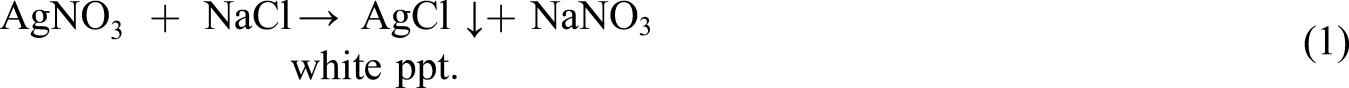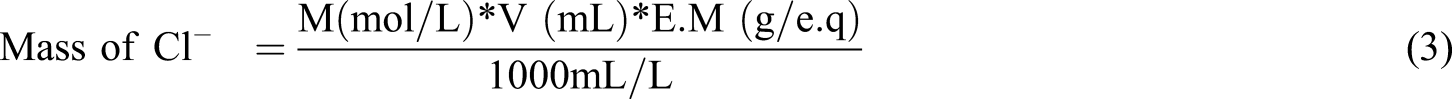Abstract
The wet treatment process was used for the dechlorination of poly(vinyl chloride) (PVC) landfill waste plastic, and the process has shown dependence on concentration of alkali. The process of dechlorination of PVC has shown dependence on the concentration of the alkaline solution and the type of organic solvent used in the preparation of the alkaline solution. Ethylene glycol (EG), diethylene glycol (DEG), and tetraethylene glycol (TEG) are the diols that have been investigated. Moreover, the temperature of the PVC dechlorination process and the time of the PVC solution reflux showed effects on the final degree of the dechlorination (DD%). These were calculated using the Mohr method, where the chloride ions were titrated with a standard solution of silver nitrate in the presence of chromate ions. The study shows that 1 M NaOH/TEG alkaline solution was the best to dechlorinate PVC waste with the wet treatment process at 160 ̊c and for 3 h reflux and the process reclaimed poly(vinyl alcohol) PVA as the main product according to the hydroxyl group substitution mechanism (SN2), besides a few units according to the hydrogen chloride elimination mechanism (E2) with a few remaining chlorinated chains after analysis.
Keywords
Introduction
Poly(vinyl chloride) PVC is a highly consumed plastic in the world. Because its products are easily manufactured with simple techniques, available, cheap, and in a high performance, the PVC became the most produced polymer, globally. 1 Accordingly, a huge amount of the PVC waste grew steadily 2 and the landfilling areas became insufficient; therefore, recycling of PVC is the most suitable solution for its environmental problems. 3 Incineration and pyrolysis of PVC waste were part of the old methods used for the recycling of PVC waste.2,3 However, they are no longer desirable today because of the toxic compounds (such as hydrogen chloride) formed and emitted to the environment in the process. 4 Other pollutants that create extra environmental problems are toxic dioxins and furans. 5 The recycling process of PVC waste using the conventional mechanical method without any changes in its chemical composition 6 was another well-known method. The major problems of the mechanical recycling process are the continuous changes in its morphological structures. 7 The mechanical method changes the original particle structure of PVC inside the network of entanglement due to the shearing stress during the process. 8 On the other hand, the chemical recycling method which breaks up the polymer waste using heat, chemical agents, and catalysts into their basic chemicals is one of the most important techniques used for recycling PVC waste.9,10
The wet treatment processes for dehydrochlorination or dechlorination of PVC have been developed, 11 where the NaOH/EG solution is used at the atmospheric pressure and the boiling point of EG (196oC). 12 To evaluate the wet treatment process used for chemical recycling of PVC waste and to select the most suitable conditions for that treatment, the degree of dechlorination (DD%) of the PVC waste has to be calculated. 13
This work aims to study the suitable conditions for dechlorinating the PVC waste collected from Mosul city landfills using the wet treatment process. In addition, the calculation of the degree of dechlorination (DD%) of the PVC waste will help in evaluating the wet treatment process and fixing its suitable conditions. The alkaline solutions of different concentrations in diols were used for dechlorination. The time and temperature of the PVC hydrolysis used in the wet treatment process have been studied.
Experimental
Materials
PVC waste materials were supplied from Mosul city landfills in Iraq. The samples were prepared for hydrolysis by cutting into small pieces, cleaned, and finally ground. Ethylene glycol (EG), diethylene glycol (DEG), and teraethylene glycol (TEG) from Fluka, Switzerland, were used as received. Silver nitrate and potassium chromate were from BDH Chemical Company, United Kingdom, also used as received. Other chemicals were of analytical grade reagents and received from Fluka.
Wet treatment process for dechlorination of PVC waste
In 250 mL three-necked reflux flask provided with nitrogen inlet tube, 50 mL of different concentrations of the alkaline solution in organic solvents were added. The alkaline solution was prepared from sodium hydroxide NaOH in one of the following hydrocarbon solvents: ethylene glycol (EG), diethylene glycol (DEG.), or teraethylene glycol (TEG) having concentrations of 0.2 M, 0.5 M, 1.0 M, and 2.0 M. The 50 mL of the alkaline solution inside the reflux flask was stirred for half an hour under nitrogen, the inert gas for expelling oxygen, and finally, 0.1 g of ground PVC waste was added. The mixture was heated under different reflux temperatures 150oC, 175oC, and 200oC and for hydrolysis time intervals of 120 min, 180 min, and 240 min. The DD% of the reflux mixture at each fixed time interval and for each fixed degree of reflux temperature was measured. Later, 5 mL was extracted from the reflux solution and washed carefully with distilled water and methanol for DD% measurement.
Standardization of silver nitrate
Silver nitrate (AgNO3) of 0.1 M concentration was prepared in distilled water. Then 0.25 g of dried NaCl was dissolved in 100 mL of distilled water into a 250 mL Erlenmeyer flask. To adjust the pH of the solutions, few milligrams of NaHCO3 were added until effervescence ceased, then 2 mL of 5% K2CrO4 of indicator was added. The prepared NaCl solution was titrated with the AgNO3 solution to the first permanent appearance of brick-red Ag2CrO4 precipitate [equations (1) and (2)].
14
Determination of unknown chloride from PVC hydrolyzed filtrate
The Mohr method was used to determine the concentration of the unknown chloride ions formed from the dechlorination process of PVC waste in the alkaline solution. The unknown chloride ion samples were taken from the PVC/alkaline/organic solvent mixture at a fixed time interval and a certain temperature. The sample was washed carefully with distilled water and methanol, and the formative white precipitate which represents the unconverted PVC sample was separated by filtration, while the filtrate was completed to 5 mL in the Erlenmeyer flask. A few milligrams of NaHCO3 were added for pH adjusting until effervescence ceased. 2 mL of 5% K2CrO4 of the indicator was added and then titrated with a standard AgNO3 solution.
14
The weight of the chloride ions in the analyzed PVC sample was calculated
15
according to the following equations (3) and (4)
Results and discussion
PVC, the third largest produced polymer in the world, is used widely in industries and many applications in our daily life. Therefore, its waste materials accumulate in a huge amount in the landfills, and this problem needs to be solved. Thermal and chemical recycling processes are considered as the some of the most brilliant recycling strategies. Thermal recycle of PVC wastes and its uses as fuel in combustion is very common; however, the presence of chlorine, with more than 50% of its mass, will lead to the corrosion of apparatus due to the formation of HCl in the treatment plants. As a result, chemical recycling is considered more acceptable. Accordingly, the present work is based on the wet treatment process for the degradation of PVC using NaOH solution in different diols and under mild condition. The DD% of PVC was calculated under variable conditions of alkaline/diol mixture concentrations, besides the time and temperature effect on DD%
PVC characterizations
FTIR study
The FTIR spectrum of pristine PVC recorded with FTIR spectrophotometer of Tensor Co. Brucker, 2003, Germany. Figure 1 and the characteristic bands in Table 1 show absorption bands at 2920 cm−1 and 2848 cm−1 which represent (ɣ(C-H)str) of PVC methylene groups. The absorption bands at 1460 cm−1 and 1335 cm−1 represent the (ɣ(C-H)def) of PVC methylene groups. The absorption band at 730 cm−1 belongs to (ɣ(C-Cl)str) of PVC. FTIR spectrum of pristine PVC waste. FTIR characteristic frequencies of pristine PVA waste and of dechlorinated PVA with 1 M NaOH/TEG solution.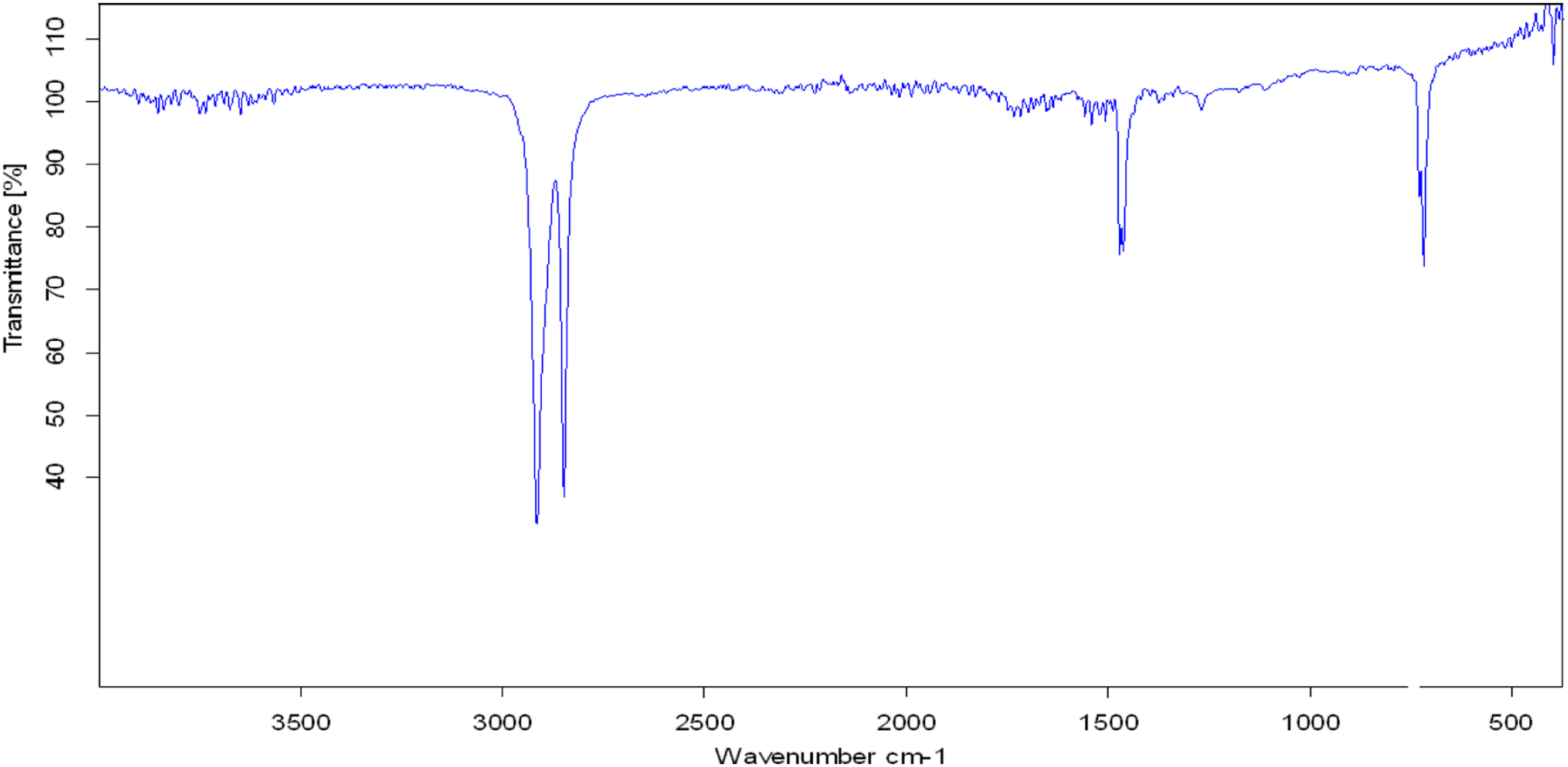

Thermal study
The thermal parameters of pristine PVC sample which was analyzed using TG analyzer type Extra TG/DTA 6300, USA, in the nitrogen atmosphere, and the data were collected from thermogram Figure 2 and described in Table 2. TG, DTG, and DTA thermogram of pristine PVC waste. TG, DTG, and DSC thermogram data of pristine PVC waste and of dechlorinated PVA with 1 M NaOH/TEG solution.

The thermogrametric analysis of pristine PVC is shown at an initial decomposition temperature IDT of 277°C and a weight loss percentage of 4.0%; while at the final decomposition temperature FDT of 500oC, a weight loss percentage was of 75%. The TG study has shown that the PVC is rigid and has moderate thermal stability; however, the polymer is decomposed within two steps. First, the polymer is dechlorinated. This is followed by the decomposition of methylene units of PVC in the second step. 16 The DTG thermogram of pristine PVC shows two maximum weight losses per unit time peaks at different temperatures.
The magnitude of the maximum weight loss of both peaks of pristine PVC refers to the high weight loss of polymer per unit time and especially in the first peak of 1.18 mg. min−1. This represents the dechlorination of PVC because the chloride ions are ordinarily present in large quantities in the polymer. The second peak of 0.25 mg. min−1 of low weight loss represents the methylene units of PVC with a low mass in comparison with chloride.
The DSC thermogram of pristine PVC shows the heat of fusion ∆Hf of two endothermic states; the first of +0.136 J mg−1 needs high energy per milligram polymer weight which represents the dechlorination of PVC. The second state of +0.019 J mg−1 needs lower energy and represents the degradation of polymer methylene units. The crystalline temperature Tcr of pristine PVC of 296oC is low, which means that the polymer is present in the low crystalline structure, whereas it’s Tg of 82oC is sufficient for PVC to resist the environmental conditions.
Effects of alkaline concentration on PVC dechlorination
The chemical hydrolyses of PVC waste with sodium hydroxide (NaOH) dissolved in ethylene glycol (EG) were prepared in different concentrations and their DD% according to Mohr method 17 was calculated. In addition, the analysis time and temperature of PVC dechlorination process have been investigated. The study shows that the concentration of NaOH as a source of hydroxyl group (OH−) has a significant effect on the dechlorination process of PVC. A competition between two mechanisms through dechlorination of PVC leads to the elimination of hydrogen chloride (E2), and the hydroxyl group substitution (SN2)18 has been concluded and confirmed by the FTIR spectrophotometric analysis. The absorption frequency at 3303 cm−1 in Figure 7 and Table 1 was fixed as the band belongs to the hydroxyl group (ɣ(O-H)str). The sharp frequency at 1712 cm−1 represents the ethylene group (ɣ(C=C)str), and the spectrum still contains the band at 723 cm−1 which represents (ɣ(C-Cl)str) and means that the dechlorination of PVC was not completed.
The hydroxyl groups of NaOH in the beginning stages of PVC dechlorination reacted with the surface and inner parts of the PVC particle. However, at high NaOH concentrations, changes probably occur in the surface morphology of the PVC particle that prevents further penetration of OH−. In this regard, the dechlorination of PVC increases dramatically with the rise of the NaOH concentration (Figure 3) and reaches its maximum DD% at 1 M concentration of NaOH. Above 1 M NaOH, the DD% decreases due to the changes in the surface morphology of PVC chains where the OH− ions could not penetrate and substitute the PVC particle.
18
Effect of the NaOH/EG concentrations and their analysis time intervals versus DD% of PVC waste analyzed at 469 K (196 c).
Dechlorination of PVC using the wet treatment process has been found to be significantly affected by the temperature of the dechlorination analysis method (Figure 4). The DD% of PVC shows a significant increase due to temperature. The dechlorination process of PVC in an aqueous solution of NaOH requires high pressure. However, because the process was carried out in ethylene glycol and because the solvent has a high boiling point of 196°C, the process was done at atmospheric pressure and has shown the highest DD% of PVC at 195°C. At a lower temperature (Figure 4), the dechlorination process of PVC has shown depression due to insufficient activation energy and the reaction entropy is as low as hydroxyl substitution.
18
Effect of the temperature of 1 M NaOH/EG solution used for the analysis of PVC waste on its DD% versus time intervals of analysis.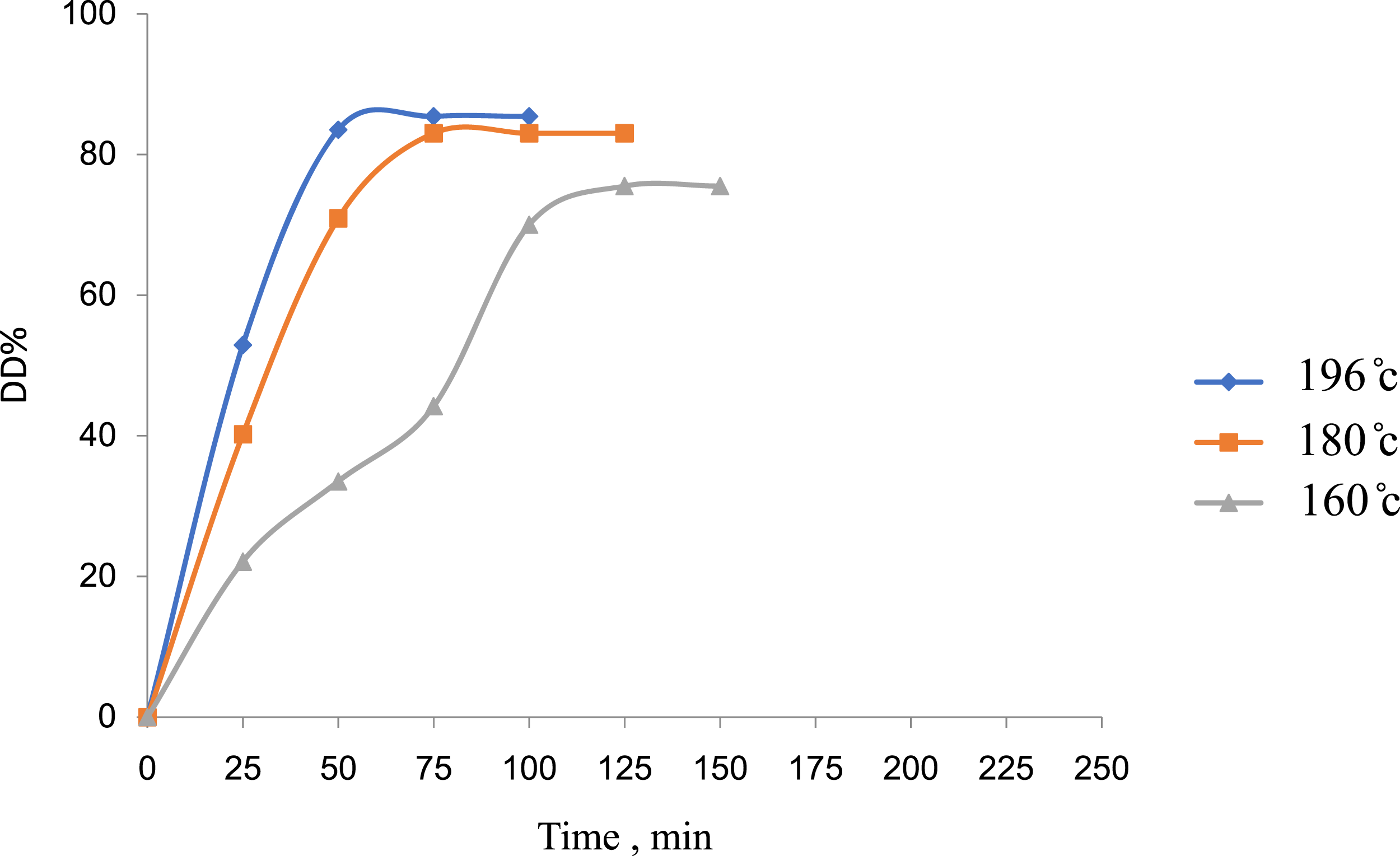
Moreover, the reaction time has shown effects on the DD% of PVC (Figures 3 and 4), although to a certain degree before it becomes useless.
Effects of diol solvent on PVC dechlorination
Three diol organic solvents were investigated for the recycling of PVC waste using the wet treatment process. Ethylene glycol (EG), diethylene glycol (DEG), and tetraethylene glycol (TEG) were tested. All examined diol solvents have functional groups and can dissolve PVC waste because of the difference in their chemical structures and their boiling points. Therefore, they have different effects on the DD% of PVC (Figure 5). The solubility parameters represent the essential difference between the examined diols which affect the PVC solubility and change its swelling behavior.
19
This represents the sum of dispersive, polar, and association forces which include hydrogen bonding and permanent dipole-induced dipole that let the tested diols in the following sequence: Effect of the type of organic solvent (diols) in 1 M NaOH solution on DD% of PVC waste versus temperature of solution analysis.
The more solubility of diol solvent is the more penetration between the polymer chains which facilitate the dechlorination process of PVC in an alkaline solution and which proceed at low temperature. In this regard, the highest DD% of PVC (Figure 5) shows the best solubility of diol solvent and the highest DD%. In other words, the dechlorination process of PVC in 1 M NaOH/TEG reached its maximum DD%, while in 1 M NaOH/EG showed a minimum according to their solubility sequences. Similarly, the time of the dechlorination of PVC (Figure 6) shows the same sequences because the solvent penetration between polymer chains needs a short time according to the high solubility of PVC waste in a 1 M NaOH/TEG solution.
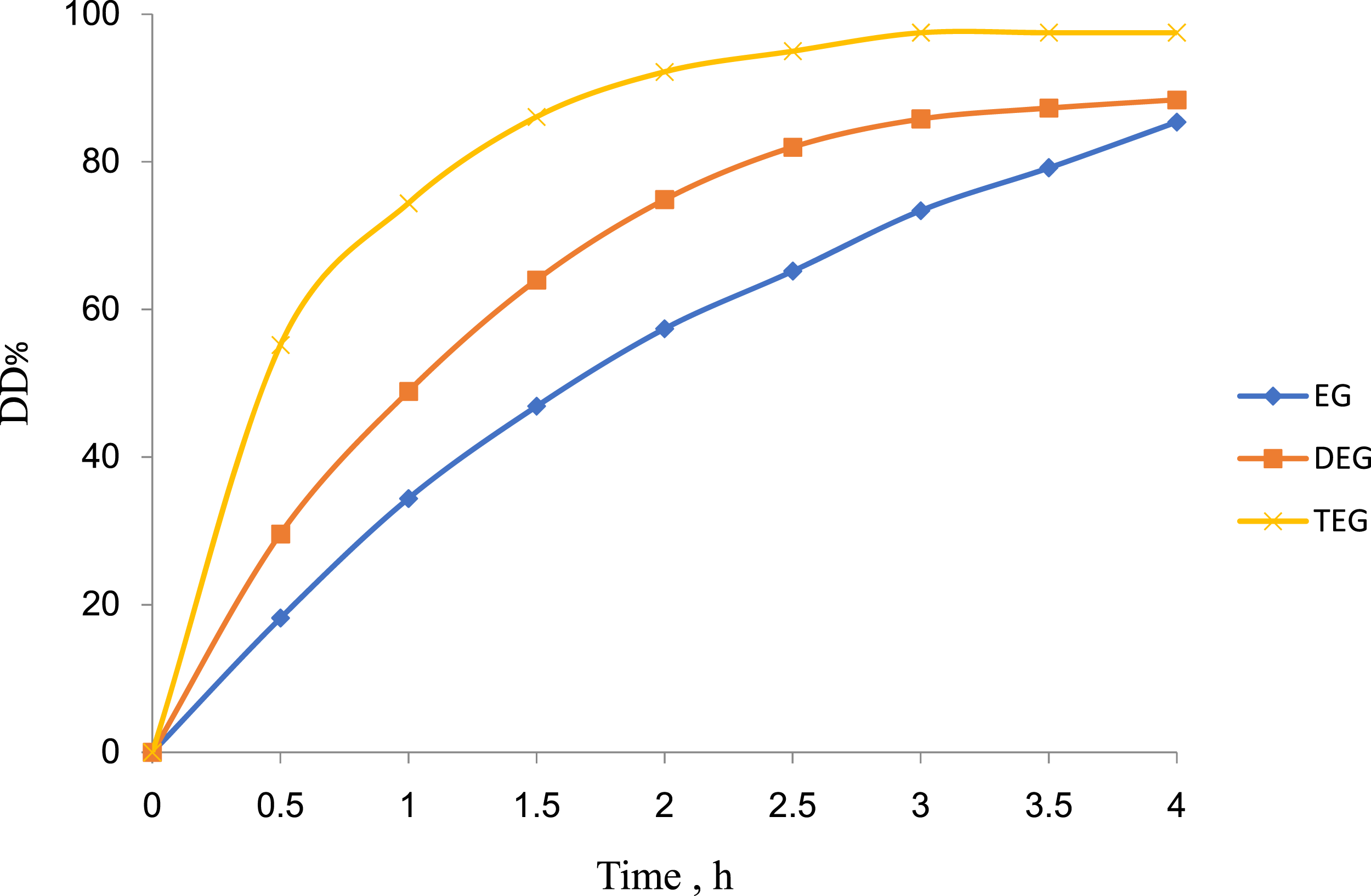
Effect of the type of organic solvent (diols) in 1 M NaOH solution on DD% of PVC waste versus time intervals of analysis.
Characterization of PVC dechlorination analysis products
The alkaline hydrolysis of PVC waste using 1 M NaOH/diol solution resulted in a white precipitate which was analyzed. The following results were recorded:
The viscosity-average molecular weight
The FTIR analysis of PVC dechlorinated product in Figure 7 and Table 1 shows absorption bands at 3431 cm−1 which belong to the hydroxyl functional group (ɣ(O-H)str) and the absorption bands 1410 and 1123 cm−1 which also belong to the hydroxyl group (ɣ(C-OH)str). According to the hydroxyl group substitution (SN2) mechanism, equation (5) has been achieved from the new absorption frequencies recorded for the PVC dechlorinated product. In addition to the absorption band at 1712 cm−1 which belongs to the unsaturated band (ɣ(C=C)str), the presence of an unsaturated group in a recycled product (Figure 7) would improve the elimination of hydrogen chloride (E2) mechanism, equation (6)
20
FTIR spectrum of dechlorinated PVC product using 1 M NaOH/TEG solution for analysis.
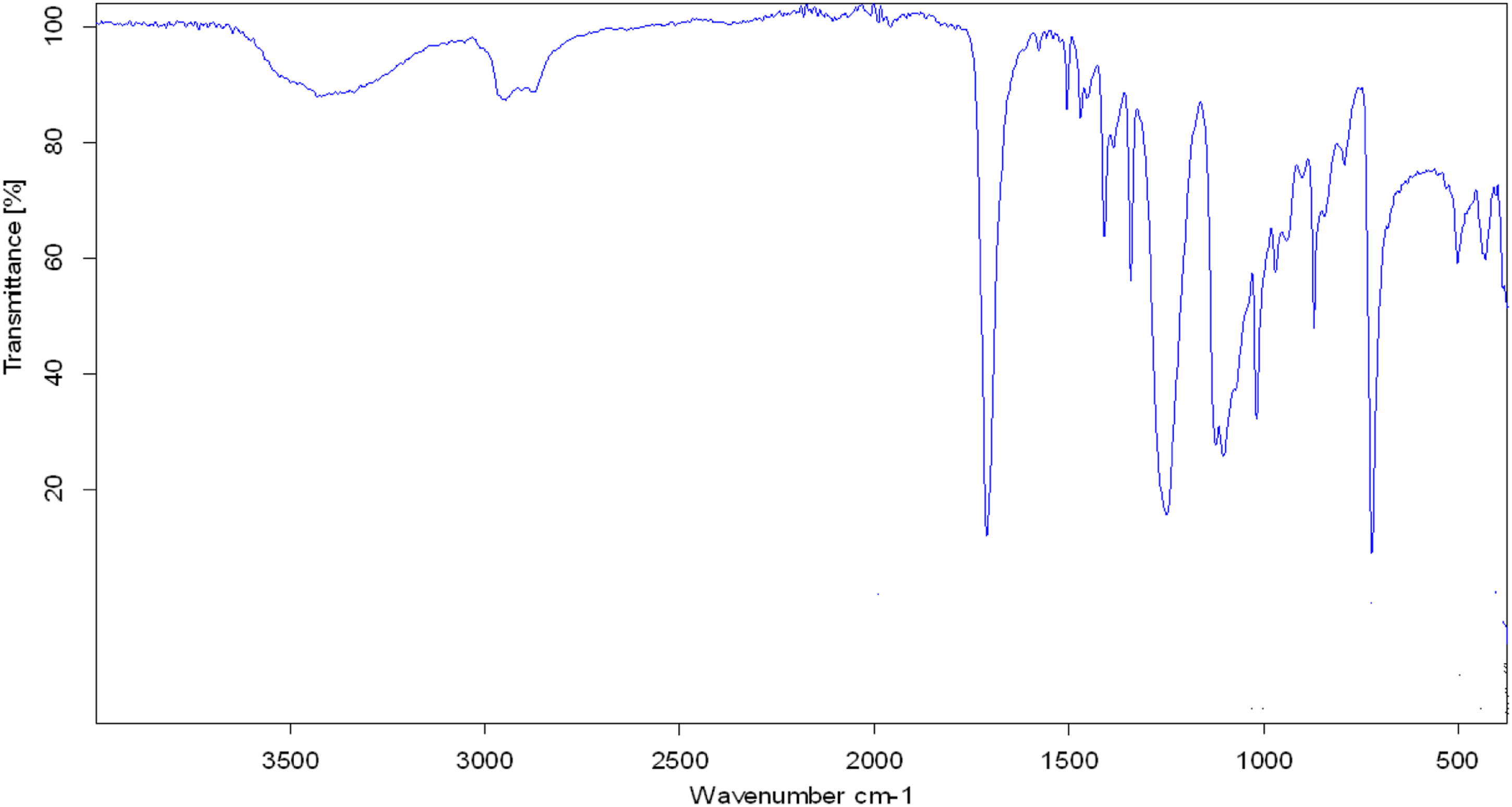
Moreover, the presence of the absorption band at 723 cm−1 (Figure 7) which belongs to the (ɣ(C-Cl)str) means that the product still contains some PVC units.
The thermogram of PVC dechlorinates product (Figure 8 and Table 2) shows completely different thermal data in comparison with the pristine PVC thermogram (Figure 2 and Table 2). Where the weight loss % at FDT is low, it means that the formed product has a high thermal stability and this is confirmed with the low weight loss per minute in DTG thermogram (Figure 8 and Table 2). Moreover, the glass transition temperature Tg and the heat of fusion ∆Hf (Figure 8 and Table 2) have proved that the PVC dechlorinated product has a stable structure and has exothermic behavior on degradation as well as stable crystalline structure according to its high crystalline temperature (Tcr) of 400oC (Figure 8 and Table 2), in comparison with 296oC of pristine PVC waste (Figure 2 and Table 2).
21
TG, DTG, and DTA thermogram of dechlorinated PVC product using 1 M NaOH/TEG solution for analysis.
Conclusions
The wet treatment process was carried out for the recycling of PVC waste using NaOH/diol solvent, and the following points were observed: 1. The concentration of alkali solution shows a significant effect on the dechlorination of PVC waste and 1 M NaOH concentration was suitable. 2. The temperature and the time of dechlorination analysis of PVC waste show a certain level of effects on DD% of PVC. 3. The type of diol solvent used in the wet treatment process shows the effects on DD% of PVC according to its solubility parameters. Accordingly, the 1 M NaOH/EG solution systems reached its maximum DD% at the temperature reaction of 196oC and after 85 min of analysis (Figures 3 and 4). The 1 M NaOH/TEG solution system has shown the highest DD% of PVC at 160oC temperature analysis and after 180 min of reflux (Figures 5 and 6). The lower temperature in NaOH/TEG system is important for there are no dissociated chains of the dechlorinated PVC product. 4. Finally, the treated poly(vinyl chloride) waste with 1 M NaOH/TEG solution at typical analysis conditions gives dechlorinated products: mainly poly(vinyl alcohol) PVA according to the FTIR studies and a small number of unsaturated ethylene units beside few units of non-decomposed (chlorinated) PVC.
Footnotes
Acknowledgments
Authors are thankful for university of Mosul, Iraq, for providing facilities to carry out this work.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This article was supported by University of Mosul.
Ethical approval
Authors have read and approved the manuscript and there are no ethical issues associated with this research. This article is original and contains unpublished materials.