Abstract
Mg/Al/Fe ternary layered double hydroxides (LDHs) were synthesized by a coprecipitation method using activated Bayer red mud as raw materials. Then, the prepared LDHs were intercalated with sodium dodecyl sulfate (SDS) and modified with (3-aminopropyl)triethoxysilane (KH550) at the surface. All samples were characterized by X-ray diffraction, thermogravimetric analysis, scanning electron microscopy, Fourier transform infrared spectrometry, and contact angle measurement. As flame-retardant filler, the KH550-modified composites showed better compatibility in comparison with both the original LDH and LDH-SDS. The composite ELDH-SDS-KH (ethylene-vinyl acetate/LDHs composites) showed not only a significant reduction in heat release rate, smoke production rate, and total heat release during combustion, but high thermal stability and less production of aliphatic hydrocarbons and carboxylic acid were also observed in comparison with other composites. Moreover, when the added amount of LDH is 50 wt%, the tensile strength value of ELDH-SDS-KH was 9.8 MPa, and the elongation at break value was 427%, which is significantly improved compared with ELDH (with the tensile strength value of 6.3 MPa and the elongation at break value 38%).
Keywords
Introduction
Ethylene-vinyl acetate (EVA) copolymer with different vinyl acetate contents is extensively used in many fields, such as wire, cable, wrapper, adhesive, and drug industries. 1 However, EVA resins are flammable and may release a large amount of toxic smoke during combustion. 2 Therefore, the applications of EVA-based materials have been limited owing to safety and environmental factors, especially in high-temperature conditions. A reasonable solution to this problem is the addition of a flame retardant which may improve the fire safety of EVA. In recent years, layered double hydroxides (LDHs) have been applied as a nonhalogenated additive in EVA because of their good fire retardancy.3,4
Hydrotalcites are also referred to as anionic clays or LDHs, which are brucite-like layered materials with a general chemical formula
Bayer red mud (BRM) as one typical kind of solid waste is formed in the production of alumina by leaching the bauxite in alkali. Our previous work has confirmed that LDHs can be successfully synthesized using BRM as raw materials.10-12 In this study, BRM was activated first by hydrochloric acid, and then Mg/Al/Fe LDHs was synthesized by a coprecipitation method. SDS was intercalated into the interlayer of LDHs during synthesis, and the surface of the intercalated LDH was modified with (3-aminopropyl)triethoxysilane (KH550). All samples were characterized by X-ray diffraction (XRD), thermogravimetric analysis (TGA), scanning electron microscopy (SEM), and Fourier transform infrared (FTIR) spectrometry. Moreover, the thermal behavior, flame retardancy, and mechanical properties of EVA and filler composites have been comparatively investigated using cone calorimeter test (CCT), tensile strength and elongation test, and thermogravimetry–Fourier transform infrared (TG-IR) spectrometry.
Experimental section
Materials
EVA18 (containing 18% vinyl acetate) was bought from Beijing Eastern Petrochemical Co., Ltd (China). BRM was kindly supplied by Aluminum Corporation of China Shandong Branch as a sponsored material. (3-Aminopropyl)triethoxysilane (KH550, 99%) was bought from Sinopharm Chemical Reagent Co., Ltd (China). Other reagents were standard laboratory reagents and used as received without further purification. The BRM was dried in an oven at 105°C for 24 h, calcined at 650°C for 4 h, and then ground and sieved to a size of <75 µm.
Synthesis of Mg/Al/Fe LDH
For BRM activation, 10 g of dried and sieved red mud was taken and 25 mL distilled water was added. After stirring for 5 min using a magnetic stirrer, 25 mL of 37% HCl was added. The sample was kept for digestion at 90°C for 90 min. Then, MgCl2·6H2O were mixed with the cooled acidic leaching liquid (with [Mg]/[Al + Fe] molar ratios of 2.0/1.0). The mixed aqueous solution was added slowly into 200 mL of deionized water, while the pH was adjusted to the range of 8.5–9.5 by drop-wise addition of NaOH-Na2CO3 mixed solution (1 mol L−1 NaOH and 0.4 mol L−1 Na2CO3), then the solution was vigorously stirred at 80°C. The resulting slurry was washed to neutral, dried at 80°C for 12 h, and ground into 500-mesh-pass particles to obtain Mg/Al/Fe LDH.
Synthesis of SDS-intercalated LDH (LDH-SDS)
The mixed aqueous solution (as mentioned before) was added slowly into 200 mL of SDS aqueous solution (0.15 mol L−1), while the pH was adjusted to a range of 8.5–9.5 by drop-wise addition of NaOH solution (1 mol L−1 NaOH), and the solution was vigorously stirred at 80°C. The resulting slurry was washed with hot water (70°C) to nearly neutral, dried at 80°C for 12 h, and ground into 500-mesh-pass particles to obtain LDH-SDS.
Surface modification of LDH-SDS
Ten grams of LDH-SDS were added to a flask containing 100 mL of toluene. The mixture was subjected to sonication in an ultra-sound bath for 15 min at room temperature. The obtained sample was reacted with 0.3 g of KH550 at 90°C. The precipitated solid was isolated and washed with toluene. Finally, the solid was dried at 120°C overnight. The powder was indicated as LDH-SDS-KH.
Preparation of EVA/LDH composites
The EVA/LDH composites were prepared via the melt blending method at 120°C under a pressure of 10 MPa for 10 min with a rotor speed of 30 r min−1. In this work, the additive level of all samples was 50%, and the samples were named as EVA (containing 100% EVA), ELDH (containing 50% LDH), ELDH-SDS (containing 50% LDH-SDS), and ELDH-SDS-KH (containing 50% LDH-SDS-KH), respectively.
Characterization techniques
Powder XRD data were recorded at room temperature on a Philips X’Pert Pro Super apparatus (Nicolet Instrument Co., Madison, Wisconsin, USA) using Cu Ka radiation with a nickel filter (wavelength = 1.5418 Å) at a scan rate of 0.0167° s−1. The SEM studies were performed using a Hitachi X650 scanning electron microscope (Hitachi, Tokyo, Japan). The TG-IR instrument consists of a thermogravimeter (TG209 F1, Netzsch Instruments, Germany), a Fourier transform infrared spectrometer (Vertex70, Bruker Optics, Germany), and a transfer tube with an inner diameter of 1 mm connected to the TG and the infrared cell. The investigation was carried out from 30°C to 800°C at a linear heating rate of 20°C min−1 under a nitrogen flow of 30 mL min−1, and the FTIR was not used when the TG behavior of the LDHs was tested. Contact angles were measured using a JC2000A contact angle/interface tensile measurer. All CCTs were carried out in a cone calorimeter (Stanton Redcroft, UK) at an incident heat flux of 50 kW m−2 according to the ISO 5660-1 standard. The elongation at break and tensile strength of all composites was performed by a GT-TCS-2000 material test machine (produced by Guangdong GOTECH Testing Machines Co., Ltd, China), according to the national standard GB/T 1040.1-2006.
Results and discussion
Powder XRD characterization of LDHs
Figure 1 shows the XRD patterns of LDH, LDH-SDS, and LDH-SDS-KH. The strong (003), (006), and (009) reflections denoting relatively good degree of crystallinity for all LDHs with layered structures. As observed from Figure 1(A) and (B) and Table 1, it can be found that after intercalated with dodecyl sulfate, the (003), (006), and (009) peaks of LDH-SDS moved to lower 2θ angles corresponding to reflections positioned around 3.11°, 7.18°, and 11.22°, respectively. And the d(003) value (2.782 nm) indicates that the interlayer space was enlarged by 1.993 nm compared with LDH. Moreover, a broad reflection in the range of 20°–23° also confirmed that the hydrocarbon chains of SDS were intercalated into the interlayer of LDHs.13,14

X-ray diffraction patterns for LDH samples. (a) LDH, (b) LDH-SDS, and (c) LDH-SDS-KH.
Structural parameters of LDH samples.

LDH: layered double hydroxide; SDS: sodium dodecyl sulfate.
It has been generally acknowledged that the thickness of a brucite-like layer is about 0.48 nm, and the interlayer space between the brucite-like layers is about 2.6 nm when the SDS anions are intercalated into LDH by means of monolayer and perpendicular mode. 15 Moreover, after surface modification by KH550, crystal texture and structural parameters of LDH-SDS-KH were essentially consistent with LDH-SDS, it indicated that the surface modification did not change the structure of the hydrotalcite significantly, similar conclusion has been found by Dong et al. 16
FTIR spectroscopy of LDH
Figure 2 illustrates the FTIR spectra of LDH, LDH-SDS, and LDH-SDS-KH, respectively. The broad absorption bands centered around 3496 cm−1 were derived from the O–H stretching vibration of the layers and the interlayer water, while the bending vibration of hydroxyl group appeared at 1623 cm−1. Typical C–H stretching modes at 2932, 2846, and 1467 cm−1 could be assigned to the CH2 scissoring vibration of alkyl chains, and –OSO3 group bands at 1208 cm−1 are observed. In the spectra of LDHs modified with KH550, characteristic bands of –NH2 group and Si–O from KH550 at 1569 and 1035 cm−1 are detected. Furthermore, stretch modes of amino group should appear at 3300 cm−1, but it is difficult to distinguish because of the coverage coming from the broad bond of hydroxyl groups.16,17

FTIR spectra of three LDHs samples.
Thermal analysis of LDHs
Figure 3(a) and (b) illustrates the TG and derivative thermogravimetric (DTG) curves of LDH, LDH-SDS, and LDH-SDS-KH, respectively. For LDH, two major mass loss steps were observed. The first region with a mass loss of 10.1 wt% from 30°C to 200°C corresponding to the loss of physisorbed and interlayer water, with the maximum mass loss rate temperature at 165°C. 18 The second step with a mass loss of 21.3 wt% (in the temperature range of 220–480°C with a strong peak at 347°C) can be ascribed to the processes of dehydroxylation and carbonates. In this step, the destruction of the layered structure of hydrotalcite and formation of the mixed-oxide structure took place. 11 For LDH-SDS, the maximum mass loss rate temperature can be observed at 144, 289, 369, and 454°C in the DTG curve, respectively. The intense and sharp peak at 369°C could be attributed to the decomposition of the SDS. Interestingly, the maximum mass loss rate temperature of hydroxyls and carbonates occurred at 454°C, which is 117°C higher than that of LDH. 19
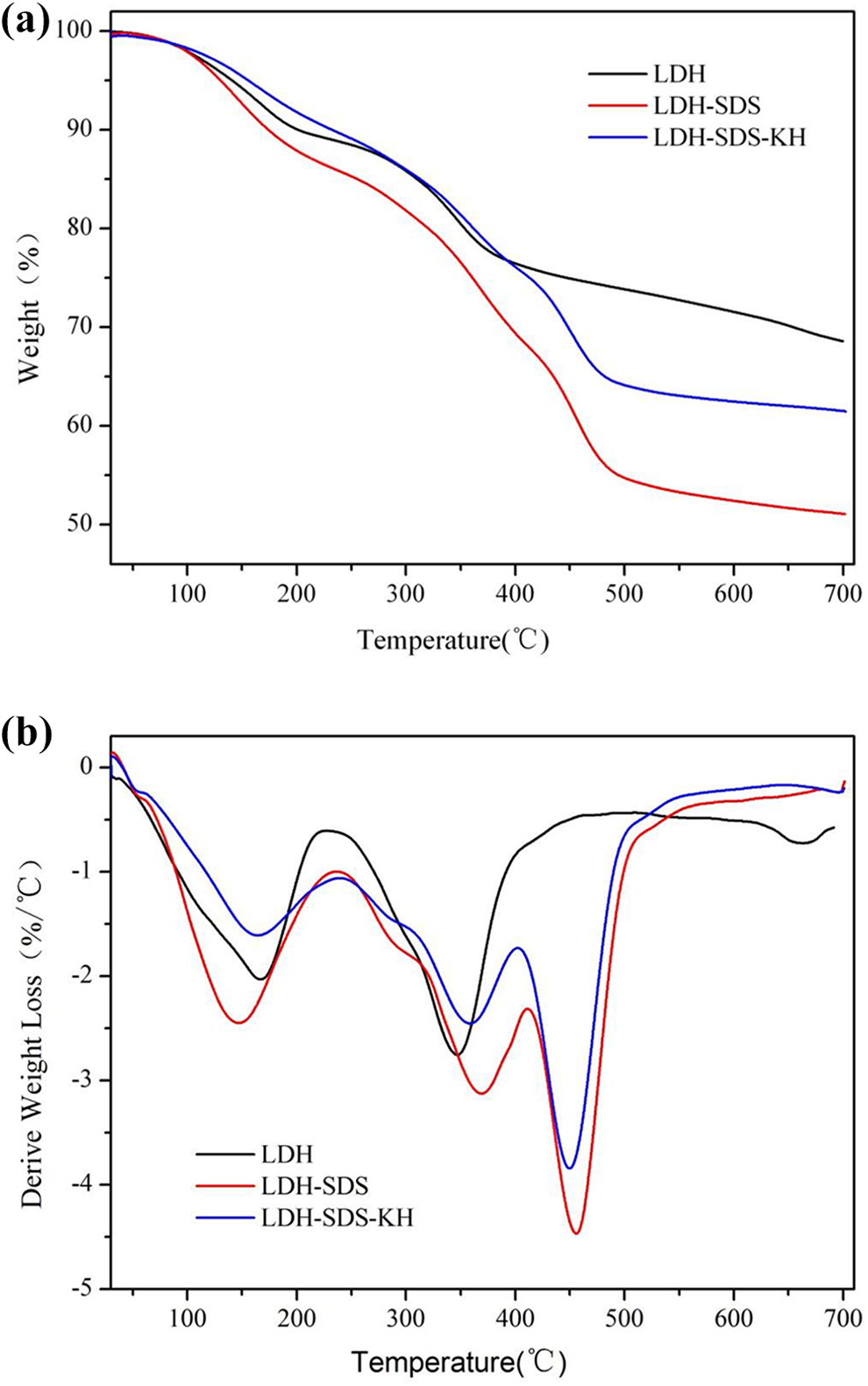
(a) TG and (b) DTG curves of three LDHs samples.
The TG and DTG curves of LDH-SDS-KH illustrate that the maximum mass loss rate temperatures are at 159°C, 285°C, 355°C, and 447°C. Comparing with LDH-SDS, the mass loss rate of LDH-SDS-KH decreased, and it demonstrates that the thermal stability of LDH-SDS-KH is increased after surface modification by KH550. In addition, the final residue masses of the three LDHs at 700°C were also different. The values are 68, 50, and 60 wt% for LDH, LDH-SDS, and LDH-SDS-KH, respectively. The above results indicate that the SDS anions intercalated into the interlayer of LDHs and surface modification have different influences on the thermal degradation temperature and the mass loss.16,19,20
Morphological properties of LDHs
Figure 4 illustrates the SEM images of LDH, LDH-SDS, and LDH-SDS-KH, respectively. It can be observed from Figure 4(a) that there are some big agglomerates, and no predominant shape for the particles can be observed. It is worth noticing that the morphologies of modified LDH (LDH-SDS and LDH-SDS-KH) are significantly different to that of LDH. For the SEM image of LDH-SDS, it can be seen that, owing to the intercalation of SDS anions, smaller agglomerates and particles were observed. After surface modification by KH550, the sizes of Mg/Al/Fe-SDS-Si particles are more uniform with flake graphics (Figure 4(c)). This result indicated that intercalation of SDS anions and surface modification by KH550 change the morphological structure of LDHs particle.

SEM images of three LDHs samples. (a) LDH, (b) LDH-SDS, and (c) LDH-SDS-KH.
Morphological structures of the composites
ELDH, ELDH-SDS, and ELDH-SDS-KH samples were brittle fractured in liquid nitrogen, and the morphologies of the fracture are shown in Figure 5. It can be seen that the unmodified LDH was homogeneously embedded in EVA matrix, and the interface between EVA and LDH was visible. However, the modified LDHs, LDH-SDS, and LDH-DDS-KH showed both homogeneous dispersion and good compatibility with the EVA matrix. Especially, the fracture surface of ELDH-SDS-KH composite was smoother, and the interface between EVA and LDH-DDS-KH cannot be distinguished clearly. It is interesting that the morphological structures of the composites are different, the reason may be ascribed to the different interfacial properties of the three samples.

SEM micrographs of fractured surface for composites with 50 wt% loading of LDH. (a) ELDH, (b) ELDH-SDS, and (c) ELDH-SDS-KH.
Contact angle analysis
Figure 6(a) to (d) shows the images of contact angle for LDH, LDH-SDS, LDH-SDS-KH, and EVA, respectively. Figure 6(a) illustrates that the contact angle of LDH is 41°, which indicated that the surface of LDH was hydrophilic. For LDH-SDS (see Figure 6(b)), the contact angle value is 58°, which is greater by 17° in comparison with that of LDH. This result revealed that the hydrophobicity of LDH-SDS was increased because of the SDS anions intercalated into LDH. 19 Moreover, the result of the contact angle measurement for LDH-SDS-KH showed that the contact angle value is 98° (see Figure 6(c)). It is demonstrated that LDH-SDS-KH were hydrophobic, having a contact angle value comparable with that of EVA (102°) (see Figure 6(d)). These results indicated that modification has changed LDH from hydrophilic to hydrophobic, which may be beneficial for its distribution in EVA.

Contact angle of the samples. (a) LDH, (b) LDH-SDS, (c) LDH-SDS-KH, and (d) EVA.
Combustion behavior of EVA and its composites
The cone calorimeter based on the oxygen consumption principle has widely been used to evaluate the flammability characteristics of materials. 7 The fire and smoke properties of EVA and its composites with 50 wt% loading of LDH were evaluated using the cone calorimeter. The results are shown in Table 2 and Figure 7(a) to (d).
Cone calorimetric results for EVA and its composites with 50 wt% loading of LDH (heat flux of 50 kW m−2).

THR: total heat release; EVA: ethylene-vinyl acetate; LDH: layered double hydroxide; SDS: sodium dodecyl sulfate.
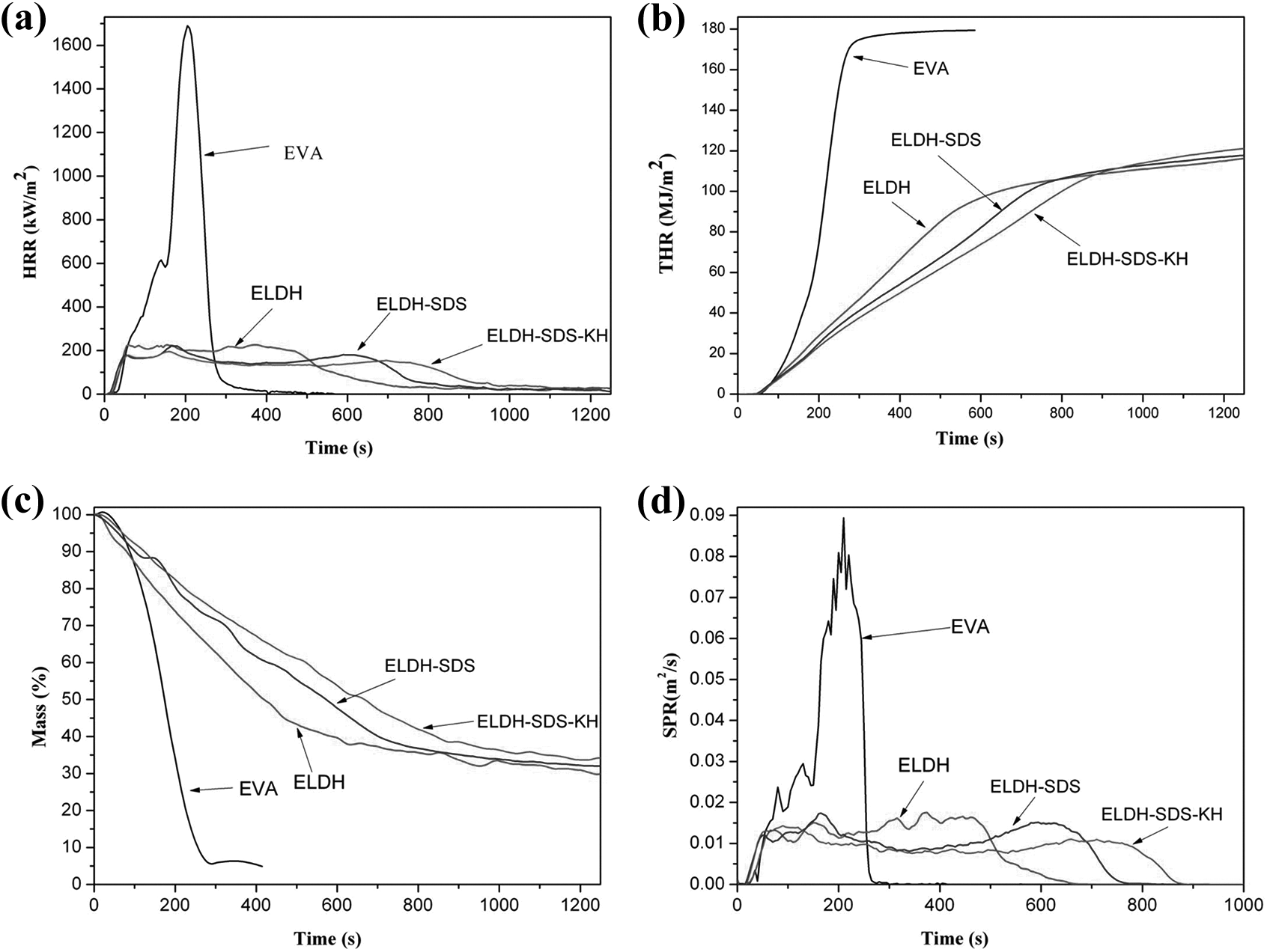
Cone calorimetric results of EVA and its composites with 50 wt% loading of LDHs. (a) HRR curves, (b) THR curves, (c) mass curves, and (d) SPR curves.
From Table 2 and Figure 7(a), it can be observed that pure EVA burns very fast after ignition, a very sharp HRR curve appears at the range of 50–300 s, and the maximal peak value of HRR for EVA is 1694.8 kW m−2; after the introduction of LDH, the HRR curve of the composites ELDH, ELDH-SDS, and ELDH-SDS-KH are much lower than pure EVA, and the pk-HRR values are 227.2, 220.6, and 195.1 kW m−2, respectively. At the same time, “time to ignition” and “combustion time” increased at different rates. In fact, once ignited, the subsequently generated carbonaceous layer, H2O and CO2 not only dilute the oxygen and volatile products but also reduce the heat transfer between the EVA matrix and the heat source by absorbing the primary heat and affecting heat transfer at the surface of the substrate.21,4 As can be observed from the THR curves in Figure 7(b), ELDH, ELDH-SDS, and ELDH-SDS-KH composites have similar THR values, which are significantly reduced in comparison with the EVA copolymer. In addition, the slope of THR curve can be assumed as representative of fire spread. 22 From Figure 7(b), it can be observed that the flame spread of samples ELDH-SDS-KH is comparatively the lowest, consistent with the result of HRR. Figure 7(c) shows the weight of the char residues. The char on the surface would act as a protective barrier and can thus limit the oxygen diffusion to the substrate or give a less dangerous low volatilization rate. In this study, ELDH, ELDH-SDS, and ELDH-SDS-KH composites have similar char residue, but pure EVA with almost nothing was left. The release of smoke is considered as another important parameter for the flame retarded materials. Figure 7(d) shows the SPR curves of the EVA and its composites with 50 wt% loading of LDH, respectively. The values of SPR for ELDH, ELDH-SDS, and ELDH-SDS-KH composites are significantly reduced compared with the EVA. The above cone calorimeter data provide evidence that the three composites perform better in flame retardancy and smoke suppression processes.
TG-FTIR characterization of EVA and its composites
Figure 8(a) and (b) illustrates the TG and DTG curves of EVA and its composites with 50 wt% loading of LDH, respectively. The weight loss of EVA showed two steps of degradation pathways in the temperature range of 300–500°C, which could be assigned to the loss of acetic acid and the degradation of the resulting unsaturated material, poly(ethylene-co-acetylene), respectively. 23 However, the other composites displayed three steps of mass loss. The first step corresponds to the removal of the interlayer water molecules; the second and third steps belong to the dehydroxylation and decarbonation of the LDH, which overlap with the decomposition of the acetate groups in EVA side chains and the scission of the main chains of EVA. 24 From the DTG curves, it can be seen that their maximum mass loss rate temperature of ethylene-based chains is almost the same as that of the pristine EVA at 470°C. Furthermore, the TGA profiles of all composites are shifted to higher degradation temperatures when mass loss exceeds 40 wt%. Moreover, when the temperature was higher than 700°C, the no residue was observed for EVA, and about 30 wt% ELDH-SDS and ELDH-SDS-KH samples were left.
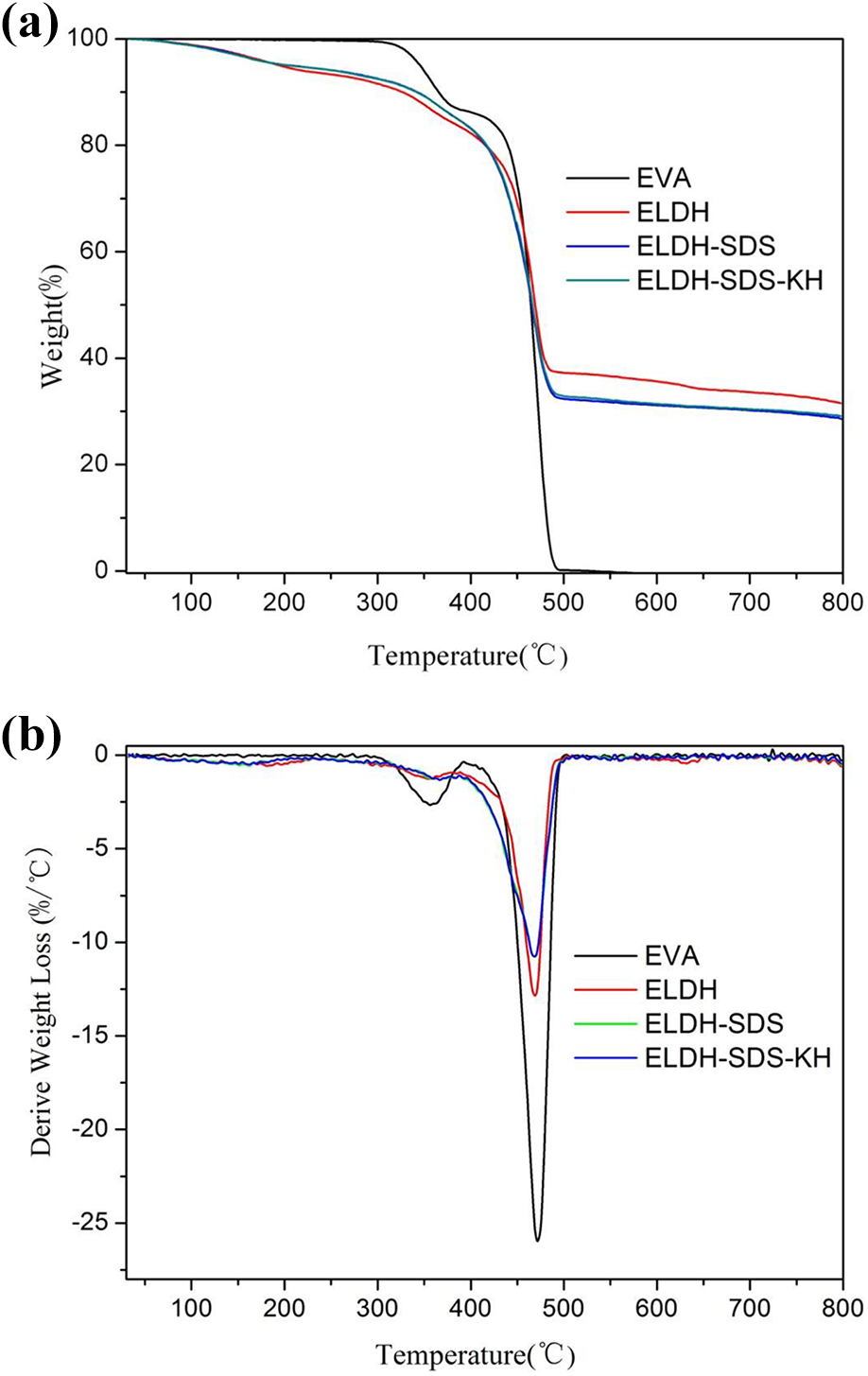
(a) TG and (b) DTG curves of EVA and its composites with 50 wt% loading of LDH.
The evolved gases from virgin EVA, ELDH, ELDH-SDS, and ELDH-SDS-KH were analyzed using TGA-FTIR. The spectra collected were found to be very similar, showing evolution of acetic acid, carbon dioxide, and water in the early stages, followed only by hydrocarbons, as shown in Figures 9(a) to (d) and 10(a) to (d). Figure 9(a) to (d) shows the 3D TG-IR spectra of pyrolysis products of the composites during the thermal degradation. The evolved gas products for the four samples assumed characteristic bands main in 3400–4000, 2800–3150, 2250–2400, 1700–1850, 1250–1500, and 950–1150 cm−1, which fitted to H2O (3400–4000 cm−1), CO2 (2300–2400 cm−1), CO (2250–2300 cm−1), carboxylic acid (1700–1850 cm−1), and aliphatic hydrocarbons (2800–3150, 1250–1500, and 950–1150 cm−1), respectively. Depolymerization is known as a process associated with thermal degradation of polymers. The main decomposition products of the composites in this research were listed as above.25-27 It can be noticed that the depolymerization processes of the four samples were different from their pyrolysis products after thermal degradation. There is nothing to be found in virgin EVA until the temperature increases to 290°C, and it then decomposes quickly with the increase of temperature. When the temperature is about 360°C, a maximum signal intensity at 1700–1850 cm−1, reflecting evolution of carboxylic acid, can be observed. A maximum signal at 2800–3150 cm−1, related to aliphatic hydrocarbons, appeared at 490°C. The signal intensity of the pyrolysis products declined gradually above 490°C, implying a slower decomposition rate of the mixture. The decomposition of the composites obviously slowed down, and the signals of aliphatic hydrocarbons and carboxylic acid were obviously less than the pure EVA. The above analysis results indicated that all the LDHs significantly enhanced the thermostability of the composites.

3D TG-FTIR spectra of the pyrolysis products of EVA and its composites with 50 wt% loading of LDHs during thermal degradation. (a) EVA, (b) ELDH, (c) ELDH-SDS, and (d) ELDH-SDS-KH.

FTIR spectra of pyrolysis products of EVA and its composites with 50 wt% loading of LDHs at various temperatures. (a) EVA, (b) ELDH, (c) ELDH-SDS, and (d) ELDH-SDS-KH.
Mechanical properties of EVA and its composites
The tensile strength and elongation at break for EVA and its composites with 50 wt% loading of LDHs are shown in Table 3. The tensile strength of ELDH, ELDH-SDS, and ELDH-SDS-KH are 6.3, 5.9, and 9.8 MPa, respectively. After SDS intercalation, the tensile strength value of ELDH-SDS is lower than that of ELDH composites. However, after surface modification with KH550, the tensile strength of the composites increased 66.1%. From Table 3, we also find that the LDH clearly affected the elongation at break of the composites, compared with that of the EVA which is 763%. It can be seen that all elongation at break values of composites with 50 wt% loading of LDH are lower than that of the pristine EVA. This result indicates that the ductility of composites was clearly reduced. And the elongation at break values of the three composites were in the order: ELDH-SDS-KH (at 427%) > ELDH-SDS (at 223%) > ELDH (at 38%). As expected, the addition of LDH-SDS-KH enhanced evidently the strength and ductility of the EVA. This is attributed to the good compatibility between the modified LDH and the EVA matrix. Therefore, LDH-SDS-KH is the best reinforcing filler in EVA.
Tensile strength and the elongation at break of composites EVA, ELDH, ELDH-SDS, and ELDH-SDS-KH.
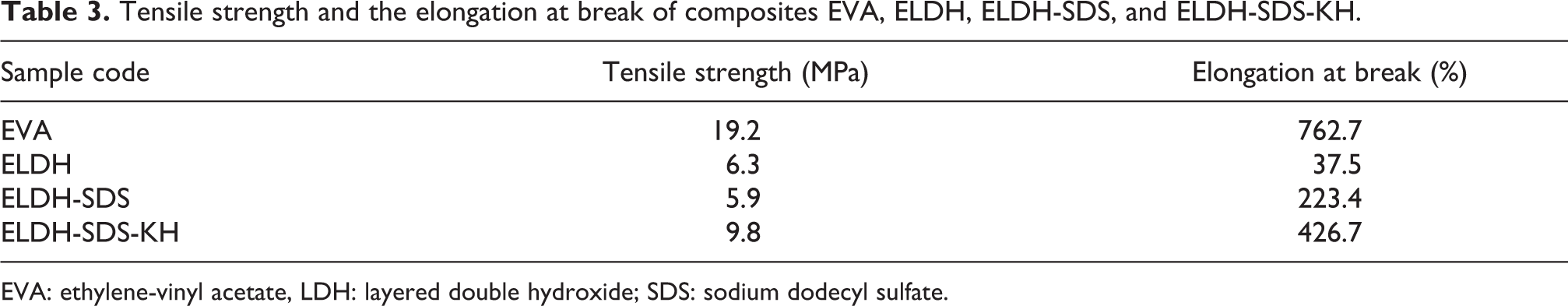
EVA: ethylene-vinyl acetate, LDH: layered double hydroxide; SDS: sodium dodecyl sulfate.
Conclusion
LDH was synthesized by a coprecipitation method based on activated BRM. The LDHs intercalated with SDS and surface modified with KH550 were prepared. The XRD and FTIR results showed all three LDHs were synthesized successfully. The results of the contact angle tests indicated that the surfaces of the modified LDH showed a change from hydrophilic to hydrophobic. The composite ELDH-SDS-KH showed the highest thermal stability. Furthermore, the three composites including ELDH, ELDH-SDS, and ELDH-SDS-KH performed excellently in flame retardancy and smoke suppression. In addition, LDH intercalated with anion surfactant SDS and surface modified by KH550 could greatly improve the compatibility of LDHs with EVA matrix, with a result of good mechanical properties.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was financially supported by the National Natural Science Foundation of China (No. 51572138), the Key R&D Project of Shandong Province (Nos 2019GSF109001 and 2019CSF109080), the Shandong Provincial Natural Science Foundation, China (No. ZR2018BB072), the Original Innovation Project of Qingdao City (No. 19-6-2-23-cg), the Foundation of State Key Laboratory of High-Efficiency Utilization of Coal and Green Chemical Engineering (Nos 2018-K09 and 2018-K43), the Key Laboratory of Coastal Environmental Processes and Ecological Remediation, YICCAS (No. 2018KFJJ02), and the Opening Project of Shandong Ecochemical Engineering Collaborative Innovation Center (No. XTCXQN02).
