Abstract
The aim of this study was to compare the fastness properties and wastewater quality in obtaining the same color on 100% woven wool fabric with natural (madder) and synthetic (1:1 metal complex) dyes. The same color, with similar wet fastness properties, was obtained on the wool fabric when dyed with either dye. On the other hand, conductivity, chemical oxygen demand (COD), and the ammonia nitrogen (NH3-N) content of dyeings carried out with the synthetic dye mixture were considerably higher than dyeing with madder root extract, and its pH was more acidic. However, the amount of volatile suspended solids and color obtained in natural dyeing was higher than the amount in the synthetic dyeing wastewater.
Introduction
The art of painting has a long history and most of the paints come from prehistory.
1
Traces of colored garments of cloth and traces of madder dye were found in the ruins of the Indus Valley civilization in Mohenjodaro and Harappa (3500 BC).1–2 The strong and almost fadeless cotton dye known as Turkish Red was developed in India and spread from there to Turkey. Madder dye powder produced in India costs about Rs 5000/kg. High quality madder root is priced at Rs 100–200/kg and about 3–4% of the dye cannot be recovered.
3
Blue indigo from the leaves of
As is generally known, the dyeing of wool fibers can be carried out using acid, chrome mordant, metal complex, and reactive dyes. 8 In the past, the use of mordant dyes in conjunction with chromium mordants on wool became widespread owing to the excellent all-round fastness properties of the dyeings. Mordant dyeing of wool suffers several disadvantages, however: color matching is difficult owing to the marked change in shade involved, and wool tendering occurs during the prolonged and relatively complicated two-stage, pre- and after-chrome dyeing processes. Consequently, in the early twentieth century, efforts were made to overcome these disadvantages, resulting in the introduction of metal-complex or pre-metallized acid dyes. As their name implies, in metal-complex dyes, one metal atom, commonly chromium, is complexed with either one (1:1 metal-complex dye) or two (1:2 metal-complex dye) molecules typically containing monoazo dye containing groups (such as hydroxyl, carboxyl, or amino groups) that are capable of coordinating with the metal. 9 Discharge of dye-bearing wastewater into natural streams and rivers from textile industries poses a severe problem, as dyes impart toxicity to aquatic life and are damaging the aesthetic nature of the environment. The textile industry consumes high amounts of processed water and produces highly-polluted discharge water. Textile industry wastewater includes organics, total suspended solids, total dissolved solids, sulfur, sulfide, ammonia nitrogen, heavy metals, and other pollutants. 10
Natural dyes are non-toxic and they can be obtained relatively easily and safely. For this reason they are said to be a good alternative to synthetic dyes in terms of their environmental and ecological effects. 11 However, the problem in dyeing wool with natural dyes is that heavy shades and high fastness values could not be obtained without mordant. To overcome these problems, metallic salts are used as mordants, which are traditionally used to improve fastness properties or exhaustion, and to develop different shades with the same dye. 12 But from the view of ecology, mordants are not desired in wastewater. The present study tried to clarify if natural dyeing without mordant usage could be an ecological alternative to 1:1 metal complex dyeing of wool fabrics.
In the literature, it is stated in many studies on natural dyes that natural dyeing is more environmentally-friendly than dyeing with synthetic dyes. However, none of them characterized the wastewater produced from obtaining the same color with natural or synthetic dyes on wool fabric. From this point of view, although there are many studies on madder, it can be said that the current study is original. Beyond this, another original point of this study is that the treatment efficiency of madder root and synthetic dye wastewater with activated carbon was examined comparatively.
The aim of this study was to compare the fastness properties and wastewater quality in obtaining the same color on 100% woven wool fabric with natural (madder) and synthetic (1:1 metal complex) dyes. The same color, with similar wet fastness properties, was obtained on the wool fabric when dyed with either dye. Afterwards, fastness properties and wastewater quality of these dyeings were compared. Furthermore, the reuse of the of natural dyeing bath was studied. Accordingly, after dyeing with madder root extract, the remaining dyebath was taken and used again for dyeing a wool fabric. Both color and fastness properties were tested. After testing, the color and fastness properties of the dyeings with natural and synthetic dyes, the wastewater characteristics, and the ecological risks of these wastewaters were also compared. The treatability of synthetic and natural dyeing wastewater was also studied. The adsorption process was selected for treatability studies.
Experimental
Material
In this study, 100% wool woven fabric (154 g/m2), kindly supplied by Yunsa Inc., was used. Warp and weft density values were 36 and 32 threads/cm. All experiments were carried out by using soft (<5°dH) water.
Natural Dye
Madder (

Initial (left) and ground (right) forms of madder root.
The most important colorants in madder are the anthra-quinones alizarin (I), purpuroxanthin (II), rubiadin (III), manjistin (IV), purpurin (V), and pseudopurpurin (VI), as shown in Fig. 2. The C.I. numbers of colorants present in madder are C.I. Natural Red 8, 9, and 14. 13
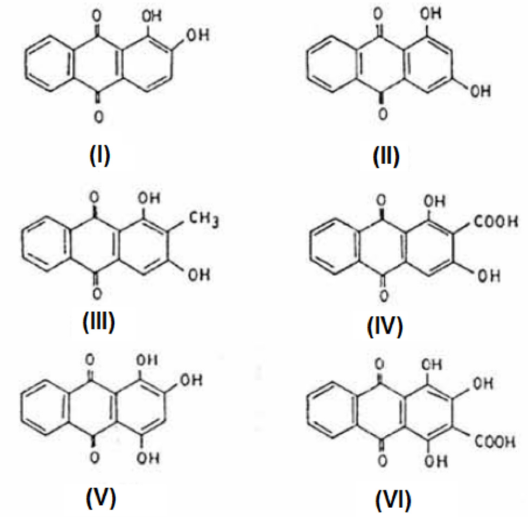
Chemical composition of madder. 3
Preparation of Extract Dye Solution
In 1L of water, 10 g of madder root in ground form was soaked. Subsequently, the solution was heated to boiling, boiled for 30 min, and filtered with gauze fabric. The volume of this filtrated solution was then adjusted to 1 L using pure water and it was used as a dyeing liquor.
Dyeing with Madder Root Extract
The filtered dye extract solution (100 mL) was used to provide a liquor ratio (LR) of 10:1 for 10 g of material. Dyeings were carried out at the extract solution pH value of 5.3. Dyeings were carried out without mordant usage on a Termal HT Dyeing Machine according to the dyeing graph given in Fig. 3. No mordant was used to obtain a more ecological dyeing. Reuse of the natural dyeing bath was also studied.
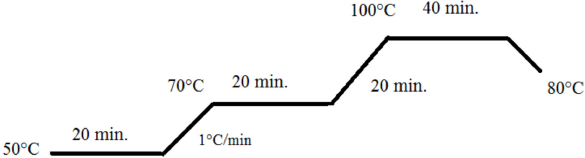
Dyeing graph used in dyeing with madder root.
Dyeing with 1:1 Metal Complex Dyes
We tried to match the color on wool obtained with madder root with that using 1:1 metal complex dyes. Many laboratory trials were carried out. Finally, the desired result was obtained. Dyeing liquor (100 mL) containing 1:1 metal complex dyes (Neolan Yellow P-0.26%, Neolan Red P-0.49%, and Neolan Blue-0.08% owf mixture from Huntsman), formic acid (Merck, 85%, 3 g/L), sodium sulfate (Merck, 10 g/L), sodium acetate (Merck, 1 g/L), wetting agent (Nevowet Dnan New Liq, Nevodis, 1 g/L), wool protective agent (Nevowop TWP, Nevodis, 4%), anticrease agent (Tanapon TS, Tanatex, 1 g/L), antifelting agent (Biavin BLI, CHT, 1 g/L), antifoaming agent (Kollasol CDS, CHT, 0.35 g/L), and levelling agent (Egalen WS/D, Prochimica, 2%) was used to provide the liquor ratio of 10:1 for 10 g of material. After dyeing, fabrics were neutralized with sodium acetate (4 g/L) and then subjected to aftertreatment with a fixing agent (Mesitol, Tanatex). Dyeings were carried out on a Termal HT Dyeing Machine according to the dyeing graph given in Fig. 4.
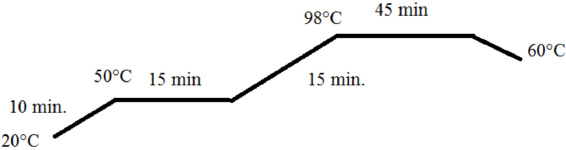
Dyeing graph used in dyeing with 1:1 metal complex dyes.
Color Measurements
Reflectance (R%) values of dyed samples were measured with a Gretag Macbeth E700 (D 65/10°) and color yields (K/S) of dyed samples were calculated by the Kubelka Munk equation (Eq. 1.). 14
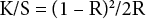
K is the absorption coefficient, R is the reflectance value at the maximum absorption wavelength (nm), and S is the scattering coefficient.
CIE
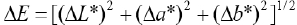
Δ
Fastness Tests
The washing (at 40 °C), rubbing (both dry and wet), and perspiration (both acid and alkali) fastness values of dyed samples were tested according to ISO-105 C06, ISO 105-X12, and ISO 105-D01 standards respectively.15–17 Then the results were assessed with a gray scale. Light fastness values of samples were also tested according to the ISO 105 B02 standard and then assessed by using a blue scale. 18
Physical and Chemical Characterization of Wastewater
Chemical parameters for wastewater samples of natural and synthetic dyeing treatments— including chemical oxygen demand (COD), ammonia nitrogen (NH3-N), suspended solid (SS) and volatile suspended solids (VSS)—were determined according to standard methods. 19 pH, conductivity, and temperature were measured by using the appropriate sensors instantaneously in the laboratory after the samples were taken. For the color and UV254 nm absorbance (aromaticity) parameters, the samples were first centrifuged at 4000 rpm for 5 min. Color was determined in accordance with TS ISO 7887 “Method B: Determination of the true color using optical instruments.” 20 Wastewater from industry doesn't show sharp and distinguished absorption maxima. According to this standard, those waters are examined using a minimum of three wavelengths (436, 525, and 620 nm). The water color was determined using a photometer or spectrophotometer. 20
In this study, the water color was determined using a UV-Vis spectrophotometer (Shimadzu UV-2401 PC instrument) at 436, 525, and 620 nm. Then the spectral absorption coefficients for these wavelengths were determined as m—1 with Eq. 3.
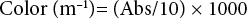
Abs is the measured absorbance value at 436, 525, or 620 nm. Color was calculated by summing these three values. UV254 was used as an indicator of aromatic compounds and measured with the UV-Vis spectrophotometer.
Treatment of Wastewater by Adsorption Process
Powdered activated carbon (PAC-Norit SA 2, Acros Organics), which had a specific surface area of 686 m2/g, was used for adsorption experiments. Batch sorption studies were carried out by shaking a series of bottles containing various amounts of PAC. The adsorption study was carried out with 50 mL of natural and synthetic dyeing wastewater samples. The pH of the 50 mL samples was adjusted to 3, 7 and 11, and 0.2 g of PAC were added. The prepared samples were then placed in a shaker and agitated at room temperature (25 °C) at 250 rpm for 150 min. After shaking, 10-mL samples were taken and centrifuged at 4000 rpm for 5 min to measure COD, color, and UV254 absorbance (aromaticity). After the optimum pH study, the effect of adsorbent doses on COD, color, and aromaticity removals were studied by a series of equilibrium experiments. For this aim, 0.1 to 1.5 g of PAC were added to the 50-mL samples and COD, color, and aromaticity were measured.
Removal rates of color, aromaticity, and COD were calculated from Eq. 4.
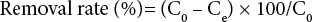
C0 and Ce are the initial and equilibrium COD, color, or aromaticity amounts, in the solution (mg/L and abs).
Results and Discussion
Results Related to Comparison of Natural and Synthetic Dyeings
Wool fabric was first dyed with madder extract solution as a natural dye. Then, taking this fabric as a reference, color matching was carried out by using 1:1 metal complex dyes to obtain approximately the same color as that with the natural dye. Color yield (K/S) and
K/S and CIE
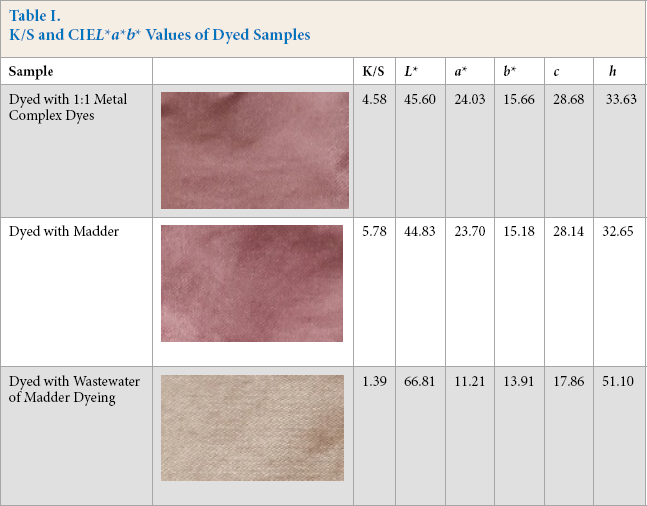
The K/S value of the sample dyed with 10 g/L of madder was moderate. By taking into consideration the fact that madder includes approximately 3-4% dye in its content, the dye concentration in experiments were 0.3-0.4 g/L. Madder has good coloring ability on wool fabric. It was also possible to increase the color depth by increasing the madder root concentration.
When
The natural dye (madder) did not contain heavy metals, neither in the dye wastewater nor on the fabric, but the 1:1 metal complex dyes were metal complexes of acid dyes.
The same color was obtained by using only one dye (madder) for natural dyeing, while with 1:1 metal complex dyes, it was obtained by using three (Yellow-Red-Blue) dyes.
In natural dyeing with madder, there was no need to use any kind of auxiliary chemi cal, but in dyeing with the synthetic dyes used in this study, many auxiliaries were added to the dyebath.
For dyeing with the natural dye (madder), pH adjustment was not required, while for dyeing with the 1:1 metal complex dyes used in this study, the pH should be adjusted to a strong acidic medium to obtain level dyeing.
For natural dyeing with madder, only cold and warm rinsing was enough, but for dyeing with the 1:1 metal complex dyes used in this study, both rinsing and aftertreatment with fixing agents were required.
On the other hand, the natural dyeing process was longer compared to dyeing with 1:1 metal complex dyes and natural dyes are more expensive. However, when the total dyeing cost is calculated, natural dyeing is cost effective due to less chemical usage and shorter after-treatment processes. This also would cause natural dyeing to be more ecological. Another important issue in natural dyeing is that the color reproducibility is problematic. In this study, both the natural and synthetic dyeings were repeated 10 times and variation coefficients (CV) were calculated over the color yield values. The CV (%) values were 4.32 and 1.78 for natural (madder) and synthetic (1:1 metal complex) dyeings respectively.
As mentioned previously, another aim of this study was to investigate the reuse possibilities of a natural dyeing bath. After dyeing with madder root extract, the remaining dyebath was taken and used again for dyeing a wool fabric. From color yield (K/S) and
Fastness is one of the most important properties in textile dyeing. Washing, rubbing, and light fastness tests were carried out. Fastness test results of dyed samples are listed in Table II.
Fastness Values of Dyed Samples a
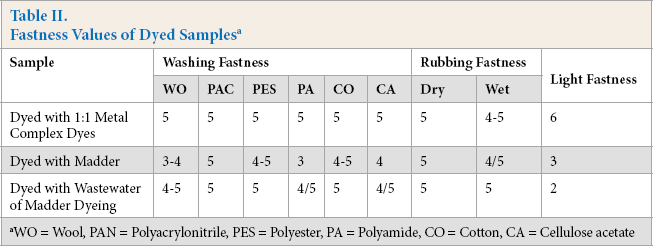
WO = Wool, PAN = Polyacrylonitrile, PES = Polyester, PA = Polyamide, CO = Cotton, CA = Cellulose acetate
Wool fabric could be dyed with madder with moderate to high wet fastness values without mordant usage (Table II). Washing fastness values of samples dyed with natural dye (madder) were lower, especially for staining values on wool and polyamide. However, there was no afertreatment with fixing agents in the case of natural dyes. The light fastness of the natural dye was considerably lower than that of the synthetic dye, especially in the case of light colors. This is already a common result for natural dyes. However, in woolen materials, light fastness is not a very critical parameter when the season in which they are used is considered. Acid and alkali perspiration fastness test results are given in Table III.
Acid and Alkali Perspiration Fastness Test Results of Dyed Samples
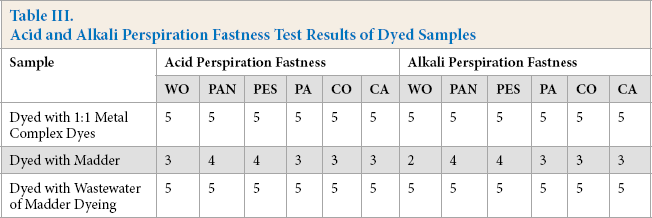
The woolen fabric was dyed with madder giving moderate levels of perspiration fastness without mordant usage. The values were lower than the sample dyed with synthetic dye. Again, however, fabrics dyed with madder were not aftertreated with any chemical that improved fastness values.
Results Related to Physical and Chemical Characterization of Wastewater
After comparing the color and fastness properties of dyeings carried out with the natural and synthetic dyes studied, the wastewater quality of these dyeings were also compared. Results are given in Table IV.
Wastewater Quality of Dyeing Carried Out with Natural and Synthetic Dyes
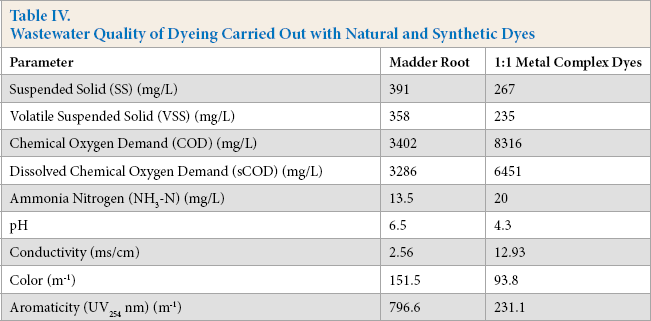
The conductivity of the synthetic dyeing wastewater was almost ten times higher than the conductivity of the natural dyeing wastewater (Table IV). This is why electrolytes and auxiliaries are used when dyeing with synthetic dyes, while for natural dyeing, only the dye and water is used without any pH adjustment.
The COD, dissolved COD, and ammonia nitrogen content of dyeings carried out with synthetic (1:1 metal complex) dyes were considerably higher than natural dyeing with the madder root extract (Table IV). Although the amount of dye used to obtain synthetic dyeing wastewater was low, the COD was high due to the added auxiliary organic chemicals. This result indicates the hazardous effects on the environment caused by synthetic dyeing.
UV254 absorbance was used to indicate the presence of aromatic compounds.21,22 The color and aromaticity values of the natural dyeing wastewater were greater than those for the synthetic dyeing wastewater sample. More dye was used in the natural dyeing process for obtaining the same color as with synthetic dyeing.
The pH of the 1:1 metal complex dye waste-water was lower than that of the wastewater from dyeing carried out with madder extract solution (Table IV). Discharge of acidic solutions are harmful to the aquatic system.
Textile wastewater is generally biologically treated. The activated sludge process operates most effectively over a pH range of 6.5 to 8.5. Neutralization may be required for wastewater that is out of this pH range. 10
In addition, very high (>9.5) or low (<4.5) pH values are not suitable for most aquatic organisms. 23 If the pH of waste-water is too acidic or too alkaline, its pH must be adjusted before discharge or biological treatment. Discharge of waste-water having low pH values are harmful to aquatic systems and should be adjusted by neutralization.
In this study, the amount of volatile suspended solids obtained by natural dyeing was slightly higher than the amount in synthetic wastewater. The organic part of the natural dyeing wastewater sample was much greater than that of the synthetic dyeing wastewater sample. To obtain the same color from synthetic dyeing using natural dyes, much more natural dye should be used. For example, in this study, the same color of 10% madder root (100 mL of filtrated dye extract, which contained 1 g of madder root used for 10 g of fabric) was obtained with 0.83% of the 1:1 metal complex dye mixture.
Results Related to Treatment of Wastewater by the Adsorption Process
Besides the determination of wastewater ecological impact/ risks, treatability is also important. The chemicals or methods used in treatability studies should be economical and feasible. Therefore, in this study, the treatability of synthetic and natural dyeing wastewater was studied. The adsorption process was selected for treatability studies.
The solution pH is one of the important control parameters in the adsorption process since it affects the surface charge of the adsorbents. The effect of pH on the COD, color, and aromaticity removal rates were evaluated with a dose of 4 g/L (0.2 g/50 mL) of PAC. The pH effects for natural and synthetic dyeing wastewater are shown in Figs. 5 and 6. The maximum COD, color, and aromaticity removal rates were obtained at pH 3. The PAC surface is positively charged at pH 3 and the adsorption of negatively-charged dye molecules in the natural (Fig. 5) and synthetic dyeing wastewaters in this study increased by electrostatic force attraction. At higher pH values, the surface of the adsorbent was negatively charged and the removal rates decreased due to ionic repulsion. 24
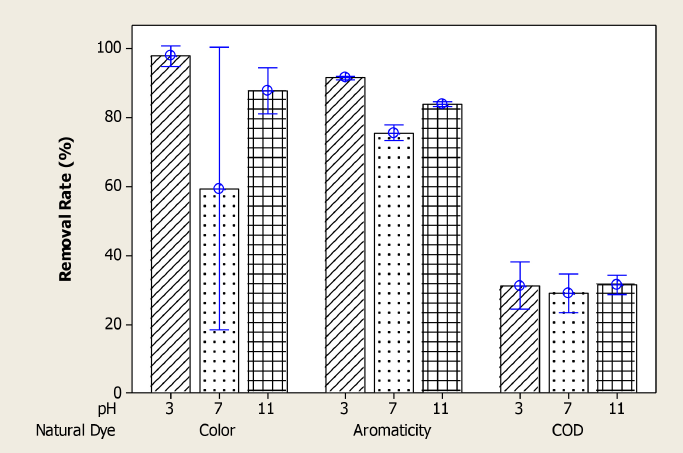
Effect of pH on color, aromaticity, and COD removal rates for natural dyeing wastewater.
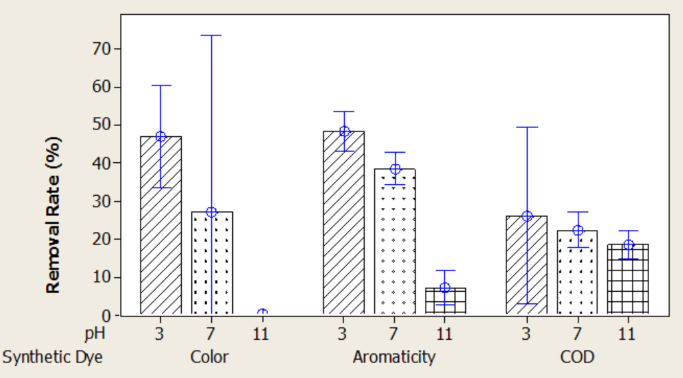
Effect of pH on color, aromaticity, and COD removal rates for synthetic dyeing wastewater.
The effect of PAC dosages on the removal rates of COD, color, and aromaticity were evaluated with a dose range of 5–30 g/L at pH 3 of the two wastewaters at a constant mixing rate of 250 rpm. Test results are shown in Figs. 7–9. As can be seen from the figures, the removal rates increased as the adsorbent dosages increased from 5 to 30 g/L. This result was due to the increase in the number of available adsorption sites and surface area. 25
As seen in Fig. 7, the equilibrium concentration in the synthetic dyeing wastewater was 16 g/L for COD, color, and aromaticity, and the removal efficiencies at this dosage were 55%, 85%, and 72%, respectively. In the natural dyeing wastewater, the equilibrium concentration for COD removal was 12 g/L and the COD removal rate at this adsorbent dosage was approximately 42%. In terms of color and aromaticity, equilibrium was reached at lower adsorbent dosages for the natural dye waste-water (Figs. 8 and 9). The equilibrium concentration for color and aromaticity was 8 g/L and the removal rates at these concentrations were 94% and 93%, respectively. The COD, color, and aromaticity concentrations in the natural dyeing wastewater were 1906 mg/L, 9.09 m–1 and 55.7 m–1 at these adsorbent doses. The concentrations of COD, color, and aromaticity in the synthetic dyeing wastewater were 2872 mg/L, 14.1 m–1 and 64.7 m–1, respectively.
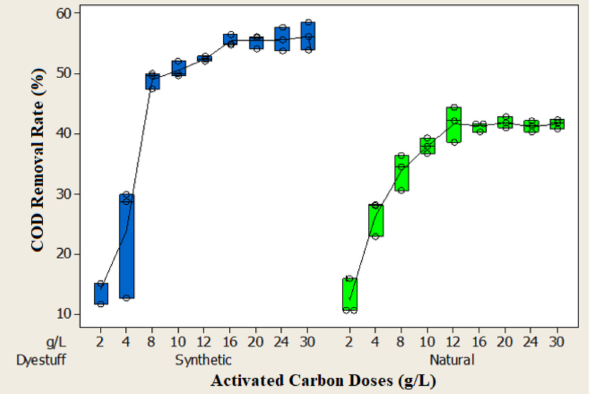
Effect of adsorbent doses on COD removal rate.
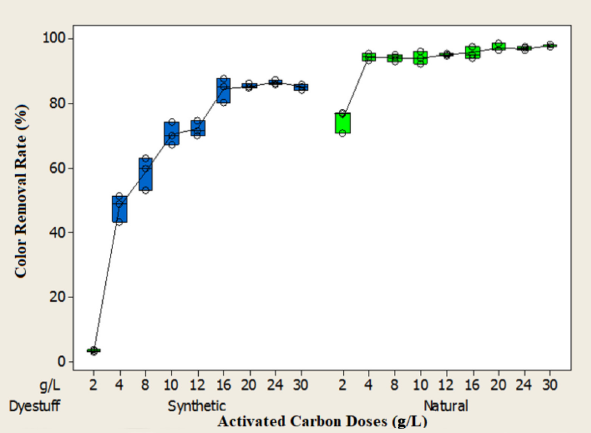
Effect of adsorbent doses on color removal rate.
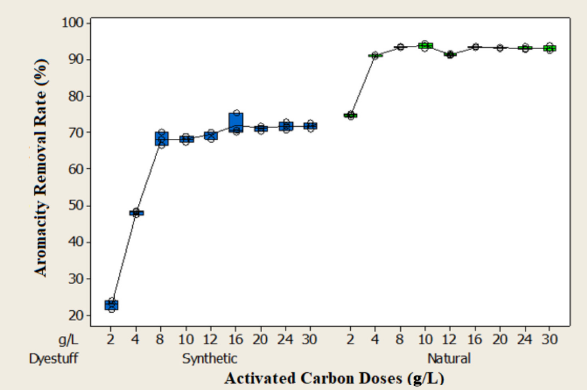
Effect of adsorbent doses on aromaticity removal rate.
As can be seen from the results, lower adsorbent doses were needed to remove COD, color, and aromaticity in the natural dyeing wastewater and the color and aromaticity removal efficiencies were very low at the same adsorbent doses in the synthetic dyeing wastewater. The most important reason for this result was the high conductivity of the synthetic dyeing wastewater. The salt ions compete with dye molecules for available adsorption sites. 26 When COD was considered, the equilibrium concentration was lower in the natural dyeing wastewater, but the removal efficiency of the synthetic dye wastewater was slightly higher. The synthetic dyeing wastewater not only contained dye molecules, but also auxiliary chemicals as organic matter that increased the COD. It is thought that these chemicals contributing to the COD are easier to remove by adsorption, and therefore the removal efficiency of the synthetic dyeing wastewater with PAC is higher.
The natural dyeing wastewater in this study needed much lower adsorbent dosages to remove color, aromaticity, and COD, making it economically more viable. After adsorption treatment, the discharge of natural dyeing wastewater was potentially less hazardous. Furthermore, the COD of the natural dyeing wastewater was less than that of the synthetic dyeing wastewater. Accordingly, lower COD values were obtained after adsorption treatment.
Conclusion
In this study, comparison of the fastness properties and wastewater quality of obtaining the same color on wool fabric with a natural (madder) and synthetic (1:1 metal complex) dyes was studied. It was possible to obtain the same color on wool fabric with the natural dye (madder) as with the synthetic dye used. Dyeing fastness values of the naturally dyed wool were lower compared to dyeing with 1:1 metal complex dyes. However, for the natural dyeing with madder root, after-treatment was not performed. Better fastness values can be obtained if after-treatment is done. The light fastness values were lower, but this is not critical for woolen materials.
In addition, after dyeing with the madder, the remaining dyebath was reused for dyeing of woolen fabrics. With the color yield (K/S) and CIE
In this study, it was determined that conductivity, COD, and ammonia nitrogen content of dyeings carried out with synthetic (1:1 metal complex) dyes were considerably higher than natural dyeing with madder root extract, and also its pH was more acidic than the wastewater of dyeing carried out with madder extract solution. However, the amount of volatile suspended solids and color obtained by the natural dyeing was higher than the amount in the synthetic dyeing wastewater. On the other hand, if residual dyebaths were used for further dyeing, this can lead to zero discharge. The determination of the ecological impacts of the dyeing waste-water and treatability studies by the adsorption method were also done. It was concluded that the natural dyeing waste-water produced in this study needed much lower adsorbent dosages to remove color, aromaticity, and COD, making it economically more viable.
Footnotes
Acknowledgement
We would like to deeply thank our student Bürhan Bugdayci for his useful assistance in the laboratory experiments.
