Abstract
This study aimed to modify the starch-based bioplastics with acceptable mechanical and water resistance properties using palm oil (PO) and epoxidized palm oil (EPO). The structural, thermal, mechanical and water resistance properties of the resulted bio-composites were evaluated. EPO had better compatibility within the starch matrix than PO. The bioplastics with PO and EPO exhibited different melting behaviors. Low concentrations of oils (<3 wt%) improved the tensile strength and elongation at break of the bioplastics remarkably, while higher concentrations of oils caused the decrease of tensile properties due to phase separation. Comparatively, EPO was more efficient at enhancing tensile properties than PO due to the presence of epoxy groups. Both oils reduced the water uptake and solubility of the bio-composites slightly. In conclusion, the addition of EPO was a more attractive way to enhance the properties of starch-based bioplastics.
Introduction
In the past few decades, aiming to substitute the synthetic petroleum-based plastics which cause environmental impact and petroleum shortage problems, there has been a growing interest to obtain biodegradable plastics from bio-based materials.1,2 Among these natural biopolymers, starch is widely used as an alternative to produce the bioplastic materials, as it is abundant, biodegradable, and renewable.3,4 Cassava starch is obtained from the roots of cassava and it is plentiful in Malaysia. It has been extensively investigated to fabricate the biodegradable films.5,6 The addition of a plasticizer is necessary to increase the flexibility because pure starch-based bioplastics are too brittle. Different polyols such as glycerol, xylitol, and sorbitol have been used. In most of the studies, starch-based bioplastics are most effectively plasticized by glycerol.7,8
However, the hydrophilic nature of starch and plasticizers generally results in poor mechanical properties of the fabricated bioplastics in humid conditions, which hinders their extensive applications.4,6 A number of methods have been attempted to enhance the water resistance properties of starch-based bio-composites, including chemical modification of starch, the addition of hydrophobic compounds, as well as blending with other polymers or reinforcement materials.1,9 Recently, plant oils, which exhibit excellent hydrophobic properties, have been introduced into the bioplastics to improve the water-proof properties because they are nontoxic and biodegradable.3,10,11 In addition, they can be chemically converted into the reactive oxirane ring by epoxidation with organic peracids. Epoxidized plant oils can easily undergo variety of reactions with many other functional groups to develop rigid and crosslinked products by nucleophilic epoxy ring opening reactions.12,13
Palm oil (PO) is one of the cheapest and most widely used plant oils in Malaysia. 14 It is of particular interest to expand its application and develop new products from PO. 15 Despite its high saturation, PO has been increasingly used to make industrial materials for pharmaceuticals, oleochemicals, detergents, and bio-resins besides food ingredients.16,17 In addition, PO can be epoxidized and utilized for many applications, such as the plasticizer for the polyvinyl alcohol, starting material for polyols, and bio-resin for bio-thermosets.
PO or epoxidized palm oil (EPO) is just developed as a potential modifier in the bioplastics recently compared to soybean oil or epoxidized soybean oil, which has been established as the component in industrial production successfully. 18 PO is increasingly used to improve the hydrophobicity of starch-based bioplastics. 13 Regarding EPO, it is mainly used as the plasticizer in poly(lactic acid) (PLA)-based composites. 19 In our previous study, we have compared the effect of EPO and epoxidized soybean oil on the properties of starch/fiber-based bioplastics. Both epoxidized oils presented favorable mechanical and water resistance properties to the composites due to the reaction between the epoxy groups of epoxidized oils and the hydroxyl groups of starch. 18
The effect of PO or EPO on the properties of starch-based bioplastic was extensively studied. To the best of our knowledge, there is no study about comparing the effect of PO and EPO on the attributes of starch-based bioplastics, which would shed light on the choice of PO or EPO to improve the properties of the bioplastics and promote the development of palm oil industry in Malaysia. PO can be combined with starch by emulsion technique. 20 EPO, similar to epoxidized soybean oil, might crosslink with the hydroxyl groups of starch through the epoxide ring. The addition of plant oils can reduce water resistance of the composites but may affect their mechanical properties.1,8 Therefore, this research explored the effect of PO and EPO as hydrophobic components on the properties of starch-based bioplastics. Fourier transform infrared spectroscopy (FTIR), scanning electron microscopy (SEM), differential scanning calorimetry (DSC), mechanical, and water resistance properties of the bio-composites were characterized.
Experimental
Materials
Native cassava starch (moisture content 11.43%, amylose content 21.20%) and commercial refined palm oil (minimum purity 99%) were provided by LGC Scientific SDN BHD located in Selangor (Malaysia). Epoxidized palm oil (EPO) was synthesized by following the methods of Kim and Sharma 21 and Yang, Ching 18 with slight modifications. Briefly, 100 g PO, 11.13 g CH3COOH and a certain amount of H2SO4 (2% of the H2O2-CH3COOH mixture) were blended in a 500 mL flask with an oil bath, a mechanical stirrer, a thermometer, a dropping funnel, and a cold-water condenser. Next, 42.05 g 30% H2O2 were added drop by drop and the reaction was conducted at 60°C for 8 h. Afterward, the solution was washed with 50°C 5 wt% Na2CO3 solution and 50°C distilled water in sequence. The oil layer was collected by decantation and dried at 60°C for 24 h. The oxirane oxygen content of EPO was measured by the AOCS Official Method Cd 9-57. Glycerol and Tween 80 were obtained from Sigma-Aldrich and used as the plasticizer and the emulsifier, respectively. Acetic acid (CH3COOH), anhydrous sodium sulfate (Na2SO4), hydrogen peroxide (30% H2O2), sodium carbonate (Na2CO3), sodium chloride (NaCl), and sulfuric acid (95-97% H2SO4) were purchased from Friendemann Schmidt Chemicals (Parkwood, Australia). The chemical reagents were all analytical grade and used as received.
Preparation of starch-based bioplastics
Formulations of the starch-based bioplastics based on dry weight.
The emulsion with 2.0000 g solid was casted on the glass plates (diameter 15 cm) and dried for 24 h under 40°C in the oven. Due to the weak strength of the produced bioplastic films, it was too difficult to peel them off the plates intact. Thus, the dry films (about 70 g) were further processed by a Hydrauic Molding Test Press (GT-7014-A30C) at 130°C for 6 min at a load of 13.79 MPa with a mold (200 mm × 200 mm × 1 mm). Before characterizing the composites, the samples were equilibrated for 2 days in a chamber with 53% relative humidity (RH).
Fourier transform infrared spectroscopy
The FTIR spectra of all prepared bioplastics were performed on a Spectrum 400 FTIR/FT-FIR spectrometer. The specimens were grinded and pressed into pellets with KBr powder for the measurement. The spectral regions were investigated over the range of 4000–450 cm−1 at a spectral resolution of 4 cm−1, with 32 scans. The FTIR spectra of PO and EPO were measured without mixing with KBr.
Scanning electron microscopy
Surface microstructure of the bioplastics was observed by a SEM (Phenom Pro X) at an accelerating voltage of 5 kV. The specimens (0.5 cm × 1.0 cm) were first fixed on the support using double side adhesive and then coated by a gold layer with a sputter-coater (Polaron SC7640). The micrographs were recorded at the magnifications of 400× and 1000× for every sample.
Differential scanning calorimetry
Dry samples (about 5–10 mg) were scanned by a DSC 1/500 (Mettler-Toledo (M) Sdn Bhd) from −50°C to 250°C with a heating rate of 10 °C/min in a sealed aluminum pan. The high purity nitrogen gas was used at a flow rate of 10 mL/min. Glass transition temperature (T g ), melting temperature (T m ), and crystallization temperature (T c ) were determined from the DSC thermograms.
Mechanical properties
The mechanical properties of the bioplastics were measured according to the ASTM D638 using an AGS-X universal/tensile tester (Shimadzu, Japan). The specimens were cut into dumbbell shape and mounted in the grips of the machine with 35 mm clamp distance. The crosshead speed was at a constant rate of 5 mm/min until breaking. The tests were performed with five replicates for each sample.
Moisture content, water uptake, and solubility
The specimens (1.0 cm × 1.5 cm) were dried at 105°C for 24 h in an oven to reach constant weights. Moisture content of each sample was calculated according to equation (1)
For water uptake, the dry samples (1.0 cm × 1.5 cm) were conditioned in a desiccator containing saturated sodium chloride solution (75% RH) for 3 days. The water uptake values were depicted as absorbed water (g) per dry matter (g).
Regarding solubility, the dry samples (1.0 cm × 1.5 cm) were accurately weighed and immersed in the test beakers with 50 mL distilled water at room temperature for 24 h. After this period, undissolved pieces were filtered, gently rinsed with distilled water, and subsequently dried in an oven at 105°C for 24 h. The solubility of each bioplastic was calculated according to the equation (2)
Statistical analysis
Data analysis was carried out by SPSS 19.0 software (SPSS Inc., Chicago, USA). Differences in the means of the results were determined via one-way analysis of variance (ANOVA) and Tukey’s test with p < 0.05 as statistical significance.
Results and discussion
Fourier transform infrared spectroscopy
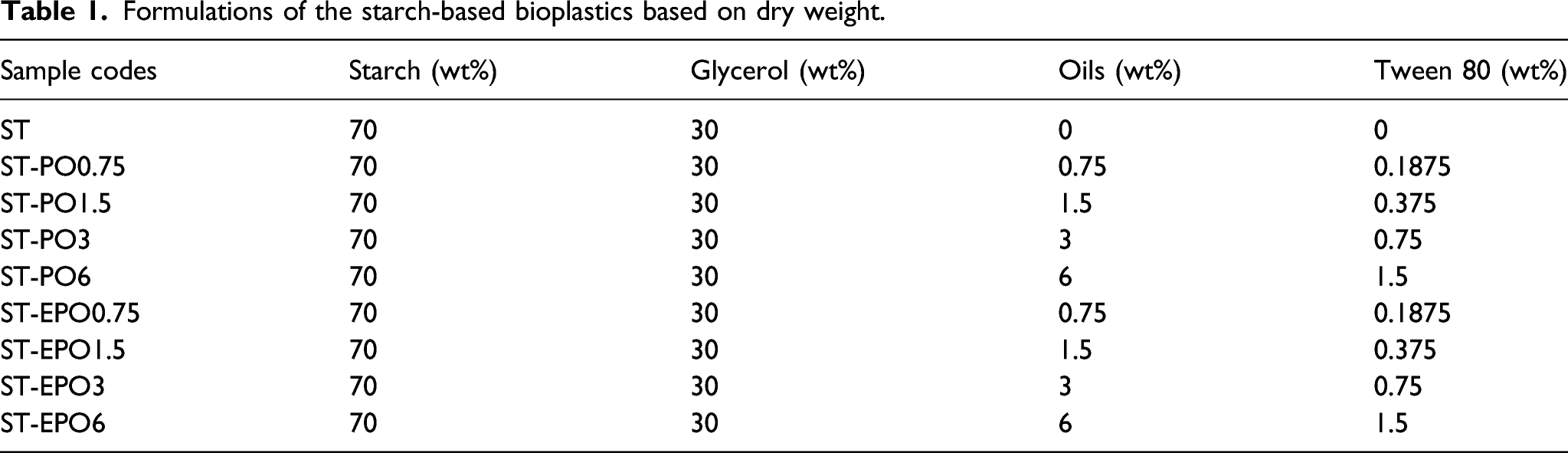
The FTIR spectra of PO and EPO are depicted in Figure 1(a). Regarding the spectrum of PO, the band observed at 3007 cm−1 is attributed to the C-H stretching of C=C-H. The peaks in the 2922 cm−1 and 2853 cm−1 regions are associated with the asymmetric and symmetric C-H stretching in methylene groups, respectively.
22
Two characteristic ester bands at 1743 cm−1 and 1158 cm−1 correspond to the stretching of C=O and C-O, respectively.
16
Moreover, the peak around the 1465 cm−1 region is assigned to the CH2 deformation of the alkane.
11
Fourier transform infrared spectroscopy spectra of PO/EPO (a) and starch-based bioplastics incorporated with PO (b) and EPO (c).
The epoxidation of PO was evident due to the disappearance of the band for C=C (3007 cm−1). Moreover, EPO showed low intensity of absorbance of epoxide groups around 846 cm−1 in the spectrum. The low peak might be attributable to low unsaturated level of PO, thus in consistent with the low oxirane oxygen content (2.95%) of EPO. 15 By-products such as polyols were likely to be formed during the epoxy reaction.21,23 Water can react with the epoxide groups by the oxirane ring opening at 80°C to give a polyol. 24
The spectra for starch-based bio-composites with PO and EPO are shown in Figure 1(b) and (c). It was apparent that the spectra of the composites containing PO and EPO were similar. The broad bands in the 3287–3301 cm−1 are characteristic of the vibrational stretching of the hydroxyl groups. In the 2923–2930 cm−1 regions, the bands correspond to the C-H stretching. 11 The peaks around 2854–2856 cm−1 intensified with PO and EPO contents increasing which confirmed the presence of oils. The composites with increasing contents of oils displayed more intense bands at wavenumbers of 1743–1748 cm−1 assignable to the C=O groups of ester bonds. The peaks from 1647–1650 cm−1 are assigned to water molecules attached to starch. The broad bands around 1077–1079 cm−1 and 993–1017 cm−1 signify the C-O stretching of the glucose ring of starch. 25
The structural interactions among the ingredients in the composites can be revealed by the changes of absorption bands. 22 The peaks typical of hydroxyl groups at 3287 cm−1 shifted to higher wavenumbers while the peaks at 2923 cm−1 changed to lower wavenumbers after the introduction of PO and EPO. It was suggested that hydrogen bond interactions among starch/glycerol and oils/surfactants occurred. 13 According to previous authors, the carboxylic or carbonyl groups of plant oils may interact with the hydroxyl groups of starch through hydrogen bonds. 11 The epoxy groups of epoxidized oils may react through the ring opening reaction with the hydroxyl groups of starch and glycerol. 2 However, the reaction was not monitored by the FTIR due to the low oxirane oxygen content and loadings of EPO. Similar observations were found in the literatures for polymers toughed with epoxidized oils.2,13
Scanning electron microscopy
The arrangement of bioplastic components (starch, plasticizer, and oils) in the composites depends on the nature of the polymers, the interactions among the components and the drying process.
7
The surface photographs of all the bioplastics were taken at magnifications of 400× and 1000×, as shown in Figure 2(a)–(j). It was observed that the surfaces of unloaded bioplastics were homogeneous and continuous without any granules, cracks, or pores (Figure 2(a) and (b)), which revealed a good gelatinization of the composites. Scanning electron microscopy micrographs of starch-based bioplastics incorporated with PO and EPO. (a-b) ST, (c-d) ST-PO1.5, (e-f) ST-PO6, (g-h) ST-EPO1.5, and (i-j) ST-EPO6.
The emulsified bioplastics containing 1.5 wt% PO (Figure 2(c) and (d)) presented a discontinuous and porous structure. This suggested that PO was not dispersed uniformly within the blends, and phase separation occurred on the interfaces between PO and macromolecular chains of starch. 8 On the surface of the composites, many small white particles were clearly observed, which could be attributed to starch particles or oil droplets. This result might indicate that PO hampered the plasticization of starch. By comparison, EPO seemed to be more uniformly distributed in the polymer matrix than PO at 1.5 wt%, giving rise to a more homogenous and compact surface with fewer holes detected (Figure 2(g) and (h)). In addition, the white particles were less evident on the surface. When 6 wt% both types of oils were introduced, the surfaces of the bioplastics showed obvious aggregates surrounded by micro-voids, resulting in less compact structures. This was due to the fact that high oil loadings favored the flocculation rate and hampered the structural formation of the bioplastics.8,26
Differential scanning calorimetry
The thermal transitions of the prepared bioplastics were determined by DSC analysis. In Figure 3, the small exothermic peak of the control bioplastic at 55.33°C (T
c
) might be associated with crystallization behavior of plasticized bioplastics.26,27 It has been found that crystallinity of starch-based bioplastics changed over time due to retrogradation.
28
In addition, the crystallization process would be enhanced in the process of compression molding because nucleation and propagation required the mobility of polymer chains.29,30 After the addition of 1.5 wt% PO, two crystallization peaks appeared and intensified at 31.50 and 67.83°C, respectively. It was also reported that two glass transition temperatures (T
g
s) occurred for the starch–lipid complex due to phase separation.
8
The integration of PO in the starch matrix was not homogeneous as shown by the SEM images, which might contribute to the presence of two crystallization peaks. Differential scanning calorimetry thermograms of starch-based bioplastics.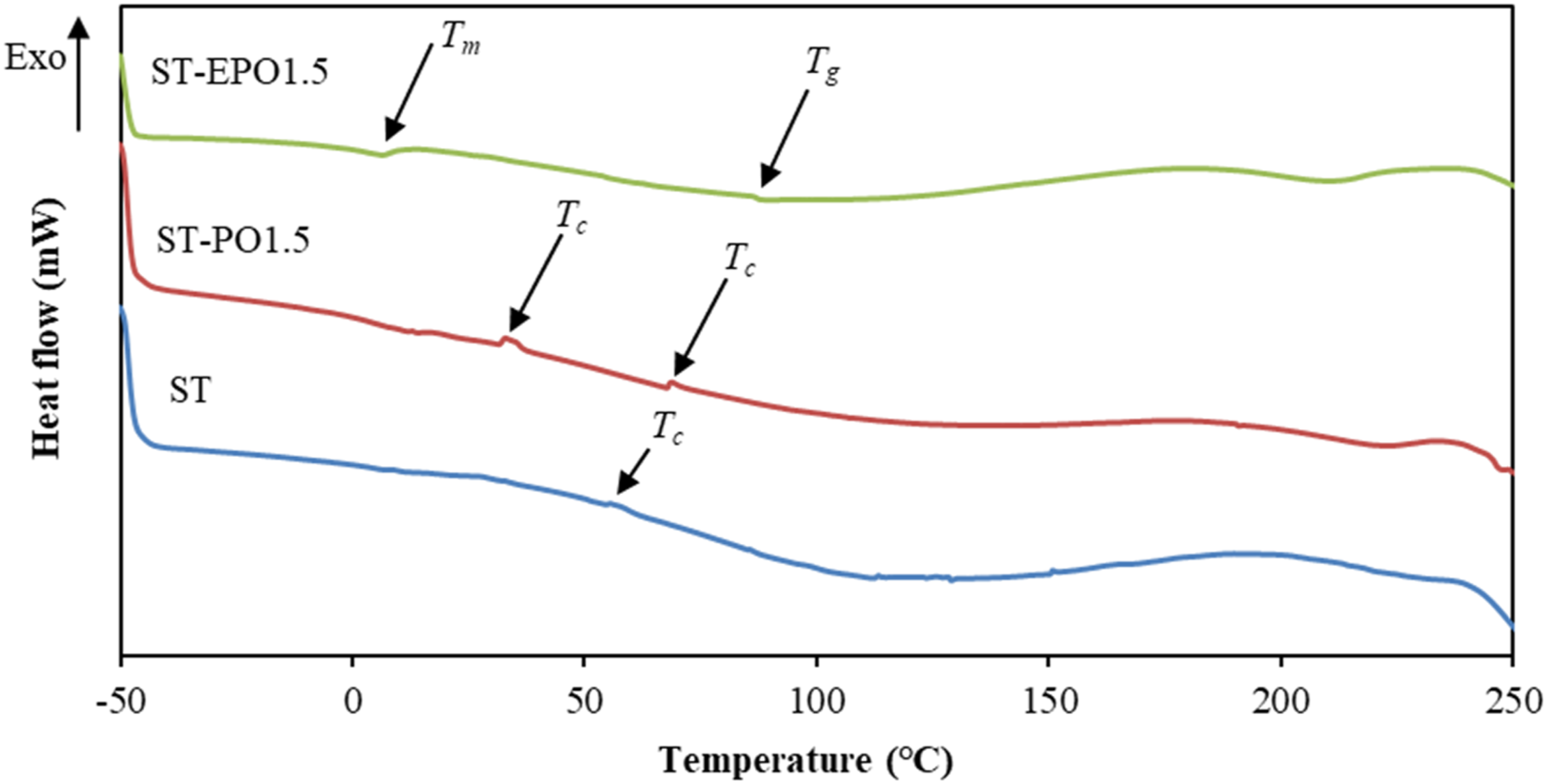
After the introduction of 1.5 wt% EPO, the crystallization peaks of the bioplastics disappeared. The endothermic peak at 90.33°C might be attributed to T g of the composites. 31 Previous studies showed that T g of starch-based bioplastics was hardly observed by DSC. 32 The appearance of T g in the study may be due to the fact that EPO improved the structural integrity of the starch complex, as demonstrated by the high tensile strength of the sample. Additionally, the endothermic peak around 6.50°C (T m ) might be assigned to the melting of free unbounded EPO. 4 However, the melting peak of PO was not observed obviously in the thermogram of ST-PO1.5. Tongnuanchan, Benjakul 16 also noticed that no melting transition was observed for pure basil essential oil in gelatin-based film containing basil essential oil. These outcomes suggested that PO and EPO exhibited different interfacial interactions with starch, which affected the horizontal reorganization and the melting behaviors of starch molecules. 3
Mechanical properties
Tensile strength and elongation at break were determined to estimate the influence of various oils on the mechanical properties of the bioplastics, as shown in Figure 4(a) and (b). Bioplastics without the addition of oils exhibited relatively low elongation at break (30.70%) and tensile strength (0.45 MPa) which were in agreement with those reported by Brandelero, Grossmann
11
that worked with cassava starch. Many studies have found the reduction in the mechanical properties of the films incorporated with lipids because the addition of lipids introduced discontinuities in the matrix and contributed to the reduction of polymer cohesion forces.1,7 However, in this experiment, the results revealed that both oils, especially EPO, significantly improved the mechanical properties of the bio-composites. Tensile strength (a) and elongation at break (b) of starch-based bioplastics. (a-d), A-E: Significant differences among samples (p < 0.05) are indicated by different letters within the same indicator. 1–2: Different numbers show significant differences between the formulations with PO and EPO at the same concentration (p < 0.05).
Initially, the composites exhibited the rising tensile strength with the loading levels of oils increasing, followed by a decreasing tensile strength upon reaching the maximum. The tensile strength reached the maximum value at 1.5 wt% oil content (p < 0.05), which revealed 353.33% and 53.33% increase for the composites with EPO and PO, respectively, as compared to the pure composites. The observed effect was likely attributed to the strengthened interactions between the carboxylic groups of fatty acids in the oils and hydroxyl groups of starch/glycerol, as was indicated in the FTIR spectra. At the same oil concentration, the bioplastics with EPO revealed significantly higher tensile strength compared to those with PO (p < 0.05). This effect was the consequence of the partial crosslinking induced by the epoxide ring of EPO with the hydroxyl groups of starch.2,13 Therefore, the epoxy groups of plant oils seemed to be key factors to obtain the blends with higher tensile strength. 2
Similarly, elongation at break showed a huge increase at 0.75 wt% oil content (p < 0.05) which increased by 158.95% for the bioplastics with PO and 103.57% for the bioplastics with EPO. The increase in the elongation at break has been previously observed, which was due to the plasticizing effect of plant oils. It was reported that incorporating plant oils in the polysaccharide-based or protein-based films may interfere with polymer interactions by facilitating polymer–oil interactions. This phenomenon increased the free volume within starch molecules and contributed to greater chain mobility, thereby enhancing the extensibility of the resulted composites.8,16 Comparatively, the composites with EPO showed lower elongation at break than the composites with PO, which can be illustrated by the possible crosslinking effect between EPO and starch/glycerol.
However, when higher contents of oils were used, a gradual decrease of the mechanical properties was found. This can be attributed to the reduced interactions due to phase separation between starch and oils, which led to the formation of discontinuous zones, as observed in the SEM micrographs.1,20 Also, the oil–oil interactions showed less bonding than the oil–starch interactions, thus influencing their mechanical properties. Furthermore, the active groups on EPO can only provide low reactivity with starch due to the steric hindrance. 2
Water resistance properties
Water resistance properties of emulsion-based composites depend on the biopolymer properties, the interactions among different components, and the structure of resulted composites.4,5,8 As shown in Figure 5(a)–(c), the moisture content, water uptake and solubility of the control sample were 8.48%, 23.44% ,and 34.27%, respectively, which were close to previously reported work.
6
After introducing PO and EPO into the composites, only slight decrease in water uptake and moisture content of the composites was observed (p > 0.05), which was ascribed to the hydrophilic character of starch and glycerol.31,33 Water solubility was significantly reduced by the incorporation of 6 wt% PO and various concentrations of EPO (p < 0.05). Moisture content (a), water uptake (b) and water solubility (c) of starch-based bioplastics. (a-b), A-B: Significant differences among samples (p < 0.05) are indicated by different letters within the same indicator. 1-2: Different numbers show significant differences between the formulations with PO and EPO at the same concentration (p < 0.05).
The enhanced water resistance properties were expected due to the hydrophobic characteristic of both oils. It was reported that the addition of plant oils can cause structural changes and form more tortuosity in the composites. Moreover, from microscopy observation in other studies, the oils may cover the starch granular surface, hamper water penetration into the matrix and increase its hydrophobicity.30,34,35Also, the great interactions among components caused the unavailability of free -OH groups with water. 3 The hydrophobic, tortuous, and compact structures slowed down the speed of water transfer and increased the path and time for water transfer, thus preventing water transfer.13,36,37 Comparatively, the addition of EPO could reduce the water solubility better than PO at the same oil concentration, especially at 1.5 wt% oil (p < 0.05). This might be explained by the enhanced interactions between the epoxy groups of EPO and hydroxyl groups of starch/glycerol, contributing to a decrease in the availability of hydroxyl groups, thus reducing starch–water interactions.13,14
At high concentrations of both oils (3-6 wt%), we did not obtain huge reduction of water uptake and solubility. This can be due to the fact that aggregation phenomena were promoted by the increasing oil concentrations as verified by the SEM micrographs below. In addition, differences in density between oils and starch provoked the migration of lipid aggregates to the material surfaces, which damaged cohesive structural integrity.7,26,27
Conclusions
This study explored the influence of PO and EPO on the properties of starch-based bioplastics. The hydrogen bond interactions between the oils and starch/glycerol mixtures were apparently observed by the FTIR. The reaction between EPO and starch was not revealed due to the low oxirane oxygen content and loading levels of EPO. The micrographs confirmed that the bioplastics with PO and EPO had less structural integrity due to the phase separation. DSC results revealed that the bioplastics with PO and EPO showed different melting behaviors. EPO showed a greater effect on improving the tensile and water resistance properties compared to PO because of the reactivity of epoxy groups. The addition of a small quantity of EPO caused a huge increase of the tensile strength presumably due to the crosslinking effect of epoxy groups. Enhanced elongation at break emphasized that the inclusion of oils produced the plasticization effect. At high contents of both oils (>1.5 wt%), the composites exhibited significant decrease of the mechanical properties which might be related to phase separation and weak oil–oil interaction. The incorporation of both oils led to the slight reduction of water uptake and solubility in comparison with the control sample. With high concentrations of both oils (3-6 wt%) incorporated, the bioplastics did not obtain huge reduction of water uptake and solubility due to phase separation. Overall, this study suggested that EPO showed a promising potential to be formulated in the starch-based bioplastics with favorable properties for packaging.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Ministry of Education Malaysia Ministry of Education Malaysia, PRGS: PR006-2019A, and University Malaya research grants ST058-2021 and GPF022A-2019. The authors expressed their gratitude to Dr Sampath for his assistance in the melt blending and compression molding operation.
