Abstract
Batch experiments were conducted to investigate the capacity and mechanisms for adsorbing Cd2+ from aqueous solutions by the composite material. The composite material was manufactured with
Introduction
The development of human life and industrial activities, many heavy metals ions are discharged into the tributary rivers. Industrial wastewater is the main cause of heavy metals pollution of shallow lakes in China, and cadmium is the main pollution element. Growing attention is being given to health hazards caused by the presence of heavy metals in aqueous environments (Deng et al., 2016). Previous studies showed metal-contaminated river can lead to the accumulation of metal ions in fish muscle and vegetables, and then affect human health directly (Kwok et al., 2014; Monferran et al., 2016; Sharma et al., 2016). Cadmium ions selectively accumulate in pancreas, bones, liver, and lungs, which affect cell proliferation, differentiation, apoptosis, and cause a series of diseases, including bone-thinning disease, osteoporosis, and respiratory tract problems (Flora et al., 2008). Many methods, including precipitation, membrane filtration, and flocculation are conventional techniques to remove Cd2+ ions from wastewater, but more conventional technologies are ineffective and unfavorable as they cause sludge disposal problems, are expensive, and do not lead to complete removal (Bai et al., 2008; Halttunen et al., 2007). Utilization of microbial adsorbents for the treatment of wastewater contaminated with heavy metals has become an alternative method to conventional treatments. In the past two decades, researchers found some microorganisms have the capacity to adsorb metal ions under the metal-contaminated environment (Abbas et al., 2014; Neethu et al., 2015; Roane and Pepper, 1999). Consequently, researchers utilized microorganisms as a new bioremediation technology to remove metal ions (Abbas et al., 2014).
Materials and methods
Manufacture composite material
The modified method was first to pretreat PAN-CF in acetone solution for 2 h and then heat material at 200℃ for 1 h. After pretreatment, we used 60% nitric acid, 5% potassium permanganate, and 15% hydrogen peroxide to modify material for 1 h, respectively. The modified PAN-CF was washed with deionized water until the washed water pH at 7. The modified PAN-CF was placed in the culture medium which contains 
The characteristics of experimental materials
Characteristics of unmodified PAN-CF and modified PAN-CF were analyzed with BET, FE-SEM, FTIR, OCA, and ADM. The OCA system was to measure the contact angle of PAN-CF and then calculate the surface free energy by Owens methods (Owens and Wendt, 1969). The surface free energy of PAN-CF was calculated using equations (2) to (4)
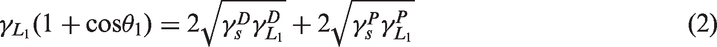


Composite material was observed under FE-SEM after freeze-dried overnight. The energy-dispersive X-ray spectrometer (EDX, Oxford, UK) was used to analyze chemical element characteristic after adsorption experiments. In Boehm’s titration experiment, 1.000 g carbon fiber material was added into NaOH, Na2CO3, and NaHCO3 solution (100 ml 0.05 molċl−1) to calculate the number of functional groups on the material surface, respectively. Zeta potential measurements part was obtained by 0.1 g composite material in 0.01 molċl−1 NaNO3 solution at pH values ranging from 2.0 to 7.0 (adjustment with 1.0 M HNO3 or NaOH). Zeta potential values were determined for each sample using an Electrokinetic Analyzer (JS94H; ZhongChen, China).
Analysis of affecting factors
The effects of various adsorbent dosages (0.25, 0.5, 1.0, 1.5, 2.0, and 2.5 g), pHs (1 2, 3, 4, 5, 6, and 7), temperature (10, 15, 20, 25, 30, 35, and 40℃), ionic strength (0.01, 0.1, and 1.0 mol l−1 NaCl), and time on the removal rate in Cd2+ solution by composite materials were studied. The composite material and the control group (modified PAN-CF) were added into Cd2+ solution (50 mg l−1, 100ml) to test the removal rate. The pH of the precipitate appearance of Cd2+ ions was 7.41, so the range of detected pHs was set from 1 to 7. Cd2+ ions concentrations were analyzed by ICP Optima 8000 inductively coupled plasma (PerkinElmer, USA) at 228.8 nm and then Cd2+ removal rate was calculated by using equation (5)

Adsorption kinetic and isotherm model simulation
The composite materials were added into Cd2+ ions solution (100 mg l−1, 50 ml) at 20℃ for 24 h. We design 24 h as the final time of adsorption kinetic experiment, because of the adsorption equilibrium time point was at 20 h in the above adsorption experiment. Adsorption kinetic models were analyzed by using Lagergren pseudo-first-order (equation (6)) and pseudo-second-order models (equation (7))


Isotherm sorption model simulations were analyzed by using Langmuir model and Freundlich model. Equations (8) and (9) are mathematical models corresponding to both sorption isotherms, respectively


Statistical analysis
All experimental data are expressed as mean ± SD and used T-test to analyze significant differences by IBM SPSS Statistics 22.0 (IBM Software Inc, New York, USA). The value of
Results
The characteristics of experimental materials
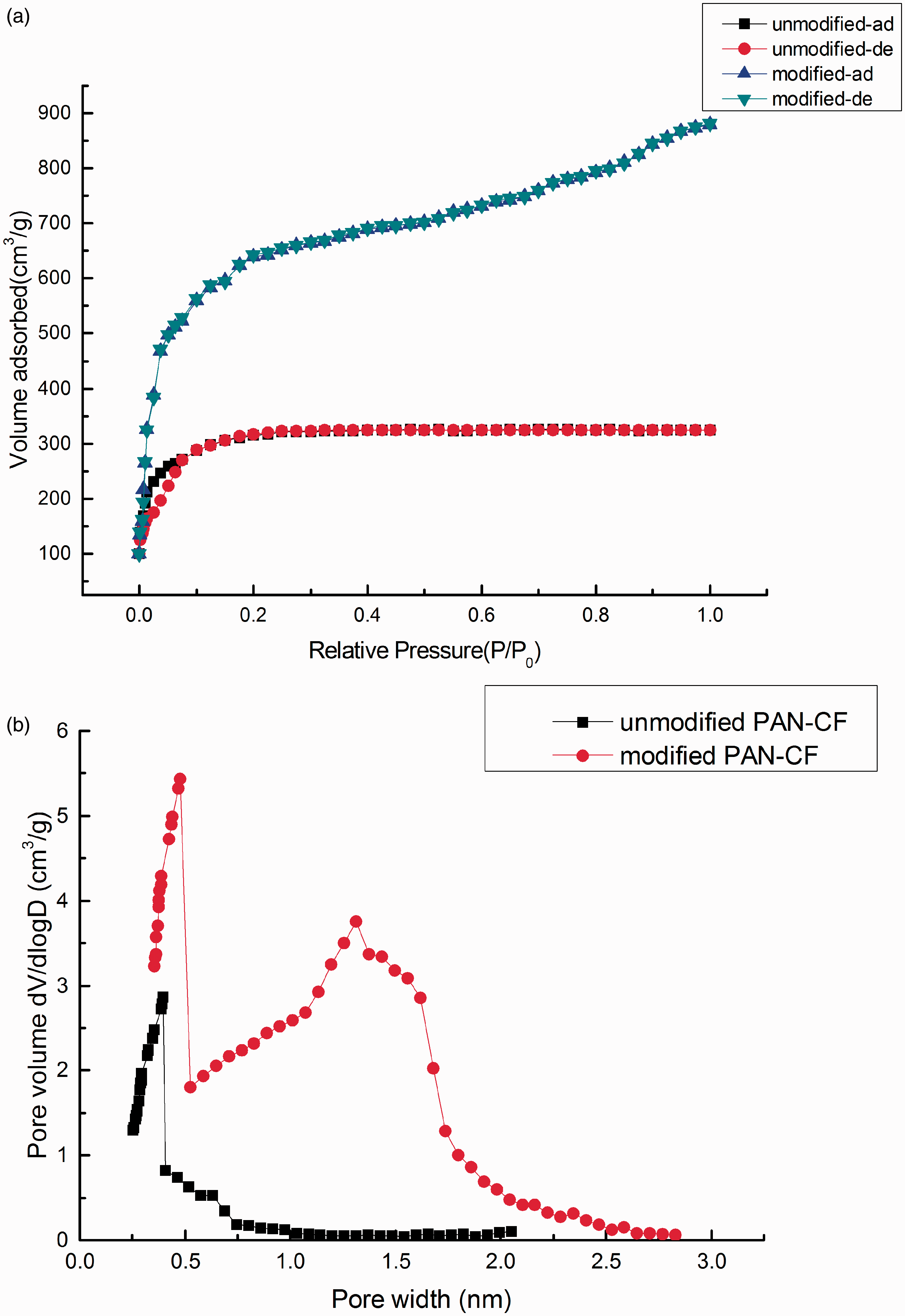
FE-SEM results reveal the surface of the modified PAN-CF has been oxidized obvious and presented delamination (Figure 1(a) and (b)). The bacteria could be immobilized on the surface of modified carbon fibers, even after deionized water washing (Figure 1(c)). FTIR result shows modified PAN-CF increased one secondary hydroxyl group at about 1082.14 cm−1 and the peak intensity at 1385.21 cm−1 increased obviously (Figure 2). Figure 3 shows the color of PAN-CF became brilliant black into gray black after chemical modification. In addition, the total surface free energy The surface of different materials was observed by FE-SEM. (a) Unmodified PAN-CF, (b) modified PAN-CF, and (c) microbial composite carbon fiber materials. FE-SEM: field emission scanning electron microscope; PAN-CF: polyacrylonitrile-based carbon fiber. PAN-CF was analyzed by FTIR system. FTIR: Fourier transform infrared; PAN-CF: polyacrylonitrile-based carbon fiber. Surface morphology of PAN-CF was analyzed by ADM system. (a) Unmodified PAN-CF and (b) modified PAN-CF. ADM: automated digital microscope; PAN-CF: polyacrylonitrile-based carbon fiber. Surface energy analysis of modified PAN-CF and unmodified PAN-CF. PAN-CF: polyacrylonitrile-based carbon fiber. Surface areas porosity and biofilm dry weight of PAN-CF samples. PAN-CF: polyacrylonitrile-based carbon fiber. BET experimental data of PAN-CF samples. (a) Nitrogen adsorption–desorption isotherms and (b) pore size distribution. BET: Brunauer-Emmett-Teller; PAN-CF: polyacrylonitrile-based carbon fiber. Surface acidity sites of PAN-CF by Boehm’ method. PAN-CF: polyacrylonitrile-based carbon fiber. Zeta potentials of microbial composite carbon fiber material. Simulation of sorption kinetic equations and corresponding parameters. Simulation of sorption isotherms equations and corresponding parameters.








Analysis of influencing factors
As can be seen in Figure 6(a), the adsorbent dosage affects removal capacity. We can see that the removal efficiency increased as the adsorbent dosage increased. However, after 0.5 g, the increasing was more stable. Thus, the dosage of 0.5 g was chosen for subsequent experiments. As can be seen in Figure 6(b), the maximum Cd2+ ions removal rate was 71.38% at pH 7. However, there was no significant correlation between the removal rate and temperature changes (Figure 6(c)). Ionic strength test results show the adsorption capacity varies no significant with the increase of ionic strength (Figure 6(d)). The consequence of cadmium removal dynamic experiment is shown in Figure 7. The equilibrium removal rate of Cd2+ by composite material and the control group was 71.56 and 24.18%, respectively. The sorption of Cd2+ ions by composite material was a rapid process and contained three stages. During the initial stage, the adsorption rate increased rapidly; after 60 min, the Cd2+ ions removal rate slowed down and then stabilized as it reached equilibrium after 1200 min. The equilibrium adsorption amount of composite material was 7.126 mgċg−1. EDX results showed that Cd2+ ions were adsorbed on the surface of composite material (Figure 8).
pH, temperature, dosages, and ionic strength effect of removal rate patterns. (a) Removal rate under different adsorbent dosages, (b) removal rate under different pH values, (c) removal rate under different temperature, and (d) the effect of ionic strength on Cd2+ adsorption on composite material. The equilibrium adsorption curve of both material ( EDX image of composite material after adsorbing Cd2+ ions. EDX: energy-dispersive X-ray spectrometer.


Adsorption kinetics and isotherms model
The linear fitting results show the adsorption process of composite material was in accordance with pseudo-second-order model ( Linear fitting curve plots of isotherms and kinetics model for Cd2+ ions removal by composite material. (a) Lagergren pseudo-first-order linear fitting curve plot, (b) pseudo-second-order linear fitting curve plot, (c) Langmuir isotherm linear fitting curve plot, and (d) Freundlich isotherm linear fitting curve plot.
Discussions
The modified PAN-CF was added one secondary hydroxyl group in 1082 cm−1 and the peak intensity increased in 1385 cm−1 which indicates that the modified process can increase the numbers of alkyl, alkene, and hydroxyl groups. Because the chemical oxidant destroyed the PAN-CF surface, so some delamination appeared on the fiber surface. At the same time, a large number of micropores arose in the delamination, which increased the specific surface area, pore width, and pore volume. In addition, the rise of surface free energy and specific surface area can enhance PAN-CF hydrophilic property to promote bacteria fixed on the PAN-CF surface efficiently. Hence, bacteria hanging amount of modified PAN-CF was increased by 77.34% compared with unmodified PAN-CF. These results indicated that the chemical modification can promote the carbon fiber material more biocompatible and increase the bacteria hanging amount. In the affecting factors experiments, we found the removal capacity of composite materials increased as the pH value increased in the solution. The pH of the biosorption solution affects both cell surface metal binding sites and metal chemistry in water. Because when the pH was 1 the H3O+ ions in the solution were at a high concentration and Cd2+ ions had to compete with them for biosorption sites. Ionic strength test results indicated the adsorption of cadmium ions on composite material is less affected by ionic strength. The whole adsorption process formed inner sphere surface complexes through surface complexation. The result of Zeta potential data shows that with the increase of pH value, the adsorbent surface absolute potential increased, which means there is more negative charge on the adsorbent surface to promote Cd2+ ions adsorbed in solution. Zeta potential results are consistent with the results of pH influencing factor. But, the experimental results of the temperature change were not related to the removal efficiency, which means no need to adjust the temperature in the actual pollution control in the future. The results of dynamic adsorption experiments show that the adsorption process consists of three stages which are same as previous studies (Zhang et al., 2015). The first is fast adsorption stage, a large number of Cd2+ ions were bound to the vacant active sites rapidly and the removal rate increased sharply in the first 60 min. The second stage, the removal rate slowed down gradually with vacant active sites decreased. The last stage is the equilibrium stage, which the vacant active sites were all combined with Cd2+ ions. This phenomenon may be due to the fact that, initially, all active sites on the surface of the adsorbents were vacant and the solution concentration was high. After that period, many surface active sites were occupied, so no increase in metal uptake was observed. The Cd2+ ions removal rate in the equilibrium phase was 71.56%, which is about three times than the control group. We can infer that the modified carbon fiber as carrier for Cd2+ ions adsorption is limited. The bacteria are still playing an important role in the whole adsorption process. The Boehm’s titration experiments also validate that conclusion; the surface acidity sites numbers of composite material were much more than modified PAN-CF. EDX results indicated that Cd2+ ions were adsorbed on the surface of the composite material as Kim et al. (2015) reported. As can be seen in Figure 9, Lagergren’s first-order kinetic equation and pseudo-second-order kinetic equation were used to simulate the whole process of Cd2+ biosorption. For the biosorption of Cd2+, the pseudo-second-order kinetic equations (
Conclusions
In summary, we have manufactured composite material with
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was financially supported by the National Science and Technology Major Project of China (No. 2013ZX07201007) and State Key Laboratory of Urban Water Resource and Environment Funding (No. 2017DX15).
