Abstract
Nowadays, most of the world’s palm oil is being produced in Malaysia and Indonesia; however, the demand for this vegetable oil as an edible oil is declining in many countries since consuming palm oil in excess can result in serious health problems. Consequently, finding new applications such as the production of bio-based polymers to make use of this cheap and abundant vegetable oil seems necessary. Herein, we report the copolymerization of palm oil with sulfur with different feed ratios via inverse vulcanization. The copolymers are then characterized using Fourier-transform infrared spectroscopy, differential scanning calorimetry and X-ray diffraction analysis. The results confirmed the formation of the polymers and their stability against depolymerization. Altogether, the obtained sulfur-palm oil copolymers showed great properties such as thermal stability up to 230°C under a nitrogen atmosphere and rubbery properties at room temperature. Although the Thermogravimetric analysis (TGA) thermograms had previously confirmed the high conversion of elemental sulfur into the polymeric structure by comparing the initial sulfur content and the final polysulfide content in the polymer, some unreacted elemental sulfur was also observed in the final product. Sulfur-palm oil (S-Palm oil) is a new green polymer that helps to find a new use for palm oil as a big industry as well as sulfur which is underutilized and left in stockpile as a byproduct in gas and petroleum refineries.
Keywords
Introduction
Modern human life cannot be imagined without polymers and plastics. Polymers play an essential role in modern technology and industry; however, it is only in recent decades that the unavoidable disadvantages of conventional polymers on the environment and as a consequence on human life are being considered very seriously.1,2 Most of the polymers produced so far are petroleum-based which are not only expensive but also extremely harmful to the environment. It is estimated that in 2018 only, around 359 million metric tons of different plastics were produced worldwide. 3 Most of these plastics are non-biodegradable and dependent on petroleum, making it difficult to come up with an appropriate solution for their waste and environmental management.4,5 Biopolymers, on the other hand, are sustainable polymers that are produced using bio-based monomers which directly decrease the dependency of polymers on petro-based monomers,6,7 and in many cases, they are also biodegradable.
Vegetable oils are a great source of bio-based monomers that have demonstrated great potential in the production of biopolymers which are mainly utilized in composites, food packaging, insulators, plasticizers, adhesives, and also medical devices.7–9 Palm oil, in particular, is a cheap vegetable oil that is being produced in high amounts in countries such as Malaysia and Indonesia both of which are responsible for 86% of the global palm oil production.10,11 In 2019, around 18,469,258 million tons of palm oil were only exported from Malaysia. The fact that the demand for this vegetable oil is decreasing as an edible oil in many countries indicates the necessity of finding new applications (such as polymer production) for this abundant green feedstock to maintain the current production level and also to boost the domestic palm oil industry. 12
Palm oil, in particular, has already been investigated in the production of polymers in a number of reports. 13 Mas’ud et al. 14 studied the polymerization of waste palm cooking oil employing cationic polymerization under microwave irradiation using a trifluoride-etherate catalyst. Also, the polymerization of 2-(vinyloxy) ethanol with palm oil using the base-catalyzed transesterification is reported by Kalita et al. 15 In another report, Tajau et al. 16 investigated the usage of palm oil in the production of epoxidized palm olein and diol as a novel polymeric precursor for drug delivery purposes. The need for initiators and catalysts together with low conversion of palm oil into polymeric structure were the main limitations of these polymerization processes.14–16 Inverse vulcanization is a recently developed technique that is proven to be very effective and economical to produce sulfur-based polymers using petro-based or bio-based monomers including vegetable oils.7,17
Nowadays, sulfur, with an annual production of almost 70 million tons, mostly comes as a byproduct of natural gas and petroleum refineries making it a very cheap and readily available material.18–20 With the high supply and low market demand for sulfur globally, elemental sulfur is, for the most part, left piled up in open spaces in gas and petroleum refineries which raises environmental concerns making it inevitable to search for a new way to convert it to less harmful materials for the environment. Inverse vulcanization first reported in 2013 is a solvent- and initiator-free polymerization method which makes it possible to produce sulfur-based polymers using either petro-based or bio-based monomer(s).21–23 The octet ring structure of elemental sulfur (S8), if heated above its floor temperature (159°C), will be opened, and forms a biradical linear chain of sulfur atoms accompanied by a color change of sulfur melt from yellow to orange. If heating continues, the opened chains of elemental sulfur will homopolymerize and form a pure polysulfide.24,25 Due to the nature of sulfur, pure polysulfide is not stable, and it undergoes depolymerization after only a short period resulting in the reformation of elemental sulfur crystals.7,26,27 Adding a comonomer with one or more vinylic C=C bonds to the sulfur melt with free radicals results in the production of stable sulfur-based polymers.21,23,27 Depending on the nature of the comonomer(s) and the reaction parameters, sulfur-based polymers have shown a wide range of properties from linear thermoplastics to highly crosslinked thermosets.7,28
So far, petro-based monomers are mostly used in the investigation of sulfur-based polymers. However, some bio-based monomers such as farnesene, 19 myrcene, 19 squalene, 29 limonene, 30 and diallyl disulfide 31 were also reported. Oil of some vegetables including corn, 32 rubber seed,33,34 canola, 35 soybean, 36 rice bran, 37 castor, 37 cottonseed, 38 sunflower, 39 linseed, 39 olive, 39 and algae 40 were also studied in the production of sulfur-based polymers. Nevertheless, the complex chemical structure of vegetable oils together with their unknown mechanism of reaction makes it practically very difficult to control the properties of the products. 23 The copolymerization of vegetable oils with elemental sulfur opens up a new approach not only to find new applications for sulfur but also to produce cheap polymers that can act as a good alternative for petro-based polymers in a number of applications.
Palm oil can be used as a comonomer to be copolymerized with elemental sulfur in an inverse vulcanization process due to its unsaturated fatty acid portion.41,42 Depending on the plant breed and also the growing environmental conditions, the percentage of different triglycerides in palm oil can vary.
28
Figure 1 shows the fatty acid composition of a typical palm oil species in which C:D represents the ratio of the number of carbon atoms to the number of C=C bonds.
11
It can be observed that palm oil is consisted of about 50 wt% unsaturated fatty acids including oleic, linoleic, and linolenic acids with 39.2, 10.1, and 0.4 wt%, respectively.
11
Fatty acid composition (wt%) of typical palm oil.
11

In our recent reported paper proceeding, we briefly presented the preliminary copolymerization of sulfur with palm oil with a limited study of different feed ratios and also a narrow investigation of the product properties. 41 Herein, a full report including the copolymerization of sulfur with palm oil using different feed ratios and a detailed investigation of their properties together with a full comparison between sulfur-palm oil and the other reported sulfur-oil polymers is presented that not only proposes a new application for palm oil and as a consequence helps the domestic palm oil industry but also suggests an innovative use of the stockpile of sulfur to reduce the environmental concern of its pollution in soil. The effect of different feed ratios on the properties of the obtained polymer is discussed. The sulfur–palm oil polymers (poly (S-Palm oil)) were produced under chosen reaction parameters (including reaction time and reaction temperature) based on the preliminary results to obtain homogenous polymeric phases without releasing a huge amount of harmful gas during the polymerization. The obtained polymers by the inverse vulcanization technique were characterized using Fourier-transform infrared spectroscopy (FTIR), differential scanning calorimetry (DSC), and X-ray diffraction analysis (XRD). A comparison study of the properties of the obtained poly (S-Palm oil) with reported bio-based sulfur polymer using other vegetable oils as monomers is outlined. Poly (S-Palm oil) suggests a potential application for the abundant and readily available palm oil which can help the domestic palm industry. The obtained poly (S-Palm oil) can be directly used for environmental-friendly applications such as mercury removal due to the affinity of sulfur to mercury,35,37 oil spill removal, 43 slow-release fertilizers, 36 and palladium capture, 30 to name a few.
Materials and methods
Materials
Elemental sulfur 99.9% and refined palm oil were purchased from PC Laboratory Reagent, Malaysia, and Malaysian local suppliers, respectively, and were used without further purification.
Copolymerization of poly (S-Palm oil)
Initial monitoring of reaction conditions helped to select the reaction conditions at which a homogenous polymeric phase can be achieved. Different wt% ratios of sulfur to palm oil, that is, 50:50, 60:40, 70:30, and 80:20, were copolymerized using inverse vulcanization. In each case, elemental sulfur was initially melted in a glass tube at 170°C employing a thermostatic oil bath with continuous stirring. After the ring-opening of sulfur chains began, observable by the color change of the molten sulfur from yellow to orange, the palm oil was gradually added to the melt to prevent a sudden decrease in the melt temperature. The reaction melt was continuously stirred for 1 h while maintaining the temperature (170°C), and subsequently, the product was let to be cooled at room temperature. As a matter of comparison, the same reaction conditions (170°C and 1 h) were utilized to produce pure polysulfide (sulfur as the only monomer) without adding palm oil as a comonomer in this case. As a reference for DSC and XRD, this pure polysulfide was tested exactly after polymerization to minimize the depolymerization of sulfur chains back to elemental sulfur. The mechanism of polymerization of sulfur with palm oil using inverse vulcanization is shown in Figure 2, based on the fact that most unsaturated fatty acid portion of palm oil consists of oleic acid and linoleic acid. Polymerization of sulfur with palm oil using inverse vulcanization.
Characterization of poly (S-Palm oil)
A PerkinElmer Frontier model spectrophotometer model equipped with Universal ATR Accessory was employed to perform FTIR in a frequency range of 4000-500 cm−1 with eight scans at a resolution of 4 cm−1. A TA Instruments Q2000 equipment was also employed to run the DSC tests under a nitrogen atmosphere in a temperature range of −50 to 200°C at a heating rate of 10 °C/min. A Malvern PANalytical X’Pert powder diffractometer provided with a Cu anode material (Kγ1 = 1.540598 Å, Kα2 = 1.544426 Å) at 40 mA in transmission geometry utilizing a capillary spinner was used to conduct the XRD measurements. The morphology of the polymers was also studied using a Zeiss EVO LS 15 microscope after the gold coating (using Emitech K550X sputter coater) of the already fractured samples in liquid nitrogen.
Results and discussion
The obtained copolymers using different feed ratios are shown in Figure 3. It can be observed that the obtained copolymers mainly consist of a dense brownish polymer phase which becomes lighter by increasing the sulfur content and a liquid phase which is assumed to be the saturated portion of palm oil that did not take part in the polymerization process. The preliminary results showed that the polymers obtained by more than 80 wt% initial sulfur content were not firm against depolymerization even in short times and thus, 80 wt% sulfur was chosen as the maximum amount of initial sulfur content. Poly (S-Palm oil)s at all studied compositions showed no solubility in conventional organic solvents including toluene, chloroform, tetrahydrofuran (THF), dimethylformamide (DMF), n-hexane, ethanol, methanol, 2-propanol, acetonitrile, and 1,4-dioxane. It is also necessary to mention that Poly (S-Palm oil)s at all percent compositions were not strongly stable against depolymerization by time, and after about a month, it can be observed that elemental sulfur is separating from the polymeric structure. This phase separation proved to be faster with increasing the initial sulfur content and in humid environments. Poly (S-Palm oil) with different feed ratios (wt%): (a) 50:50, (b) 60:40, (c) 70:30, and (d) 80:20.
Composition of poly (S-Palm oil)
Figure 4 illustrates the FTIR spectra of poly (S-Palm oil) with different feed ratios together with the spectrum of pure palm oil as a reference. The FTIR spectrum of palm oil is indistinguishable from those of other vegetable oils due to the similarity of the composition of different varieties of vegetable oils.32,39 In the case of pure palm oil, the tiny peaks at 1655 cm−1 and 3006 cm−1 are due to the vinylic C=C double bond stretching and vinylic C-H stretching vibration, respectively, while the copolymers do not show such peaks.36,39 Instead, a small peak at 965 cm−1 appeared in the case of copolymers which represents the C-H rocking vibrations in the vicinity of C-S bonds.32,39 The disappearance of the vinylic peaks together with the appearance of C-H rocking vibrations in the vicinity of C-S bonds confirmed the consumption of the vinylic C=C bonds and the formation of the Poly (S-Palm oil).36,39 Furthermore, the variation of molar ratios does not affect the FTIR spectra as the S-palm oil polymers are well-formed for all compositions. Fourier-transform infrared spectroscopy spectra for (a) palm oil and the obtained poly (S-Palm oil) copolymers: (b) 50:50, (c) 60:40, (d) 70:30, and (e) 80:20.
Thermal properties of poly (S-Palm oil)
Elemental sulfur starts to degrade at around 210°C. It was previously shown by the TGA thermograms that the degradation onset temperatures of the copolymers with 70 and 80 wt% sulfur take place sooner at around 200°C, whereas copolymers with 50 and 60 wt% initial sulfur contents show higher onset temperatures (about 230°C) compared with elemental sulfur. 41 Poly (S-Palm oil) with all feed ratios showed a different weight-loss trend in DTG curves compared with elemental sulfur which itself confirms the formation of a polymeric structure. 41 This trend for poly (S-Palm oil), regardless of its feed ratio, consisted of a rapid first degradation step (sulfur content of the polymer) accompanied by a slight second step and finally a gradual reduction in the weight percentage.31,39,41
Poly (S-Palm oil) 70:30 and poly (S-Palm oil) 80:20 due to their distinct TGA trends together with fresh pure polysulfide as a reference were selected for DSC analysis (Figure 5). Sulfur in its elemental state demonstrated two melting peaks at around 109 and 119°C due to the presence of two different crystalline structures.44,45 The copolymers also show these two peaks suggesting the incomplete conversion of elemental sulfur into the polymeric structure. The presence of these peaks might also be due to the depolymerization of some sulfur chains after some time.19,35,46 Fresh pure polysulfide, on the other hand, only reveals the second peak. Poly (S-Palm oil) 70:30 and poly (S-Palm oil) 80:20 demonstrate different melting peak areas which is a result of different amounts of unreacted sulfur in their structures.19,35,36,46 No glass transition temperature (Tg) can be observed for the copolymers in the experimental temperature range of −50 to 250°C. The copolymers are rubbery at room temperature, and hence, it is expected that poly (S-Palm oil) exhibits Tg of less than −50°C. The lack of presence of any melting and crystallization peaks related to the copolymer structure also suggests the possible amorphous structure of Poly (S-Palm oil). Differential scanning calorimetry thermograms for (a) fresh polysulfide (100:0) and the obtained S-Palm oil copolymers: (b) 70:30 and (c) 80:20.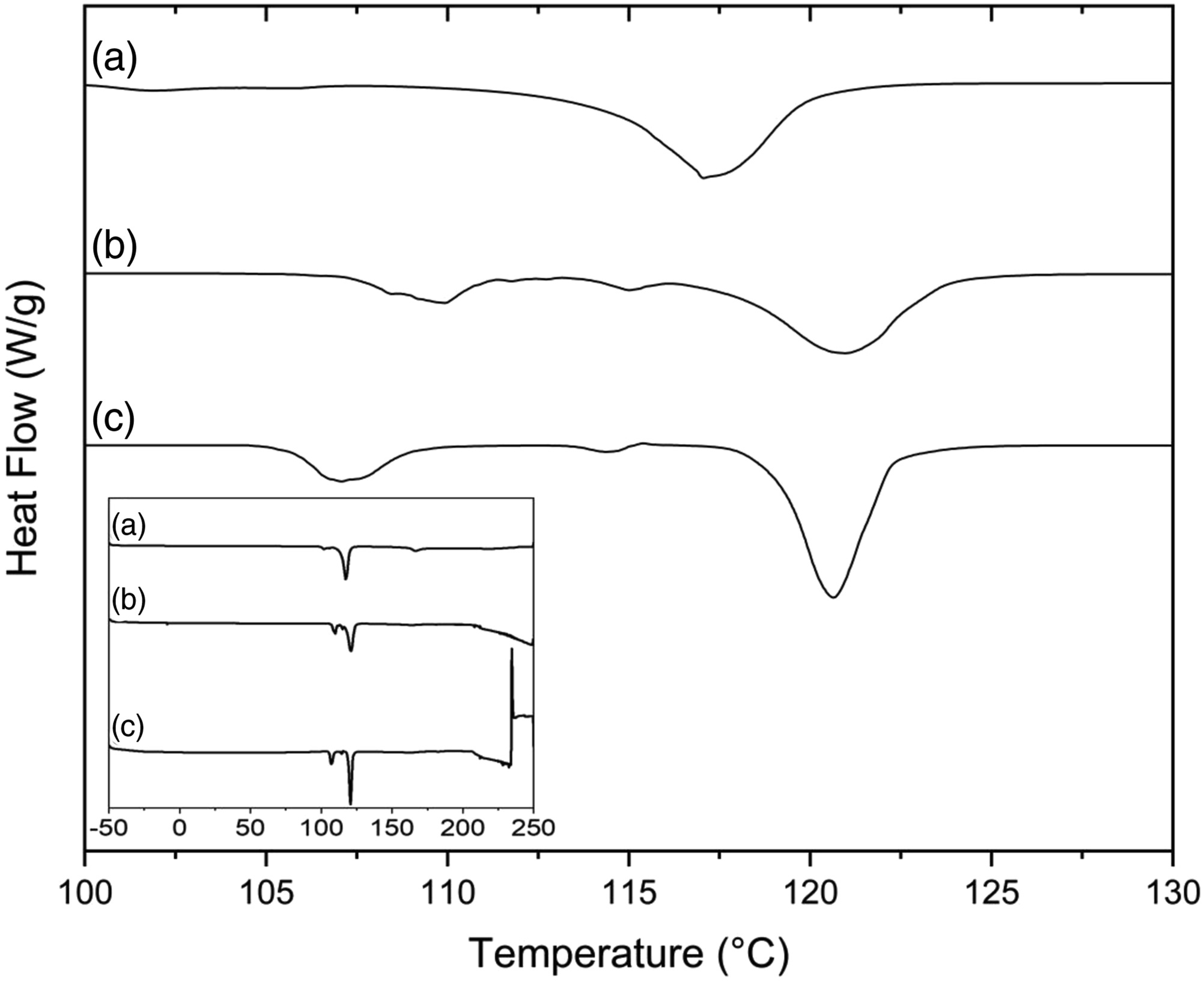
Structural properties of poly (S-Palm oil)
The XRD diffractograms of poly (S-Palm oil) with different feed ratios, elemental sulfur, and fresh pure polysulfide are presented in Figure 6. Poly (S-Palm oil) with all percent compositions and fresh polysulfide shows similar XRD patterns to that of elemental sulfur. The crystalline peaks that appeared in the case of the copolymers are due to the unreacted elemental sulfur and also that sulfur content that was initially reacted into the polymeric structure but depolymerized after some time. The absence of any crystalline peak other than those of elemental sulfur in the XRD patterns of the copolymers confirms the amorphous structure of the polymeric structure similar to other comparable sulfur-based polymers.29,47,48 The presence of elemental sulfur peaks in XRD patterns also indicates that palm oil, regardless of its feed ratio, cannot stabilize all of the initial sulfur content into the polymeric structure.19,47 X-ray diffraction analysis patterns for (a) elemental sulfur, (b) fresh polysulfide and the obtained S-Palm oil copolymers: (c) 50:50, (d) 60:40, (e) 70:30, and (f) 80:20.
Comparing poly (S-Palm oil) with other reported inverse vulcanized polymers using vegetable oils
Comparing the properties of poly (S-Palm oil) with other reported inverse vulcanized polymers using vegetable oils.

DSC: differential scanning calorimetry; XRD: X-ray diffraction analysis; THF: tetrahydrofuran; DMF: dimethylformamide.
Except for algae oil which yields a black color product in the case of low sulfur loadings, all the investigated sulfur-based polymers using vegetable oils have demonstrated different shades of brownish color which becomes lighter by increasing the initial sulfur content.32,40 Poly (S-Palm oil) showed zero solubility in conventional organic solvents such as toluene, chloroform, THF, DMF, n-hexane, ethanol, methanol, 2-Propanol, acetonitrile, and 1,4-dioxane. Besides rubber seed oil which was fully soluble in THF and partially soluble in chloroform, 34 poly (S-Canola oil) which was proved to be soluble in pyridine, 43 poly (S-Algae oil) in CHCl3, THF, and C2H2Cl4, 40 and also polymers made using sunflower, olive, and linseed oils which were partially soluble in H3C(CH2)5CH3, 39 no solubility was reported for other sulfur-based polymers using vegetable oils. In general, vegetable oils with more linolenic acid and linoleic acid portions result in highly crosslinked sulfur-based polymers.
Due to the composition similarity of different vegetable oils, the reported sulfur-based polymers using vegetable oils demonstrated similar FTIR spectra confirming the consumption of the vinylic bonds and the formation of the polymeric structure. 27 TGA thermograms of poly (S-Palm oil) with different percent compositions showed a two-step degradation pattern similar to most of the reported polymers.32,37,38,40,41 However, polymers made by using rubber seed, sunflower, olive, and linseed oils showed a three-step weight loss pattern.33,34,39 The first weight loss onsets at around 200–230°C for all the reported polymers. Just like poly (S-Corn oil), 32 poly (S-Soybean oil), 36 poly (S-Sunflower oil), 39 poly (S-Linseed oil), 39 poly (S-Olive oil), 39 and poly (S-Cottonseed oil), 38 no Tg was detected in the case of poly (S-Palm oil) in the experimental temperature range; nevertheless, poly (S-rubber seed oil),33,34 poly (S-canola oil), 37 poly (S-algae oil), 40 poly (S-Rice bran oil), 37 and poly (S-Castor oil) 37 (all with 50 wt% initial sulfur content) exhibited glass transition temperatures at −6.41, −17, 32, −13, and −24°C, respectively. Regardless of the feed ratio, vegetable oils, in general, are not capable to stabilize all the initial sulfur content against depolymerization over time, and thus, melting peaks resembling the phase transition of different crystalline structures of elemental sulfur can be detected in the DSC thermograms of their resultant sulfur-based polymers. The area of these peaks can be utilized to calculate the exact amount of elemental sulfur left embedded in the copolymers.32,36,37 Except for poly (S-algae oil) 40 with 50 and 70 wt% initial sulfur contents, other reported sulfur-based polymers using vegetable oils showed peaks identical to elemental sulfur in their XRD patterns which are in line with the DSC results. This indicates the amorphous nature of these polymers and the presence of unreacted or depolymerized sulfur in the structure of the polymers. All in all, utilizing vegetable oils as comonomers in inverse vulcanization results in brown color amorphous highly crosslinked thermosetting polymers with some unreacted or depolymerized sulfur particles embedded inside the structure.
Conclusion
The copolymerization of palm oil with sulfur using inverse vulcanization was reported. 170°C and 1 h were chosen as reaction conditions based on the preliminary results to copolymerize sulfur with palm oil using different percent compositions (S:Palm oil), that is, 50:50, 60:40, 70:30, and 80:20. Fourier-transform infrared spectroscopy, DSC, and XRD were employed to investigate the properties of the obtained copolymers. Poly (S-Palm oil) demonstrated a brownish color solid phase, which became lighter by increasing the initial sulfur load, and a liquid phase consisting of the saturated portion of palm oil. The obtained polymers showed no solubility in conventional organic solvents. Poly (S-Palm oil) is not firm against depolymerization after a long period and only after a few months, the phase separation of the reactants can be visually observed. Fourier-transform infrared spectroscopy confirmed the successful reaction of the monomers and the formation of the polymeric structure. In general, the obtained polymers with all percent compositions demonstrated a high thermal stability. Poly (S-Palm oil) with all feed ratios showed some amount of unreacted or depolymerized elemental sulfur confirmed by DSC and XRD indicating that palm oil regardless of its weight ratio is not able to stabilize all the sulfur content into the polymeric structure. The obtained poly (S-Palm oil) suggests a new application for the openly stored elemental sulfur in gas and petroleum refineries and thus helps reducing its environmental effects while also proposes a new application for palm oil that directly boosts the palm oil industry. Poly (S-Palm oil) as it is can be utilized in several applications such as mercury removal, oil spill removal, slow-release fertilizers, and palladium capture.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by Yayasan Universiti Teknologi PETRONAS (Y-UTP) through cost centers 015LC0-080 and 015LC0-065.
