Abstract
Ag-Clinoptilolite/Polyethersulfone (PES/Clin/AgNPs) nanofiber was synthesized through the electrospinning method. The effect of solvent, the amount of Ag-Clinoptilolite, and PES were investigated. Parameters such as electric field, spinning distance, and concentration of the dope solution were studied in order to demonstrate their effects on the electrospinning ability and morphology of the nanofiber. The structure of PES/Clin/AgNPs nanofiber was characterized by X-ray diffraction (XRD), scanning electron microscopy (SEM), and energy-dispersive X-ray spectroscopy (EDX) analyses. In the optimum conditions, the nanofibers could be prepared at the size of 250–800 nm. Then, their ability to remove chemical oxygen demand (COD) from real wastewater was studied. The result revealed about 85% removal of COD at pH = 10 and in 10 min for PES/Clin/AgNPs (25%). A successful fabrication method using low-cost natural zeolite and the green polymer was introduced. The reusability of the column was assessed.
Introduction
Nowadays, maintaining quality and adequate water resources to meet a society’s needs is a critical issue. Changes in lifestyle have caused different kinds of pollutions such as bacteria, organic materials, dyes, anion, and cation introduced in water. On the other hand, finding a best solution for the elimination of these pollutions has become a challenge for scientists. One of the most important issues in environmental pollution is the chemical oxygen demand (COD). COD is an important parameter to evaluate the concentration of organic contaminants in the water and wastewater. 1 Higher COD levels mean a greater amount of oxidizable organic material in the sample, which can reduce dissolved oxygen (DO) levels. 2 Residual food waste such as bottles and cans, antifreeze, and emulsified oils are all rich in COD and familiar sources of COD for industrial wastewater. 3 Influent COD in normal domestic sewage is generally 600–900 mg/L, and it is then treated to at least 75–100 mg/L before discharge to minimize pollution potential. Treating of the COD is very challenging 4 ; some processes can help to reduce wastewater’s COD, such as using chemical materials. 5 Nowadays, nanoparticles such as copper, zinc, silver, and zirconia are great and effective antibacterial agents 6 that reduce COD due to their high particular surface area and interfacial activity. Literature reviews show that introducing it to solid substrates such as titanium dioxide, zinc oxide, and silica is helpful to reduce pollutants in large-scale industrial applications. 7 Possessing some disadvantages, such as high energy requirements, 8 toxic sludge, 9 releasing of nanoparticle to environment, 10 stability of adsorbents, and agglomeration of the nano-adsorbents during adsorption 11 process are still challenging issues for this absorbent. Zeolites have been applied in various procedures such as separation 12 and filtration 13 due to their specific porous structures and mobility 14 being useful to purified water.
In recent decades, a new type of membrane has recently been designed by combining polymeric materials with nanoparticles for water treatments15,16 to overcome this challenge. The feed solution, which contains the species removed, passes through the membrane, and the impurities are retained at the surface or pores of the material in the membrane.17–20 The hybrid membranes could control many parameters, such as their pore size, porosity, charge density, and mechanical stability.21–24
Some polymers such as Polyethersulfone (PES), Polytetrafluoroethylene, Polypropylene, Polyvinylidene fluoride, Chitosan, and Cellulose acetate are popular hydrophobic membranes.25–26
One of the individual engineering plastics is PES, which has higher efficiency. 27 Furthermore, PES membranes have good conductivity and selectivity. 28 So those features are a cause of using PES for hemodialysis, 29 hemofiltration, 30 and water treatment stages. 31 Moreover, filtration often needs the membranes to provide sufficient mechanical stability and chemical and physical resistance in the diverse situations such as a broad range of temperature, humidity, mechanical vibration, shock, and reactive strength. 32
Today, electrospinning is known as an effectively produced nanofiber structure for a broader range of membranes. 33 A high-performance filter applied in the treatment and recycling of wastewater is electrospun due to the suitable fiber diameter being a significant parameter to determine the filter’s filtration feature. 34 The electrospun nanofibers have diverse individual attributes such as superior mechanical properties, particular surface area, significant porosity, and continuous fibers. 35 In addition, simplicity, low-cost effectiveness, and relatively excellent production rate are other advantages of this procedure. Therefore, electrospinning has been used in plenty of usages such as textile, catalyst, membrane, sensor, biomedical, 36 filtration, template, and removal of various heavy metal ions.37,27
In this aim, for the first time, silver nanoparticle (Ag NPs) as a low-cost natural zeolite was added to clinoptilolite (Clin). Then, Clin/Ag NPs hybrid was introduced to the PES electrospun nanofibers using a sol–gel process to modify it. The PES/Clin/AgNPs were characterized with different techniques. Finally, we considered the removal of COD of wastewater in real wastewater.
Materials and methods
Materials
PES (Mw = 48,000 g/mol and density 1.37 g/cm3) and N, N-dimethylformamide (DMF) were purchased from Sigma-Aldrich. Natural zeolite (clinoptilolite) is from Semnan Iran. Sodium hydroxide (NaOH) pellets and silver nitrate (AgNO3) (99.9%), and sodium tetrahydroborate (NaBH4) (99%) were purchased from Merck company.
Characterization
The chemical compositions of the samples were evaluated by EDX (XFLASH 6130). The PES/Clin/AgNPs nanofiber structure was analyzed by FT-IR spectra Galaxy series FT-IR 5000 spectrometer with KBr tablet. The crystalline structures of samples were studied with the XRD, Model X-ray diffractometer (PW-1840) manufactured by Philips with a CuKα anode, and λ = 154 nm radiation. The morphologies of samples were investigated by SEM Emcrafts 1000 model. Sputter Coater for gold (Au) coating of non-conducting samples for SEM imaging was used. The thermal behaviors of sample were studied with a thermogravimetric analyzer (PerkinElmer TG/DT 6300 PYRIS TM Diamond model) in the temperature range of 30–600°C in a nitrogen atmosphere. The N2 adsorption-desorption was analyzed by BET (Belsorp-Mini II). The elemental composition of sample was analyzed by XRF ARL 8410 model.
Methods
Synthesis of Ag/zeolite nanoparticles
To prepare Na/Clinoptilolite, 10 g of Clinoptilolite was added to a 2 Schematic illustration of the synthesis of Ag/zeolite nanoparticles.
Preparation of Ag/zeolite/PES electrospun nanofibers
Based on Figure 2, PES was dissolved in DMF solvent at 60°C for 24 h until it formed a homogeneous solution.
39
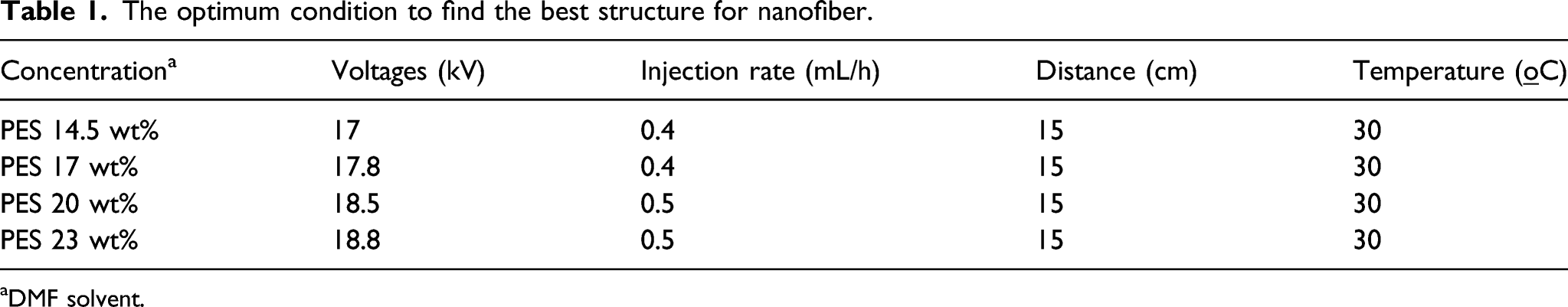
Different concentration of (23, 20, 17, and 14.5 w/w %) PES solution were prepared, and the resulting material was mixed with 0.2 g nano-Ag/Clinoptilolite, which was homogenized by ultrasonication and stirred at 60°C for 12 h. The prepared solution was packed in a syringe as the spinneret and was injected into the spinneret with a needle. For output of direct-current (DC) voltages, the spinneret was fixed by high voltage (from 15 to 22 kV). Schematic illustration preparation of Ag/zeolite/PES electrospun nanofibers.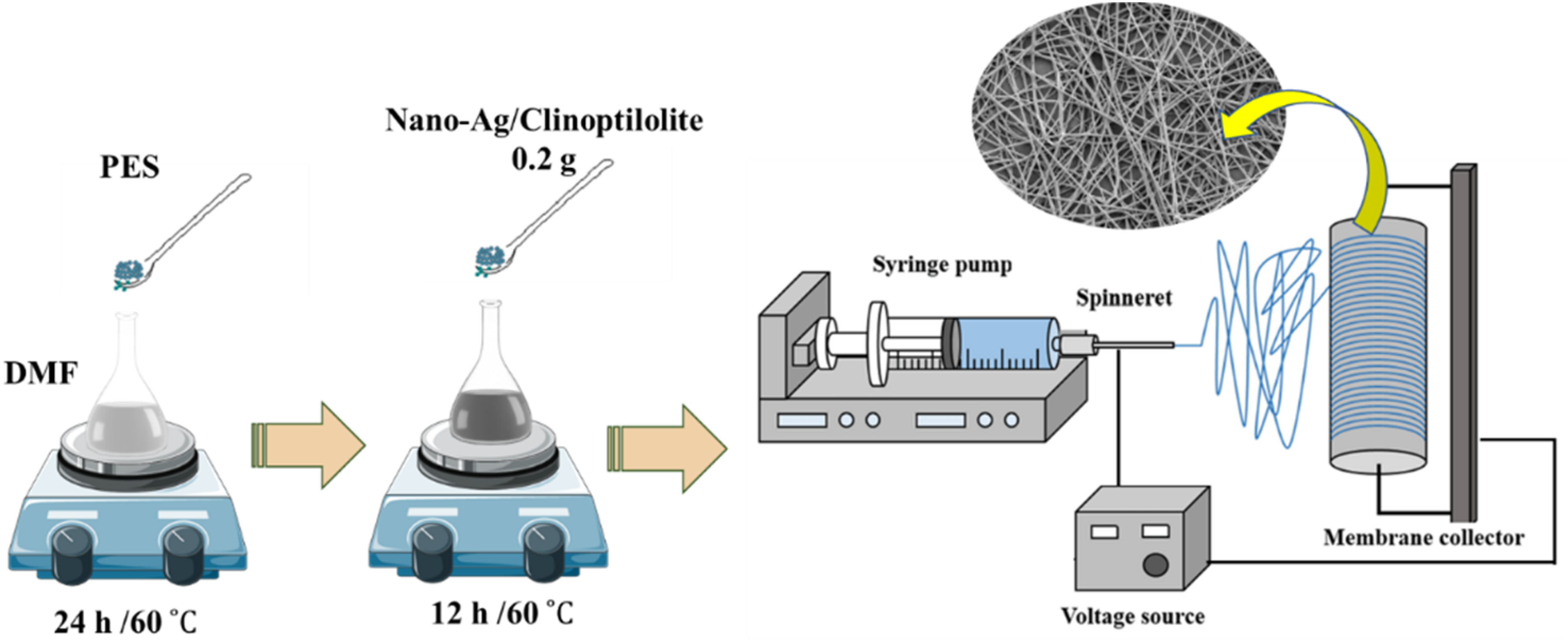
The optimum condition to find the best structure for nanofiber.
aDMF solvent.
Reduction of chemical oxygen demand parameters
To measure COD parameters, the ASTM D1259 method was used, which is a valid global reference in laboratories approved by the environment organization. 38 This method’s basis is the oxidation of organic pollutants by K2Cr2O7 and mercury catalyst HgSO4 in the presence of silver sulfate (Ag2SO4). The vials, which contained a sample, were refluxed at 150°C for 2 h, and Cr (VI) was reduced to Cr (III). The oxidant excess was distinguished by a UV–Vis spectrophotometer set at 420–600 nm. 41
Results and discussion
FT-IR analysis
Figure 3(a)–(c) show FT-IR spectra for the PES, nano-Ag/Clinoptilolite, and PES/Clin/AgNPs nanocomposite. The FT-IR PES nanofiber showed the bands at 1578 cm−1, 1484 cm−1, and 1406 cm−1, ascribing the stretching vibration of integral benzene rings that are chemically stable. Moreover, PES nanofiber had weak absorbance near 1670 cm−1, attributable to the stretching vibration of C = C of benzene rings. According to the spectrum of PES, two bands occurred about 1300 cm−1, representing the asymmetrical stretching vibration of S = O bonds, and the symmetrical stretching vibration of those bonds appeared at 1146 cm−1.
42
The infrared spectrum of nano-Ag/Clinoptilolite (Figure 3(b)) consists of broadband in the range of 3300–3700 cm−1 belonging to the surface hydroxyl groups. Moreover, the other area is in the field of 400–1200 cm−1, which is due to the zeolite network’s tensile vibrations. FT-IR spectra of PES (a), nano-Ag/Clinoptilolite (b), PES/Clin/AgNPs nanocomposite (c).
In addition, it includes Si-O-Al flexural vibrations at 461 cm−1 frequencies, symmetric vibrations at 741 cm−1 and 795 cm−1, and Si-O-Al chain vibrations at 1074 cm−1. 43 The comparison of FT-IR spectra of PES/Clin/AgNPs with pure nano-Ag/Clinoptilolite and PES are show there aren't significant change respect to PES, which may be due to fine dispersing of zeolite on the polymer. 41
X-ray diffraction analysis
X-ray diffraction patterns of Ag/Clinoptilolite and PES/Clin/AgNPs and pure PES are shown in Figure 4(a)–(c). Figure 4(a) shows the XRD pattern for nano-Ag/Clinoptilolite and lines appear at 2θ =9.9°,11.2°, 22.4°,31°, and 34.2° match with what was used to identify the phases present 1-25-1349 card numbers of Clinoptilolite (NaKCa)6(Si6Al30)O72 as principal crystalline phase about 80%. In addition, the weak lines related to nano-silver can be seen at 2Θ = 33.75° and 44.75° that are show with a circle. After introducing nano-Ag/Clinoptilolite to the polymer, which has an amorphous phase (Figure 4(c)), the official phase is amorphous due to the presence of large amount of PES, and some weak lines related to nano-Ag/Clinoptilolite are observed, which was the same as the result of other work.44,45,46 X-ray diffraction patterns of Ag-Clinoptilolite (a), PES/Clin/AgNPs (b), PES nanofiber (c).
Scanning electron microscopy images of the Ag-Clinoptilolite and PES/Clin/AgNPs nanocomposite with different concentrations of Ag-Clinoptilolite are shown in Figure 5(a)–(e), respectively. In the case of Ag-Clinoptilolite, the particle size is about 30–35 nm. PES/Clin/Ag nanocomposite shows the average fiber diameter increased from 250 nm to 800 nm which increased the percent from 23 to 14.5. SEM images of PES/zeolite with different PES/DMF ratio: Ag-Clinoptilolite (a), PES (b), 14.5 wt% (c), 17 wt% (d), 20 wt%(e), 23 wt%(f), (the scale bar is 1–10 μm, 0.5 mL/h Injection rate in 15 cm distance, 17–18.5 keV.
The images depict that Ag-Clinoptilolite particles are non-uniformly dispersed within the nanofibers. These images show that surface contact will increase and be suitable for applications requiring a high activity level. The best percentage of sulfone/Ag-Clinoptilolite polyether was found, indicating that high concentrations will increase the diameter of nanofibers and nozzle clogging and reduce the contact surface, and low concentrations will cause rosary grain structure and not suitable fibers. The chemical composition of the related compounds was evaluated by EDX for pure Clinoptilolite, Ag-Clinoptilolite, and PES/Clin/Ag nanocomposite (Figure 6(a)–(c)). The result shows that the Si/Al ratio for all samples is about four confirmed the presence of Clinoptilolite which is very close to XRF results (3.8). After introducing Ag and PES to Clinoptilolite, the peaks related to Ag, C and S can be observed indicating the presence of 20.96% Ag. EDX of (a) Clinoptilolite, (b) Ag-Clinoptilolite (c) PES/Clin/AgNPs.
Thermogravimetric analysis
Thermal analysis was carried out to examine the thermal stability of composite, and Figure 7(a) and (b) compare PES and PES/Clin/AgNPs nanocomposite. In two samples, the first step shows weight loss, which is corresponding to desorption of physical and chemical water before 180°C. In Figure 7(a), the second steps are related to the deformation and change of the polymer’s structure and thermal stability at about 420°C with weight loss of approximately 2.2%. Finally, a high weight lost after 420°C shows weight loss by the polymer chains' carbonization at about 600°C.
46
After the addition of nano Ag-Clinoptilolite to the polymer’s nanofiber, the degradation temperature rises from 420°C to 485°C. These results indicate that the thermal stability of the polymeric network increased by adding Ag-Clinoptilolite. This growth in thermal stability could be ascribed to the high thermal constancy, uniform distribution of zeolite particles, and the existence of an interaction between zeolite particles and polymer matrix.27,47,48 Thermogravimetric of PES (a), PES/Clin/AgNPs nanocomposite (b).
N2 adsorption-desorption
The N2 adsorption–desorption curves for Ag-Clinoptilolite and PES/Clin/AgNPs nanocomposite are illustrated in Figure 8. The result in the case of Ag-Clinoptilolite nitrogen adsorption–desorption isotherms shows a hysteresis loop at 0.6 to 0.9, and exhibiting characteristics of type IV isotherms for the sample confirmed the presence of mesoporous structures. In addition, a step can be observed at N2 absorption/desorption isotherms diagrams, (a) Ag-Clinoptilolite, (b) PES/Clin/AgNPs nanocomposite.
Determination of chemical oxygen demand
Figure 9 shows the schematic illustration of the experiment column designed. Three real wastewater samples were related to Halal Poyan Company (Arak, Iran) with COD 500, 480, and 420 ppm were used. First, the wastewater samples crossed through the column with the different nanofiber layers, and COD was estimated by the mentioned method and the following formulate Schematic illustration of experiment column designed.

where COD0 and CODt are the initial and equilibrium concentration of pollutants that are characterized by the use of COD, respectively.
% Removal of COD with use of polyethersulfone/Clin/Ag.

aNumber of nanofiber composite layers.
bNanofiber of PES/Clin/Ag nanocomposite containing 20% w/w of Ag-Clinoptilolite (0.2 g).
In fact, the presence of adsorbent increases free and active hydroxyl radicals and superoxide anions, which can significantly reduce the COD of wastewater because nanosilver was dispersed enough in the substrate. In addition, the sulfonyl group of the polymer and hydroxyl group of zeolites are suitable groups for trapping organic pollutants and interaction with nanosilver more comfortable with respect to powder (Table 2 entire 4). The pH of the wastewater solution is one of the essential factors in the adsorption or removal process. The effect of pH on the removal of COD is shown in Figure 10(a) for three samples with different COD of wastewater and using three layers of nanofiber of PES/Clin/AgNPs nanocomposite. The results show that the basic pH is better for decreasing COD (above 84% at pH = 10). It might be due to hydrogen ions themselves are strongly competing with organic pollutants and because pH determines the valence state of the ions and their precipitation process. (a) The effect of pH on the presence of removed COD for samples 1, 2, and 3 with COD are 500,400, and 420 ppm. (b) reusability of the column after three runs.
In addition, the deprotonations form of reactive sites such as sulfonyl groups of polymer and hydroxyl group of zeolite are responsible for the binding of pollutants (Scheme 1). To consider the reusability of PES/Clin/Ag nanofiber, the column was washed with distilled water three times, dried at room temperature (25°C), and used in three cycles. The result in Figure 10(b) shows that there is no significant change in the percent of removed COD (about 5%) which confirms the high activity of absorbent, and this result can prove the importance of this low-cost adsorbent in industry. Mechanism for removal of COD
Conclusion
In this study, different amounts of Ag-Clinoptilolite are incorporated onto polyethersulfone. High-quality electrospun PES method used in the presence of DMF as a solvent. Uniform fiber morphology (diameter: 250–800 nm) was obtained at PES concentrations from 23 to 14.5 wt% in DMF. With introducing Ag-Clinoptilolite, the average fiber diameter was slightly increased, and it trapped in polyethersulfone, confirmed by XRD and SEM result.
The PES/Clin/AgNPs nanocomposite has been used to remove COD in a real wastewater sample. Results showed that the removal percent of COD was approximately % 85 at pH = 10 in 10 min. In addition, PES/Clin/Ag nanocomposite could be used for several runs. This indicates the preservation of the nanoparticle-modified natural zeolite property and the addition of new and desirable properties to the nanofibers, being useful to wastewater industry.
Footnotes
Acknowledgments
Thanks to Arak University, and the Iranian Chemical Society’s zeolite and porous materials committee for supporting this work.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.

