Abstract
Sulfur-based polymers are unique renewable materials that are receiving a growing attention. The utilization of elemental sulfur with a variety of monomers in their preparation in the absence of solvents using the inverse vulcanization are granting them green nature and unique properties. Several characterization techniques have been used to evaluate the properties of sulfur-based polymers. However, the complex structure and lack of solubility undermine the applicability of some standard characterization techniques in the usual manners. This article reviews the characterization methods used for the evaluation of various properties of sulfur-based polymers such as chemical, morphological, structural, thermal, rheological and mechanical properties, all of which vary depending on the type of comonomer involved in the reaction and heat treatment conditions. The successful applications of different characterization techniques including Fourier transform infrared spectroscopy (FTIR), Raman spectroscopy, nuclear magnetic resonance (NMR), scanning electron microscopy/X-ray energy dispersion (SEM-EDX), X-ray diffraction (XRD), mechanical tester, rheometer, thermal gravimetric analyzer (TGA) and differential scanning calorimetry (DSC) are discussed. The challenges to the evaluation of the properties of sulfur-based polymers and the innovative applications of the conventional techniques to overcome them are also deliberated.
Keywords
Introduction
The heavy reliance on plastics from petro-based monomers in various life aspects of modern societies continues to attract growing environmental concerns. 1 According to the Statista, about 359 million metric tons of plastic were produced in 2018. 2 This situation has led to a rise in the interest to develop alternative sustainable polymers, and thus, immense efforts have been carried out to search for monomer substitutes from renewable and waste sources. Several monomers such as carbon dioxide, lactic acid, tannins, terpenes, vegetable oils and bacterial cellulose have been proposed as feedstocks for preparation of a variety of sustainable polymers and products.3–6
Sulfur-based polymers or inverse vulcanized polymers are a new class of polymers that have been recently emerged after realizing that elemental sulfur behaves as a monomer initiating polymerization when heated to its molten state with a hydrocarbon source having diene groups through a mechanism called inverse vulcanization. 7 This technique is the opposite of vulcanization, which is the most famous and widely used process for crosslinking of natural rubber in medical gloves industry. 8 The key feature of inverse vulcanization is in the direct polymerization of melted elemental sulfur with vinylic monomers to form copolymers with unique properties. 9 The obtained polymers can be used directly or serve as intermediates for further functionalization treatments leading to new functional polymeric materials for various energy and environmental applications.10,11
Sulfur powder is continuously produced in many petroleum and natural gas refineries in various parts of the world. Thus, it is highly abundant and cheap (100 USD/Tonne).12,13 Sulfur is currently under-utilized despite its various applications in manufacturing multiple products such as sulfuric acid, matches, lead-acid batteries, acidic drain cleaners, fertilizers, fungicides, pesticides, firecrackers and pharmaceuticals.14,15 However, the excessive quantities of the sulfur powder that continues to pile up in oil and gas refineries are still causing environmental and health concerns when stored in open huge pile stock. 16
Elemental sulfur is a challenging raw material for chemical reactions due to its presence in an octet structure, causing its inherently inferior physicochemical property such as inertness and low solubility in most common solvents. 17 Therefore, the development of chemistry and processes allowing the conversion of elemental sulfur to a variety of new materials can eliminate its environmental concern and open new venues for utilization as a feedstock for the development of useful materials with added values in addition to fostering green chemistry approach.10,18
Elemental sulfur primarily exists as eight-membered ring that melts at about 159°C forming thiyl radical chains with bi-radical ends forming linear sulfur polymers (polysulfanes). 19 These polysulfanes are chemically unstable and readily depolymerize back to sulfur octet rings. However, polysulfane could be stabilized by applying the inverse vulcanization method in which the thiyl radical chains in liquid molten sulfur could directly copolymerize with the unsaturated part of added diene-containing hydrocarbon co-monomers of different types.7,10,20 Several petro-based monomers such as 1,3-diisopropenylbenzene (DIB), 7 divinylbenzene (DVB),21–23 1,3,5-triisopropenylbenzene (TIB), 24 cycloalkenes, 25 styrene, 26 1,4-diphenylbutadiyne (DiPhDy), 27 bismaleimide, 22 vinyl aniline, 28 and vinylbenzene chloride, 29 were reacted with elemental sulfur to develop various sulfur-based polymers. Renewable monomers such as farnesene, farnesol, 30 myrcene, 31 perillyl alcohol, 5 squalene, 5 cardanol 32 and vegetable oils33–38 were also copolymerized with elemental sulfur. More details on the types of various monomers that have been copolymerized with elemental sulfur by inverse vulcanization can be found elsewhere.11,20
Even though sulfur-based polymers are in their infancy, they have been found promising in various applications such as environmental remediation (removal of hydrocarbon 38 and heavy metals30,35,36,39,40 from wastewater), energy storage in a form Li-S batteries,23,32,33,26 fertilizers,34,37 optical devices, 41 healable polymeric materials,42,43 in addition to many unfolded applications. These copolymers are challenged by several issues that need to be resolved to get them well established and propagate their applications. For instance, the complex structures of sulfur-based polymers and their existence in different types depending on reacting co-monomers, i.e. elastomers, 29 thermosets21,24,33–35 and thermoplastics7,21,42,44 made it difficult to establish their properties using standard characterization techniques. Particularly, the common problem among most sulfur-based polymers is the lack of solubility in common solvents and the challenging processability, both of which could have limiting impact not only on the selection of characterization methods but also on applications of these polymers.
Few reviews summarizing the previous studies related to developments of sulfur-based polymers and their applications have been published.9–11 Particularly, these reviews were dedicated for describing the inverse vulcanization method, 9 types of reacting monomers for copolymerization of sulfur and the applications of various sulfur-based polymers.18,20 However, there is no publications devoted explicitly for reviewing the characterization methods used to evaluate the properties of these copolymers and their suitability for providing a complete picture needed not only to establish the properties of the inverse polymerized polymers and identify their appropriate applications but also to determine their operating conditions and disposability as well as recycling.
The objective of this article is to review the current polymer characterization techniques used for the evaluation of the properties of the sulfur-based polymers and discuss the challenges undermining the establishment of clear understanding of physical and chemical picture of these polymers. The scope of this article includes an overview of the recent advances in sulfur copolymerization by inverse vulcanization method and the effect of the sulfur composition on the properties of copolymers. Summaries of the properties of the investigated inverse vulcanized polymers are also presented. The challenges to the characterization methods and future directions to establish sulfur-based polymers’ properties are also discussed. This review provides the polymer community with a vital information to identify suitable characterization techniques for evaluation of the properties of the newly emerging sulfur-based polymers.
Recent advances in sulfur polymerization by inverse vulcanization
The development of sulfur-based polymers has gained an increasing attention after they have been found not only contributing to environmental remediation and waste utilization but also adding value and promoting green chemistry for the development of sustainable polymers. Many studies have reported the development of these polymers using inverse vulcanization method. Various types of monomers have been copolymerized with elemental sulfur at molten conditions and yielded polymers with diverse properties.
The inverse vulcanization reaction involves the reaction between molten sulfur and vinylic monomer without any solvent or initiator. This method can be extended to any di-vinylic, multi-vinylic, or alkynyl monomers, which are suitable for reaction with thiyl radicals (linear bi-radical sulfur chains). 9 1,3-Diisoproenylbenzene (DIB) was the first monomer investigated for this process by Chung et al. 7 The mechanism for the reaction of DIB and molten sulfur via inverse vulcanization is shown in Figure 1. The copolymer of DIB and S8 was found to be chemically stable and allowed processing high sulfur content with controllable structural and thermal properties by just manipulating the comonomer feed ratio. 7 For instance, a copolymer with a low DIB composition (0–15 wt%) has a semi-crystalline structure and a low glass transition temperature (Tg). However, the increase in the DIB content, increased the Tg and the obtained copolymer became amorphous. Chung et al. 7 also used α-methyl styrene as a comonomer to prepare another sulfur enriched copolymer. The copolymer was unstable and found to have a linear structure. The instability of this copolymer was investigated by size exclusion chromatography (SEC), which revealed that a large amount of unreacted sulfur in the copolymer caused a depolymerization followed by a crystallinity increase. These findings confirmed that this process preferably work with di-vinylic, multi-vinylic or alkynyl monomers, which can undergo thiolene/thiolyne reactions with molten sulfur. 10 Inverse vulcanized copolymers obtained from reaction of elemental sulfur with different comonomers are shown in Figure 2.

Mechanism of formation of chemically stable sulfur-based copolymer by inverse vulcanization of elemental sulfur and DIB. 18

Photographs of (a) poly(S-r-vinylbenzene chloride), (b) poly(S-r-limonene), and (c) poly(S-r-Palm oil). 39
Later, many researchers got attracted to this new polymerization method and investigated different types of monomers, which can be classified as petro-based monomers, renewable monomers, synthetic hybrid monomers and vegetable oils as alternative monomers.
A semi-interpenetrating network structured copolymer was obtained by inverse vulcanization of 1,3-diethyl benzene with sulfur, which improved the electrochemical properties and suppressed the dissolution of polysulfide out of the cathode. 45 Properties of the copolymers resulted from inverse vulcanization could be modified using post-polymerization techniques, which is another merit of this technique as reported by Dirlam et al. 46 These authors reacted styrenic functional propylenedioxythiophene (ProDoT-Sty) and DIB as a commoner followed by the electro-polymerization of ProDoT side chain to form interpenetrating polymer with low electrical resistance. Hasell et al. 47 reported an increase in the surface area of the copolymer resulted from reaction of dicyclopentadiene (DCPD) and sulfur by KOH activated carbonization. Other methods to increase the surface area includes foaming by supercritical CO2, 48 electrospinning 49 and through slat porogen. 40
Copolymerization of the 1,3,5-triisopropenylbenzene with sulfur by inverse vulcanization was also reported. The obtained copolymer was highly crosslinked and had high glass transition temperature (Tg = 68–130°C). 24 The thermomechanical properties of the resulted copolymer could be improved by introducing more functional groups in the reacted comonomer, which allowed high sulfur content and formation of S–S dynamic covalent bonds. 24
Renewable monomers including cardanol benzoxazine (agro-waste from cashew nut), 32 limonene (waste of citrus industry),39,50 myrcene, farnesene (essential oils of plants), farnesol (animals and plants), 30 squalene (shark liver oil), 51 perillyl alcohol (essential oils of plants), 51 eugenol allyl ether (clove) 52 and diallyl disulfide (onion and garlic) 31 were also investigated for inverse vulcanization. The resulted copolymers were found to be chemically stable and possessed tunable properties allowing them to be investigated for water purification 30 and making polymeric cathode for Li-S batteries 32 Despite the green nature of these renewable monomers and their promising products, they are currently expensive, and the cost to extract them in practical quantities remains the main hurdle.
Alternative cheap monomers were inverse vulcanized with sulfur such as vegetable oils (linseed, olive, sunflower, canola, and soybean oils)33–35,37 in several occasions. Nevertheless, these interesting alternative monomers have an obvious limitation caused by their incomplete copolymerization with sulfur. Notably, 40–50% sulfur remained unreacted and left embedded in the structure of the copolymer.35,36 The reason for this observation is not well known because of the lack of information available for the mechanism of the reaction. Nevertheless, the unique properties of these green copolymers were explored and investigated for many applications after evaluation of their properties.
Li-S battery, which is considered to be an attractive alternative to conventional batteries with a high theoretical capacity (1672 mAh g−1) and specific energy (2500 Wh kg−1) but unfortunately facing many challenges. This is due to the intractable nature of sulfur, active material loss and diversity in solubility and volume of the Li-S redox products during phase conversion reactions between S8 and Li2Sx.53,54 The promising features of the inverse vulcanized copolymers address these problems by suppressing the deposition of the intractable low-order lithium polysulfides (Li2Sx (x ≤ 2)) due to the formation of organosulfur discharge products. 55 Semi-interpenetrating network structure of these copolymers obtained by inverse vulcanization diethylbezene (DEB) with liquid sulfur were found reduce the loss of the cathode active material 45 and the cathodes fabricated using these copolymers showed low electrical resistance as compared to S8. 46
The presence of heavy metals like mercury, palladium, and iron in water or soil are posing a threat to both aquatic and terrene life with terrible effects on the environment, human health, and economy.56–58 Inverse vulcanized copolymers with canola oil, 35 limonene 39 and perillyl alcohol or squalene 51 were investigated as adsorbents for environmental decontamination.35,39,51 These copolymers showed high selective affinity for mercury, palladium, and iron. 30 The merit of this new copolymers is using ingredients (sulfur and limonene) obtained from industrial waste.35,36 The adsorption kinetics of such copolymer was improved by increasing the surface area. 47 Few methods were proposed to increase surface area by creating porous structures in these copolymers by using porogen salt (NaCl),35,38 supercritical CO2, 48 and electrospinning. 30 The copolymers were used for coating silica gel and the obtained adsorbent showed improved kinetics and high adsorption capacity for mercury. 59
Inverse vulcanized copolymers from sulfur and 1,3,5-triisopropenylbenzene, 24 DIB, 41 tretravinyltin 60 and selenium with DIB 61 were also reported to have high refractive index ranging from n = 1.865 to 1.845. They can transmit light of near-infrared (1550 nm) and mid-infrared (3–5 micrometer) wavelengths compared to other polymeric materials. This feature provides an opportunity to extend the uses of these copolymers to optical applications such as thermal imaging. 60 Moreover, these copolymers can be also healed by thermal annealing at 100°C,42,44 which allows the fabrication of repairable lenses. 62 These copolymers can be also used in development of controlled-release fertilizers, 37 biodegradable polymers, 34 adsorbents for CO2 capturing 63 and oil spill remediation 38 and antimicrobial surfaces. Indubitably, as this field swiftly grows, these copolymers will find more new and unique applications.
Characterization of inverse vulcanized copolymers
Inverse vulcanized copolymers has recently received an increasing attention after realizing their potentials in various applications leading to publication of many reports in literature. 11 However, all the published reports did not present a complete evaluation of the properties of the copolymers and primarily focused on the preparation under sulfur contents in the range of 50–90 wt% followed by partial characterization. Later, the authors extended their attention for testing the obtained copolymers for specific applications without displaying a complete picture of structure and properties. The progress in the evaluation of the polymer properties and the related characterization techniques are reviewed in the following sections.
Solubility
The solubility of the inverse vulcanized copolymers depends on the monomer’s nature and the overall structure of the developed copolymer.10,11,18 Practically, the solubility decreases with the increase in the unsaturation in the monomer used for copolymerization with melted sulfur. For example, the copolymers obtained from the reaction of sulfur with DIB (which possesses two vinylic groups) are soluble in common organic solvents (tetrahydrofuran and chloroform). 7 Unlikely, the copolymers resulted from the reaction of sulfur with TIB (which possesses three vinylic groups) are insoluble in all solvents. 24 The solubility of the copolymers also decreases with the increase of the sulfur content, as in the case of poly(S-r-DIB). For instance, poly(S-r-DIB) copolymer with ≤50 wt% sulfur in the reaction mixture is soluble in organic solvents, but with >50 wt% sulfur, they are insoluble in all solvents.7,48
The solubility of inverse vulcanized copolymers also depends on the level of crosslinking in their structures. For example, the linear copolymer obtained from the reaction of sulfur with styrene was soluble in organic solvents (toluene, chloroform, tetrahydrofuran, and dichloromethane) 26 unlike those obtained by copolymerization of DCPD and 5-ethylidene-2-norbornene (ENB) with sulfur, which were found highly crosslinked and insoluble in all solvents.30,64
The copolymers resulted from renewable monomers are mostly soluble in organic solvents like those obtained from copolymerization of sulfur with farnesene, farnesol, myrcene, and cardanol. Particularly, these copolymers were found soluble in an organic solvent such as tetrahydrofuran suggesting that the structure is linear or not highly crosslinked.30–32 The solubility of these copolymers explains the reason for having low glass transition (Tg) temperature and low apparent molecular weights. On the other hand, copolymers from sulfur and vegetable oils exhibited poor solubility in some organic solvents (n-hexane and CDCl3) suggesting high levels of crosslinking.33–35,38 A summary of the solubility studies reported for inverse vulcanized copolymers reported in literature is shown in Table 1. It can be concluded that the number of vinylic groups in the monomer copolymerized with sulfur dictates the level of the crosslinking in the structure of sulfur-based polymers and their solubility in most solvents a function of the degree of crosslinking in turn. Thus, the poor solubility of these polymers is always associated with higher crosslinking levels and vice versa.
Summary of previous solubility studies for the inverse vulcanized copolymers reported in literature.

Molecular weight
Polymers are characterized by their molecular weight, which represents the sum of masses of individual atoms comprising the polymer molecules present at different lengths and thus it is defined as distribution instead of a specific number. The molecular weight of a polymer determines its physical properties including melt viscosity, mechanical strength, viscosity, chemical and thermal resistance and glass transition temperature. It is normally characterized by weight average molecular weight (MW), number weight average molecular weight (Mn) and polydispersity (MW/Mn). The molecular weight of the inverse vulcanized copolymers has been evaluated using gel permeation chromatography (GPC) and size exclusion chromatography (SEC) which require the copolymers to be solubilized in an organic solvents such as tetrahydrofuran (THF).7,31,52,65,66 The majority of inverse vulcanized copolymers showed an inverse relationship between molecular weight and the copolymers’ solubility, i.e. as the molecular weight of the copolymer increases because of intensified crosslinking, the copolymers become insoluble in solvents disallowing GPC and SEC analysis. 66 Only, few inverse vulcanized copolymers are known to be soluble in organic solvents, which allows molecular weight evaluation.
For example, the copolymer obtained by inverse vulcanization of sulfur and DIB at equal mass ratio was found to be soluble in THF allowing SEC analysis against polystyrene standards. The obtained copolymer was found to possess low number average molecular weight and high polydispersity. 7 Other copolymers obtained using renewable monomers such as farnesene, limonene, cardanol, eugenol allyl ether (EAE), perillyl alcohol, diallyl disulfide (DADS) and diallyl sulfide (DAS) were evaluated using GPC technique, which showed that these copolymers possess low molecular weights which slightly decreases with the increase of sulfur content.30–32,39,51,52,66 A summary of studies on evaluation of the molecular weight of the inverse vulcanized is presented in Table 2. It can be observed that the investigated inverse vulcanized copolymers apparently possess low molecular weights as compared to conventional polymers. However, the majority of these copolymers have heavily crosslinked structures that rendered them insoluble in solvents and thus their characterization using SEC and GPC techniques becomes rather difficult.
Summary of previous molecular weight studies of the inverse vulcanized copolymers.
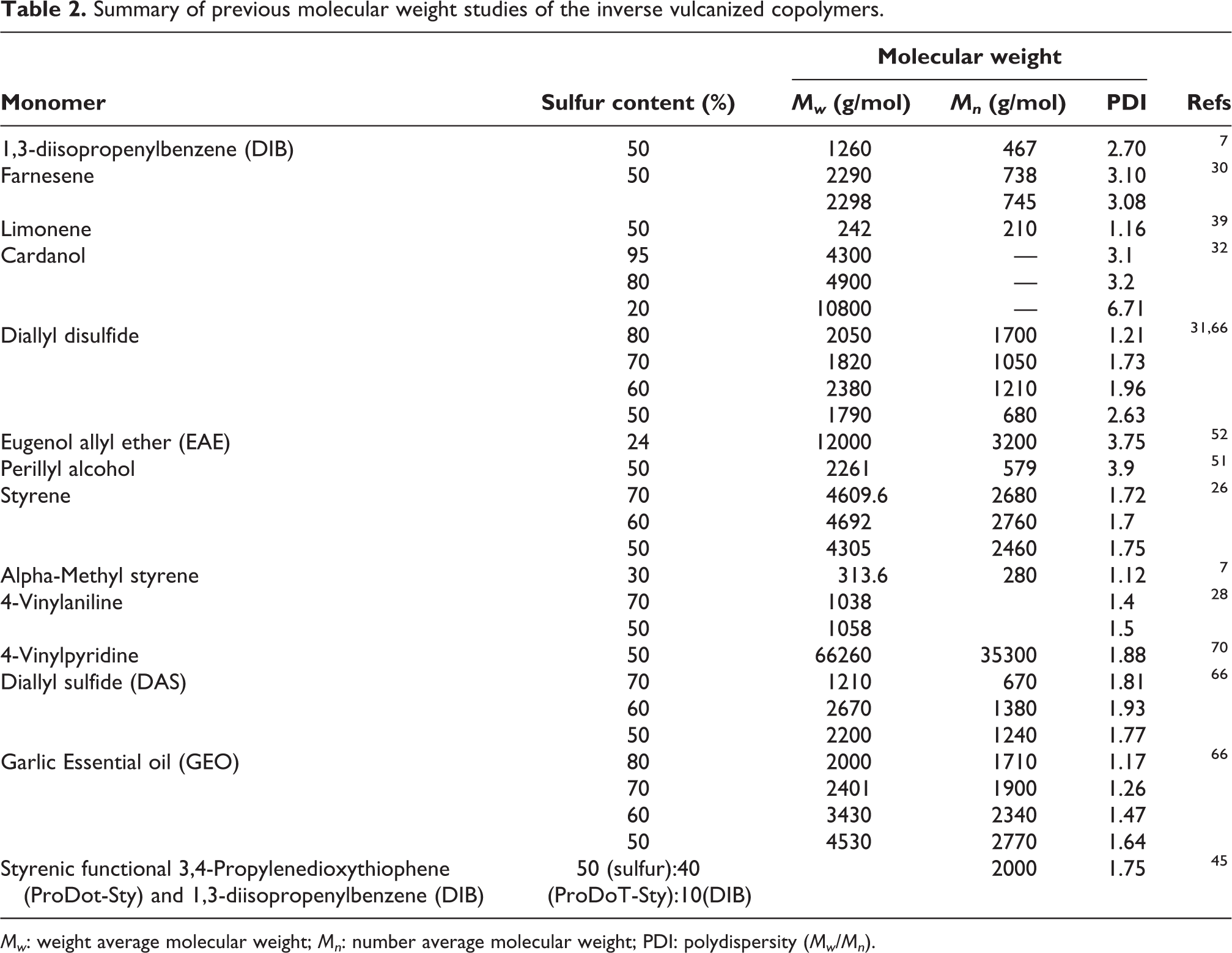
Mw: weight average molecular weight; Mn: number average molecular weight; PDI: polydispersity (Mw/Mn).
Chemical structure
The chemical composition and changes in the structure of the polymers are well known to determine not only physical properties such as crystallinity, glass-transition and melting temperatures but also their thermal stability, rheological behavior, mechanical response and operating conditions of the polymers. FTIR, Raman spectroscopy and NMR have been used to evaluate the changes in the chemical structure of the inverse vulcanized polymers.
Particularly, these techniques were used to confirm the reaction between the sulfur and the vinylic groups originated from hydrocarbon monomers and provide evidence for the formation of the sulfur-rich copolymers. Explicitly, FTIR has been widely used to confirm the reaction between the unsaturated part of the organic comonomer with sulfur, which was evident by the disappearance of the vinylic signals and the appearance of the new signals associated with C–S bond formation in the copolymer. The FTIR spectra of monomers or vegetable oils normally show signals in the range of 1600–1658 and 3000–3100 cm−1, which are attributed to C=C and C=C−H stretching, respectively. These features are depicted from the typical FTIR spectra of the copolymer obtained by inverse vulcanization of soybean oil and molten sulfur, which is compared with that of pure soybean oil as shown in Figure 3(a). The disappearance of the cis-alkenes characteristic signals in the spectra of the sulfur-rich copolymers provides an evidence for the successful formation of the copolymers.25,30–32,34–38,45,64,66,68,69,71,72
Despite the effectiveness of FTIR spectra in showing the signals for the functional group, it failed to illustrate the location of the functional group. For instance, divinylbenzene (DVB) and 4-vinyl pyridine (4-VP) are two monomers that have been investigated for the inverse vulcanization with sulfur. The spectra of these two pristine monomers showed signals at 1650 cm−1, which are associated with the C=C stretching vibration. Interestingly, these signals did not vanish and appeared weaker at the same range in the spectra of their corresponding copolymers. This is due to the presence of C=C of benzene rings in the monomers’ structures, which did not take part in the reaction as shown in Figure 3(b).70,71 This was coupled with the difficulty in detecting C−S bonds, and this suggests that FTIR is not a sufficient to provide strong evidence for the formation of the sulfur-based polymers by inverse vulcanization method using such aromatic monomers.70,71

1H NMR technique was used to characterize the copolymer obtained by inverse vulcanization of 1,3-diisopropenylbenzene (DIB) and sulfur. Pristine DIB contains signals for methyl and methylene groups attached to the benzene ring. After copolymerization, the structure of the copolymer showed signals for the aromatic protons (6.8–7.8 ppm) (due to benzene ring), and methyl proton (2.9–3.4). However, the signals for methylene proton were shifted downward (increasing chemical shift), which is likely caused by the C–S bond formation. 7 1H NMR was found effective in distinguishing the aromatic C=C from the aliphatic C=C as illustrated in Figure 4 which shows 1H NMR resonance spectra poly(S-r-DVB) compared to pristine DVB. The resonance spectrum of pristine DVB show signals for both the aliphatic C=C and C=C for benzene rings whereas the spectrum for poly(S-r-DVB) shows signals for the benzene ring only. The disappearance of the aliphatic C=C signals indicates the successful formation of the copolymer. Although 1H NMR is effective in providing evidence for formation of the copolymers but it is limited for the few sulfur-based polymers which are soluble in organic solvents. Thus, 1H NMR analysis is challenged by the poor solubility of the majority sulfur-containing copolymers. For instance, the copolymers obtained from sulfur with DCPD, TIB and vegetable oils have poor solubility in organic solvents and thus their characterization 1H NMR was undermined.24,30,33–35,38 To overcome this problem, solid-state NMR was employed to characterize such insoluble sulfur enriched copolymers. The signals for C–S bond were more potent to confirm the formation of the copolymer obtained from reaction of DIB with molten sulfur. Comparatively, no signals for C–S bond were detected by liquid 1H NMR instead, it was rather confirmed by the signals for downfield shifted methylene. However, the resonance spectra of the same copolymer obtained using solid-state NMR showed strong signals at 48 ppm, which is ascribed for the C–S bond.7,42,44 The chemical structures of other sulfur-based copolymers obtained using eugenol allyl ether (EAE), styrene, algae oil, DCPD, and vegetable oils as comonomers, were also investigated using solid-state NMR, which confirmed the consumption of the vinylic group and formation of C–S bonds in the copolymers.26,30,33,52

1H NMR spectra of (a) pristine DVB and (b) poly(S-r-DVB). 23
Raman spectroscopy is another technique used to investigate the inverse vulcanized copolymers. It is more powerful to detect the homonuclear bonds than FTIR, which makes this technique useful to distinguish between the C–C, C=C and C≡C bonds.73–75 Chalker and coworkers 36 used this technique to characterize the copolymer obtained by inverse vulcanization of sulfur and canola oil and the spectra as shown in Figure 5, canola oil alone showed signals for C=C and C=C–H at 1613 cm−1 and 3035 cm−1, respectively. However, the copolymer showed no signal for the cis-alkene character. Instead, it showed signals at 432 and 470 cm−1, which are associated with S–S bonds and additional characteristic signals at 1400 and 2900 cm−1, which are due to the oil domain in the copolymer. 36 It can be concluded that a combination of spectroscopic analysis including FTIR and Raman or NMR should be used to give a strong evidence for the formation of the inverse vulcanized polymers.
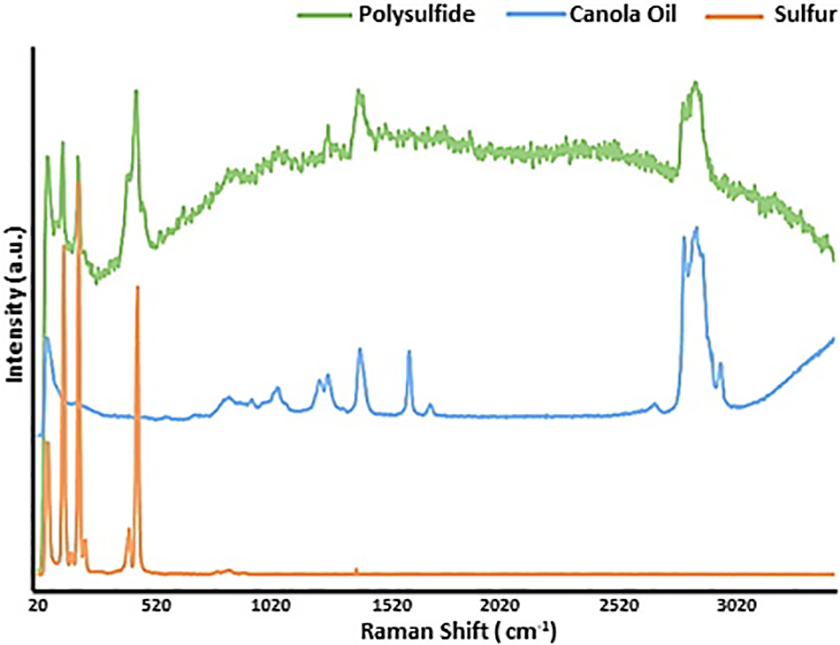
Raman spectra of elemental sulfur, canola oil and poly (S-r-Canola oil). 38
Morphology
The inverse vulcanized copolymers are mostly amorphous in nature with majority of them having crosslinked structures and crystals of embedded unreacted sulfur depending on the sulfur content and reaction conditions. The crystal fraction was found to increase with the increase of sulfur content in the copolymer. The structure and morphology of the inverse vulcanized copolymer were studied using powder X-ray diffraction (XRD) and scanning electron microscopy (SEM). Several studies have reported the use of XRD to investigate the structure of the inverse vulcanized copolymers obtained by reacting sulfur with monomers such as DCPD, cycloalkenes, diallyl disulfide (DADS), squalene, perillyl alcohol, algae oil, cardanol benzoxazines, soybean oil, farnesene, farnesol and myrcene.25,30–32,34,51,68 Elemental sulfur was found to have a crystalline structure and showed a number of crystallinity peaks at 2θ = 23, 27, 28, 53, and 56. 76 On contrary, the XRD pattern of the inverse vulcanized copolymer showed no crystalline peaks. Instead, it showed peaks representing unreacted sulfur trapped in the copolymer increasing the crystallinity depending on the sulfur concentration in the reaction mixture and the type of the monomer used. For example, the copolymers containing sulfur ≤80 wt% S possess an amorphous structure, as XRD showed no crystallinity peaks.25,51 However, sulfur-crystallinity peaks were observed in diffractogram presented in Figure 6(a). These peaks resemble α-S8, β-S8 and γ-S8 in the copolymers, which their intensities increase with higher sulfur contents (≥90 wt% S in case of petro-based monomers) and lower sulfur content (≥70 wt% S) in case of renewable monomers. 51 The copolymers obtained using vegetable oils as comonomers showed a different behavior as shown in Figure 6(b) where the crystallinity peaks were observed at all sulfur concentrations with their intensities rising with the increase in the sulfur content in the reaction mixtures.25,30–32,34,51,66,68 This further confirms that inverse vulcanized copolymers are mostly amorphous regardless the type of comonomers and suggests the apparent crystallinity representing the unreacted sulfur in the copolymers depends mainly on the sulfur contents in the reaction mixtures.

SEM images of the inverse vulcanized copolymers resulted from copolymerization of sulfur with DADS, BA-ala, 4-vinylpyridine and cycloalkenes monomers were investigated and showed smooth surfaces with the absence of phase separation and crystals, which indicates the successful conversion of the crystalline sulfur to amorphous copolymers. Unlikely, the SEM images of the inverse vulcanized copolymer obtained from vegetable oil and sulfur shown in Figure 7(a) and (b), revealed the presence of two different phases in forms of smooth surface with embedded isolated sulfur particles similar to original elemental sulfur. This observation further confirms XRD findings pertaining unreacted sulfur representing crystalline fraction detected in sulfur-based polymer. Similar findings were observed from confocal Raman spectroscopy, which revealed the presence of high sulfur containing areas in the copolymer when sulfur reacted with vegetable oils. 35 The energy dispersive X-ray spectroscopy (EDS) analysis of these areas also showed very high sulfur content.33–38 These findings suggest that SEM is an effective tool to confirm the level of sulfur conversion in the copolymers and it is rather more useful for the investigation of the morphology of the sulfur-based polymer obtained from vegetable oils.

SEM images of (a) elemental sulfur and (b) copolymer of sulfur and vegetable oil (50:50). 34
Mechanical and rheological properties
Mechanical and rheological properties of the inverse vulcanized copolymers are highly unexplored. The mechanical properties of the inverse vulcanized copolymers depend on the level of unsaturation of the monomer and the organic content in the copolymer. 21 Poly(S-r-DIB) with a 70 wt% sulfur showed a tensile strength of 2.33 (±0.15) MPa and requires 2.77 × 105 (±1.34 × 105) J/m3 tensile energy to break. 7 However, the poly(S-r-TIB) with the same composition of sulfur showed a tensile strength of 19.5 (±3.9) MPa which requires 36.13 × 105 (±1.35 × 105) J/m3 tensile energy to break. 24 This indicates that the increase in the vinylic groups of the monomer increases the crosslinking, which results in better mechanical strength.
Inverse vulcanized poly(S-r-DIB) was found to be more ductile than poly(S-r-DVB), as indicated by the ultimate stress and fracture point. Poly(S-r-DIB) with 70 wt% S can bear 0.04 MPa stress without failing and fracture point is 246.9% as compared to poly(S-r-DVB) with 70 wt% S, which showed 0.47 MPa ultimate stress and 54.51% fracture point. 21 The stress–strain curves for poly(S-r-DIB) shown in Figure 8 displays a non-linear elastic range. 21 The elongation % was found to increase with the increase in the amount of the DIB monomer in the reaction mixture. Inverse vulcanized copolymers become brittle with the increase of sulfur content. Moreover, the stress–strain curves show (Figure 8) that poly(S-r-DIB) with 85 wt% S is brittle with very little elongation, whereas the copolymer with 70 wt% S is ductile with the strain reaching the value of >400%. No obvious trend was observed between the incorporated amount of sulfur and shore hardness of these copolymers. 21 Thus, sulfur-based polymers are softer in terms of mechanical properties than conventional polymers and possess low toughness, low strength and high ductility. 21 Mechanical properties of inverse vulcanized copolymers reported in literature are summarized in Table 3. It can be concluded that very little investigations were dedicated to mechanical and rheological properties, despite a large number of studies on preparation sulfur-based polymers. Therefore, more work is highly needed to uncover the mechanical properties of these new copolymers.

Stress–strain curves of inverse vulcanized poly(S-r-DIB) with different wt% of DIB. 21
Summary of mechanical properties of inverse vulcanized copolymers reported in previous studies.

The rheological properties of inverse vulcanized copolymers were not given research attention. A rare report on rheological characterization of poly(S-r-DIB) revealed that it bears a thermoplasticity. This was indicated by the terminal regions at low frequency revealing that the structure of poly(S-r-DIB) becomes hyperbranched with 50 wt% sulfur compared to 70 and 90 wt% loadings in reaction mixture. 7 This is supported by the observed values of zero shear viscosity’s (ηo), plateau modulus’s (GN°) and relaxation time’s (τ), which were found to be 0.427 MPa.s, 0.198 MPa and 2.152 s, respectively. The corresponding values with 70 wt% sulfur was found to be ηo = 3.102 MPa.s, GN° = 0.861 MPa and τ = 3.603 s whereas those for 90 wt% sulfur were 0.152 MPa.s, 0.28 MPa and 0.542 s for ηo, GN° and τ, respectively. 7
Thermal properties
Thermal properties such as Tg, melting enthalpy and melting temperature are essential in determining the behavior and type of polymers, which are measured by differential scanning calorimetry (DSC). The thermal properties of sulfur-based polymers were studied using DSC in many occasions.6,7,24,30,34,35,39,45,51,68,71,77 It was reported that no melting peaks could be observed in the DSC thermograms of the inverse vulcanized copolymers indicating that these copolymers are amorphous. Thus, attention was focused on the evaluation of Tg of these copolymers. On contrary, the copolymer prepared by the inverse vulcanization reaction of sulfur with diallyl disulfide was the only copolymer of its kind to have melting transitions as shown in DSC thermograms shown in Figure 9 For instance, the thermogram of the elemental sulfur that was used as control sample shows two melting peaks at 109 and 119°C corresponding to the melting transitions from the orthorhombic and monoclinic phases, respectively. 76
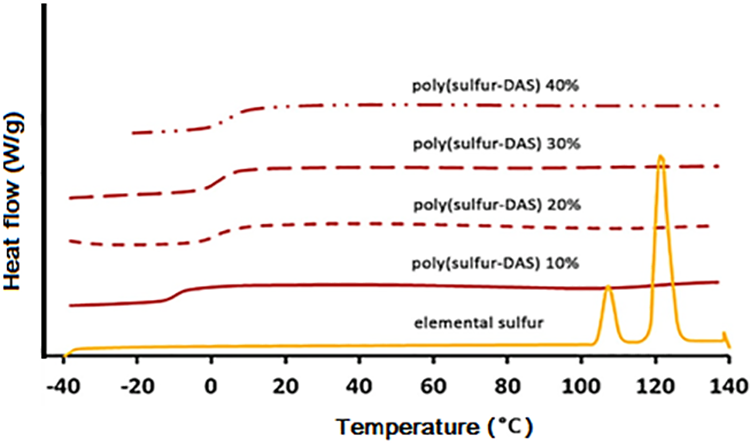
DSC thermograms of elemental sulfur and poly(S-r-DADS) with different wt% of DADS. 31
The degree of unsaturation of the comonomers and the organic content used in copolymerization reaction with molten sulfur was found to affect the thermal properties of the inverse vulcanized copolymers. For example, poly(S-r-DIB) with a 10 wt% of DIB in reaction mixture has a Tg of −13.7°C, which increases with the increase in the organic feed ratio of the copolymer until reaching a value of 28.4°C for poly(S-r-DIB) with 50 wt% DIB. 7 DIB and TIB have close structures but TIB is more unsaturated than DIB. Thus, the increase in the degree of the unsaturation of the monomer increases the crosslinking in the copolymer structure resulting in a dramatic increase in Tg to a value of 116°C for poly(S-r-TIB) with a 50 wt% sulfur compared to just 28.4°C for poly(S-r-DIB) with same reactants’ weight ratio.7,24 A similar high Tg value of 116°C was reported for densely crosslinked poly(S-r-DCPD) with 50 wt% sulfur in the reaction mixture. 64 On the other hand, inverse vulcanized copolymers with linear structures such as poly(S-r-myrcene) and poly(S-r-farnesene) showed lower Tg of 9.8°C and 0°C, respectively. 30 These observations further confirm the direct relation between crosslinking and glass transition temperatures of such copolymers and their solubility in organic solvents. In other words, higher the crosslinking structure in inverse vulcanized copolymers lead higher glass transition temperatures leaving them insoluble in organic solvents.
Interestingly, DSC was also used to calculate the amount of unreacted/free sulfur left in the copolymers, which leave peaks representing its crystalline nature. The heat of melting (ΔHm) was found to increase linearly with the increase in sulfur content in the copolymer. Calibrating ΔHm against different weights of sulfur allowed estimation of the unreacted sulfur amount left in the copolymer. No melting peak related to unreacted sulfur was observed in the DSC thermogram of the copolymers containing ≤30 wt% sulfur, which indicates the complete conversion of the sulfur.33–35,37,38 However, the unreacted sulfur was observed in thermograms of all inverse vulcanized copolymers obtained using vegetable oils as comonomers containing ≥30 wt% sulfur, which was increased with the increase of sulfur content in the reaction mixture. For example, poly(S-r-Canola oil) and poly(S-r-Soybean oil) with 50 wt% reacting sulfur showed 9.0 and 9.6% unreacted sulfur with ΔHm of 4.408 and 4.988 J/g. respectively. Such unreacted sulfur reached a value of 38.1% with ΔHm of 18.721 J/g for poly(S-r-Canola oil) and 42.6% with a ΔHm = 22.77 J/g for poly(S-r-Soybean oil), when sulfur content in both reaction mixtures increased to 70 wt%.33–38
The thermal properties of inverse vulcanized copolymers based on organic monomers having similar molecular masses and degrees of unsaturation were found to be different. Copolymers obtained reacting sulfur and monomers like Limonene, DCPD, DIB and ENB (5-ethylidene-2-norbornene), which have similar number of double bonds showed greatly different properties. Particularly, these polymers vary from viscous liquids to glassy materials. Smith et al. 64 explained this trend by showing a relation between the thermal properties of the copolymer, the degree of polymerization and crosslinking. For example, poly(S-r-DCPD) and poly(S-r-ENB) have high molecular weight and heavily crosslinked structure accompanied by insolubility in organic solvents and Tg as high as 115°C and 89°C, respectively. On the other hand, poly(S-r-DIB) showed lower Tg of 28°C whereas poly(S-r-Limonene) displayed a sub-ambient Tg of −21°C in addition to partial solubility. 64 To illustrate this observation, Hasell and coworkers 64 calculated the Fukui indices of these four monomers. The norbornene C=C of DCPD was found to be more reactive as indicated by the high value of the Fukui function (Fukui indices 0.201), whereas the Fukui values for ENB of all C=C were relatively similar (Fukui indices 0.209) making all C=C reactive and susceptible to attack by the radicals, which explains the reason of high degree of crosslinking and Tg of DCPD.10,11,64 The Fukui indices of limonene (value of Fukui indices 0.186), which is similar to ENB in terms of the degree of unsaturation and molecular masse also indicate that all C=C of the limonene are susceptible to attack by thyl radicals resulting in a crosslinked copolymer. However, the poly(S-r-Limonene) was found to be a linear copolymer with very low Tg of −21°C, which may be due to the H-atom abstraction of limonene during the copolymerization reaction, which results in an aromatic compound as indicated by the large volume of H2S released compared to others.39,64 A summary of previous studies of DSC results of the inverse vulcanized copolymers is presented in Table 4. It can be concluded that the use of DSC was limited in evaluating the main thermal properties of the sulfur-based polymers due to the complexity of the polymer structures and the absence of clear thermal melting despite being useful in determination the amount of unreacted sulfur and Tg of the copolymer. This can be harnessed in the determination of the conversion% of sulfur and yield of the copolymer.
Summary of thermal properties of inverse vulcanized copolymers reported in previous studies.

T5%: temperature at which 5% weight loss happens.
Thermal stability
Thermal stability of the polymeric materials is defined as the resistance to heat, which sets the limit of polymers’ operating temperature. Thermogravimetric analysis (TGA) of inverse vulcanized polymers such as poly(S-r-DVB) have been investigated under N2 atmosphere in comparison with elemental sulfur, which starts to thermally decompose at around 200°C and the maximum weight loss happens until 300°C as shown in Figure 10. 16 However, when sulfur undergoes bulk copolymerization reaction with DVB, the thermal stability and decomposition behavior changes. For instance, the decomposition of poly(S-DVB) took place in a two-step pattern with the first step initiated at around 210°C is due to sulfur degradation.45,46 The weight loss in the first step increased with an increase in the sulfur content in the copolymers. 10 Subsequently, the organic part of the copolymer started to decompose at around 300°C. This step varied depending on the type of comonomer.7,49 These results indicated that the inverse vulcanized copolymers have higher thermal stability than elemental sulfur and did not depolymerize back to S8 at room temperature, as observed in the case of polysulfane.9,11 The thermal stability of poly(S-r-Canola oil) was also evaluated under N2 flow, which showed a similar two-step degradation pattern with the first onset transition at 230°C is due to loosely-bonded and free S degradations. The second decomposition step at 340°C associated with the degradation of organic content of copolymers.35–38 Other petro-based and renewable monomers show similar thermal degradation trends under N2 atmosphere7,10,21,22,40,72 except for copolymer from sulfur with styrene, which showed a single step degradation pattern starting at around 200°C. 26
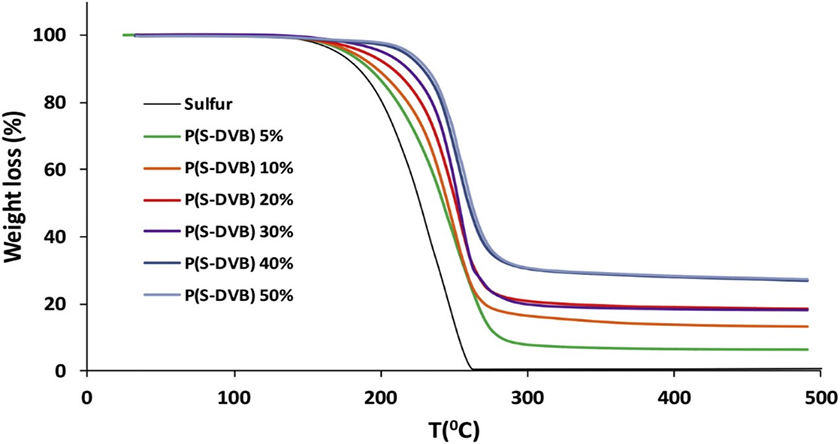
TGA thermograms of poly(S-r-DVB) with different contents of DVB. Sulfur was used as a reference. 23
On the other hand, TGA analysis under the ambient environment of the inverse vulcanized copolymers obtained from sulfur and sunflower, olive and linseed oil showed a three-step degradation pattern. 34 Coupling TGA with mass spectrometer for the analysis of the evolving gases during the different degradation steps. The sulfur decomposition at around 200°C released SO2 gas followed by the decomposition of the organic content of the copolymer releasing CO and CO2. The third step released traces of CO and CO2 along with SO2 from the sulfur that is directly bonded to C-atoms. 34 A summary of previous studies of TGA analysis and thermal degradation patterns of the inverse vulcanized copolymers are presented in Table 4. It can be observed that most of the studies used TGA analysis to evaluate the thermal stability of the inverse vulcanized copolymers under N2 atmosphere. The results revealed that the investigated copolymers do not show a complete decomposition leaving complex residues from C, S and N. On contrary, the inverse vulcanized copolymers showed a complete thermal degradation under ambient airflow that is likely to allow determination of sulfur and organic component contents in these copolymers.
Challenges and future directions
Preparation of sulfur-based polymers via inverse vulcanization provides an alternative route to develop new types of polymers. These polymers have interesting properties suitable for specific applications. However, their synthesis and processing are facing several challenges, which are discussed together with future directions.
The inverse vulcanized copolymers apparently possess low molecular weight compared to conventional counterparts. However, the limited solubility of the majority of the inverse vulcanized copolymers has imposed a number of challenges to their processing and characterization. Particularly, heavily crosslinked copolymers obtained from reaction of sulfur and vegetable oils are mostly insoluble in organic solvents imposing difficulties to the evaluation of their molecular weight with techniques such as SEC and GPC. Thus, finding appropriate solvents that can dissolve these copolymers and using possible alternative techniques is of particular interest.
The evaluations of the various properties of sulfur-based polymers with FTIR, NMR, DSC, TGA and XRD reported in literature are mostly qualitative. Only few studies reported quantitative uses of these techniques, which creates a gap in this field that must be filled in future research. It is also highly recommended to use other powerful characterization techniques like X-ray photoelectron spectroscopy (XPS), Raman spectroscopy and TG-FTIR7,35,42,46,63,78 to further explore the properties of these copolymers.
The diversity in the structures of the inverse vulcanized copolymers poses challenges in the characterization of these copolymers. The copolymers obtained from reaction sulfur and vegetable oils are in the form of thermosets with highly crosslinked structures and thus, their rheological properties cannot be evaluated using rheometers, which requires re-melting of the copolymer. Dynamic mechanical analyzing (DMA) can be a good alternative technique provided that the standard protocols are followed. They include the synthesis of the pre-polymer first followed by its molding and subsequent curing at the same temperature of synthesis.7,21,79
The use of spectroscopic analysis is very popular in for proving for formation sulfur-based polymers. Particularly, FTIR is more sensitive to vibrations of hetro-nuclear functional groups whereas Raman spectroscopy is more power for detection of the homo-nuclear bonds. However, some FTIR models operate only within the wavelength ranges of 500–4000 cm−1, which is not suitable to confirm the formation of C–S bond. Thus, it is more effective to use Raman to distinguish between C–C, C=C and C≡C bonds, which is also more accurate in sensing the C–S bonds. Although FTIR and Raman spectroscopy techniques can detect the functional group present in the molecules, but the exact group location cannot be determined. NMR is more powerful characterization tool not only to detect the functional group but also to determine the exact location of the functional group. However, the limited solubility of sulfur-based copolymers poses challenges limiting the use of 1H NMR. Alternatively, solid-state NMR is more suitable for characterization of sulfur-based polymers.73–75
DSC technique was frequently employed to evaluate Tg and the amount of unreacted sulfur in the copolymers obtained using vegetable oils as comonomers. However, the DSC thermograms of such copolymers didn’t show the transition between the glassy states to a rubbery state unlike DMA, which was found to be more sensitive and is useful in determination of Tg. So, it is highly recommended to use DMA as a primary characterization technique for these copolymers.34–38
Thermal decomposition of the inverse vulcanized copolymers investigated by TGA revealed that it depends on the nature of the organic comonomers and content of sulfur. TGA analysis proved that sulfur containing copolymers are more thermally stable than elemental sulfur and do not undergo depolymerization as polysulfane, which depolymerize to S8 at room temperature. The thermograms show a two-stage decomposition pattern with a thermal stability up to 200–210°C. However, the majority of reported TGA investigations were performed under a N2 atmosphere, leaving complex residues at the end of the heating runs. It would be more interesting to conduct TGA analysis under ambient or oxygen atmosphere to unveil the real content of reacted sulfur and organic components in these copolymers.
Preparation of sulfur-based polymers requires a high-temperature state (160–200°C) to bring sulfur to reaction at a molten state with monomers. Such high temperatures provide a narrow temperature window for inverse vulcanization to avoid any initiation for the side reactions such as H-atom abstraction, oxidation, or chain transfer, formation of random and equilibrated microstructures caused by thermal rapturing and recombination of S–S bonds in the backbone of the Sulfur-based polymers and release of H2S gas release infringing the rules of green chemistry. Furthermore, the high-temperature requirement of inverse vulcanization limits the selection of alkene monomers. Particularly, the monomers with a high boiling points are only feasible for the reaction. The use of catalyst reduces the reaction temperature, which paves the way to investigate new monomers with exciting properties and minimize the risk of H2S gas release. However, the catalysts used were either expensive or toxic, and this creates an opportunity for researchers to look for new green catalysts or alternative methods to reduce the temperature requirement of molten state copolymerization.9–11,21,59,65,66
Monomers investigated for inverse vulcanization are either petroleum driven (DIB, DVB and styrene), renewable monomers (limonene, myrcene and farnesene) or edible oils (canola, sunflower, or olive oil). The diminishing abundance and inherently non-renewable nature of the petro-based monomers, the high prices for renewable monomers and the food market demand for edible oils necessitate the search for new alternative cheap and sustainable monomers of non-edible oils for inverse vulcanization.26,30,35,39,71
High unreacted amount of sulfur that is observed in copolymers obtained by inverse vulcanization of sulfur and vegetable oils is the root cause of their undesirable mechanical properties and low solubility. The reason behind the presence of considerable of amount of unreacted sulfur is still unknown due to the complex structure of the vegetable oils. Thus, there is a need to address this properties and look for an improved preparation method to perfectly align the reaction with green chemistry or use green solvents to increase the conversion of the sulfur.34–38
The basic credo of green chemistry is to manufacture non-toxic materials employing facile and safer preparation methods. Elemental sulfur itself is non-toxic but there is no sufficient information available on the toxicity of the inverse vulcanized copolymers. Chalker and coworkers 39 reported that the polysulfide obtained by bulk-copolymerization of sulfur and limonene release nothing toxic in water as indicated by cell assessment of the HepG2 and Huh7 liver cells. However, no significant toxicity studies have been dedicated to inverse vulcanized copolymers. It is expected that the toxicity profiles of these copolymers vary with the nature of the organic comonomer. To further explore the feasibility and viability of these copolymers for environmental applications on industrial scales, more toxicological studies are highly sought. 39
The non-biodegradable nature of the polymers is always of high concern. Therefore, future investigations on the biodegradability and recyclability of the inverse vulcanized copolymers are highly important. The mechanism and products of the biodegradation depend on the stability of the polysulfide loop (S–S) and the nature of the organic monomer. Unlike conventional polymers, the backbone (S–S) of the polysulfide is labile to degradation and therefore it is worth finding methods to reproduce the starting materials from the industrially produced polysulfides, which can be further used to produce other polysulfides. 34
Conclusions
Sulfur-based polymers are potential alternatives to petro-based polymers that embrace green chemistry. The progress taking place in preparation of various sulfur-based polymers is driven by the desire to enhance the utilization of sulfur accumulated in oil and gas refineries and address its related environmental concerns. The number of potential applications of these copolymers, together with their characterization methods, are still in the infancy stage. The selection of the characterization techniques depends not only on the type of the monomer reacted with the molten sulfur and reaction conditions but also on the nature of the obtained copolymers and their complexity. Most of the inverse vulcanized copolymers have limited solubility and this poses challenges to SEC, GPC and 1H NMR investigations. FTIR is useful to provide evidences for copolymerization of sulfur with monomers, but there is an obvious difficulty in detecting C–S bonds and such a problem can be overcome by using Raman spectral analysis and solid-state NMR. SEM was found to be effective in the investigation of the morphology of the sulfur-based polymers obtained from vegetable oil compared to those prepared from petro-based monomers and this due to the presence of certain amount of unreacted sulfur giving the copolymer a composite nature. The inverse vulcanized copolymers were well investigated by XRD and found to be amorphous as a result of their crosslinked structure. DSC was not effective for the detection of melting behavior of the inverse vulcanized copolymers and was mainly used for the determination of Tg and the amount of unreacted sulfur in the copolymer, which is more profound when molten sulfur reacts with vegetable oil. The inverse vulcanized copolymers are more thermally stable than elemental sulfur with a two-step degradation pattern and a thermal resistance up to 200–210°C depending on the type of reacted monomer as revealed by TGA analysis. Other techniques like XPS, DMA and TGA-FTIR could be useful in the verge of exploring the various properties of these copolymers. It can be finally concluded that the evaluation of the whole properties of sulfur-based polymers is far from complete and more investigations are needed to uncover the merit of these polymers and their full properties. The selection of suitable characterization methods and the interpretation of the collected data must be carefully made. More efforts are also sought not only to determine the operating conditions of the inverse vulcanized polymers and prove that they are environmentally safe but also to evaluate their production viability at commercial scale.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by UTP/Institute of Future Energy (Universiti Teknologi Malaysia) project, cost center no. 015MC0-002.
