Abstract
Generally, polyamide cannot be used as film blowing material because of its unsuitable properties. In this study, polyamide 6 clay nanocomposite (cPA) and styrene maleic anhydride copolymer (SMA) were mixed in various ratios for the preparation of modified polyamide 6 clay nanocomposite SxcPAy resins by reactive extrusion. The S1cPA14 resin was blended with recycled maleic anhydride polyamide (rPA) to form the (S1cPA14)x rPAy resins. Finally, they were mixed with LDPE in 1:9 ratio to afford (SxcPAy)1LDPE9 and ((S1cPA14)x rPAy)1LDPE9 resins, respectively, followed by film blowing and the analyses of the physicochemical properties of resins. The FTIR spectrum illustrated that the C=O symmetric and asymmetric absorption fingerprint peaks in the anhydride (-OC-O-CO-) group of SMA disappeared and the new characteristic absorption peak of-CO-N-CO- of imides was observed. The anhydride functional group of SMA underwent reactive extrusion with the terminal amino group of cPA to generate the imides structure. The thermal properties showed that the glass transition temperature and crystallinity of SxcPAy and (S1cPA14)x rPAy resins increased with increasing SMA and S1cPA14 contents. The Tg (85.4.0°C) of (S1cPA14)12 rPA1 resin were enhanced significantly, with 30°C higher than cPA. In terms of tensile mechanical properties, S1cPA14 test pieces demonstrated the highest Young’s modulus and tensile strength. After mixing with LDPE, the tensile mechanical properties of (SxcPAy)1LDPE9 and ((S1cPA14)x rPAy)1LDPE9 resins and films were both higher than that of LDPE. ((S1cPA14)12 rPA1)1LDPE9 film shown the best tensile properties and barrier performance compared with other films due to the optimal rPA content could assisted SMA as a better compatibilizer to improve the dispersion and compatibility of cPA in HDPE. It was worth noting that (SxcPAy)1LDPE9 and ((S1cPA14)x rPAy)1LDPE9 resins were formed by film blowing at the processing temperature of 140°C followed by successful preparation of the film.
Introduction
Polyamide (PA) is one of the most widely engineering plastics due to its outstanding properties, such as excellent chemical resistance, barrier performance, high tensile strength, and flexibility. 1 But the price of PA is high, and cannot be used as film blowing material. Low-density polyethylene (LDPE) is one of the most commonly plastics because of good processability and low cost, and it is a good for blowing film. 2 By mixing PA and LDPE, the advantages of the two polymers can be combined to obtain a resin with blowing film, low cost, and better barrier property.
The development of polymer blends usually successfully enhanced the physical properties of single polymer material and it is a simple and quick method. The technical development of polymer blends is a market trend to serve as an economical and effective method for developing new resins.3,4 The polymer blending methods can be categorized as: 1. miscible blends, such as polyphenylene oxide/polystyrene and poly(vinyl chloride)/selected polyesters. The properties of such blends are usually additive. Moreover, mutual interactions occur between the polymers in the blends with the same glass transition temperature (Tg).5–8 2. Immiscible blends: The mutual compatibility of such blends is extremely low, requiring the addition of another functional polymer such as surfactant or a compatibilizer. The addition of a compatibilizer can reduce the surface tension between incompatible polymer blends1–11 It can prevent the coagulation of disperse phases and improve their stability in the matrix along with the fine distribution characteristics.12–14
In our previous study, PA, EVOH, and HDPE were blended. As the PA and EVOH materials contain the polar amide functional group and 55–75% of polar ethanol functional group, respectively, they were incompatible with nonpolar HDPE. During the co-extrusion or laminar blow-molding processes, these PE/PA, PE/PVA, PE/EVOH, and PE/NYC blends were thermally immiscible and mechanically incompatible. The compatibilizer precursor (CP) resin is usually used to improve the surface property of PE to PA, PVA, and/or EVOH and to enhance the barrier property of PE/PA/CP blends.15–18 The carboxylic acid group on CP and the amine group on PA underwent reactive extrusion to afford the CP-grafted-PA copolymers similar to the cross-linked CP/PA copolymers. Such CP/PA copolymers significantly enhanced the melting-shear viscosity of Modified PA. As a result, its viscosity was increased with increasing CP percentage. 16 Furthermore, in related studies, Lim et al. 19 blended polyamide 12 (PA12)/styrene ethylene/butylene styrene (SEBS) and PA12/maleic anhydride grafted SEBS (SEBS-g-MA) individually by the double screw method. The differential scanning calorimetry (DSC) results indicated that PA12/SEBS-g-MA showed improved thermal stability than PA12/SEBS while scanning electron microscope (SEM) revealed favorable phase morphology of PA12/SEBS-g-MA. Padwa et al. 20 used PE and maleic anhydride for grafting modification to give PE-g-MA as CP followed by blending of nylon6/PE-g-MA. The results showed that, in the compatibilization study, identical outcome on the impact property to those in the publications of grafting maleic anhydride onto the polyethylene phase21–25 were achieved.
Upon the arrival of the era of high oil prices, the prices of petroleum cracking products have increased dramatically. On the other hand, the plastic degradation requires as many as a hundred years and are associated with numerous issues such as the impact on the environment. As a result, the government of each country adopts various promotion measures with rewards to encourage the industries and the public to recycle waste, with satisfactory outcomes achieved. Nowadays, waste recycling has created tremendous profits in each country around the world. In 2012, the total income of waste recycling in Taiwan reached NTD $7,030,000,000, 498,000 tons of waste recycled. From 1998 to 2012, the volume of garbage clearance per capita per day was reduced from 1.14 kg to 0.39 kg (Environmental Protection Administration, Executive Yuan, the R.O.C.). Therefore, the objectives of waste reduction and transformation of garbage to gold were achieved. In the United States, recycled plastics rapidly grow by an annual rate of 5.9%. It is forecasted to reach 3,400,000,000 pounds in 2016 (market report by the Freedonia Group). PET and HDPE plastics account for the majority of recycled plastics market. The research and development of recycled products has also become mature. The sports apparels of 9 football teams including Holland and Germany in the 2010 Football World Cup were made from textiles produced from recycled PET in Taiwan. However, there were few studies and reports on the related products of recycled nylon 6 plastics, which is deemed as important as PET plastics in terms of synthetic plastics. Recycled maleic anhydride PA (rPA) is one of polymer waste from plastic processing factory. However, there was no study reports in rPA, it might be a good filler for polymer technology. Polyamide 6 clay nanocomposite (cPA) is an excellent barrier packaging material, which has been reported in previous study. 15 However, cPA is very expensive, and cannot be used as blown material. Therefore, there are three main purpose of the study, the first is to reduce the cPA price, because the more LDPE is added, the lower the cost. The second is to improve the film blowing processability of cPA. The third is that the recycling of rPA can reduce environmental hazards and reduce material costs. It was surprising that rPA could also be used as a compatibilizer in a suitable proportion. Therefore, the present study investigated the blending of polyamide 6 clay nanocomposite (cPA) and styrene maleic anhydride copolymer (SMA) in varied ratios to prepare the modified polyamide 6-clay nanocomposite (SxcPAy) resin by reactive extrusion. The S1cPA14 resin and rPA were blended in different ratios to afford the (S1cPA14)x rPAy resin. Finally, they were mixed with LDPE in 1:9 ratio to afford the (SxcPAy)1LDPE9 and ((S1cPA14)x rPAy)1LDPE9 resins attributed to LDPE is cheap, and easy to blow film, but poor barrier properties. The ratio of PA to LDPE is a continuation of previous study, 15 all resins followed by film blowing for molding. The FTIR spectroscopy, differential scanning calorimetry (DSC), tensile mechanical properties, morphological properties, and optical properties of resins were also investigated and discussed.
Experimental
Materials and sample preparation
The Polyamide 6 Clay Nanocomposite (cPA), compatibilizer precursor (SMA) and LDPE used in this study were obtained from Nanopolymer Composites Corporation (Tainan, Taiwan), wherein cPA resins with a trade name of NF3020 and SMA is a Styrene Maleic Anhydride copolymer. The modified cPA (SxcPAy) resin was prepared by reactive extrusion of the melt blending of SMA and cPA in various ratios. The SMA, recycled maleic anhydride polyamide 6 (rPA) and the low-density polyethylene resins (LDPE NA207-66) used in this study were obtained from Chembridge International Corporation (Taipei, Taiwan), Liang Haw Technology Corporation (Taipei, Taiwan) and USI Corporation (Taipei, Taiwan), respectively.
The SMA compatibilizer, cPA resins and rPA were prepared by reaction extrusion, the dried SMA, cPA and rPA resins at varying ratios using a Nanjing Jiant SHJ-20 twin-screw extruder. Before melt blending, SMA, cPA and rPA resins were dried in a vacuum oven at 80°C for 16 hrs. cPA resins were mixed with SMA and S1cPA14 resins were mixed with rPA by dry blending in varying ratios separately followed by twin-screw extruder operating at 240°C in the feeding zone and 230°C toward the extrusion die with a screw speed of 200 rpm. The compositions of the SxcPAy and (S1cPA14)xrPAy resins prepared in this study are summarized in Table 1.
Compositions of the SxcPAy and (S1cPA14)xrPAy resins.
The SxcPAy and (S1cPA14)xrPAy pellets prepared from the twin-screw extruder was dried at 80°C for 16 h, and then dry blended with LDPE at a 10:90 weight ratios. The mixed (SxcPAy)1LDPE9 and ((S1cPA14)x rPAy)1LDPE9 pellets were then blown films in a Jonh Huah TPH-550 extrusion blown film molding machine using an extrusion temperature of 140°C and a screw speed of 50 rpm, respectively.
Attenuated total reflectance Fourier transform infrared spectroscopy (ATR-FTIR)
ATR-FTIR measurements of cPA, SxcPAy, (SxcPAy)1LDPE9 and ((S1cPA14)x rPAy)1LDPE9 specimens were recorded on a Jasco-460 ATR-FTIR spectrophotometer at 25°C and 20% relative humidity, wherein 32 scans with a spectral resolution of 4 cm−1 were collected during each spectroscopic measurement. The cast films used in this study were prepared sufficiently thin enough to obey the Beer–Lambert law.
Thermal properties
The thermal properties of cPA, SxcPAy, (SxcPAy)1LDPE9 and ((S1cPA14)x rPAy)1LDPE9 resins were determined at 25°C and 20% relative humidity using a Maia 200-F3 differential scanning calorimetry (DSC) machine. All scans were carried out at a heating rate of 10°C/min and under flowing nitrogen of a flow rate of 25 mL/min. The instrument was calibrated using pure indium. Samples weighing about 7 mg were placed in standard aluminum sample pans for each DSC experiment.
Mechanical properties
Tensile strength tests of cPA, SxcPAy, S1cPA14)x rPAy, (S1cPA14)x rPAy)1LDPE9 and (SxcPAy)1LDPE9 sheets and films were carried out in an HT-9501 Universal Testing Machine Indicator, according to ASTM-D 638 at a cross head speed of 100 mm/min. The results reported are the average of 10 tests.
Morphology properties
In order to observe the deformation structures of cPA and S1cPA14 resins in the cPA1LDPE9 and (S1cPA14) 1LDPE9 sheets, respectively, these sheets were first sectioned by a scalpel, and then etched with formic acid. The etched samples were then gold-coated and examined by a scanning electron micrograph (SEM, JSM-6335F JEOL, Tachigawa, Japan).
Water vapor leakage test
The water barrier properties of the LDPE, cPA1LDPE9, (SxcPAy)1LDPE9 and ((S1cPA14)x rPAy)1LDPE9 films were evaluated by measuring the weight loss of water contained in the 200 ml conical flask with 100 g water. The film sample was cut into 10 × 10 cm2, the thickness of the film was 0.05 mm, adhered to the mouth of the conical flask with vacuum glue, and put into the oven at 40°C for 14 days. For each experiment, at least 3 samples are taken to calculate the average value. The water vapor permeation rate was calculated using the following equations:

where W0 was the weight before putting into the oven (conical flask + 100 g water); Wx was the weight of (conical flask + water) on the x day after placing in the oven.
UV-VIS transmission properties
Ultraviolet-visible (UV-vis) spectra of the (SxcPAy)1LDPE9 and (S1cPA14)x rPAy)1LDPE9 films were recorded on a Hitachi U3900 spectrophotometer at room temperature with the scanning range of 200 to 800 nm.
CIE LAB color intensity measurement
The color intensities of (SxcPAy)1LDPE9 and (S1cPA14)x rPAy)1LDPE9 thin films were measured by X-rite Color SP60 spectrophotometer with the standard D65 light source at the angle of 8 degrees.
Results and discussion
ATR-FTIR spectroscopy
Typical Fourier transform infrared spectra (ATR-FTIR) of SMA, SxcPAy, and cPA samples were shown in Figure 1. The distinguished absorption bands of SMA sample centered at 1225, 1600, 1779/1860 and 3433/3630 cm−1 are most likely corresponding to the motions of C-O-C stretching of the anhydrides, C=C stretching of the styrene, O=C-O-C=O symmetric and asymmetric stretching of the anhydrides and O-H stretching vibration present in SMA molecules, respectively. Similar to those found by Yeh et al., 14 the FT-IR spectra of cPA exhibit three distinguished absorption bands centered at 1542, 1642, and 3295 cm−1, which are attributed to the motions of N-H bending vibration, C=O stretching vibration, and N-H stretching of PA molecules, respectively. In contrast, the FT-IR spectra of SxcPAy are very similar to that of the cPA specimen, wherein the three main absorption bands centered at 1542, 1636, and 3298 cm−1 were also found in the spectra of SxcPAy. However, the 1779/1860 (O=C-O-C=O symmetric and asymmetric stretching of the anhydrides) absorption bands originally shown on the FT-IR spectra of SMA sample almost disappeared completely, and found the new imides group absorption bands centered at 1734 cm−1 in the SxcPAy samples. Presumably, this disappearance is due to the for formation of cPA-grafted-SMA copolymers through the reaction of carboxylic acid groups of anhydrides of SMA with the terminal amine groups of cPA molecules during the reactive extrusion of SxcPAy resins.26,27 Typical ATR-FTIR of rPA, S1cPA14 and (S1cPA14)xrPAy samples are shown in Figure 2. After blending, the ATR-FTIR of the (S1cPA14)xrPAy samples were similar to that of the S1cPA14 sample. The possible mechanisms of the reaction extrusion of the SxcPAy resins are suggested in the Figure 3.
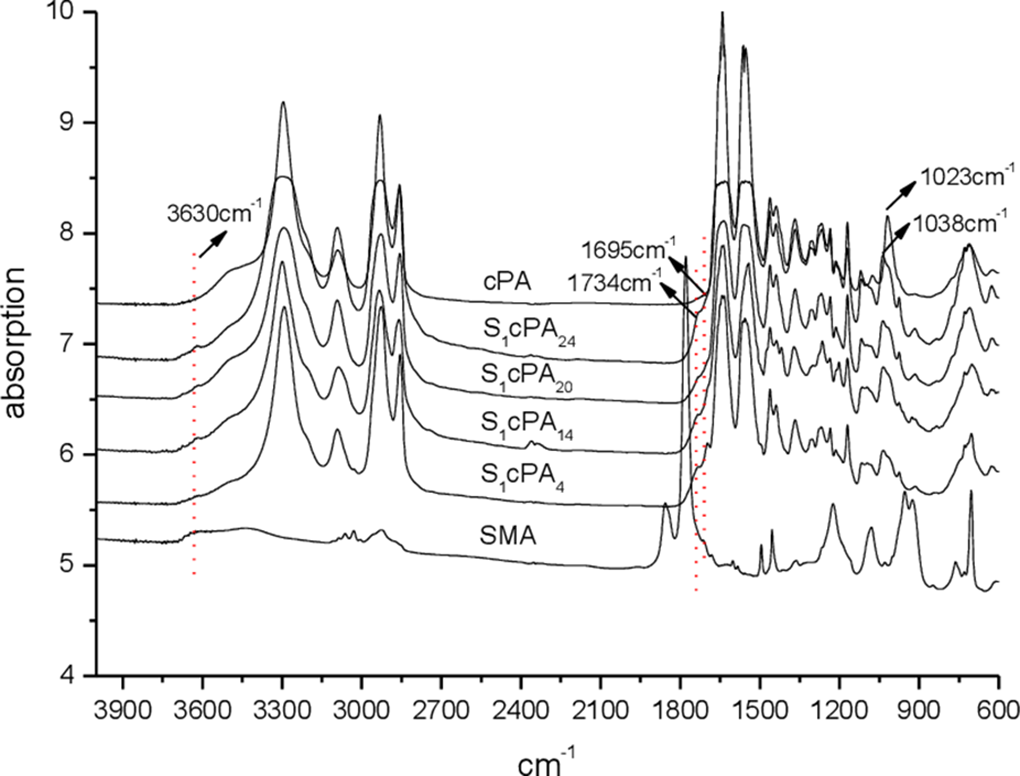
ATR

ATR

The possible mechanisms of the reaction extrusion of the SxcPAy resins.
Typical ATR-FTIR of (SxcPAy)1LDPE9 and ((S1cPA14)12 rPA1)1LDPE9 samples are shown in Figure 4. The distinguished absorption bands of (SxcPAy)1LDPE9 and ((S1cPA14)12 rPA1)1LDPE9 samples centered at 1437, 1542/1642, 1726 and 3295 cm−1 were most likely corresponding to the motions of C-H bending of the LDPE, NH/C=O stretching of the cPA, -OC-N-CO- stretching of imides group of the cPA-grafted-SMA copolymers and N-H stretching vibration present in (SxcPAy)1LDPE9 molecules, respectively.

FTIR-ATR spectra of (SxcPAy)1LDPE9 and ((S1cPA14)x rPAy)1LDPE9 samples determined at 35°C.
Thermal behavior
Figure 5 summarized the DSC thermograms of the second heating of SMA, cPA, SxcPAy, (S1cPA14)x rPAy and rPA resins, including the glass transition temperature (Tmid) graph of cPA, and (S1cPA14)12 rPA1 resins.

DSC thermograms of the second heating scan for SMA, cPA, SxcPAy, (S1cPA14)x rPAy and rPA resins.
In addition, the crystallinity, Xc, was calculated as follows 28 :

where △Hf is the apparent enthalpy of fusion (indicated in DSC thermograms as the melting enthalpy per gram of blends) corresponding to the component, ω is the total weight fraction of the cPA in the blend, and △Hf0 is the average enthalpy of fusion per gram of the component in its completely crystalline state (240 J/g for PA; α form=241J/g and γ form=239J/g). 29 PE completely crystalline state (289 J/g for LDPE). 27 The Tg, Tm, △Tm, and Xc of cPA, SxcPAy, and (S1cPA14)x rPAy samples were illustrated in Table 2. As shown in the results, the Tg, Tm, △Tm, and Xc of cPA are 55.4°C, 219.1°C, 15.0°C, and 18.1%, respectively. It was found that the Tg of SxcPAy blending sample increased with increasing SMA, with the Tg (72.24°C) of S1cPA4 (small graph in Figure 5) was 16.8°C higher than that of CPA. In the (S1cPA14)x rPAy resin, it was observed that the sample with added edge trim showed higher Tg and Xc than S1cPA14 sample. Among them, (S1cPA14)12 rPA1 demonstrated the highest Tg and Xc of 85.4°C and 21.7%, respectively (small graph in Figure 5 and Table 2).
Melting behavior of cPA, SxcPAy, (S1cPA14)xrPAy and rPA resins.

*△Tm is the half-height width of the Tm peak.
Figure 6 and Table 3 illustrated the DSC thermograms and thermal properties of the second heating of LDPE, (SxcPAy)1LDPE9, ((S1cPA14)x rPAy)1LDPE9, and cPA resins. The results revealed that the Tm and Xc of LDPE were 109°C and 42.9%, respectively. In the blending sample of (SxcPAy)1LDPE9, the Tm values of LDPE samples were found to be little changed. On the other hand, the Tm and Xc of SxcPAy samples were found to decrease significantly, with S1cPA14 showing the most significant decreases by 13.6°C and 13.59%. Based on the aforementioned results, the modified SxcPAy resin, following being blended in the LDPE matrix, aided in the crystallization of LDPE. Yet, the crystal plates of its own decreased, with S1cPA14 dropping the most (4.51%, Table 3). In the ((S1cPA14)x rPAy)1LDPE9 samples, there were little changes in the Tm values of LDPE samples, yet Xc values were found to decline. In addition, the crystal melting points of nylon in the ((S1cPA14)4 rPA1)1LDPE9 and ((S1cPA14)12rPA1)1LDPE9 disappeared (Table 3).
Melting behavior of LDPE, (SxcPAy)1LDPE9, ((S1cPA14)x rPAy)1LDPE9 and cPA resins.

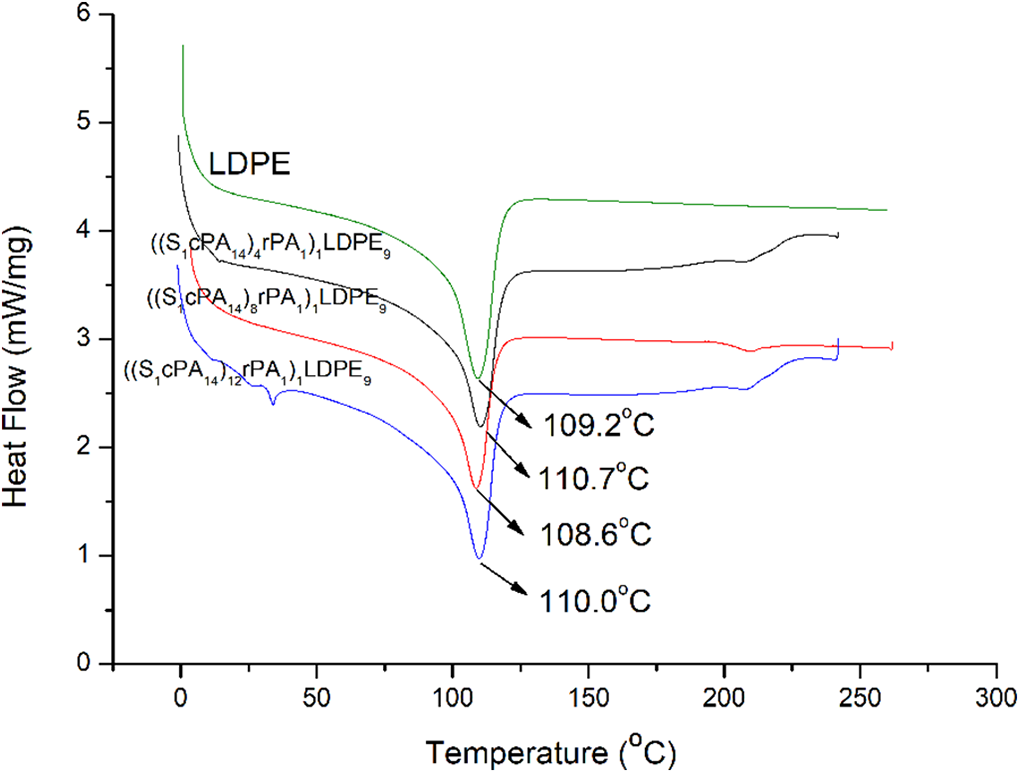
DSC thermograms of the second heating scan for LDPE, (SxcPAy)1LDPE9, ((S1cPA14)x rPAy)1LDPE9 and cPA resins.
The results of thermal analysis showed that the Tg value of (S1cPA14)12 rPA1 resin was 30°C higher than that of cPA, which may be attributed to the formation of dense polyimide structure under the optimum rPA content in S1cPA14, which leads to the decrease of molecular chain fluidity and the increase of glass transition temperature of the resin.30,31
Mechanical properties
The assessments of mechanical properties of the injected pieces of cPA, SxcPAy, (S1cPA14)x rPAy, LDPE,(SxcPAy)1LDPE9, and ((S1cPA14)x rPAy)1LDPE9 are summarized in Table 4. The results show that the Young’s modulus (E), Tensile strength (TS), and Elongation at break (ε (%)) of the cPA test pieces were 0.491 GPa, 40.3 MPa, and 8.30%. Among SxcPAy resins, those were found that the E values were all greater than that of cPA except S1cPA24 resin, with S1cPA14 resin having the highest E, TS, and ε (%) of 0.82 GPa, 42.8 MPa, and 5.59%, respectively. With respect to the mechanical properties of LDPE test pieces, the E, TS, and ε (%) were 0.081 GPa, 7.85 MPa, and 39.3%. For (SxcPAy)1LDPE9, the E and TS of all samples were higher than those of LDPE. (S1cPA14)1LDPE9 showed the highest E and TS of 0.191 GPa and 11.1 MPa, 1.47 and 1.41 times higher than those of LDPE, respectively. Moreover, (S1cPA14)1LDPE9 resin also showed the highest elongation at break of 48.2%, about 1.23 times higher than that of LDPE. Compared to cPA and (S1cPA14)x rPAy resins showed lower TS and E, but higher ε (%). The better ε (%) was also one of the reasons for improving the blowing properties. 32 Therefore, the resins were blended with LDPE before blown film.
Mechanical properties of the blends prepared by injection
E = Young modulus; TS = Tensile strength; ε = Elongation at break.
Table 5 summarizes the mechanical properties along the X and Y axes of LDPE, (SxcPAy)1LDPE9, and ((S1cPA14)x rPAy)1LDPE9 thin films from blowing process. It was shown that all thin films of the same test pieces possessed larger elongation at break along the X-axis than the Y-axis. However, the E and TS values of the Y-axis were generally higher than those of the X-axis. The Young’s modulus (E), Tensile strength (TS), and Elongation at break (ε (%)) of the LDPE thin films along the X and Y axes were 0.122 GPa, 8.271 MPa, 169.9%, 0.260 GPa, 11.08 MPa, and 69.25%, respectively. The data along the X and Y axes of (SxcPAy)1LDPE9 were all larger than those of LDPE. Among them, the TS and Elongation at break along the X and Y axes of (S1cPA24)1LDPE9 thin film were 16.58 MPa, 182.5%, 17.76 MPa, and 104.6%, respectively. The E and TS values along the X and Y axes of the ((S1cPA14)x rPAy)1LDPE9 thin film were also higher than those of LDPE, with the ((S1cPA14)12 rPA1)1LDPE9 thin film showing the largest TS values along the X and Y axes of 16.80 MPa and 19.39 MPa, respectively.
Mechanical properties of the blends prepared by blown molding (blow-up rate of 3.5 and film thickness of 0.05 mm).

X-Axis (horizontal); Y-Axis (vertical).
In summary of Tables 4 and 5, the Young’s modulus and tensile strength of resins were increased in the injection or thin film test pieces containing either 10% modified nylon or recycled modified nylon. Blown film with optimal rPA ratio presented the best E and TS values, this may be because cPA could further enhanced the compatibility of S1cPA14 in LDPE. 33
Morphological properties
The morphological property analysis of the cPA1LDPE9, (S1cPA14)1LDPE9 and ((S1cPA14)12 rPA1)1LDPE9 were summarized in Figure 7. As the results indicated, the cPA without the addition of SMA was blended in the LDPE matrix to show larger cPA streaking dots (0.50–2.00 μm) in the Figure 7(a). The smallest size of S1cPA14 dots in the (S1cPA14)1LDPE9 test pieces was approximately 0.05–0.1µm (see Figure 7(b)), improved by 10–20 times as compared to the cPA1LDPE9 pieces. When rPA was added with (S1cPA14)1LDPE9, the cross section of the film becomes smoother, and its streaking dots almost disappear (see Figure 7(c)). This indicated that the addition of rPA could cooperated with SMA as compatibilizer. These results are consistent with the analysis of mechanical properties, which proved that the compatibility, TS and E of the blown film could be further improved by the combination of rPA and SMA.34,35

SEM photos of (a) cPA1LDPE9, (b) (S1cPA14)1LDPE9, (c) ((S1cPA614)12 rPA1)1LDPE9.
Analysis of water vapor leakage properties
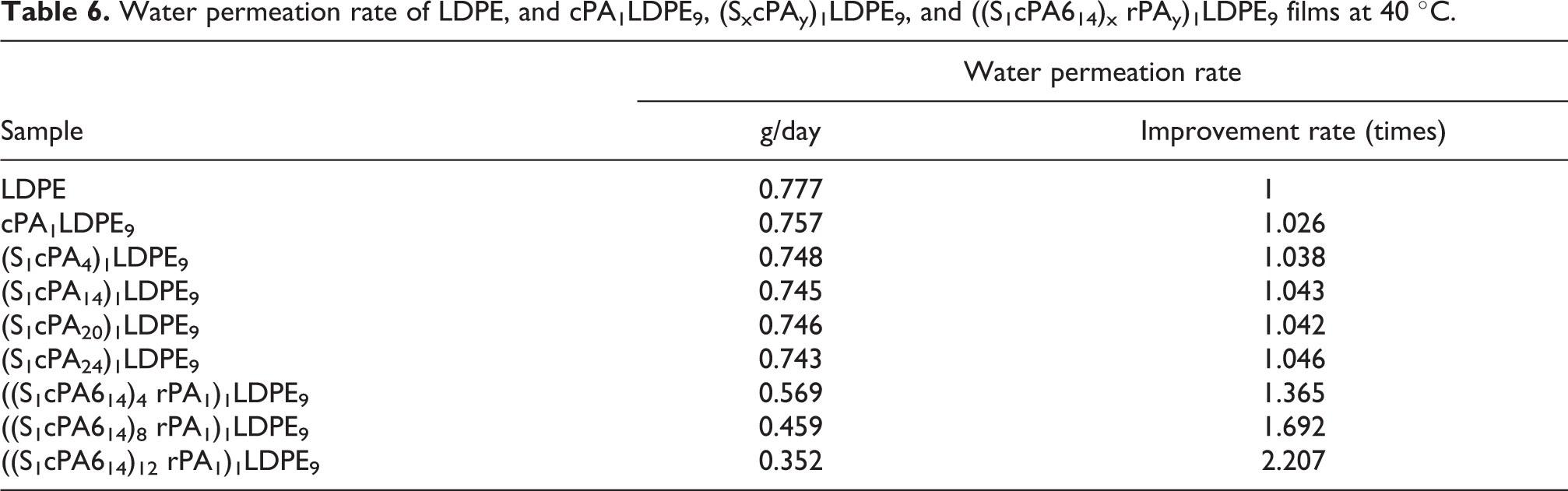
Table 6 summarizes the water vapor leakage properties of LDPE, cPA1LDPE9,(SxcPAy)1LDPE9 and ((S1cPA14)xrPAy)1LDPE9 films. The results show that the water vapor leakage of cPA1LDPE9 film was similar to that of neat LDPE. ((S1cPA14)12rPA1)1LDPE9 film has the smallest water vapor leakage (0.352g/day), and the improvement rate of water vapor leakage was more than twice that of LDPE. From the results of the thermal properties (Table 2), mechanical properties (Table 5) and morphology analysis (Figure 7) indicated that the presence of optimal rPA content could enhanced the interfacial compatibility of various materials. It might be attributed to rPA plays the role of compatibilizer, which reduced the defects of the resins and improved the water vapor barrier ability. 36
Water permeation rate of LDPE, and cPA1LDPE9, (SxcPAy)1LDPE9, and ((S1cPA614)x rPAy)1LDPE9 films at 40 °C.
Optical transparency
Figure 8 illustrates the optical transparency (thickness of 40–60 μm) (SxcPAy)1LDPE9 and ((S1cPA14)xrPAy)1LDPE9 films in the UV-VIS spectrum. 37 These optical transparencies include cutoff wavelength (absorption edge, λ0) and transmittance of various wavelengths as shown in Table 7. The results indicate that the cutoff wavelengths of all films were below 226 nm and transmittance increased with increasing wavelength. The transmittance of LDPE at 500 nm was 59% as a translucent film. The transmittances of films following modification all decreased, with (S1cPA24)1LDPE9 showing the highest transmittance of 51% at 500 nm. The transmittances of films after the addition of recycled nylon were found to decline further.
Optical transparency of LDPE, (SxcPAy)1LDPE9 and ((S1cPA14)xrPAy)1LDPE9 films.

λ0: UV cutoff wavelength; T400–700: transmittance at 400–700 nm, respectively.
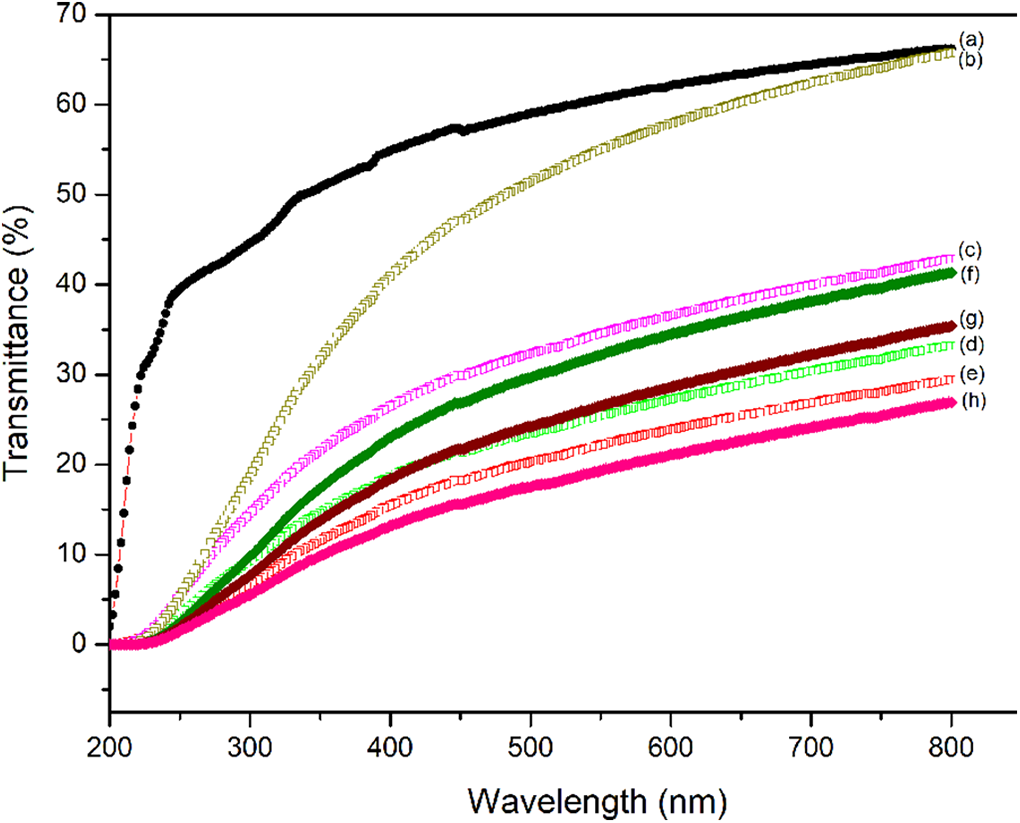
UV-vis spectra of (a) LDPE, (b) (S1cPA24)1LDPE9, (c) (S1cPA20)1LDPE9, (d) (S1cPA14)1LDPE9, (e) (S1cPA4)1LDPE9, (f) ((S1cPA14)4rPA1)1LDPE9, (g) ((S1cPA14)8rPA1)1LDPE9, (h) ((S1cPA14)12rPA1)1LDPE9 films.
CIE LAB system
The CIE LAB color coordinates of LDPE, (SxcPAy)1LDPE9, and ((S1cPA14)xrPAy)1LDPE9 films are illustrated in Figure 9 and Table 8. 37 LDPE was a bluish green film with more blue tone. Following modification, (SxcPAy)1LDPE9 films were mostly bluish green films with more green tone. With the addition of rPA, ((S1cPA14)xrPAy)1LDPE9 films were converted to yellowish green on appearance. The (S1cPA14)1LDPE9 and ((S1cPA14)12rPA1)1LDPE9 film were demonstrated as an example (Figure 10).
Color coordinates of LDPE, (SxcPAy)1LDPE9 and ((S1cPA14)xrPAy)1LDPE9 films.a

a The color parameters were calculated according to a CIE LAB equation. L* refers to lightness; 100 means white, while 0 indicates black. A positive a* means red color, a negative a* indicates green color. A positive b* means yellow color, a negative b* indicates blue color. H* refers to hue angle. 29

CIELAB coordinates for the LDPE, (SxcPAy)1LDPE9 and ((S1cPA14)xrPAy)1LDPE9 films in the a*–b* plane.

Photographs of (a) (S1cPA614)1LDPE9 and (b) ((S1cPA614)12 rPA1)1LDPE9 films.
Conclusions
The application of reactive extrusion to prepare SxcPAy resins showed new characteristic absorption peak of –CO-N-CO- of imides at 1734 cm−1 in ATR-FTIR. The thermal properties indicated that the glass transition temperatures and crystallinity of SxcPAy and (S1cPA14)xrPAy increased with increasing SMA and S1cPA14. The Tg (85.4.0°C) of (S1cPA14)12rPA1 resin were enhanced significantly, showing 30°C higher than that of cPA. The tensile mechanical properties revealed that the Young’s modulus and tensile strength both increased but the elongation at break declined with the addition of 10% of modified or recycled nylon in either injection or thin-film test pieces, with the exceptions of (S1cPA14)1LDPE9 and (S1cPA20)1LDPE9 injection pieces and (S1cPA24)1LDPE9 film test pieces, which also showed favorable elongation at break. The morphological properties demonstrated that the SxcPAy resins were scattered as smaller spherical particles in the LDPE matrix than unmodified cPA resins. Further blending with rPA, (S1cPA14)1LDPE9 film could hardly be seen in LDPE, which indicated that rPA could assist SMA to improve the dispersion and compatibility of SxcPAy in HDPE. The optical transparency test showed that the transmittance of LDPE was 59% at 500 nm as a translucent film. Following modification, the transmittance of (S1cPA24)1LDPE9 thin film at 500 nm was 51%, the highest among all. The color coordinates suggested that LDPE and (SxcPAy)1LDPE9 films were bluish green thin films while ((S1cPA14)xrPAy)1LDPE9 films, with the addition of recycled nylon, were altered to yellowish green. Compared with all the resins, (S1cPA14)12 rPA1 resin showed the highest Tg value, and the blown film after blending with LDPE also showed the highest E, TS and barrier performance, indicated that the optimal content of rPA could enhanced the compatibility between S1cPA14 and LDPE. It is worth noting that, both (SxcPAy)1LDPE9 and ((S1cPA14)x rPAy)1LDPE9 resins were prepared by blow-molding at the processing temperature of 140°C to complete the preparation of films. The reuse of recycled nylon edge trims, improve thermal and tensile mechanical properties, the application of simple and affordable double screw extruder, and the energy-saving blow processing are important indicators for the market applications.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This present research was supported by the Oriental Institute of Technology and Industry Cooperation Support Program (103-5-01-105, 103-5-01-104).
