Abstract
The public concern for industrial pollution is urging for an economical solution to remove industrial effluent that increases the risk of human cancers. Currently, there are a few successful techniques implemented for dye removal; however, little has been done in the field of using reusable biological material extracted from waste. We propose a new procedure to fabricate biocompatible magnetic collagen for dye removal. Our study was conducted on crystal violet (CV) removal from industrial effluent using fish scales collagen. In this study, removal of dye by adsorption on various types of collagen was studied using soft, coarse and magnetic collagen nanocomposite. The magnetic collagen nanocomposite was synthesized by coprecipitation method followed by mixing of iron nanoparticles with collagen. Characterization of the prepared materials was performed using transmission electron microscopy (TEM) and Fourier transform infrared spectroscopy (FTIR). Soft collagen, coarse collagen and magnetic collagen nanocomposite were proved to adsorb crystal violet dye at concentrations up to 1000 ppm. Uptake capacity was 92% at 100 ppm of CV while cytotoxicity was reduced to 15%. The ability of collagen and magnetic collagen nanocomposite to adsorb the dye and thus reducing the polluted water cytotoxicity, as well as accumulating under the action of the magnetic field was proved experimentally.
Introduction
At the early stages of the 20th century, synthetic dyes replaced dyes from animals and plants to for use in color substances such as in fabric, dying, printing, pharmaceuticals, paper production, leather, foodstuffs and medicine generating a large amount of colored wastewater. Textile industry is considered as the greatest consumer of dyes constituting 60% of total dyes.1,2 Generally, dyes are classified based on either their application or chemical behavior into acidic, basic, direct, scatter, metallic, penetrating, pigments, and reactive dyes. Industrial dyes are one of the most difficult contaminants to be removed from wastewater as they are aerobically indigestible, light stable and biologically undegradable. 3
Factory-discharged dyes are major environmental contaminants that damage ecosystems enormously when released without proper treatments. Harmful dyes can be acquired through inhalation, ingestion or through direct contact with skin which may cause cancer, eye irritation, skin irritation and allergic reactions.4,5 Even at very low concentration (1 ppm), untreated dyes may cause water to vary from its natural color, 6 rendering it not only unacceptable for drinking but also shielding light from the deeper water and consequently altering the fauna of the aqueous ecosystems.2,7
While contaminated water can be treated physically, chemically or biologically, conventional wastewater treatment methods were always considered as inefficient for synthetic dyes due to their chemical stability. 3 Physical treatment method based on adsorption is considered as the most inexpensive convenient method. 8 The latter method can be considered as the safest azo dye’s removal method with no azo bond breakage, reducing the toxic aromatic amines generation. 9 Synthetic dyes such as crystal violet (CV) (N, N, N’, N’, N”, N”-hexamethylpararosaniline) dye has been extensively used in medicine and in textile industry.10,11
Crystal violet is considered as a severe environmental damaging agent when it is released into water, as it is absorbable into fish tissue. Furthermore, some reports suggested that CV is a mutagen and mitotic poison that induces renal, hepatic and lung tumor in mice and may increase the risk of human bladder cancer.12,13 Unfortunately, traditional wastewater treatment methods have failed to eliminate crystal violet from contaminated wastewater, thus increasing pollution of aqueous ecosystem.14,15 Eco-friendly Biomass-based materials exhibiting excellent absorption capacities were used for removal of CV including esparto grass removing 43.47 mg/g, 16 ginger [227.27 mg/g], 17 shrimp shell (39.56 mg/g 18 and mango stone 352.79 mg/g. 19 The aim of the present study was to evaluate the application potential of fish scale collagen as a low-cost biomass–based adsorbent for removal of pollutants in harmless forms.
Collagen is widely used as a biomaterial for many clinical and industrial applications. 20 Biological characteristics of collagen such as biocompatibility, biodegradability and weak antigenicity, have made this polymer an ideal candidate for various applications. While maturated collagen was considered as a good biopolymer adsorbant material that is highly crosslinked and insoluble in water, it can be mixed with metals, polymer and/or biopolymer to improve efficiency and recovery. 21
Moreover, other materials are used for colored effluent decolorization such as metal nanoparticles. Physical and chemical modification methods are used to control the size and shape of these nanoparticles. These nanoparticles have been coated with polymeric compounds of low molecular weight to prevent aggregation which reduces the activity of these particles.22,23
Magnetic iron oxide nanoparticles (MIONs) have been good candidates for mixing with polymers in various applications due to their extensive characteristics of chemical stability, biodegradability, biocompatibility, small size, ease of action, low toxicity, low cost and high affinity to many chemical substances.24,25 Owing to the above properties, magnetite particles are widely used in various fields such as magnetic resonance imaging,26,27 separation and adsorption28,29 and as implants for controlled drug delivery systems.30,31
Application of the magnetic particles depends on methods of preparation such as precipitation from solution, microemulsions, coprecipitation, sol-gel method, thermal decomposition of organic precursors, sonochemical decomposition method and hydrothermal method.32-34 The most commonly used method is coprecipitation which is the simplest and most efficient chemical pathway for synthesizing magnetic particles.34,35
Combination of polymers of biological origin with various types of materials in a single composite is desirable to optimize its properties for the required application. 36 Nanobiocomposites of nanoparticles and biologically degradable polymers have been promoted in various biomedical and environmental applications.37-39 Interestingly, combination of metal nanoparticles such as magnetic Fe3O4 nanoparticles with collagen polymer could exploit new properties for collagen biopolymer magnetic nanoparticles interaction. 40
In this study, collagen fiber extracted from fish waste was optimized for dye removal. To stabilize collagen waste fibers, magnetic iron oxide (Fe3O4) nanoparticles were utilized selectively to facilitate dye removal from wastewater by magnetic tracking. Furthermore, this composite can be reused many times due to its magnetic properties.
Materials and methods
Crystal violet was gathered from local textile factory. It was defined as N, N, N’, N’, N”, N”- hexamethylpararosaniline, and its mono-, di-, and tri-N-demethylation products are referred to as N, N, N’, N’, N”- penta-, N, N, N’, N”-tetra-, and N, N’, N”-trimethylpararosaniline, respectively. All other chemicals used were of highly purified analytical grade (≥ 97%).
Preparation of fish scale collagen (soft and coarse collagen)
Collagen was extracted from fish scales collected from fresh fish and washed with running water. The scales were washed with distilled water and stored at −25°C until used. The lyophilized scales (5.0 g) were treated with 0.1 N NaOH (Merck Chemicals; Darmstadt, Germany) to get rid of non-collagenous proteins and pigments for 3 days by changing the solution once a day, then washed with distilled water, dried, and stored at 80°C until used. The matter was extracted with 0.5 M acetic acid (Merck Chemicals; Darmstadt, Germany) for 3 days, and the extract was centrifuged at 50 000 g for 1 h. For soft collagen, the supernatants were pooled and salted out by adding NaCl (Merck Chemicals; Darmstadt, Germany) to a final concentration of 0.9 M. The pellet was then washed and reprecipitated three times with distilled water to remove any salt.
The pellets were suspended in 0.5 M acetic acid and then lyophilized. Collagen was then precipitated and lyophilized. Thereafter, the lyophilized samples were grounded to powder using a personal mill and sieved with a mesh (0.15 mm) sieve. 41 For coarse collagen preparation, the waste of fish scale after 3 times preparation of soft collagen was lyophilized to be used as coarse collagen. Collagen fibres were examined under light microscope.
Decolorization assay via UV-Vis spectroscopy
The dye decolorization capacity of collagen was studied. All the decolorization experiments were done in triplicate. Wavelength for maximum absorbance of crystal violet was determined through spectrophotometric analysis and was found to be at 590 nm. Calibration curve was prepared by plotting known concentrations of crystal violet dye versus corresponding absorbance values. Decolorizing activity was expressed in terms of decolorization percentage and was determined by monitoring the decrease in absorbance at 590 nm by UV/VIS spectrophotometer (Jenway UV/Visible-2605 spectrophotometer, England). Distilled water was used as blank, samples containing crystal violet were used as reference (control) and each experiment’s soft and coarse collagen was used as sample, then decolorization was calculated using the following equation. 42
where C= control, S= sample.
In order to calculate the dye adsorption capacity, the adsorption amounts per gram of materials were evaluated by applying the following equation:
where Q is the adsorption capacity (mg/g), V is the volume of the solution (mL), Co is the initial concentration, Ce is the final concentration and M is the mass of material or the adsorbent (g).
2.3. Decolorization of crystal violet by two kinds of collagen at different contact times Soft and coarse collagen were added at different concentrations (5, 10, 15, 20, 25, 30 g/L) to 200 ppm of crystal violet prepared in deionized water, and decolorization experiments were performed in an open batch system at room temperature, 28 ± 2°C. Dye solutions were stirred using a shaker (Rossari Labtech, Mumbai) at a speed of 150 rpm to keep collagen suspended. Samples were withdrawn at fixed time intervals (30, 60, 90, 120 min) and were subsequently spinned for 5 min at 3000 rpm. Clear solution was then pipetted out, and rate of decolorization was measured by measuring the absorbance at maximum wavelength (590 nm) using UV-VIS spectrophotometer (UV-VIS 8500, TECHCOMP Limited, Hong Kong). Dye decolorization percentage was calculated from dye solution calibration curve.
Effect of static or shaking conditions
This experiment was carried out using 20 mL (200 ppm) crystal violet mixed with 25 g/L of soft collagen monitored at different agitations. Tubes (50 mL) were then incubated under different shaking (50, 150, 250 rpm) and static conditions. After 60 min incubation at room temperature, aliquot of 5 mL was withdrawn, centrifuged at 3000 rpm for 15 min to separate collagen mass. The supernatant was then measured spectrophotometrically to measure dye decolorization.
Effect of pH on decolorization
Soft collagen (25 g/L) in 20 mL solution was incubated with 200 ppm of crystal violet at different pH (5, 7, 9 and 11 adjusted with 1 M HCl or 1 M NaOH) under static conditions at room temperature, 28 ± 2°C. The effect of pH on dye decolorization was then checked spectrophotometrically after 60 min incubation.
Effect of initial dye concentration
Various concentrations of dye were tested (100, 200, 300, 500, 600, 800 and 1000 ppm) in a 20 mL solution. The various concentrations were added to 25 g/L soft collagen. The tubes (50 mL) were then incubated under static conditions at room temperature for 60 min.
Preparation of magnetic iron oxide nanoparticles
Magnetic Iron Oxide Nanoparticles (Fe3O4) were synthesized by coprecipitation method, 2:1 molar ratio solution of Fe+3 and Fe+2 ions with very basic solutions. 43 In this method, a solution of FeCl3 (Merck Chemicals; Darmstadt, Germany) and Fe2SO4 (Merck Chemicals; Darmstadt, Germany) mixture (molar ratio 2:1) was prepared, followed by dropping aqueous NH4OH 25% (Merck Chemicals; Darmstadt, Germany) solution with vigorous stirring for 30 min until complete precipitation of the black magnetite was achieved at pH 9–11, under N2 protecting. The generated Fe3O4 NPs were filtered and washed three times with distilled water by magnetic separation. 44
Magnetic collagen nanocomposites formation
Collagen/Fe3O4 composite was prepared by using collagen as soft powder and Fe3O4 solution (0.05 g/mL) in the ratio of 100: 50 at room temperature to be mixed for 2 h with water (2000% based on collagen weight) as the matrix to obtain magnetic nanocomposites. In a typical experiment, 1 g of hide powder was mixed with 20 mL water and further treated with 0.25 g MIONs (5 mL of 0.05 g/mL Fe3O4 solution) for 2 h at room temperature with magnetic stirring. The reaction mixture was washed immediately with distilled water and magnetic collagen nanocomposite was separated from solution by magnetic separation; thereafter, it was dried and stored. Collagen-magnetic nanocomposites were examined for dye removal and were track under magnetic field. 44
Testing magnetic collagen nanocomposite efficiency for repetitive dye removal
The efficiency of immobilized magnetic collagen nanocomposite for many cycles against CV dye removal was tested. CV dye (20 mL) was added every 60 min for three cycles at pH 7, static condition, room temperature and 200 ppm concentration. The magnetic collagen nanocomposite material (25 g/L) was separated from the treatment water using external magnetic field, washed and reused for many cycles and the decolorization of dye was determined spectrophotometrically at 590 nm after each cycle.
Characterization of the magnetic collagen nanocomposite
The properties of the synthesized collagen nanocomposite were studied using different techniques such as FTIR, TEM, and light microscope.
Fourier transform infrared spectroscopy (FTIR) spectrum analysis
FTIR spectroscopy of freeze-dried sample of collagen nanocomposite relied on a Bio-Rad FTIR-40, USA. Sample (10 mg) was mixed with 100 mg of dried potassium bromide KBr (Merck Chemicals; Darmstadt, Germany) and compressed to prepare as a salt disc (10 mm in diameter) for reading the spectrum further. Spectra were collected between wave numbers of 4000 and 500 cm−1.
Transmission electron microscopy (TEM)
The morphology and particle size of the synthesized collagen nanocomposite was investigated using transmission electron microscope. In order to prepare TEM sample, the NPs solution was sonicated for 5 min for better dispersion. A drop of the sample was placed with a carbon-coated copper TEM grid (200–300 mesh) and kept at room temperature to dry and then, imaging.
Cytotoxicity test with peripheral blood mononuclear cells (PBMC) cells
PBMC were isolated from peripheral blood using Ficoll-Paque, as previously reported. 45 Anticoagulated blood was centrifuged at 800 × g for 30 min and the top layer containing plasma was removed. The remaining blood was diluted with an equal volume of phosphate-buffered saline, pH 7.4 (PBS), containing 0.05 M ethylenediaminetetraacetic acid (EDTA; Invitrogen). A 12.5 mL of diluted blood was layered above 25 mL of the Ficoll-Paque PLUS (GE Healthcare). Gradients were centrifuged at 400 × g for 30 min at room temperature in a swinging-bucket rotor without the brake applied. The PBMC interface was carefully removed by pipetting and washed with PBS-EDTA by centrifugation at 250 × g for 10 min. Cells were then resuspended in RPMI medium GIBCO BRL (Grand Island, NY) (COMPONENTS g/L: Calcium Nitrate H2O, 0.1; Magnesium Sulfate (anhydrous), 0.04884; Potassium Chloride, 0.4; Sodium Chloride, 6.0; Sodium Phosphate Dibasic (anhydrous), 0.8; L-Arginine, 0.2; L-Asparagine (anhydrous), 0.05; L-Aspartic Acid, 0.02; L-Cystine, 0.0652; L-Glutamic Acid, 0.02; Glycine, 0.01; L-Histidine, 0.015; Hydroxy-L-Proline, 0.02;L-Isoleucine, 0.05; L-Phenylalanine, 0.015; L-Proline, 0.02; L-Serine, 0.03; L-Threonine, 0.02; L-Tryptophan, 0.005; L-Tyrosine, 0.02883; L-Valine, 0.02; D-Biotin, 0.0002; Choline Chloride, 0.003; Folic Acid, 0.001; myo-Inositol, 0.035; Niacinamide, 0.001; p-Amino Benzoic Acid, 0.001; D-Pantothenic Acid (hemicalcium), 0.00025; Pyridoxine•HCl, 0.001; Riboflavin, 0.0002; Thiamine•HCl, 0.001; Vitamin B-12, 0.000005; D-Glucose, 2.0; Glutathione (reduced), 0.001; Phenol Red (sodium), 0.0053) to reach 1.34 × 106 cells/mL. Isolated PBMC were grown as a monolayer at 37°C in 5% CO2 atmosphere in RPMI medium GIBCO BRL (Grand Island, NY) supplemented by 1% glutamine, 10% fetal bovine serum GIBCO BRL (Grand Island, NY) and 1% penicillin/streptomycin solution GIBCO BRL (Grand Island, NY). The test was carried out in 6-well plastic plates seeded with 2 mL cell suspension (5 × 104 cells/mL). After growing cells for 24 h, the culture is usually about 60% confluent. The cells were exposed to the sterile filtered dye (100 ppm) treated with collagen and collagen nanoparticles composite for 24 h. Non-exposed cells with medium were used as negative control and all results were compared to them. Cells treated with dye and not treated with adsorbent served as the reference cells. The viability of the cells was observed by light microscopy to obtain preliminary information.
Results
Soft and coarse collagen flakes were prepared from fish scale collagen, solidified product of soft collagen fibrils can be revealed under light microscopy (Figure 1).

Light microscopy image (10×) of the extracted fish scale collagen fibers.
Effect of collagen concentration as a substrate for dye removal
To investigate the correlation of collagen amount with crystal violet removal, various amounts (5–30 g/L) of collagen at pH 7 and room temperature were added into 200 ppm solutions of crystal violet. The results are shown in Figure 2, from which it can be established that crystal violet removal percentage increased upon increasing soft and coarse collagen amount through different contact times.
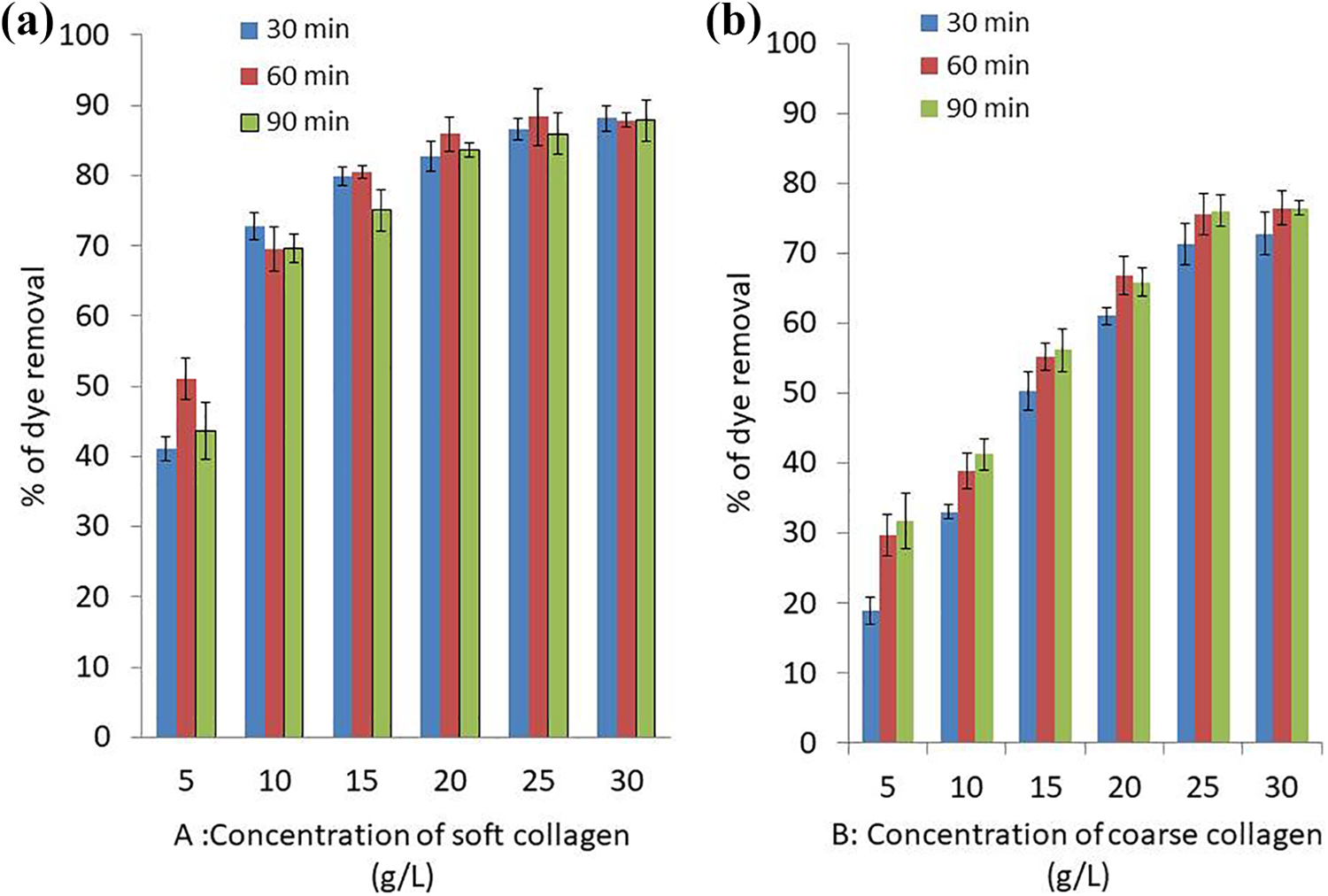
Effect of different concentrations and contact time of soft collagen (a) and coarse collagen (b) on removal of crystal violet dye at pH 7, room temperature and 200 ppm of dye concentration.
As the amount of soft and coarse collagen increased from 5 g/L to 25 g/L, the efficiency of removal of crystal violet increased significantly and p-value was <0.00001 for both collagen types using chi-square test. No significant increase of crystal violet removal could be detected as the amount of soft and coarse collagen increased from 25 g /L to 30 g/L, where the p-value was 0.674108 and 0.875527, respectively. Among the studied collagen concentration values, the optimum removal efficiency was standardized at 25 g/L collagen concentration.
Effect of contact time on dye removal
Collagen was set to contact CV dye at different time intervals of 30, 60 and 90 min. The highest removal efficiency of CV was obtained using 25 g/L soft and coarse collagen concentration and contact time of 60 min as shown in Figure 2 showing 88.3 and 75.5% dye removal with 7.08 and 6.04 mg/g adsorption capacity for soft and coarse collagen, respectively. The equilibrium was reached after 60 min.
Effect of pH on dye removal by collagen
Solution’s pH affect speed of the reactions that occur on the surface of collagen particles by influencing the properties of the surface charge. The influence of pH on removal of 200 ppm crystal violet dye by 25 g/L of collagen was investigated at pHs 5 to 11, while keeping the other parameters constant.
Removal of CV increases up to 78.99% at pH 5 and slightly decreased to 77.67 at pH 7 and dropped to 72.3% at pH 9 with adsorption capacity of 6.312, 6.22 and 5.78 mg/g, respectively while collagen precipitated at pH 11 (Figure 3). Among the studied pH values, the best removal efficiency was obtained under acidic conditions with no significant difference between dye removal at pH 5 and pH 9 p-value was 0.327187.
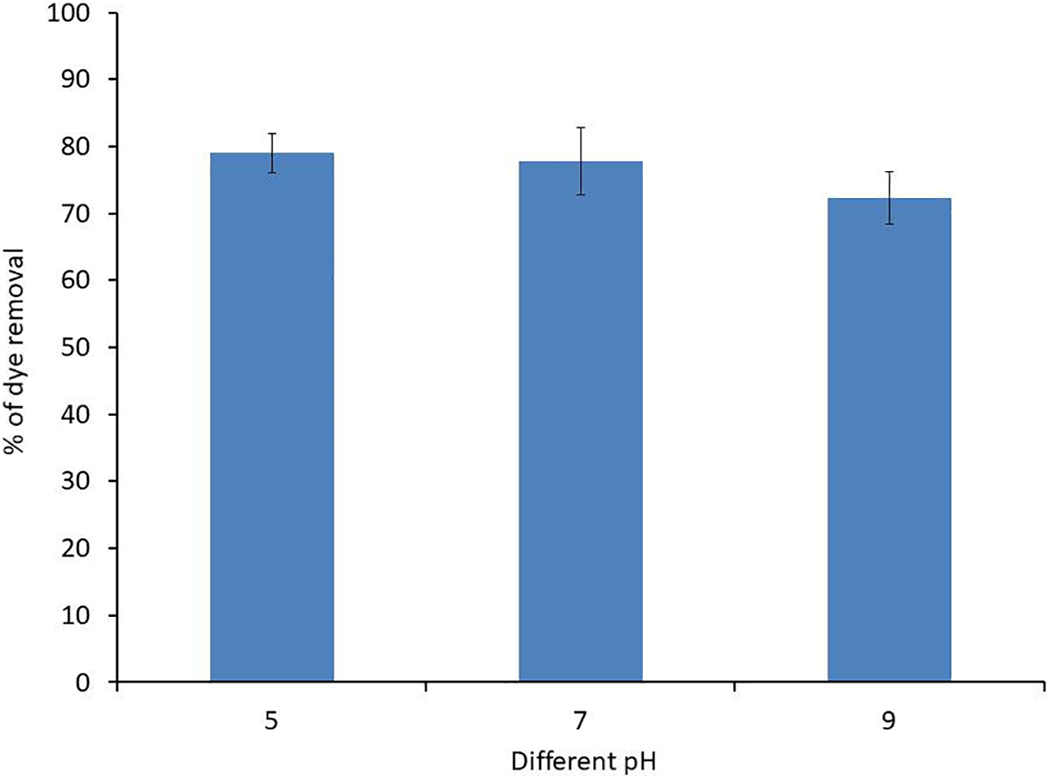
Effect of different collagen acidity (pH) on removal of 200 ppm crystal violet dye at room temperature for 60 min.
Effect of shaking speed (rpm) on dye removal
The effect of shaking speed on removal of crystal violet (200 ppm) dye by collagen (25 g/L) at pH 7 was investigated by varying shaking speed from 0 to 250 rpm. The results in Figure 4 indicated that the removal percentage of dye decreased insignificantly from 86.09% to 84.52% with p-value of 0.692061 as the shaking speed increased from 0 to 50 rpm, whereas the percentage of dye removal decreased insignificantly to 83.88% at 150 rpm with p-value of 0.557772 when shaking speed increased from 50 to 150 rpm. Dye removal percentage increased insignificantly (p-value 0.83607) to 87.62% with adsorption capacity of 7.01 mg/g at 250 rpm.

Effect of different shaking speed (rpm) on removal of 200 ppm crystal violet dye at room temperature for 60 min by collagen (25 g/L) at pH 7.
Effect of different initial dye concentrations
Different initial dye concentrations in the range of 100 to 2500 ppm were investigated. As shown in Figure 5, at concentration range of 100 to 1000 ppm with 100 intervals, the removal percentage of CV dye was almost stable around 88.6% to 92.1% showing non-significant change in dye removal efficiency with p-value of 0.469392 using chi-square test.

Effect of different dye concentrations on its removal by collagen (25 g/L) at pH 7 for 60 min, the right image showing light microscopic image of collagen with different dye concentrations.
On the other hand, at concentration range of 1000 to 2500 ppm with 500 intervals, the dye solution was oversaturated with CV and the dye presentation occurred.
Magnetic collagen nanocomposite formation
Nanoscale magnetic Fe3O4 was synthesized successfully, then it was mixed with collagen. Collagen nanoparticles with super-paramagnetic behavior was attracted to a magnet.
Desorption and recycling efficiency
Recycling efficiency of magnetic collagen nanocomposite 25 g/L was investigated for the removal of 200 ppm CV at static condition, pH 7 after 60 min. Three cycles of dye removal with magnetic collagen nanocomposite were done and the adsorption efficiency of collagen was reduced from 83.86 to 47.31 and 39.49%, respectively.
Magnetic collagen nanocomposite characterization using TEM, FTIR analysis
Micrograph images (TEM) of magnetic collagen fibers exposed collagen surface as rough and corrugated (Figure 6). This composite was revealed as a heterogeneous material consisting of magnetic particles of spherical shapes having considerable layers with grooves of fibers of varying sizes.

TEM image of magnetic collagen nanoparticles shows agglomerates magnetic nanoparticles on a collagen polymer matrix.
The adsorption of CV dye and nanoparticles was analyzed using data obtained by FTIR analyzing the change of collagen molecular structure before and after dye adsorption (Figure 7).
Collagen was characterized by FTIR spectrum after dye adsorption, with amide I absorption bands mainly at ∼1650 cm−1, amide II at ∼1560 cm−1, and amide III represented by three weaker bands with vibration modes centered at ∼1245 cm−1 (Figure 7). Additional peaks were associated with dye and nanoparticles adsorption to collagen as shown in Table 1 and Figure 7, respectively.

FTIR spectra analyzing the change of the collagen molecular structure showing the alteration in spectra after CV dye adsorption to collagen, collagen spectra after attachment of the magnetic nanoparticles and spectra of magnetic collagen nanoparticles adsorbing the CV dye.
IR spectra of collagen adsorbing CV, magnetic collagen nanocomposite, magnetic collagen nanocomposite adsorbing CV and control collagen.
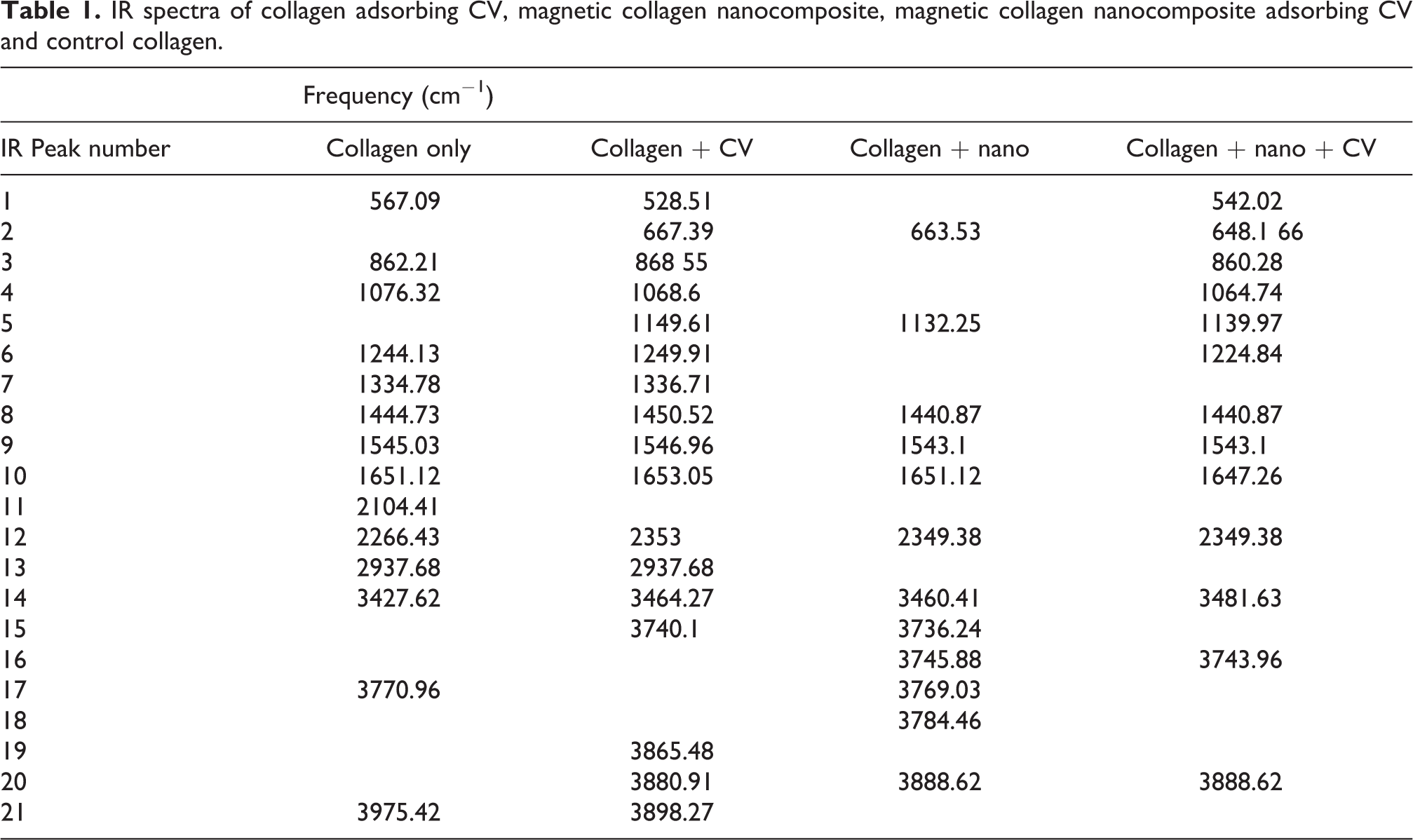
Peaks in the control dye spectrum represented the deformation of C-H at 675 cm−1, CH stretching at 2949.16 cm−1, 2922.16 cm−1 and 2864.29 cm−1. The stretching vibrations at 1112.93 cm−1, 1049.28 cm−1 and 1031.92 cm−1 showed C-O stretching at 1643.35 cm−1, the stretching vibrations also showed N-H bending.
Ameliorating cytotoxicity of crystal violet using magnetic nanocomposite
PBMC saturated with 100 ppm CV dye served as the control reference cells, treatment of cells with collagen only and magnetic collagen nanocomposite reduced cytotoxicity significantly with p-value of 0.00001 for both treatments as shown in Figure 8. Cell cytotoxicity was considerably high in the range of 75% for reference cells which was reduced to 40% for cells treated with dye solution mixed with collagen and in case of magnetic collagen, nanocomposite cytotoxicity was reduced to 15%.

Light microscope image of PBMC subjected to 100 ppm CV dye (a) PBMC subjected to 100 ppm CV dye treated with collagen (b) and PBMC subjected to 100 ppm CV dye treated with magnetic collagen nanocomposite (c).
Discussion
Extraction of collagen from fish scale was achieved through primary decalcification step which exposes collagen fibrils to facilitate dissolution through direct solute–solvent interaction. Collagen fibres observed through an optical microscope would later serve as a polymer for environmental applications.
The adsorbent amount was studied as one of the most important factors that influence adsorbent capacity for a given initial amount of collagen. 46 The increase in removal percentage for crystal violet at low adsorbent amount of collagen (5–25 g/L) might be due to better dispersal of collagen in an aqueous solution. 47 The adsorbent active sites (charged amino acid of collagen) on the surface are entirely vacant, which could accelerate the contact of dyes molecules to adsorbent active sites. On the other hand, as the adsorbent concentrations of collagen increases above 25 g/L, the contact of adsorbent active sites decreases and a larger fraction of the active sites become occupied, with a consequent decrease in the adsorption capacity. 48 The amount of water consumed in textile industries was considered in this experiment as the traditional textile finishing industry consumes about 100 L of water in the processing of a kilogram of textile material. Consequently, the potential of water re-use was an objective when applying collagen for removal of high concentration of crystal violet.
An amount of 25 g/L of collagen was chosen as the optimal dosage for further study. By analyzing the effect of effective collagen dosage, it illustrates the ability of crystal violet dye to be adsorbed with the minimum amount of collagen, to establish the ability of dye removal from economical point of view.
The alteration in adsorption rate with increasing contact time is due to the vacancy of most of the adsorbent sites at the start where the solute is highly concentrated. Thereafter, adsorption rate was decreased at 60 min due to decrease in the number of unoccupied sites of collagen as an adsorbent and CV dye concentrations. The declined adsorption rate at the end of the experiment indicates that CV has possibly molded a monolayer on the adsorbent surface.49,50 This reduces the number of unoccupied active sites for further uptake after reaching equilibrium. 51
Given that the magnitude of electrostatic charges of collagen and dye molecules is controlled by the solution pH, the rate of dye removal varied with acidity of the medium used. Acidic condition proved suitable for the maximum removal efficiency. The affinity between collagen as an adsorbent and crystal violet dye could be due to acid-base properties, as expected by the presence of ionizable functional groups on the adsorbent collagen and the target adsorbates CV dye. 52 This would be explained as the attraction between positive and negative charges between collagen amino acids and CV dye. These charges vary according to the solution acidity in which CV and collagen are in contact.
The isoelectric point of collagen is pH 6.6–6.8 where the amino acids are positively and negatively charged making the net charge of the molecule nearly zero. At a basic pH (7–11), collagen amino acids are negatively charged, therefore collagen molecule as a whole is negatively charged. 53 On the other hand, crystal violet is a positively charged dye which is known to stain down to 16 ng of protein through interaction with the carboxyl group of the negative charged amino acids. 54
A little decrease in the removal percentage of dye using collagen was recorded with increasing shaking speed that might be due to desorption of dye’s molecules from the surface of collagen at higher rpm. 52
Interestingly, the percentage removal of crystal violet at the highest shaking speed (200 rpm) increased a little in percentage which could be due to collision of the adsorbent with the dye. The removal percentage of the dye wasn’t affected so much by shaking as the highest removal percentage of CV was 86.09 at static condition (0 rpm), whereas the removal percentage of CV was 87.62 at 250 rpm. For the sake of energy saving, static condition was selected as the best condition to be used for further experiments.
The removal percentage of CV dye was almost stable at different dye concentrations showing non-significant change in dye removal efficiency. This can be explained by CV reaching saturation before the addition of collagen as an adsorbent and the unoccupied active sites needed for the adsorption of dye were reduced. 55 The non-soluble particles compensate for CV dye saturation after adsorption by the collagen, thus CV dye removal was almost stable at that range. On the other hand, at concentrations of 1000 to 2500 ppm with 500 intervals, dye solution was oversaturated with CV and dye presentation occurred.
Nanoscale magnetic Fe3O4 was synthesized and then mixed with collagen as a surface polymer, helping to obtain collagen nanoparticles with super-paramagnetic behavior. In the present study, nanoparticles composite synthesis required no protecting gas and was easily implemented because it is simple and environmentally friendly. This magnetic characteristic was essential for collagen dye removal recycling which was tested and approved for three cycles of dye removal. The spherical shapes of magnetic particles with considerable layers and grooves of fibers of varying size as demonstrated by TEM analysis may provide a fair chance for the dye to be attached to the composite and hence adsorbed. Collagen fibers composition provides a proper active site for the adsorption of other molecules. FTIR analysis was used to identify some characteristic functional groups of collagen before and after attachment of the magnetic nanoparticles; shift in the absorption peak suggested the interactions of dye and nanoparticles molecules with the functional groups of biosorbent.51,56
Biological effectiveness of collagen and magnetic collagen nanocomposite were evaluated using cytotoxicity test since these tests are frequently used for toxicity screening of pollutants (air, soil and water). Previous reports by Rane et al. 57 suggested that after dye removal by several methods, toxicity may stay as a matter of major concern as residues/degradation intermediate compounds and end product could be more toxic than the parent compound. The present study showed that collagen and magnetic collagen nanocomposite treatment has the ability to detoxify dyes along with decolorization which reduced cell toxicity by 80%. In line with our results, the toxicity of industrial effluents treated by ligninolytic enzymes toxicity was reduced by 98%. 58 Industrial effluents treated with collagen would be enriched with protein which is used frequently as food supplements; on the other hand, magnetic collagen nanocomposite can be magnetically eliminated after dye removal.
Conclusions
In this present work, efficient removal of CV dye was established using fish scale collagen and magnetic collagen nanocomposite for the first time. Additionally, the material could be reused for up to three cycles. This would be of particular importance economically as collagen was extracted from low-cost waste by simple efficient method. We have developed newly synthesized collagen magnetic nanocomposites with the potential for scaling up to meet the larger quantity requirements for environmental and medical applications. Excellent dispersion of the magnetic nanoparticles in the polymers has been accomplished. The newly synthesized nanocomposite was employed as a highly efficient adsorbent for dyes from industrially polluted water reducing toxicity by 80% in one cycle. Few studies have been performed on adsorption of dyes by collagen, here we provided a simple and environmentally friendly method for dye removal. The present study is promising in terms of applying nanoscale dye removal devices that have wide environmental applications.
Footnotes
Data availability statement
The authors confirm that the data supporting the findings of this study are available within the article [and/or] its supplementary materials.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Patents
A patent with number 688956 was applied at the Egyptian patent office.


