Abstract
Electrospinning can be used to create nanofiber mats with high material purity and a large inner surface, applicable for medical or biotechnological filters, cell growth, and so on. Not each polymer which can be dissolved, however, can be spun in this way. Depending on the material, solution, and spinning parameters, especially on the molecular weight and the polymer concentration in the solution, either fibers or droplets can be formed. Both fibers and droplets are of technological interest for different applications. This article examines the possibilities of electrospinning or electrospraying poloxamer 188 (formerly known as Lutrol F 68, BASF, Germany) in combination with different biopolymers (dextran, gelatin, and agarose) as well as polyacrylonitrile (PAN) which belongs to the polymers spinnable from nontoxic solvents. Due to the similarity of the chemical structures of poloxamer and poly(ethylene glycol), a well-known spinning agent, and the relatively low molecular weight of poloxamer 188, it can be expected to work as a spraying agent. Our results show that electrospraying poloxamer/biopolymer blends is indeed possible, with the unexpected effect that for higher polymer concentrations, combining poloxamer 188 with dextran, fibers are formed additionally on parts of the substrate. Co-spinning poloxamer 188 with PAN, on the other hand, results in creation of a nanofiber mat whose morphology is mainly defined by the PAN content. The study shows that poloxamer can indeed be used as a spraying/spinning agent in electrospraying/electrospinning diverse biopolymers.
Introduction
Electrospinning can be used to create fine fiber mats, usually with diameters of some hundred nanometers. 1 –5 Different technologies can be used to perform electrospinning, using a syringe to extrude the polymer into an electrical field, a rotating drum, a pair of wires one of which is continuously coated with a polymer solution, and so on. Spinning is possible from polymer solutions or melts, depending on the material and the equipment used.
Finding a suitable solvent for a desired polymer often causes problems since several solvents are toxic, corrosive, or may cause other problems, making them inappropriate for use in academic or non-specialized industry environments. Biopolymers, on the other hand, are often water soluble and can thus be spun from aqueous solutions. 6,7 A prominent example is poly(ethylene glycol) (PEG, also called poly(ethylene oxide) (PEO)) which can be used as a spinning agent, allowing for co-spinning polymers which do not form fibers themselves. 8,9
Interestingly, poloxamers have a similar chemical structure as PEO but are scarcely described for electrospinning. Poloxamers consist of a poly(propylene oxide) block in the middle of the chain which is insoluble and soluble PEO blocks at both ends of the chain. Thus, the ends of the chain are identical to PEG, with an additional insoluble part in the middle. This may lead to the idea to use poloxamers as spinning agents, too.
Additionally, aqueous solutions of poloxamers show an unusual thermo-gelling effect, being fluid at low temperature and forming a gel at higher temperatures. The temperature range of this reversible transition between both regimes depends on the exact polymer composition. 10
In spite of these interesting properties, poloxamers are only scarcely used for electrospinning. Poloxamers were blended with poly(epsilon-caprolactone) and used for drug delivery. 11 In combination with poly(epsilon-caprolactone-co-lactide), they were tested for applications in skin tissue engineering 12 as well as wound healing. 13
On the other hand, fiber blends are often used in electrospinning or electrospraying. For wound dressing applications, honey (which is naturally antimicrobial) was co-spun with poly(1,4-cyclohexane dimethylene isosorbide terephthalate) in different ratios, showing variations of the fiber morphology, mechanical properties, and contact angle, resulting in an ideal ratio for honey release. 14 Blending polyacrylonitrile (PAN) fibers with pitch increased conductivity and crystallinity of carbon nanofibers after carbonization. 15 Blending PAN with cellulose acetate increases the pore characteristics for adsorption performance of activated carbon nanofibers. 16 NIH/3T3 fibroblast cells could be grown on nanofiber mats from ternary blends of poly(lactic acid), polycaprolactone, and cellulose acetate butyrate (CAB), using blending to increase the surface smoothness due to CAB working as a compatibilizer. 17 A gel electrolyte was prepared blending poly(vinylidene fluoride), thermoplastic polyurethane, and PAN. 18 Even the creation of shape memory fibers by electrospinning polymer blends was shown in the literature. 19
Due to the significant increase of possible applications by blending different materials for fiber creation, we have investigated the possible use of poloxamers for electrospinning or—since they usually have smaller molecular weights than the PEG which is used as a spinning agent—for electrospraying. This technique can be used, for example, for microencapsulation 20 or coating 21 in biotechnological or medical applications. For PEG, fiber formation was found to strongly depend on the molecular weight, with a minimum value of approximately 100 kDa necessary to form fibers. 7 The poloxamer used here has a molecular weight of less than 10 kDa and was thus expected to form droplets instead of fibers.
Experimental
For electrospinning, the needleless electrospinning machine Nanospider Lab from Elmarco (Czech Republic) was used. This machine works in the following way: Between two wires in a changeable distance (here 240 mm), a strong electric field is created, using a high voltage of up to 80 kV. One of the wires is coated by a polymer solution, using a nozzle (here with diameter 0.9 mm) being moved along the wire with a velocity of 50–300 mm/s (here 200 mm/s). The electric field drags the dissolved polymer to the counter electrode which is shielded by a substrate on which the polymer fibers or droplets impinge. Depending on spinning and solution parameters, the polymer solution droplets carried away from the first wire can either be stretched on their way to the substrate and arrive there in the shape of fine fibers or—especially for lower molecular weights and lower polymer concentrations in the solution—they stay droplets. In the latter case, electrospraying instead of electrospinning occurs.
The poloxamer 188 “Lutrol F 68” (7680–9510 Da, 2 × 40% hydrophilic parts, purchased from BASF) was used as part of all aqueous spinning solutions. Additionally, Dextran 500 (for biochemistry, 500 kDa, purchased from Carl Roth GmbH + Co. KG, Karlsruhe, Germany) was used, as well as agarose and gelatine. These biopolymers were chosen since they cannot be spun solely and are often used in biotechnology as substrates for cell growth.
As a non-water-soluble polymer, PAN was partly used as a first layer below the water-soluble polymer layer or for co-spinning with the water-soluble materials. PAN (14%) was dissolved in DMSO (minimum 99.9%, purchased from S3 Chemicals, Germany).
In all experiments, care was taken to reduce the relative humidity in the chamber to 32–33% to increase the possibility of fiber formation. The temperature in the spinning chamber was 21–22°C.
Optical examination of the nanofiber mats and electrosprayed materials was performed using a confocal laser scanning microscope VK-9000 (Keyence, Neu-Isenburg, Germany) with a nominal magnification of 2000×. All scale bars have dimensions of 10 µm.
Results and discussion
First tests were performed combining poloxamer with PAN to ensure fiber creation. Here, poloxamer is not meant to work as a spraying agent but to add new functionalities to the PAN nanofiber mat.
Figure 1 depicts PAN/poloxamer co-spun nanofiber mats with different amounts of poloxamer. In both cases, the main structure of the fiber mat is defined by the PAN, which nevertheless shows some irregularities. It can be assumed that mixing both polymers does not result in a completely homogenous solution.

Nanofiber mats, electrospun from PAN 11.6% and poloxamer 6.9% (left panel) or 13% (right panel), respectively. PAN: polyacrylonitrile.
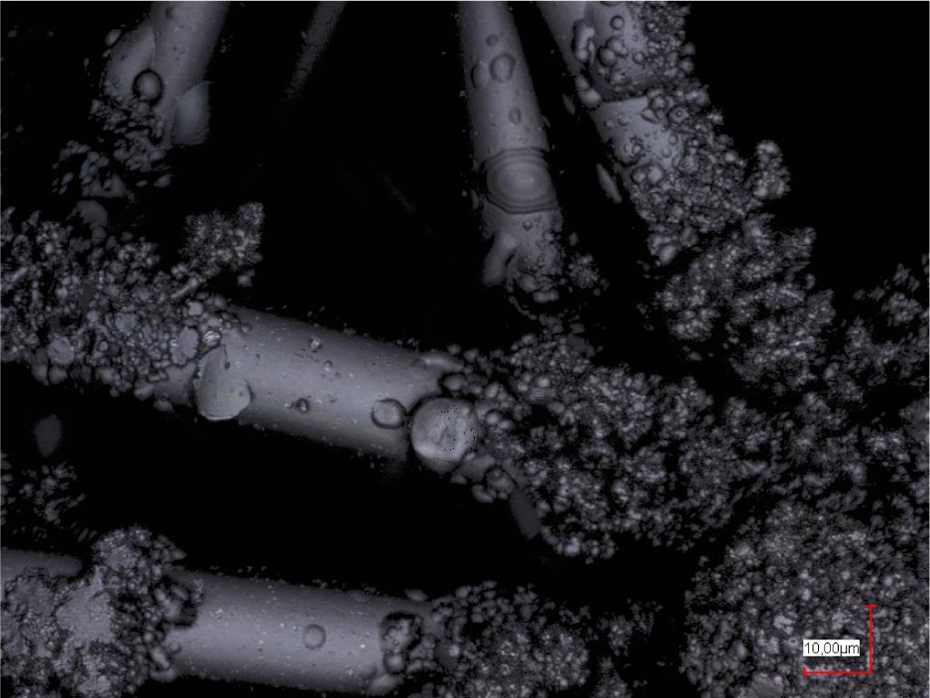
Thus, poloxamer was added as a second layer on a previously spun PAN nanofiber mat. Figure 2 depicts the results gained by electrospraying poloxamer in combination with dextran or gelatin, respectively, from an aqueous solution. In both cases, droplet formation on the PAN nanofiber mat is clearly visible. No differences of the morphologies gained by electrospraying poloxamer with dextran and with gelatin are visible.

Electrospraying on a PAN nanofiber mat (spun with 80 kV high voltage), using poloxamer/dextran (left panel) or gelatin 32%/poloxamer 2.25% (right panel) in the sprayed layer. PAN: polyacrylonitrile.
To examine the electrospraying process more in detail, different experiments were performed using the typical nanospinning substrate (polypropylene (PP)) as a base. Figure 3 depicts poloxamer (with a high concentration of 40%) electrosprayed on the PP nonwoven. The sprayed material seems to agglomerate at some positions, with the droplet diameters being relatively broadly distributed. Apparently, in this way no fine, homogenous electrosprayed layer is formed.
Electrosprayed poloxamer (40%) on a PP nonwoven. PP: polypropylene.
Thus, combinations of poloxamer with different biopolymers were tested next. Figure 4 shows the results of electrospraying/electrospinning poloxamer in combination with dextran since this blend showed a relatively homogenous droplet layer on PAN (cf. Figure 1). Here, the poloxamer concentration is lower than in the experiment depicted in Figure 3 but significantly higher than in the previous electrospraying test shown in Figure 2.

Electrospraying/electrospinning poloxamer 14% and dextran 13%. Images taken in the middle of the substrate (left panel) and near the border (right panel).
Firstly, in the middle of the substrate, a fine electrosprayed nano-droplet layer is visible, in the left part of the image shielded by a higher PP fiber during spinning. Unexpectedly, this behavior changes completely in the outer area of the substrate (Figure 4, right panel). Here, a nanofiber mat is visible, built from fibers of diameters approximately 1 µm. It should be mentioned that only for co-spinning poloxamer and dextran, fiber formation could be observed, while pure dextran did not have any effect at all (not even droplet formation) and pure poloxamer showed only electrospraying, as expected (cf. Figure 3).
The difference between both positions is that in the middle of the substrate, the electric field is stronger, forcing the dissolved polymer faster toward the substrate. Thus, here is usually less time between evaporation of the solvent and impinging on the substrate. Apparently, in this case, there is not enough time for fiber formation in the middle of the substrate, while this process has time to take place in the outer parts. This means, on the other hand, that it may be possible to tailor the transition between droplet and fiber formation by modifying the high voltage—if this value is slightly reduced, there should be more time for fiber formation, while increasing it (or decreasing the distance between the polymer-coated electrode and the substrate) should result in pure electrospraying along the whole substrate.
In the next step, the concentrations of poloxamer and dextran were reduced to perform pure electrospraying. Figure 5 shows the results of electrospraying with 7% poloxamer and 7.2% dextran on different parts of the PP substrate. In both images, the sprayed microparticles show similar diameters of approximately 1 µm. The distribution is significantly more even than for pure poloxamer with 40% concentration (Figure 3) and, as expected, less dense than for the case of doubled polymer concentrations (Figure 4). Depending on the planned application, it must be decided which droplet distribution should be preferred.
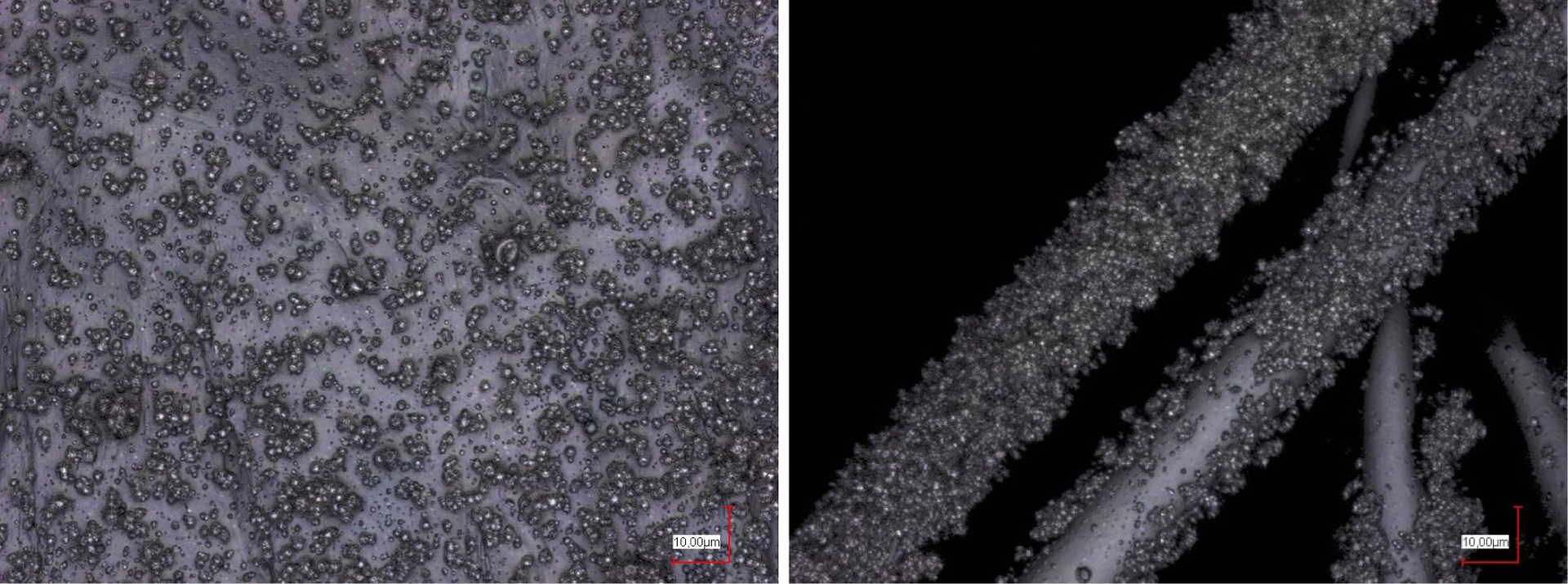
Electrospraying on plane (left panel) and fibrous parts of the PP substrate (right panel), using poloxamer 7% and dextran 7.2%. PP: polypropylene.
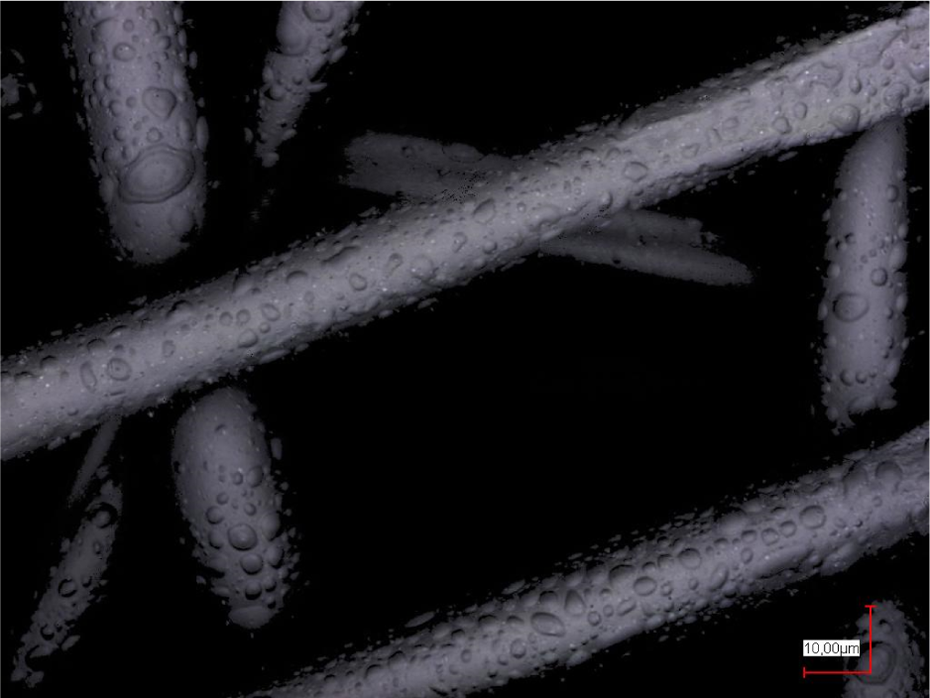
In the next step, the poloxamer concentration was further decreased, combined with the identical dextran concentration. The result (Figure 6) shows that decreasing the poloxamer concentration to 1.89%, while the dextran concentration of 7.2% is retained, results in a droplet distribution on the nonwoven substrate which is quite similar to the results depicted in Figure 5 (right panel), obtained with a significantly higher poloxamer concentration.

Poloxamer 1.89% and dextran 7.2%, electrosprayed on a PP nonwoven. PP: polypropylene.
Although dextran cannot be spun or sprayed solely, these experiments show that it is an important factor, influencing the electrospinning or electrospraying properties of poloxamer significantly. This is the reason why other biopolymers were examined in combination with poloxamer.
Figure 7 depicts the results of co-spinning poloxamer 1.75% and agarose 1.8%, using different nozzle diameters. While the larger nozzles usually lead to impaired fiber morphologies in electrospinning, here the larger nozzle apparently enables finer and more uniformly distributed polymer droplets. This shows the possibility that smaller poloxamer concentrations may also be suitable for electrospinning with different biopolymers if the nozzle diameter or possibly other geometrical parameters are optimized.
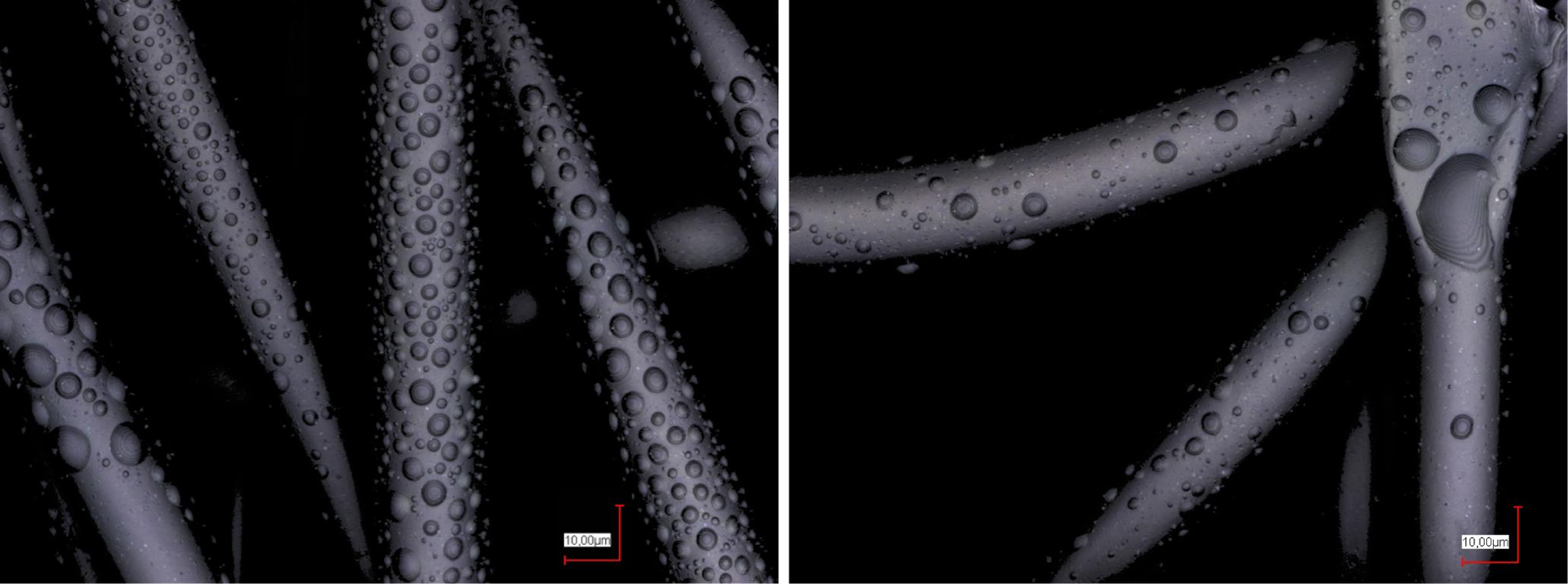
Poloxamer 1.75% with agarose 1.8%, electrosprayed using nozzles of 1.5 mm nozzle (left panel) or 0.9 mm diameter (right panel).
In Figure 8, the result of electrospinning the combination of poloxamer and agarose with approximately halved concentrations is depicted. Here, no electrosprayed droplets are visible anymore. Instead, a fine coating can be recognized. This effect may be useful for medical or biotechnological applications in which only very small amounts of different functional materials are needed.

Poloxamer 0.83% and agarose 0.85% after electrospraying.
With a higher poloxamer concentration and gelatin as the co-spinning material, the structure of the droplets electrosprayed on the PP substrate changes again (Figure 9). Instead of the rough, crystal-like droplets depicted in Figure 6 (left panel) or the round, thick droplets depicted in Figure 7 (left panel), here flat droplets with an uneven border are visible. Apparently, the spinning partner also has a significant impact on the resulting droplet morphology.
Poloxamer 2.25% and gelatin 32% after electrospraying.
Conclusions
Comparing results of electrospraying and electrospinning poloxamer with different biopolymers (dextran, gelatin, and agarose) shows a broad variety of possible morphologies, from droplets of different shapes and sizes to complete fibers. Only the combination of poloxamer and dextran resulted in fiber formation along the outer areas of the substrate, while none of the single materials could be electrospun solely. The parameters influencing the droplet morphology are poloxamer concentration, biopolymer concentration, and choice of the biopolymer. Here, only some examples from the broad range of possible combinations could be shown, suggesting further more detailed tests in the future to detect all possibilities of electrospinning or electrospraying different material combinations with different morphologies to extend the possible applications of poloxamer blends with respect to recent research areas. 11 –13 In this way, an electrospraying process of different biopolymers can be established, using poloxamer as a spraying agent, similar to PEG as an electrospinning agent, 8,9 to add functionalities to medical or biotechnological materials based on the intrinsic properties of the respective blended biopolymers.
Footnotes
Acknowledgement
The authors acknowledge gratefully the program FH Basis of the German federal country North Rhine-Westphalia for funding the “Nanospider Lab” and the Internal Project Funding of Bielefeld University of Applied Sciences for partly funding this project.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author received no financial support for the research, authorship, and/or publication of this article.
