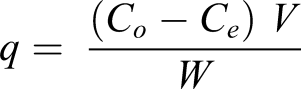Abstract
The objective of this study is to prepare a super absorbent hydrogel based on polyacrylic acid and an environmentally friendly material such as shellac to remove malachite green dye from aqueous solution using gamma radiation. The adsorption of malachite green (MG) dyes using polyacrylic acid (PAA)/shellac (SH) hydrogels was studied. These hydrogels were prepared by mixing aqueous solutions of PAA and SH at differing molecular ratios (10%, 20%, and 30% SH content in the final reaction mixture) and varying doses of gamma radiation (10–50 kGy). The water absorption by the PAA/SH hydrogels prepared at a 30-kGy radiation dose increased with an increase in the SH content. The removal of the MG dyes from an aqueous solution using PAA and PAA/SH hydrogels was investigated at different values for selected parameters, such as pH, contact time, SH concentration, and adsorbent dosage. The highest MG adsorption percentage (95.5%) was obtained for the PAA/SH hydrogel with the highest SH content (30%). As the adsorbent dosage increased, the dye adsorption capacity increased, accordingly. The combination of SH and gamma radiation caused the PAA to undergo structural changes verified by X-ray diffraction and Fourier transform infrared spectroscopy. Scanning electron microscopy was employed to investigate the morphology of the PAA/SH hydrogel and revealed that the SH belonging to the PAA matrixes exhibited a homogeneous dispersion. The thermal stability of the hydrogels was investigated by thermogravimetric analysis. The amount of dye adsorbed by the PAA/SH hydrogel was calculated by subtraction based on the ultraviolet–visible (UV–VIS) spectroscopy determination of the concentration of leftover dye in solution using a UV–VIS spectrophotometer.
Introduction
Superabsorbent polymers (SAPs) are a class of hydrophilic three-dimensional functional polymer network systems that can adsorb large amounts of water. Certain SAPs exhibit good water-holding capacity even under high pressures or high temperatures. 1 Typically, ionic functional groups along crosslinked polymer chains encourage water diffusion within the network, without allowing the polymer to dissolve in water. 2 Superabsorbent polymers are widely used in many applications, such as drug delivery, hygiene, food, cosmetics, and agriculture.3-5 Polymer irradiation leads to various modifications in both structural and optical behavior. Molecular chain separation, intermolecular linkage, and formation of C = C are some of the structural changes that can occur in a radioactive polymer, all of which modify the physical properties of the material.6,7 The use of ionizing radiation is an extremely convenient method for preparing SAPs. The synthetic procedure for the radiotherapy method does not require an initiator, stimulator, or crosslinker because the ionizing radiation is highly active. 8
Radiation treatment has many other advantages, such as the ease of process control; the technology is environmentally friendly as it does not produce any environmental residues or pollutants.9,10 SAPs contain carboxylic acids, partially neutralized carboxyl groups, carboxylate salts, and carboxamide groups. Polyacrylic acid (PAA) is characterized by the presence of an ionizable group on each repeat unit (–COOH). Moreover, PAA chains are crosslinked. SAPs are prepared from been applied acrylic acid and a crosslinker by suspension or solution polymerization. By controlling the type and amount of the crosslinker, the stretch ability and swelling capacity can be controlled.
Shellac (SH) is a natural thermosetting resin of animal origin secreted by Lac insects (Kerria lacca and Laccifer lacca), which grow mostly on host trees in China, India, Myanmar, and Thailand.11–14 SH comprises an intimate mixture of several polar and nonpolar components; a resinous material is always associated with an odoriferous compound, waxy component, and mixture of dyes, such as erythrolaccin and desoxyerythrolaccin, which are hydroxyanthraquinone derivatives
15
(Scheme 1). SH is soluble in solvents containing alcoholic hydroxyl groups and in an aqueous alkaline solution or acetone in the presence of a polar solvent; however, it is insoluble in water, esters, ether, hydrocarbons, and chlorinated solvents.
16
For centuries, SH has been widely used in the art sector mainly to varnish and protect the surface of wooden artworks, in the field of wooden furniture restoration and musical instruments, owing to its appealing advantages. These advantages include good film-forming, ease of application, high adhesion to the wood surface, aesthetical excellence of the coatings, protective properties, and nontoxicity.
17
Recently, SH has in different fields (the pharmaceutical, food treatment, children’s toys, and electrical insulation industries) because of its peculiar characteristics, such as thermoplasticity, insulation, and low toxicity.
18
SH is one of the most elegant finishes for furniture, and it is currently used for restoring and refinishing wooden antiques. In particular, it is one of the most used varnishes for string musical instruments.
19
Another application of Shellac is its use as a matrix material in water treatment
20
as well as an antibacterial.
21
Structure of (a) shellac (R=CHO, R’ =CH2OH) (b) malachite green (malachite green) (c) Polyacrylic acid.
The presence of dyes in wastewaters due to the activity of textile industries, industrial effluents, and water supplies can cause environmental damage and health problems, considering the known resistance of these products to natural degradation and their allergenicity and carcinogenicity. 22 The removal of various dyes to reduce sewage pollution caused by industrial activities is an important area of basic and applied research. Malachite green (MG), a chemical from the triphenylmethane family, is among the most cationic dyes soluble in water. 23 It is used in the manufacturing and processing of wool, textiles, paper, leather, cotton, acrylic fibers, silk, and jute. 24 MG exhibits properties that render its removal from aqueous solutions difficult. In addition, it is toxic to major microorganisms and highly cytotoxic to mammalian cells. Incidences of lung, breast, and ovary tumors have been reported in rats exposed to MG. 25 Various treatment processes are known to separate dyes from wastewater, including biological treatment, oxidation, coagulation, and membrane filtration. 26 Adsorption is one of the best dye removal techniques because of its simple design, low cost, high efficiency, simplicity of the recovery step, and possibility of recycling the adsorbents. 27
Here, a novel type of superabsorbent hydrogel comprising a blend of two polymers, PAA and SH at different ratios, was prepared using gamma radiation. Notably, hydrogels of this type were synthesized for the adsorption removal of MG dye from aqueous media. The various PAA/SH blends obtained were characterized by Fourier transform infrared (FTIR) spectroscopy, X-ray diffraction (XRD), and thermogravimetric (TG) analysis. The adsorption capacity of the MG dye was determined by ultraviolet–visible (UV–VIS) spectroscopy. The effects of pH and contact time on dye adsorption were investigated.
Experimental 2.1. Materials
Polyacrylic acid (molecular weight: 100,000) was purchased from Wako Pure Chemical Industries, Ltd (Osaka, Japan). SH was supplied by Lac Research Station, Bangladesh Agricultural Research Institute (Chapai Nawabganj, Bangladesh). Sodium hydroxide, ammonium hydroxide, and glacial acetic acid were supplied by El-Nasr Pharmaceutical Chemicals Co., (Egypt). MG dye with an absorbance wavelength of ∼617 nm was supplied by Bayer (Germany). The molecular structure of the dye is depicted in Scheme 1. All the chemicals were of analytical grade, and all the solutions were prepared using distilled water.
Preparation of the superabsorbent PAA/SH blend
The PAA was mixed with distilled water in a beaker (250 mL) using an electric stirrer for 1 h at room temperature until a paste-like slurry was obtained. Thereafter, SH was added in various concentrations (10–30%) to the paste-like slurry. The SH solution was obtained by dissolving it at a pH of 8 with ammonium hydroxide. The obtained mixture of PAA and SH was poured into glass test tubes, which were sealed and irradiated at room temperature in atmospheric air with gamma rays emitted by a Co-60 source (radiation dose, 10–50 kGy; dose rate, 1.1 kGy/h). The product of PAA/SH mixtures was washed with ethanol and distilled water for several times to remove any impurities. The PAA/SH SAP units obtained, which exhibited a cylindrical shape, were cut into small pieces, air-dried, and dried in a vacuum oven until a constant weight was attained. The processed SAPs underwent measurements aimed at determining the gel fraction and swelling properties of the polymer.
Characterization of PAA and PAA/SH blends
Water absorbency measurements
A weighed quantity of a particular SAP sample was immersed in distilled water at room temperature until the swelling equilibrium was attained. The swollen SAP samples were separated by screening, and they were weighed after draining on a sieve for 15 min. This procedure was performed twice for each sample to determine its average weight. The swelling ratio was calculated using the following equation
Determination of the gel fraction
The SAP samples dried to a constant weight were immersed in distilled water for 24 h to remove the soluble fraction. They were subsequently removed from the distilled water, air-dried, and further dried to a constant weight in an oven. This procedure was performed three times for each sample, and the average weight of the extracted sample was measured. The gel fraction was calculated using the following equation
FTIR measurements
The PAA and PAA/SH blends were characterized by performing FTIR measurements within a wide wavenumber range (400–4000 cm−1) using an FTIR spectrometer (ATI Mattson-Genesis series, England). The spectra were recorded at a resolution of 4 cm−1 and averaged over 25 scans.
XRD measurements
X-ray diffraction measurements were conducted at room temperature using a Philips (PW 1390) instrument with Nickel-filtered Cu-Kα radiation.
Scanning electron microscopy (SEM)
Scanning electron microscopy experiments were performed using a JEOL 6060LV variable pressure scanning electron microscope (JEOL UK Ltd.,). Specifically, before the samples were inserted in the microscope chamber, they were cut in half through the coated areas, mounted onto carbon disks, and coated in gold for 120 s using a Leica EM SCD005. Images were recorded at ×40, ×250, ×370, or ×650 magnification.
Thermal behavior
The thermal behavior of the samples was investigated by TG analysis using a TG-50 instrument from Shimadzu (Japan). In detail, the samples were encapsulated in platinum pans and heated from room temperature to 600°C at a constant heating rate of 10 °C/min, according to ASTM E1641-07. The measurements were conducted in an atmosphere of nitrogen gas flowing at a rate of 20 mL/min.
Batch adsorption studies and quantification of the adsorbed dye
Batch adsorption experiments were conducted to investigate the influence of the contact time and pH on the MG adsorption onto the PAA/SH hydrogel utilizing MG solutions of initial concentrations of 50 mg/L. Aliquots (4.0 g each) of the adsorbent were added to 75 mL of the 50 mg/L MG dye solutions. The temperature was maintained at 27°C. The series of conical flasks containing the various mixtures were shaken on a rotary shaker at a constant speed of 100 r/min. Samples were drawn for spectrophotometric analysis at predetermined contact times (15–1440 min). The experiments were conducted at different solution pH levels (3–9). Notably, the amount of dye adsorbed by the PAA/SH hydrogel was calculated by subtraction based on the UV–VIS spectroscopy determination of the concentration of leftover dye in solution using a UV–VIS spectrophotometer (Unicom UV2 series).
The relationship between different known concentrations of MG dye and the corresponding light absorption was investigated, and the data collected enabled the drawing of a standard curve, which was used as a reference to determine the unknown dye concentrations in the analyte samples.
28
Dry weighed PAA/SH blends were impregnated in dye solutions contained in flasks with shaking. The amount of adsorbed dye, q (mg/g), was determined using the following equation
The percentage removal of the dye from the solution was calculated using the following equation
Results and discussion
Gel content
The gel content of the PAA/SH polymer blend is indicative of the degree of crosslinking in the hydrogel. Notably, a high degree of crosslinking will render the hydrogel structure rigid and reduce its ability to absorb water.
30
Figure 1 shows data reflecting how the gel fraction in the PAA and PAA/SH blends varied with radiation dose (10, 20, 30, 40, and 50 kGy) and SH content (10%, 20%, and 30%). Importantly, the gel fraction in the PAA and PAA/SH hydrogels increased with radiation dose and SH content. A high SH content (30%) in pure PAA resulted in a high crosslinking density of PAA at 30 kGy under conditions where crosslinking by gamma-ray radiation successfully transformed the linear polymer into a three-dimensional lattice, thereby reducing the water solubility of the hydrogel.
31
The data shown in Figure 1 indicated that the gel fractions of the PAA/SH hydrogels exhibited higher values than those of the PAA hydrogels. Considering that the PAA/SH hydrogel comprises hydrogen bonds that form between the PAA and SH backbones, the gamma radiation produced intermolecular bonds in the hydrogel. Alternatively, the gel content (%) increased with radiation dose, until a maximum stable level (97.7%) was attained for PAA/30%SH (the hydrogel with a 30-kGy gamma radiation dose). No significant change in gel content (%) was observed at gamma radiation doses above 30 kGy. Moreover, by increasing the radiation dose and SH content, the gel content increased to a maximum at 30 kGy and 30% SH. Thereafter, any increase in the gamma radiation dose resulted in stable values for the gel level (%). Effect of the irradiation dose on the gel content of irradiated polyacrylic acid and polyacrylic acid/shellac.
Effect of gamma radiation dose on water absorbency
An important trait for evaluating the properties of hydrogels is their ability to absorb water without their component polymers dissolving as a result. Figure 2 shows data reflecting the effect of the gamma radiation dose on the water absorbency of the PAA and PAA/SH hydrogel of different compositions (SH: 10%, 20%, and 30%). As shown, the hydrogel swelling increased with a radiation dose of up to a maximum of 30 kGy. Afterward, it decreased until it practically attained the same absorbency at 50 kGy. At low radiation doses (below 30 kGy), the decrease in water absorption may be due to a possible deterioration in crosslinking density. This trend correlated with the results of the experiments conducted to determine the gel content of the samples. Furthermore, at high gamma-ray doses (above 30 kGy), the density of crosslinking in PAA/SH hydrogel is high, which can lead to the formation of narrow pores in the hydrogels and reduce the space for water molecules to enter the polymer networks, resulting in low water absorption. Fig. 2. Effect of irradiation dose on water absorbency of polyacrylic acid and polyacrylic acid/(10,20, and 30%)shellac.
Conversely, when the radiation dose was increased, the water absorbency decreased probably because of the formation of excessive three-dimensional crosslinked structures in the PAA and PAA/SH hydrogel. In addition to the increased swelling associated with an increase in the SH content, full swelling was obtained at 30 kGy for PAA/30%SH. In addition to the hydroxyl groups of the SH macromolecules in the presence of gamma rays, carboxylic groups of PAA were expected and formed a complex. This might be caused by the enhancement and increase in the swelling of PAA/(10%, 20%, and 30%)SH at 30 kGy. This mechanism is depicted in Scheme 2. The maximum condition occurred because of the hydrophilic group in the hydrogel, which was involved in a hydrogen bonding interaction with water, or because the hydrophilic group in the hydrogel attained equilibrium. Mechanism of combination of polyacrylic acid and shellac by gamma radiation.
It may be concluded that the addition of SH to PAA in different proportions (10%, 20%, and 30%) and subjecting the two-polymer reaction mixture to a gamma radiation dose up to 30 kGy resulted in a greater hydrogel swelling than that for the pure PAA hydrogel. This phenomenon can be attributed to the increased porosity of the PAA/SH hydrogel, which led to an increase in the ease of loosening of the polymer and a consequent increase in water absorption. The water absorbency of a polymer is indicative of the extent of the crosslinking within it. Notably, as the crosslinking density in the polymer increased past the level attained when a dose of 30 kGy was applied, the water absorbency of the hydrogel decreased because the pore space in the crosslinked network for water to enter began to decrease.
FTIR spectroscopy
Fourier transform infrared spectroscopy is an important tool for tracking and indexing the homogeneity and compatibility between PAA and SH under gamma radiation. Figure 3 shows the FTIR spectra of PAA, SH, and their blends obtained at the 30-kGy dose. As shown, the PAA spectrum was characterized by a unique absorption peak in the 2850–3300 cm−1 wavenumber range, which was assigned to the carboxylic group (–COOH). An intense peak was observed at 1705 cm−1, which was attributed to the carbonyl group, and characteristic peaks at 2897 and 2968 cm−1 were observed, which corresponded to the aliphatic C–H group. Oppositely, the SH spectrum included a characteristic absorption peak in the 2720–3210 cm−1 wavenumber range, which was assigned to the carboxylic group; extremely intense absorption peaks at 2903 and 2986 cm−1, which corresponded to the aliphatic C–H group; a weak absorption peak at 1605 cm−1, which was attributed to the cyclic aliphatic unsaturated carbon (C = C); and a strong intense absorption peak at 1716 cm−1, which was due to the carbonyl group. After adding SH to PAA and subjecting different PAA/SH hydrogel to a 30-kGy radiation dose, the intensities of the absorption peaks due to the aliphatic C–H group at 2897 and 2968 cm−1 were observed to increase with an increase in the SH content (Figure 1). In addition, the intensity of the peak due to the unsaturated cyclic aliphatic carbon (C = C) was observed to increase with an increase in the SH content in the PAA/SH hydrogel (90%PAA/10%SH, 80%PAA/20%SH, and 70%PAA/30%SH). These observations confirmed the successful compatibility and homogeneity of PAA and SH at different concentrations, resulting in a higher molecular weight of the resulting polymer. Fourier transform infrared of PAA, SH, and PAA/SH at different concentrations under 0 and 30 kGy irradiation dose. PAA: polyacrylic acid; SH: shellac.
X-ray diffraction (XRD)
XRD is an effective technique for revealing amorphous materials and the microstructure of crystalline materials. As shown in the XRD patterns of the PAA, PAA hydrogel, SH, and PAA/10%SH, PAA/20%SH, and PAA/30%SH hydrogels in Figure 4, a sharp characterized peak was observed at 2θ = 20.8°, indicating the highly crystalline nature of pure SH. The broadness of the XRD pattern of the PAA powders indicated the amorphous nature of these samples, while the XRD pattern of the PAA hydrogel exhibited the same broad peak with low intensity, indicating the amorphous state of the PAA gel, which is the result of crosslinking. Broad bands observed at 2θ = 21.5, which were typical of amorphous phases, were present in the XRD patterns of the PAA/10%SH, PAA/20%SH, and PAA/30%SH hydrogels, and they were ascribed to the crosslinking between the PAA and SH polymers. A decrease in the intensity of the characteristic peak of the blend hydrogels was evident because of the introduction of the SH polymer, which had a low crystal lattice. Oppositely, owing to the increase in the ratio of SH in the PAA/SH hydrogel, a slight increase in the crystalline phase content was observed owing to the complex structure of the SH with a high crystal lattice. X-ray diffraction scans of 100%PAA, (90%PAA/10%SH), (80%PAA/20%SH), and (70%PAA/30%SH) hydrogels. PAA: polyacrylic acid; SH: shellac.
Thermogravimetric analysis
Figure 5 shows the thermograms and corresponding thermal decomposition rates for the PAA and PAA/SH hydrogel characterized by different compositions. The temperature parameters are listed in Table 1. The appearance of the TG curves indicated that the thermal decomposition of the samples occurred in three or four stages. The first decomposition occurred between 200°C and 280°C for all samples, indicating that it comprised the decomposition of additives and raw materials. The second and third degradations occurred between 280°C and 355°C for PAA and 280°C and 520°C for the various PAA/SH hydrogel; it comprised the thermal decomposition of the molecular chain and crosslinking structure of these polymers. Finally, a fourth degradation occurred between 520°C and 650°C and was attributed to the oxidation of residues formed as a result of the thermal decomposition of the crosslinked polymer. In general, the TG analysis results demonstrated that thermal stability increased for the PAA/SH hydrogel at different concentrations, accompanied by a decrease in the rate of thermal decomposition, compared with that of the blank sample (PAA). This observation was attributed to the presence of strong intermolecular bonding interactions between the crosslinked PAA and SH in the PAA/SH hydrogel prepared with a gamma radiation dose under 30 kGy. As listed in Table 1, the thermal stability of the PAA/SH hydrogel, as reflected by the value of Tmax, was superior to that of PAA, and it increased with an increase in the SH content. Thermogravimetric analysis thermograms and the rate of thermal decomposition for PAA and PAA/SH at different concentrations. PAA: polyacrylic acid; SH: shellac. The Temperature parameters of polyacrylic acid(PAA) and PAA/shellac blend at different concentrations. PAA: polyacrylic acid; SH: shellac.

Dye adsorption by PAA and PAA/SH hydrogels
The primary objective of this study was to develop novel biodegradable hydrogels that can be employed as adsorbents capable of removing dyes from aqueous solutions. The adsorption of dye molecules by PAA and PAA/SH hydrogel of different compositions, which possess a porous structure (resulting in a high surface area), occurs by physical adsorption, where the dye molecules accumulate on the surface of blend polymers through intermolecular penetration. The attractive interactions between the polymer surfaces and the adsorbed dye molecules are electrostatic and are derived from van der Waals forces. 32 Considering the weakness of these forces, dye adsorption is reversible.
Adsorption selectivity of PAA and PAA/SH hydrogels toward the MG dyes
The adsorption selectivity of PAA and PAA/SH hydrogels of different compositions toward the MG dyes varied according to the nature of the polymer and adsorbed dye. Importantly, the adsorption depended on the nature of the interactions of the adsorbate with the functional groups of the polymers.
The results of the experiments conducted to determine the adsorption selectivity of the PAA and PAA/SH hydrogels toward MG dyes are shown in Figure 6. The different hydrogels exhibited different degrees of swelling (PAA/30%SH > PAA/20%SH > PAA/10%SH > PAA). The hydrogels were covalently crosslinked and comprised–CO−, –OH, –COOH, and –COO− functional groups in different ratios. Notably, the PAA/30%SH hydrogel exhibited maximum swelling. This may be because PAA/30%SH comprised free–COOH functional groups from PAA and–OH,–COOH,–CO, and –COO− functional groups from SH. The hydrophilicity of the blend increased with the number of–OH,–COOH, and–COO− groups. These groups had a higher swelling capacity than that of the other hydrogels.33,34 Strong electrostatic interactions existed between the positively charged MG dye molecules and the functional groups with negative charges, such as–COO−, –COOH, –CO, and –OH. This could be attributed to the effect of the nature of the SH polymer and its high swelling and additional negative sites (–OH, –COOH, –CO, and –COO−). Therefore, as the SH content increased in the polymer blends, the speed at which the dye was adsorbed increased, thereby providing great access to active functional groups of blend polymers. Therefore, PAA/30%SH was observed to exhibit the highest MG dye adsorption capacity because of its high swelling value. Conversely, PAA exhibited the lowest adsorption capacity because of its low swelling value. The adsorption capacity of malachite green dye by PAA and different ratios of (PAA, SH) hydrogels. PAA: polyacrylic acid; SH: shellac.
Mechanism of adsorption of MG dyes by PAA and PAA/SH hydrogels
The adsorption mechanism depends on the nature of the dye and adsorbent. MG is a cationic dye comprising two amine groups, where positive single charges are localized on the nitrogen atoms. Adsorption occurred by physical adsorption in the form of monolayer formation, followed by multilayer formation.
35
The two most probable mechanisms for dye adsorption were evaluated here, as shown in Scheme 3. One comprised the electrostatic interaction between the negatively charged adsorption sites of the polymer (–OH,–CO,–COO−, and–COOH), which demonstrated different hydrophilicities, and the positively charged adsorbent dye. The other mechanism depended on the hydrogen bonding interactions between the–OH or–COOH functional groups on the polymer surface and the–NR2 amine groups in the basic dye. Considering that the number of ionizable PAA groups increased because of the crosslinking process triggered by the introduction of SH, the prepared PAA/SH hydrogels comprised many additional carboxyl groups with respect to PAA. This can increase the intensity of the interaction between the cationic groups of the dye and the anionic groups on the surface of the prepared polymer blends. Therefore, under basic conditions, the adsorption of the MG dye will result from the ionic interaction between the negatively charged groups (–OH ,–CO, –COO−, and –COOH) present on the surfaces of the PAA and PAA/SH hydrogel and the positively charged MG dye molecules. However, the electrostatic interaction between the hydrogel matrix and dye molecules appeared to be the main intermolecular interaction in this method.
36
Schematic representation of the adsorption process and its mechanism.
Effect of pH on dye adsorption
The initial pH of the dye solution is a key parameter that influenced the adsorption of MG (q) onto the PAA and PAA/SH hydrogels. The pH range that we chose to investigate for the MG dye solution was 3–9 because of the high sensitivity of MG and to avoid using a pH range that may lead to fading and loss of color intensity. Therefore, concentrated solutions of HCl and NaOH were diluted with distilled water to achieve the desired acidic or basic pH level.
37
Figure 7 shows data reflecting the effect of the pH on the adsorption of MG onto the PAA and PAA/SH hydrogels and its effect on the MG removal percentage. The maximum and minimum MG adsorptions were observed at pH levels of 5 and 3, respectively. The MG dye became positively charged when dissolved in water. Therefore, the low adsorption of the basic MG dye at a low (acidic) pH was suggested to be due to the presence of excess H+ ions competing with the cationic dye for the interaction with the negatively charged adsorption sites. Under such pH conditions, the carboxylate groups on the surface of PAA and PAA/SH will probably undergo protonation (to produce –COOH), hindering the cationic MG dye from binding to the activated sites of the adsorbent. On one hand, the hydrogen bonding among the –COOH and –OH groups was enhanced, and additional physical cross bonds were produced. On the other hand, the electrostatic repulsion between the –COO− and –OH groups was limited by the effect of screening counter ions, such as Cl−, in the swelling medium; therefore, the graft hydrogel network continued to slightly shrink, becoming partially hydrophobic and reducing swelling values. As the pH of the system increased to a pH of 5, the number of positively charged sites decreased, while the number of negatively charged sites increased. Notably, the presence of negatively charged sites has been suggested to favor the adsorption of the cationic MG dye because of strong electrostatic attractions.
38
At pH > 5, few carboxylate groups on the surface of the adsorbents were ionized; therefore, their hydrogen bonding interactions were broken and the electrostatic repulsion between the –OH and –COO− groups caused an increase in the swelling capacity.
33
At pH > 7, the swelling efficiency decreased in the swelling media because of the charge screening effect afforded by excess Na+ ions in the solution because of the addition of NaOH to tune the pH of the medium. These ions shielded the carboxylate anions and –OH and prevented efficient anion–anion repulsion.
39
However, as the concentration of Na+ ions increased, the cations competed with the cationic dye for interaction with the negatively charged sites of the adsorbents. In this context, the optimal pH was determined to be 5. At this pH, the binding sites of the adsorbent remained unprotonated (as OH− and –COO− groups), and the dye binding was maximized. Therefore, a pH of 5.5 was selected for the adsorption solution of the basic MG dye. Effect of different pH ranges on adsorption capacity and removal percentage of malachite green dye by different ratios of (polyacrylic acid/shellac) hydrogels.
Effect of contact time on dye adsorption
The effect of the contact time on the adsorption capacity (q) and removal percentage of MG by the PAA and PAA/SH hydrogels was investigated. Relevant experiments were conducted at room temperature and pH 5, and the contact time was made to vary between 15 and 1440 min. The relevant results are shown in Figure 8. The results indicated that the amount of adsorbed dye by the PAA and PAA/SH hydrogel significantly increased after the 120-min mark; however, the rate of the increase in MG dye adsorption progressively reduced until the contact time reached the 360-min mark. After this point, the MG dye adsorption tended to level off. An increase in the reaction time increased the chances for the adsorbent hydrogels and MG molecules to interact with each other.
40
The adsorption capacity was observed to rapidly increase in the initial phase and decrease or remain unchanged in the subsequent stage. This general trend was due to the great number of linking sites ready for adsorption during the initial step of the reaction. Subsequently, the binding sites progressively approached saturation, when the adsorption equilibrium was attained.
41
The rapid adsorption rate was mainly attributed to the electrostatic interaction between the oppositely charged adsorbate and adsorbent species, in addition to their π–π interaction.
42
The time required for the MG dye to attain adsorption equilibrium with the adsorbent was determined to be 360 min. Effect of contact time on the adsorption capacity and removal percentage by different ratios of (polyacrylic acid, shellac) hydrogels, at constant pH 5.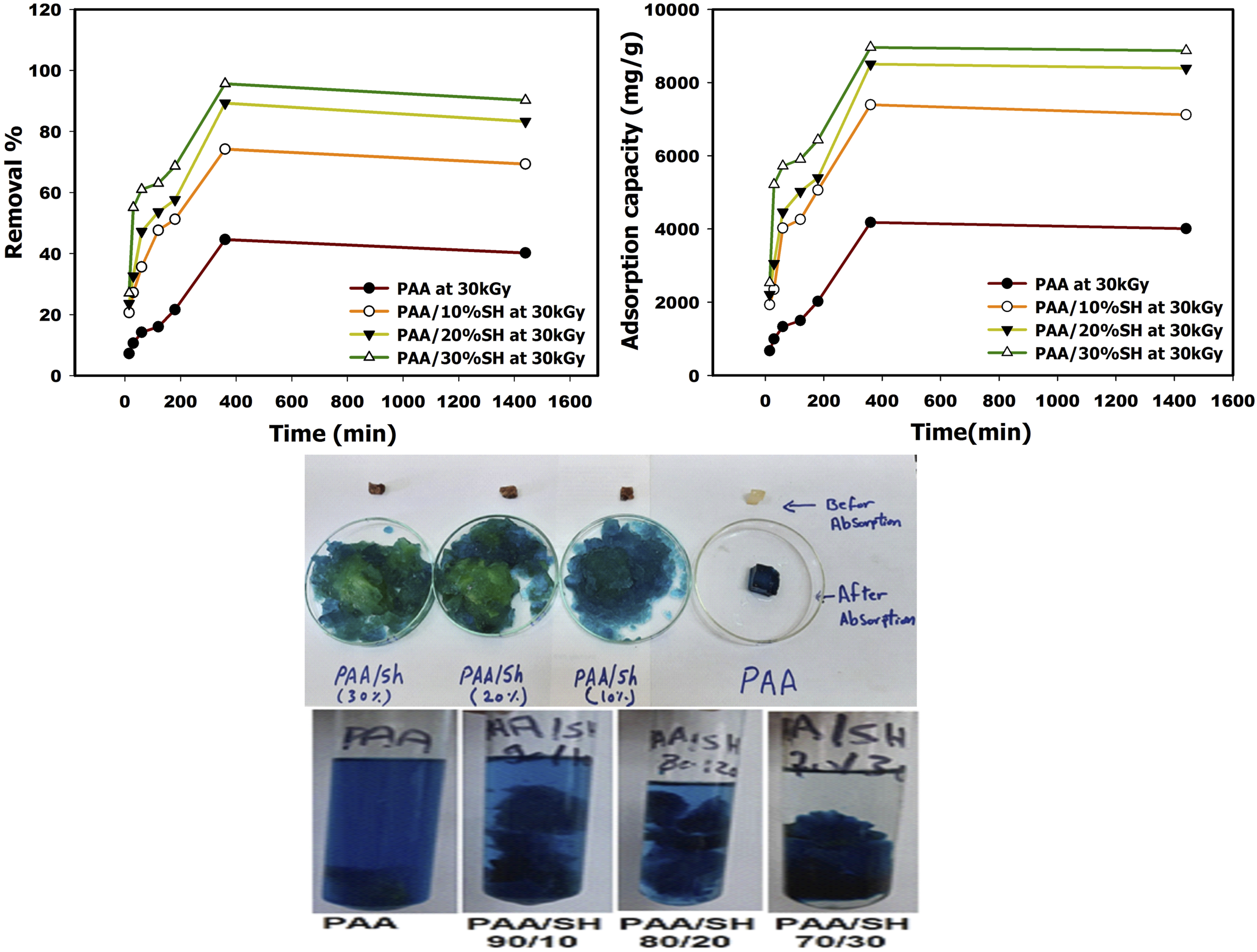
Morphological analysis
The morphology of the as-prepared PAA/SH hydrogel was investigated by SEM. The results of the SEM analysis indicated the state of the SH blend in the PAA matrix, which had a significantly visible homogenous phase, confirming the compatibility between the PAA and SH polymers. Figures 9(a)–(d) show the cross-section morphology of the hydrogels with PAA and varying SH contents. The purpose of the test was to investigate the effect of SH loading on the pore size of the PAA hydrogels. The cavity and morphological construction of the PAA/SH hydrogels irradiated at 30 kGy were inspected. The cavity of polymeric materials mainly depends on factors that influence the absorption and morphological properties of hydrogels and sorely affect morphology, which are the radiation dose and copolymer fabrication.
43
As shown in the micrographs, all the hydrogels exhibited extremely porous and sponge-like structures. Interestingly, the changes in pore size correlated with the swelling test results, in which the 70%PAA/30%SH hydrogel exhibited the largest pore size, compared with those of the other samples, and relevant results are shown in Figure 5(d). Furthermore, it was observed that the pore diffusion in the 70%PAA/30%SH hydrogel was more regular than that in the other hydrogels, which indicated a more ordered polymer network. Scanning electron microscopy micrographs of cross-section hydrogels at 30 kGy before and after adsorption malachite green dye of (a) PAA, (b) PAA/10% SH, (c) PAA/20% SH, (d) PAA/30% SH, (e) PAA; (f) PAA/10% SH, (g) PAA/20% SH, and (h) PAA/30% SH. PAA: polyacrylic acid; SH: shellac.
The micrographs shown in Figures 9(e)–(h) represent PAA and PAA/SH after the adsorption of the MG dye. The micrographs show the PAA and PAA/SH hydrogel of different compositions (SH: 10%, 20%, and 30%) obtained at a 30-kGy gamma radiation dose. The images show that a highly MG dye-adsorbing porous hydrogel was produced because the SH blended with the PAA. With this structure, a high amount of MG dye can be retained in the hydrogel structure. The higher the adsorption of the MG dye, the higher the disappearance of the hydrogel pores, and the surface was coated by the MG dye.
Conclusion
The results obtained in this study indicated that the prepared PAA/SH hydrogels can be used as superabsorbent materials for the removal of MG dye from aqueous solutions. Among the hydrogels prepared, the highest MG dye adsorption capacity was exhibited by the hydrogel with the highest SH content (PAA/30%SH), which was prepared using a 30-kGy gamma radiation dose. Regarding the other investigated parameters, the highest dye adsorption was observed at a pH of 5, and the adsorption was observed to increase with the contact time up to the 6-h mark. In sum, the prepared PAA/30%SH hydrogel is a hydrophilic functional polymeric network system that can be used as a good novel superabsorbent material for waste and industrial water treatment.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.