Abstract
In this study, alkali lignin modified by ethylene glycol and lignin nanoparticles was prepared through acid precipitation technology. Lignin nanoparticle-g-polyacrylic acid adsorbent was prepared using copolymerization reactions between lignin nanoparticle and polyacrylic acid in the presence of potassium persulfate as the radical initiator. Then, lignin nanoparticle-g-polyacrylic acid adsorbent was used to remove Safranin-O from an aqueous environment. The adsorbent structures and morphologies of lignin nanoparticle and lignin nanoparticle-g-polyacrylic acid adsorbent were investigated using scanning electron microscopy, Fourier transform infrared spectroscopy, and thermogravimetric analysis. Nanoparticle sizes were assessed using dynamic light scattering. Equilibrium isotherms were compared using the Langmuir, Freundlich, and Temkin adsorption models. Both adsorbent lignin nanoparticle and lignin nanoparticle-g-polyacrylic acid adsorbent showed good agreement with the Langmuir isotherm. The maximum adsorption capacities of lignin nanoparticle and lignin nanoparticle-g-polyacrylic acid adsorbent reached 99 and 138.88 mg g−1 using model-like, pseudo-second-order, and intraparticle diffusion, respectively. Experimental results showed that adsorbent lignin nanoparticle and lignin nanoparticle-g-polyacrylic acid adsorbent followed the pseudo-second order kinetic model.
Introduction
An increase in color reproduction using dyes and their numerous applications worldwide have led to wastewater production, resulting in dangerous pollution (Kaur and Singh, 2007; Nadi et al., 2012). Most substances in dye wastewater are toxic and carcinogenic (Entezari, 2007; Gong et al., 2005). Therefore, limitations and strict rules have been imposed by the government to remove wastewater containing organic compounds before discharge.
Basic Red 2 dye is highly soluble in water and is nonvolatile. This compound is red in color and is a cationic dye of the imine group. Safranin-O is dangerous and toxic (Korbahti and Rauf, 2008; Vinod et al., 2006), and it is used in painting, fashion, research, and paper industries (Malekbala et al., 2012; Marahel et al., 2015). Nevertheless, different methods, including physicochemical, biological, and chemical methods, can be used to remove cationic dyes (Gomez et al., 2007; Zhu et al., 2007). Unfortunately, many of these methods are costly and difficult to control under normal conditions. However, adsorption techniques using natural materials are highly regarded as extremely effective (Amin, 2008; Río et al., 2011).
Adsorption processes comprise physical activities that feature advantages compared with other waste technologies; they are low cost, accessible, and can treat dyes in high concentrations. A variety of natural and synthetic absorbents are used to bleach dyes; natural adsorbents are environmentally friendly, inexpensive, exhibit a high adsorption capacity, lack toxicity, and cause a few problems in the final desorption phase (Badii et al., 2010; Batzias and Sidiras, 2007; Bulut et al., 2008; Crini and Badot, 2008; Haji Aghaei1 et al., 2015; Mall et al., 2005; Mohan et al., 2002; Nigam et al., 2000).
After cellulose, lignin is the second most abundant natural polymer; it consists of phenylpropane structural units with carbon–carbon and ether connections. Lignin is an amorphous and a super complex structure that still requires full investigation (Camilla et al., 2017; Daliang et al., 2017; Sarocha et al., 2017; Sixiao and You-Lo, 2015). Lignin constitutes 15–36% of the weight of wood; thus, lignin sources are abundant and suitable for use in the wood and paper industry (Göran, 2015). Lignin contains aromatic and aliphatic parts and various functional groups, including phenolic, hydroxyl, carboxyl, benzyl alcohol, methoxy, and aldehyde groups (Daliang et al., 2017; Guo et al., 2008; Jun-Seok et al., 2015). Recently, alkali lignin (AL), cellulose, and hemicellulose have been proven to exhibit characteristics that are suitable for adsorption and ion exchange. However, the adsorption capacity of lignin macromolecules is relatively low. Therefore, chemical modification of lignin from natural materials is essential.
Some researchers have attempted to increase adsorption capacity and efficiency of lignin by adding polymers containing specific functional groups, such as polyacrylic acid, polyacrylamide, and polyacrylonitrile, to its structure (Fang et al., 2010; Ibrahim et al., 2010). Kishor (2012) measured Acid Blue 9 dye adsorption using a chitosan-g-polyamide amine dendrimer copolymer (Kishor et al., 2012). Genlin et al. (2014) investigated the removal of methyl orange, disperse blue, and malachite green chloride dyes using a cellulose-g-polyacrylic acid copolymer in 2013 and observed that the adsorption capacities of these dyes in optimal conditions were as follows: 1111, 1096, and 555 mg g−1, respectively. Li et al. (2011) also studied the removal of methylene blue dye using chitosan–polyacrylic acid–attapulgite composite in 2011; its maximum adsorption capacity on the Langmuir isotherm model totals 1870 mg g−1.
This research aimed to prepare lignin nanoparticle-g-polyacrylic acid adsorbent (LN-g-PAA) for the removal of Safranin-O. After the preparation of LN through acid precipitation technology, PAA chains containing abundant carboxylic acid groups were connected to LN. Considering that the grafting percent in the synthesis of LN-g-PAA nanocomposites is an effective agent during dye adsorption. Thus, optimal conditions for PAA amount, reaction temperature, reaction time, and initiator amount were investigated to graft lignin. pH, adsorbent dosage, dye concentration, and contact time were studied as factors in the adsorption process. Pseudo-first order, pseudo-second order, and intraparticle diffusion were also investigated in the Langmuir, Freundlich, and Temkin models to evaluate the adsorption processes.
Materials and methods
Materials
AL was prepared by acidification of a black liquor obtained from the Pars Khuzestan paper factory in Iran. Safranin-O dye (99.5%), sodium hydroxide (99%), hydrochloric acid (36 wt%), ethylene glycol (98%), potassium persulfate (K2S2O8), and acrylic acid (AA) were purchased from Merck in Germany. Ethanol (99.9%) was prepared by Bidestan in Iran.
Devices
Fourier transform infrared spectroscopy (FT-IR) was used to determine the chemical structures of the compounds in the range of 400–4000 cm−1 using a Tensor 27 FT-IR spectrophotometer (Brucker, Germany). Safranin-O dye was used to determine the amount of absorbed dye. To determine the amount of ultraviolet–visible adsorption, a Model M350 Double Beam Spectrophotometer (UK) was used. pH was determined using a Model Metrohom 827 pH meter. To study the structural changes in the synthesis of compounds and heat resistance up to 500°C, a thermogravimetric analysis–differential thermogravimetric (TGA–DTG) device (Model Perkin Elemer, UK) was used. A Zetasizer PSS0012-22 (Malvrn, USA) was used for dynamic light scattering (DLS) analysis, and scanning electron microscopy (SEM) device (Model LEO 1455VP, UK) was used to determine the particle size and surface morphology of the absorbents. Gel permeation chromatography was performed with a Shimadzu 6-A (USA) to determine the molecular weight of AL.
Preparation of AL
To precipitate AL, the acidic black liquor from a pulp-and-paper factory in Khuzestan was placed in 0.1 M hydrochloric acid at a pH range of 2.5–3. The impure lignin precipitate sediment was centrifuged and isolated. The sediment was washed with distilled water until the filter effluent reached a pH of 7. For purification, lignin was dissolved in relatively warm ethanol (50–60°C). Ethanol-soluble fractions were separated by filtration, and the solid residue was discarded. The bulk of ethanol (75%) in the evaporator was evaporated at low pressure. Distilled water was poured on the remaining solution and the sediment of lignin in colloidal form. After centrifugation for 10 min at a speed of 4500 r min−1 at room temperature, a considerable amount of purified lignin was obtained (Mirshokraie, 2011).
Preparation of LN
AL (0.28 g) was dissolved in 50 ml of polyethylene glycol (0.56 wt%) and was stirred for 2 h at 40°C. Then, the mixture was filtered with 0.4 µm filters for purification. Next, 0.25 M hydrochloric acid was slowly dropped in 45 ml of the filtered solution at pH 4. LNs were formed in this solution. After centrifugation, the nanoparticles were washed with distilled water and dried under a vacuum at a temperature of 50°C (Frangville et al., 2012). Frangville et al. (2012) proposed similar mechanisms to produce LN (Frangville et al., 2012).
Preparation of LN-g-PAA nanocomposites
First, 0.8 ml of AA monomer and 40 ml of distilled water were poured into a three-neck round-bottom flask equipped with a reflux condenser and were stirred by a magnetic stirrer until complete dissolution. Then, 0.2 g of LN was added; using a sodium hydroxide solution (10 wt%), the solution pH was increased to 11. The reaction temperature was adjusted to 70°C, and the solution was stirred for 30 min. Next, 3 mg of potassium persulfate was added to the contents of the flask as the initiator, and nitrogen gas was flowed through the flask for 15 min to eliminate the damaging effects of oxygen in the reaction. After 5 h, to stop the reaction, reflux was stopped, and the flask was cooled at room temperature. Then, using 0.1 M hydrochloric acid, the pH of the solution was decreased to 2. LN-g-PAA copolymers were precipitated by acidification. The sediment was separated by centrifugation and washed several times with distilled water to remove the monomers and homopolymers of AA. Finally, the sediment was dried at room temperature (Rong et al., 2013; Yufang et al., 2016). Figure 1 presents the mechanisms used for the preparation of LN-g-PAA nanocomposites.

Mechanisms used to prepare the LN-g-PAA Nanocomposites.
Polymerization reactions were evaluated to determine the optimal reaction conditions, which were as follows: 40–90°C temperature after 1–6 h and using 0.2–1.2 ml of AA and 1–5 mg of potassium persulfate.
To prepare the LN-g-PAA copolymers, efficiency and grafting were calculated using equations (1) and (2), respectively (Run et al., 2009)
Safranin-O dye adsorption
A stock solution with a concentration of 200 mg l−1 was prepared and maintained at a temperature of 4°C. All experiments were carried out with 100 ml of the dye at 25°C on a shaker at 70 r min−1. The samples were filtered after each step to measure dye concentration, and the adsorption rate was determined using ultraviolet–visible adsorption spectroscopy at a wavelength of 517 nm. Each sample was tested thrice, and the averages were recorded. The effects of various factors, including pH, contact time, adsorbent dosage, and initial dye concentration, were evaluated in terms of adsorption percent and capacity. The amount of dye adsorbed at equilibrium (qe (mg g−1)) and dye removal efficiency (R%) were determined using equations (3) and (4), respectively (Fanchiang and Tseng, 2009; Madrakian et al., 2012)
Isoelectric range
The point of zero charge (pHpzc) of LN and LN-g-PAA was determined using solid addition (Yufang et al., 2016). The experiment was conducted in a series of 100 ml flasks with glass stopper. Each flask was filled with 20 ml of 0.1 M potassium nitrate solutions of different initial pH values (pH0) and 0.20 g of LN or LN-g-PAA. pH values of the KNO3 solutions were adjusted to 2–10 with 0.1 M sodium hydroxide or diluted hydrochloric acid. The suspensions were then sealed and shaken for 24 h at 100 r min−1 and 24°C. The final pH values (pHf) of the supernatant liquid were noted. The differences (
Results
Determining LN size using DLS
As a function of the percentage of nanoparticles, the particle size distribution of LNs at pH 4 was studied. At pH 4, LN size between 40 and 60 nm, and the average diameter was 52.7 nm. Yuanyuan et al. (2014) estimated the average diameter of the AL nanoparticles produced by nitric acid and pH reductions to reach 278 ± 13 nm (Yuanyuan et al., 2014).
Morphology of AL and LNs
SEM was used to study the morphology and approximate particle size of AL, LN, and LN-g-PAA. Figure 2 shows the morphologies of AL and LN before and after adsorption of Safranin-O. AL macromolecules featured homogeneous and interconnected particles that were relatively uniform and flat. LN exhibited an average diameter of less than 80 nm and highly porous surfaces. By creating a copolymer bond between PAA and LN, the particles became non-nano and intertwined and formed a relatively uniform distribution on the surface. In addition, the dimples on the copolymer surface deepened and the area widened.

SEM images of AL, LN, and LN-g-PAA before and after the adsorption of Safranin-O dye.
FT-IR spectra
Figure 3 displays the FT-IR spectra of AL, LN, LN-g-PAA, and PAA. AL and LN yielded very similar FT-IR spectra. No change was observed in the chemical structure of AL during nanoparticle preparation. The results showed the stability of phenol propane rings in the synthesis of nanoparticles. The weak peak at 1709 cm−1 indicated the good vibration of carbonyl bonds in the carboxylic acid groups. The peak at 1315 cm−1 in the LN showed the stretching vibration in connecting syringyl (S) and guaiacyl (G) and appeared as a result of nanoparticle formation. The stretching vibrations of S and G resulted in longer wavelengths measuring 1377 and 1351 cm−1, respectively. The adsorption peak at 1158 cm−1 resulted from the –C–O vibration in para-hydroxyphenyl (H); this peak was more intense for LN than AL and was attributed to the interaction of –OH phenolic groups in H units. As for AL, the peak at the 3405 cm−1 area was related to the vibration of –OH bonds, which in LN compound was due to the connection with ethylene glycol, moved to 3394 cm−1, and increased in intensity. The increased peak intensity at 1077 cm−1 was attributed to overlapping –C–O vibrations in the connection of carboxylic acid groups with ethylene glycol and –C–H vibrations in connection with alcohol type 2. Shengli et al. (2016) provided similar information about extracting lignin from rice (Shengli et al., 2016).
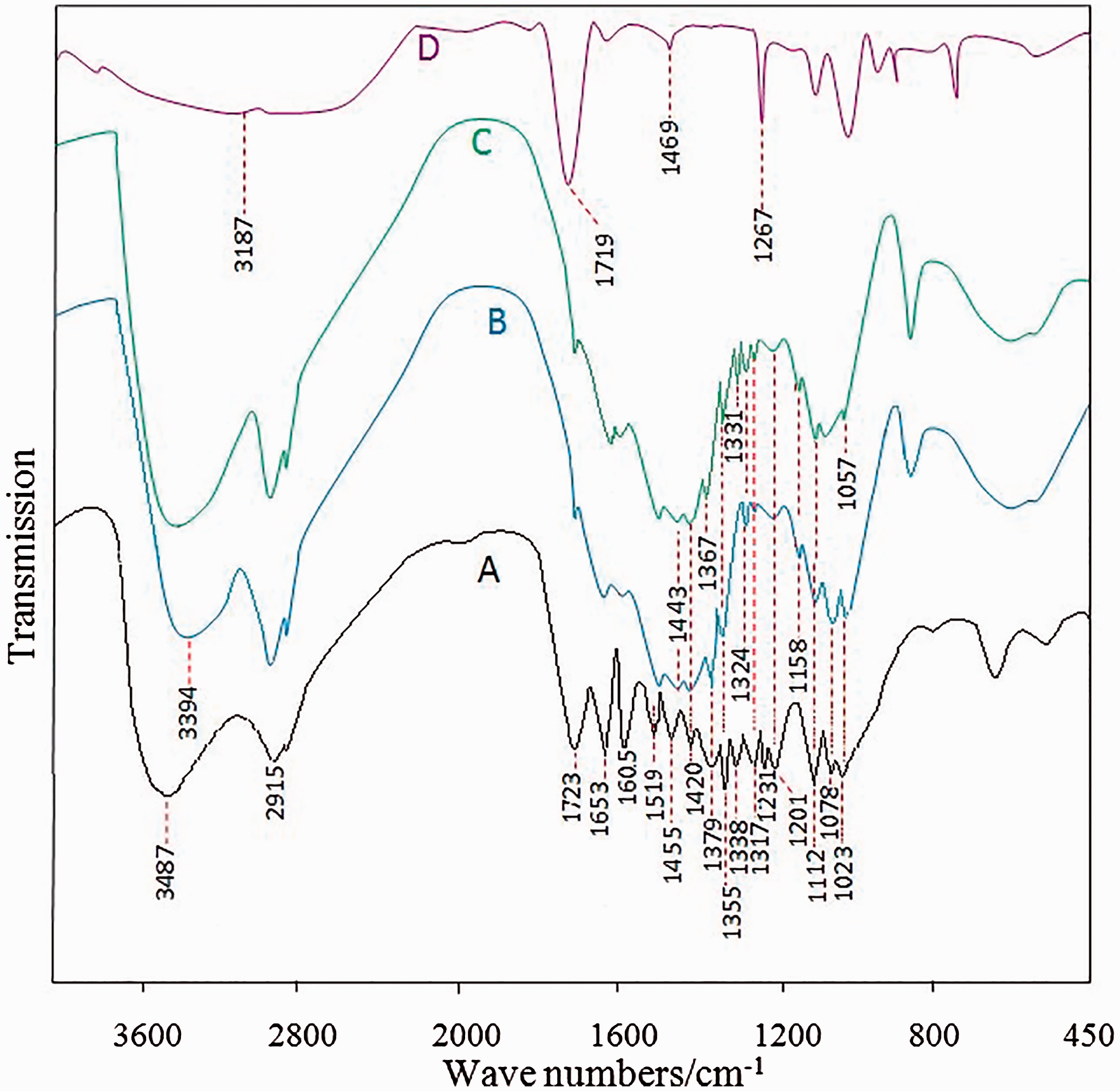
FT-IR spectra of (a) LN-g-PAA, (b) LN, (c) AL, and (d) PAA.
The stretching vibration of O–H in the PAA aliphatic groups transferred from the 3187 cm−1 area to higher wavelengths in the 3487 cm−1 area and overlapped with the stretching vibration of O–H in the phenol groups. The bending vibration of –CH2 in PAA appeared in the 1469 cm−1 area, moved to higher wavelengths at 1455 cm−1 as a result of copolymerization, and overlapped with the bending vibration of CH3. In the LN-g-PAA nanocomposite, owing to copolymerization, the carbonyl group in the carboxylic acid transferred from the 1709 cm−1 area to the 1723 cm−1 area. Further, the stretching vibration of –C–O in the LN-g-PAA carboxylic acid transferred from the 1158 cm−1 area to the 1231 cm−1 area. Thus, copolymer grafting was achieved in the LN-g-PAA nanocomposites.
Thermal stability
Figure 4 shows the TGA and DTG spectra of PAA, LN, and LN-g-PAA. The weight loss of LN occurred in two stages. The first weight loss occurred at temperatures of 38–141°C and equaled to 4.3%, and this result was attributed to the evaporation of water contained in the LN compounds. The second occurred at 141–503°C with weight loss percentages of 47.7%; these findings were attributed to the breakage of S, G, and H polymer units in LN (Shoujuan et al., 2016).

TGA and DTG curves in the PAA (A1 and A2), LN (B1 and B2), and LN-g-PAA (C1 and C2).
However, the weight loss in PAA samples and LN-g-PAA nanocomposites was achieved in three stages. The first weight loss occurred at temperatures of 29–122°C and 28–162°C with weight loss percentages of 1.3 and 12%, respectively; these results were attributed to evaporation of water in the compounds. The second occurred at temperatures of 167–275°C and 162–283°C, respectively, and equaled 12 and 10%, and these results were attributed to the decomposition of carboxylic acids in the PAA samples and LN-g-PAA. The third occurred at temperatures of 327–514°C and 310–524°C, yielding weight loss percentages of 37 and 32%, respectively; these values were attributed to the breakage of the main chain in PAA and S, G, and H polymer units in the LN-g-PAA nanocomposites.
In the LN-g-PAA nanocomposites, increasing the temperature to 310°C resulted in the breakage of carboxylic acid groups in the PAA, preventing heat transfer to the main structure of LN (Shoujuan et al., 2016). The results showed more structural changes in the LN-g-PAA nanocomposites compared with LN due to copolymerization with PAA. The PAA, LN, and LN-g-PAA nanocomposites stabilized at temperatures less than 167, 146, and 162°C, respectively, indicating that heat resistance in LN increased due to copolymerization with PAA. In addition, most thermal decomposition of LN and LN-g-PAA nanocomposites occurred at 361 and 376°C, respectively, further confirming the increased thermal resistance of the copolymers.
Factors affecting the yields and grafting ratio in LN/PAA copolymerization
Effect of AA dosage on copolymerization
Figure 5(a) shows the effect of the amount of AA on grafting and yield in LN/PAA copolymerization. Increasing the amounts of AA from 0.2 to 0.8 ml resulted in increased grafting and yield during copolymerization. With 0.8 ml of AA, grafting and yield reached 167 and 74%, respectively. In summary, grafting and yield showed no significant increase and remained relatively constant.
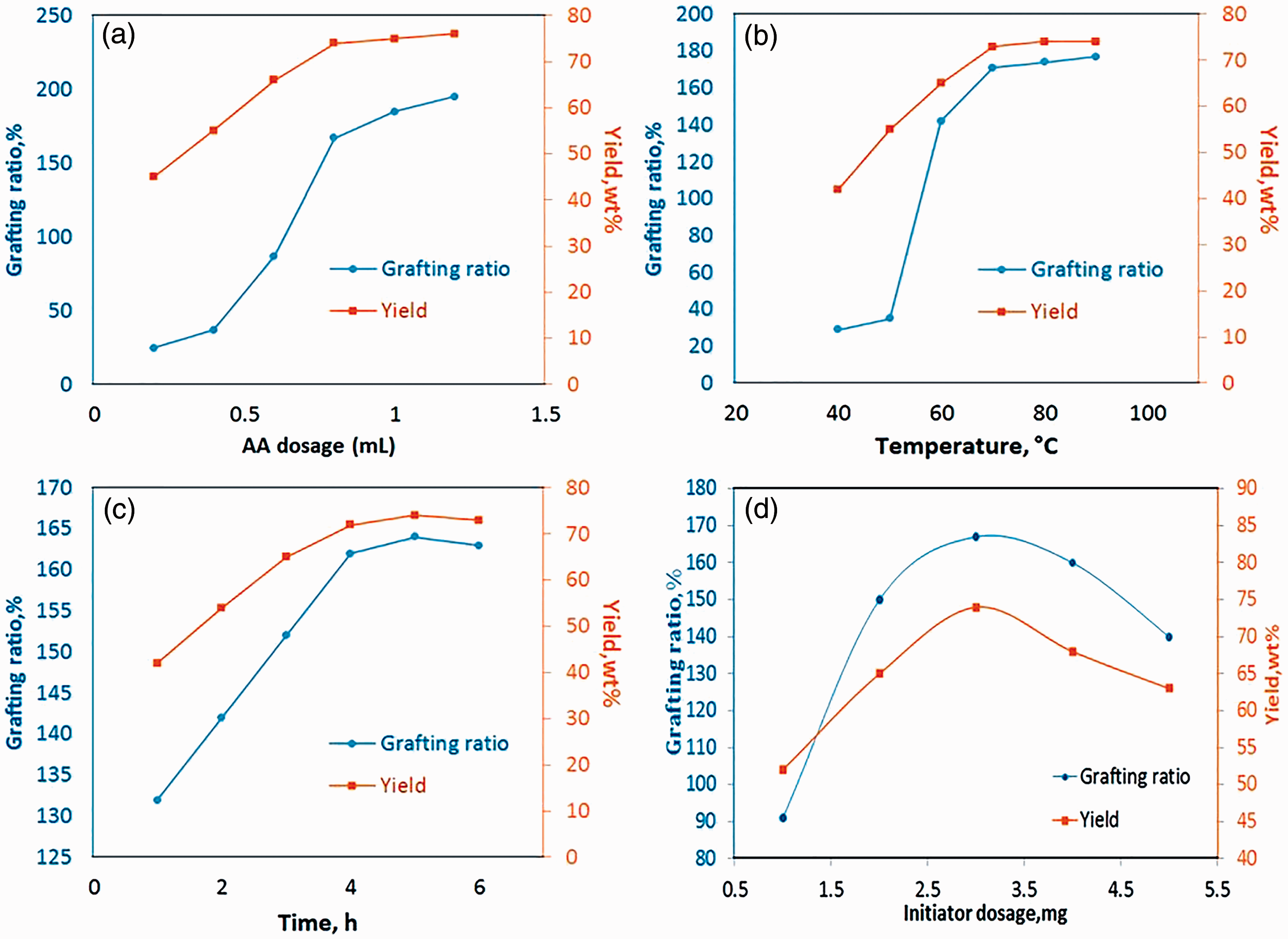
Effect of (a) AA dosage, (b) temperature, (c) time, (d) initiator dosage on the grafting and yield in the LN/PAA copolymerization (acrylic acid dosage = 0.8 g, temperature = 70°C, time = 5 h, initiator amount = 3 mg).
With increasing AA concentrations, the increased available sites for connection to the LN and increased grafting and yield. Further increasing AA to the optimal amount of 0.8 ml increased the AA homopolymers but caused no significant increase in grafting and yield (Shoujuan et al., 2016).
Effect of reaction temperature on copolymerization
LN/PAA copolymerization was carried out at temperatures of 40–90°C; the results are presented in Figure 5(b). With increasing temperatures, grafting and yield in copolymerization also increased due to the increase in free radicals. At temperatures above 70°C, grafting and yield increased to negligible amounts. Therefore, 70°C was determined to be the optimal temperature. At 70°C, copolymerization was terminated, and the free radical rate remained constant (Shoujuan et al., 2016).
Effect of reaction time on copolymerization
Figure 5(c) presents the effect of contact time on grafting and yield in LN/PAA copolymerization. By increasing the time from 1 to 5 h, grafting and yield increased rapidly. After 5 h, a significant increase was not observed in either variable. The initial increases in grafting and yield were attributed to the increase in the number of active sites in LN and extension in the propagation step during copolymerization. After 5 h, the reaction entered the termination stage, the number of active sites decreased, and grafting and yield decreased (Shoujuan et al., 2016).
Effect of initiator dosage on copolymerization
Different amounts of potassium persulfate were used to evaluate the effects of the initiator on grafting and yield in LN/PAA copolymerization; the results are presented in Figure 5(d). Potassium persulfate created phenoxy active radicals in LN and provided suitable conditions for grafting of AA radicals. By increasing the amount of initiators from 1 to 3 mg, grafting and yield also increased; this result is attributed to the increase in the number of phenoxy radicals and competition among AA radicals to connect to active phenoxy sites. With a further increase in initiators, the phenoxy radicals connected to one another. As a result, the termination stage began in the radical chain. However, by increasing the amount of initiators, the formation of homopolymer chains of PAA intensified. As a result, consumption of AA monomers also increased. Hence, grafting and yield decreased (Shoujuan et al., 2016).
Factors affecting adsorption
Effect of initial pH
Figure 6(a) shows the optimal pH; Safranin-O dye is stable at a pH of 2–10 (5–8). Given its aggregate structure, LN-g-PAA nanocomposites dissolve in alkaline environments at a pH higher than 10 and loses its efficiency at a pH of less than 3 (Frangville et al., 2012). Thus, according to the stability of LN-g-PAA nanocomposite, adsorption of Safranin-O dye was evaluated in a pH range of 3–10.

Effect of (a) pH, (b) adsorbent dosage, (c) contact time, (d) initial dye concentration on the removal of Safranin-O dye (pH = 7, dye concentration = 150 mg l−1, absorbent amount = 0.1 g, contact time = 100 min (for LN) and 225 min (for LN-g-PAA)).
The isoelectric points (
At
Effect of adsorbent dosage
The effect of adsorbent dosage on the adsorption of Safranin-O dye was investigated (Figure 6(b)). Increases in the amounts of LN and LN-g-PAA increased the percentage of Safranin-O dye removal, and optimum adsorbent dosage for both LN and LN-g-PAA composition equaled 0.1 g. The percentage of Safranin-O dye removal for both LN and LN-g-PAA reached 59.66 and 83.66%, respectively, in optimal conditions. In other words, the removal rate of the dye increased almost 1.4-fold, and this result was attributed to the increased active sites of LN-g-PAA. By increasing the amount of adsorption, the adsorption capacity reduced slightly given that with low amounts of absorbent, more competition existed between dye molecules to connect to the adsorbent (Amin, 2008; Gomez et al., 2007; Río et al., 2011; Zhu et al., 2007). Thus, with the use of 0.02 g of absorbent, the adsorption capacities of LN and LN-g-PAA reached 104 and 134 mg g−1, respectively. By increasing the amount of adsorbent to 0.1 g, the adsorption capacities of LN and LN-g-PAA compounds reduced.
Effect of contact time
The effect of contact time on Safranin-O dye adsorption was evaluated (Figure 6(c)). The adsorption capacity and percentage of Safranin-O dye removal for both LN and LN-g-PAA nanocomposition were initially rapid. After about 100 min for LN compound and 225 min for LN-g-PAA composites, they reached equilibrium and remained almost constant. According to Figure 6(c), the intensity increase during adsorption of LN-g-PAA nanocomposites was higher than that of LN; this result was attributed to the increase in the number of active sites resulting from the quantitative increase in carboxylic acid groups. Therefore, the optimal times for LN combination and LN-g-PAA nanocomposition were determined to be 100 and 225 min, respectively.
Effect of initial dye concentration
The effect of initial dye concentration on the adsorption capacity and percentage of dye removal was investigated using adsorbent LN and LN-g-PAA (Figure 6(d)). With increased initial dye concentrations, a higher amount of dye interacted with the adsorbent surface, thereby increasing the adsorption capacity. In addition, by increasing the concentration of the dye, the number of dye molecules per unit area increased, and the percentage of Safranin-O dye removal decreased. Therefore, by increasing the dye concentration from 110 to 150 mg l−1, the percentage of Safranin-O removal in the LN and LN-g-PAA compounds decreased from 70 to 57.33% and 94 to 83.66%, respectively.
Evaluation of the Langmuir, Freundlich, and Temkin isotherm models
Adsorption isotherms provide equations to describe the adsorption equilibrium between solid and fluid phases (Shoujuan et al., 2016). The Langmuir and Freundlich equations are shown in equations (5) and (6), respectively (Yuzhong et al., 2013)
In the Langmuir model, adsorption is assumed to occur in a number of homogeneous locations within the absorbent. An essential characteristic of the Langmuir isotherm is a constant and dimensionless parameter called RL, which can be obtained from equation (7) (Shoujuan et al., 2016):
C0 (mg l−1) represents the initial dye concentration in the solution.
Isotherms describe the actions of adsorbents and absorbents. In the studied Langmuir model, a layer of dye molecules was absorbed, and an equal amount of adsorption was observed in all surfaces of the absorbent. Adsorption bonds in the Langmuir model were assumed to be reversible. In the Freundlich model, the surface areas of absorbents were nonuniform and differed in their adsorption abilities (Yuzhong et al., 2013). KF represents the adsorption capacity, and 1/n represents the adsorption intensity.
The Temkin isotherm is linear and can be determined by equation (8) (Caner et al., 2009; Mahmoud et al., 2013)
Using the curve qe in Ln Ce, Bt and Kt were determined by the slope and intercept of the curve.
The Langmuir, Freundlich, and Temkin isotherm models were evaluated, and their data are shown in Table 1. The RL data in the Langmuir model and 1/n in the Freundlich model for were between 0 and 1 both LN and LN-g-PAA. Safranin-O dye adsorption of both LN and LN-g-PAA was assessed desirably using the Langmuir and Freundlich models. The correlation coefficient R2 values in the LN and LN-g-PAA absorbents reached 0.997 and 0.987 for the Langmuir isotherm, 0.73 and 0.61 for the Freundlich isotherm, 0.77 and 0.59 for the Temkin isotherm, respectively. The higher R2 in both LN and LN-g-PAA showed that dye adsorption followed the Langmuir isotherm. Surface energy absorbents were homogeneous. The maximum adsorption capacity of LN reached 81.96 mg g−1 and that of LN-g-PAA totaled 138.88 mg g−1 in the Langmuir model; both results were close to the experimental values. The high adsorption capacity of the LN-g-PAA nanocomposite was attributed to the quantitative increase in carboxylic acid groups in the polymer chain of the AA connected to the phenoxy radicals in LN during polymerization. Therefore, by forming –COO− groups in a pH higher than 5.4, the electrostatic attraction to the cationic dye increased. As a result, the adsorption capacity also increased.
Effect of the Langmuir isotherm, Freundlich isotherm, and Temkin isotherm on Safranin-O dye adsorption (dye concentration = 150 mg l−1, pH = 7, adsorbent dose = 0.1 g, contact time = 100 min (for LN) and 225 min (for LN-g-PAA)).

LN: lignin; LN-g-PAA: lignin nanoparticle-g-polyacrylic acid adsorbent.
Table 2 compares the adsorption capacity derived from cationic dye adsorption using the LN-g-PAA nanocomposite adsorbent with that found in other studies.
Reported maximum adsorption capacities for Safranin-O adsorbed onto different adsorbents.

Evaluation of pseudo-first-order, pseudo-second-order, and intraparticle diffusion kinetic models
Adsorption kinetics were studied to determine the factors that influenced the rate of adsorption. Pseudo-first-order and pseudo-second-order models are used to convey quantitative expressions in adsorption kinetics. The pseudo-first-order kinetic model shows that infiltration occurred from within a layer and is proportional to the number of unoccupied locations in the surface of an adsorbent. The linear pseudo-first-order kinetic model is shown in equation (9) (Gil et al., 2011)

The pseudo-second-order kinetic model indicates that chemical adsorption is the retarder stage. The linear pseudo-second-order kinetic model is shown in equation (10) (Gil et al., 2011)

According to the intraparticle diffusion model, adsorption transpired in three distinct phases. First, adsorbate molecules were transferred from the soluble phase to the adsorbent surface. In this step, adsorbate molecules must pass through the surrounding film solvent. This stage is known as the penetration process. Then, the absorbed molecules were transported to porous locations in the adsorbent. This step is called the influence on porosity. Finally, the particles attached to the surface of the adsorbent and were absorbed. The intraparticle diffusion model is presented in equation (11) (Siddique et al., 2009)

Table 3 shows the K، qe and R2 in the pseudo-first-order and pseudo-second-order kinetic models and Kp and C in the intraparticle diffusion model. The correlation coefficient (R2) in the pseudo-second-order kinetic model is higher than that in the pseudo-first-order and intraparticle diffusion kinetic models. Therefore, the bleaching process can be described by the pseudo-second-order model. Considering the pseudo-second-order model, chemical adsorption affected the connection of Safranin-O dye to LN and LN-g-PAA.
Pseudo-first-order, pseudo-second-order, and intraparticle diffusion kinetic models for BR2 dye adsorption (dye concentration = 150 mg l−1, pH = 7, adsorbent dose = 0.1 g, contact time = 100 min (for LN) and 225 min (for LN-g-PAA)).

LN: lignin; LN-g-PAA: lignin nanoparticle-g-polyacrylic acid adsorbent.
Coefficient C in the intraparticle diffusion kinetic model is nonzero, and the curve does not pass the origin of the coordinates. Therefore, dye concentration and diffusion are not the only rate-determining step in dye adsorption; other processes, such as complex formation or ion exchange, effectively connect Safranin-O to LN and LN-g-PAA and can control adsorption (Siddique et al., 2009). In the pseudo-second-order kinetic model, the adsorption capacities of LN and LN-g-PAA measured 92.59 and 136.98 in the computational results and 85 and 125.5 in the experimental results, respectively. The proximity of the experimental and theoretical results shows that the pseudo-second-order kinetic model is appropriate to determine Safranin-O dye adsorption in LN and LN-g-PAA.
Adsorption/desorption
To determine the potential reuse of LN-g-PAA adsorbent, the five steps of the adsorption and desorption test were performed. As shown in Figure 7, adsorption rate of hydrogel decreased with reuse. Thus, in the initial adsorption/desorption test, the percentage of removal of the dye was 84%. After the five experimental stages, the percentage removal of the dye reduced to 55%. After five desalinating steps, the hydrogel absorber retained its ability to reabsorb of more than 50% of the dye. This result can be attributed to the saturation of adsorption sites in hydrogels and the reduction in porosity at absorber levels.

Determine the potential reuse of LN-g-PAA adsorbent for five steps of the adsorption/desorption test.
Conclusion
This study was conducted to assess the efficacy of LN-g-PAA as cheap bioadsorbents that are environmentally friendly in the removal of cationic Safranin-O from aqueous solutions. The results showed that the adsorption capacity of LN-g-PAA increased to almost 1.4-fold in LN. The most important factor is the electrostatic attraction. The presence of polar groups of –COOH in the structure of natural absorbents is the most important factor that creates electrostatic attraction for BR2 dye adsorption. With the increase in electrostatic attraction, hydrogen bonding, and surface area, the adsorption capacity and percentage of dye removal also increased.
Adsorption using the pseudo-second-order model for LN and LN-g-PAA absorbents occurred according to the principles of two-stage adsorption kinetics. In the first stage, dye removal rate was increased by adsorption. In the second stage, the dye removal rate decreased due to the influence of the porous surface. Thus, LN-g-PAA achieved a suitable adsorption capacity and compatibility with the environment, implying that this compound is suitable for the removal of wastewater containing Safranin-O dye.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The authors are grateful for the financial support from Vice-Presidency for Science and Technology (Fund for the support of Researchers and Technologists of the Country).








